Introduction
Morphodynamics refers to the biomechanical effects involved in development. There are several researchers who continue to build on concepts developed by Blechschmidt and others describing developmental events in terms of the physics involved in stresses and fluid movement within the embryo as important factors involved in establishing embryonic structures.
Mechanism - "a process, technique, or system for achieving a result".
This page is an attempt to include concepts related to development based upon the physics (stresses, strains, gravity and fluid movement) occuring during growth. In some respects this is in response to the very dominant "molecular" nature of recent studies in comparison to the many other ways of describing developmental events. The two area appear more recently to be converging using new molecular findings to be incorporated or married with the morphodynamic descriptions.
Some Recent Findings
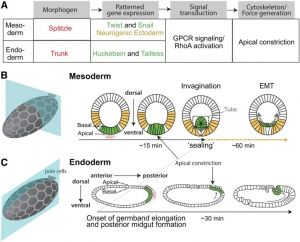
Drosophila gastrulation
[1]
- Review - Heterochromatin Morphodynamics in Late Oogenesis and Early Embryogenesis of Mammals[2] "During the period of oocyte growth, chromatin undergoes global rearrangements at both morphological and molecular levels. An intriguing feature of oogenesis in some mammalian species is the formation of a heterochromatin ring-shaped structure, called the karyosphere or surrounded "nucleolus", which is associated with the periphery of the nucleolus-like bodies (NLBs). Morphologically similar heterochromatin structures also form around the nucleolus-precursor bodies (NPBs) in zygotes and persist for several first cleavage divisions in blastomeres. Despite recent progress in our understanding the regulation of gene silencing/expression during early mammalian development, as well as the molecular mechanisms that underlie chromatin condensation and heterochromatin structure, the biological significance of the karyosphere and its counterparts in early embryos is still elusive. We pay attention to both the changes of heterochromatin morphology and to the molecular mechanisms that can affect the configuration and functional activity of chromatin. We briefly discuss how DNA methylation, post-translational histone modifications, alternative histone variants, and some chromatin-associated non-histone proteins may be involved in the formation of peculiar heterochromatin structures intimately associated with NLBs and NPBs, the unique nuclear bodies of oocytes and early embryos."
- The Physical Mechanisms of Drosophila gastrulation: mesoderm and endoderm Invagination[1] "A critical juncture in early development is the partitioning of cells that will adopt different fates into three germ layers: the ectoderm, the mesoderm, and the endoderm. This step is achieved through the internalization of specified cells from the outermost surface layer, through a process called gastrulation. In Drosophila, gastrulation is achieved through cell shape changes (i.e., apical constriction) that change tissue curvature and lead to the folding of a surface epithelium. Folding of embryonic tissue results in mesoderm and endoderm invagination, not as individual cells, but as collective tissue units. The tractability of Drosophila as a model system is best exemplified by how much we know about Drosophila gastrulation, from the signals that pattern the embryo to the molecular components that generate force, and how these components are organized to promote cell and tissue shape changes. For mesoderm invagination, graded signaling by the morphogen, Spätzle, sets up a