Developmental Mechanism - Morphodynamics: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 19: | Line 19: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Review - Heterochromatin Morphodynamics in Late Oogenesis and Early Embryogenesis of Mammals'''{{#pmid:32575486|PMID32575486}} "During the period of oocyte growth, chromatin undergoes global rearrangements at both morphological and molecular levels. An intriguing feature of oogenesis in some mammalian species is the formation of a heterochromatin ring-shaped structure, called the karyosphere or surrounded "nucleolus", which is associated with the periphery of the nucleolus-like bodies (NLBs). Morphologically similar heterochromatin structures also form around the nucleolus-precursor bodies (NPBs) in {{zygote}}s and persist for several first cleavage divisions in blastomeres. Despite recent progress in our understanding the regulation of gene silencing/expression during early mammalian development, as well as the molecular mechanisms that underlie chromatin condensation and heterochromatin structure, the biological significance of the karyosphere and its counterparts in early embryos is still elusive. We pay attention to both the changes of heterochromatin morphology and to the molecular mechanisms that can affect the configuration and functional activity of chromatin. We briefly discuss how DNA methylation, post-translational histone modifications, alternative histone variants, and some chromatin-associated non-histone proteins may be involved in the formation of peculiar heterochromatin structures intimately associated with NLBs and NPBs, the unique nuclear bodies of oocytes and early embryos." | |||
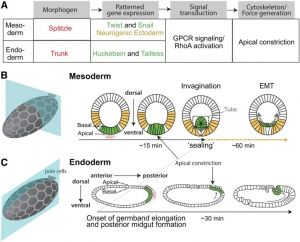
* '''The Physical Mechanisms of {{Drosophila}} {{Gastrulation}}: {{Mesoderm}} and {{Endoderm}} Invagination'''{{#pmid:32132154|PMID32132154}} "A critical juncture in early development is the partitioning of cells that will adopt different fates into three germ layers: the ectoderm, the mesoderm, and the endoderm. This step is achieved through the internalization of specified cells from the outermost surface layer, through a process called gastrulation. In Drosophila, gastrulation is achieved through cell shape changes (i.e., apical constriction) that change tissue curvature and lead to the folding of a surface epithelium. Folding of embryonic tissue results in mesoderm and endoderm invagination, not as individual cells, but as collective tissue units. The tractability of Drosophila as a model system is best exemplified by how much we know about Drosophila gastrulation, from the signals that pattern the embryo to the molecular components that generate force, and how these components are organized to promote cell and tissue shape changes. For mesoderm invagination, graded signaling by the morphogen, Spätzle, sets up a gradient in transcriptional activity that leads to the expression of a secreted ligand (Folded gastrulation) and a transmembrane protein (T48). Together with the GPCR Mist, which is expressed in the mesoderm, and the GPCR Smog, which is expressed uniformly, these signals activate heterotrimeric G-protein and small Rho-family G-protein signaling to promote apical contractility and changes in cell and tissue shape. A notable feature of this signaling pathway is its intricate organization in both space and time. At the cellular level, signaling components and the cytoskeleton exhibit striking polarity, not only along the apical-basal cell axis, but also within the apical domain. Furthermore, gene expression controls a highly choreographed chain of events, the dynamics of which are critical for primordium invagination; it does not simply throw the cytoskeletal "on" switch." {{fly}} | * '''The Physical Mechanisms of {{Drosophila}} {{Gastrulation}}: {{Mesoderm}} and {{Endoderm}} Invagination'''{{#pmid:32132154|PMID32132154}} "A critical juncture in early development is the partitioning of cells that will adopt different fates into three germ layers: the ectoderm, the mesoderm, and the endoderm. This step is achieved through the internalization of specified cells from the outermost surface layer, through a process called gastrulation. In Drosophila, gastrulation is achieved through cell shape changes (i.e., apical constriction) that change tissue curvature and lead to the folding of a surface epithelium. Folding of embryonic tissue results in mesoderm and endoderm invagination, not as individual cells, but as collective tissue units. The tractability of Drosophila as a model system is best exemplified by how much we know about Drosophila gastrulation, from the signals that pattern the embryo to the molecular components that generate force, and how these components are organized to promote cell and tissue shape changes. For mesoderm invagination, graded signaling by the morphogen, Spätzle, sets up a gradient in transcriptional activity that leads to the expression of a secreted ligand (Folded gastrulation) and a transmembrane protein (T48). Together with the GPCR Mist, which is expressed in the mesoderm, and the GPCR Smog, which is expressed uniformly, these signals activate heterotrimeric G-protein and small Rho-family G-protein signaling to promote apical contractility and changes in cell and tissue shape. A notable feature of this signaling pathway is its intricate organization in both space and time. At the cellular level, signaling components and the cytoskeleton exhibit striking polarity, not only along the apical-basal cell axis, but also within the apical domain. Furthermore, gene expression controls a highly choreographed chain of events, the dynamics of which are critical for primordium invagination; it does not simply throw the cytoskeletal "on" switch." {{fly}} | ||
Revision as of 10:03, 13 September 2020
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Morphodynamics refers to the biomechanical effects involved in development. There are several researchers who continue to build on concepts developed by Blechschmidt and others describing developmental events in terms of the physics involved in stresses and fluid movement within the embryo as important factors involved in establishing embryonic structures.
This page is an attempt to include concepts related to development based upon the physics (stresses, strains, gravity and fluid movement) occuring during growth. In some respects this is in response to the very dominant "molecular" nature of recent studies in comparison to the many other ways of describing developmental events. The two area appear more recently to be converging using new molecular findings to be incorporated or married with the morphodynamic descriptions.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Morphodynamics |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
References
- ↑ 1.0 1.1 Martin AC. (2020). The Physical Mechanisms of Drosophila Gastrulation: Mesoderm and Endoderm Invagination. Genetics , 214, 543-560. PMID: 32132154 DOI.
- ↑ Bogolyubova I & Bogolyubov D. (2020). Heterochromatin Morphodynamics in Late Oogenesis and Early Embryogenesis of Mammals. Cells , 9, . PMID: 32575486 DOI.
- ↑ Labernadie A & Trepat X. (2018). Sticking, steering, squeezing and shearing: cell movements driven by heterotypic mechanical forces. Curr. Opin. Cell Biol. , 54, 57-65. PMID: 29719271 DOI.
- ↑ Smutny M, Ákos Z, Grigolon S, Shamipour S, Ruprecht V, Čapek D, Behrndt M, Papusheva E, Tada M, Hof B, Vicsek T, Salbreux G & Heisenberg CP. (2017). Friction forces position the neural anlage. Nat. Cell Biol. , 19, 306-317. PMID: 28346437 DOI.
- ↑ Sutherland AE. (2016). Tissue morphodynamics shaping the early mouse embryo. Semin. Cell Dev. Biol. , 55, 89-98. PMID: 26820524 DOI.
- ↑ Varner VD, Gleghorn JP, Miller E, Radisky DC & Nelson CM. (2015). Mechanically patterning the embryonic airway epithelium. Proc. Natl. Acad. Sci. U.S.A. , 112, 9230-5. PMID: 26170292 DOI.
- ↑ Imuta Y, Koyama H, Shi D, Eiraku M, Fujimori T & Sasaki H. (2014). Mechanical control of notochord morphogenesis by extra-embryonic tissues in mouse embryos. Mech. Dev. , 132, 44-58. PMID: 24509350 DOI.
- ↑ Kim HY, Varner VD & Nelson CM. (2013). Apical constriction initiates new bud formation during monopodial branching of the embryonic chicken lung. Development , 140, 3146-55. PMID: 23824575 DOI.
Textbooks
Reviews
Heyn R, Makabe S & Motta PM. (2001). Ultrastructural morphodynamics of human Sertoli cells during testicular differentiation. Ital J Anat Embryol , 106, 163-71. PMID: 11732573
Articles
Search PubMed
Search Pubmed: Embryo Morphodynamics | Morphodynamics
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Developmental Mechanism - Morphodynamics. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Mechanism_-_Morphodynamics
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
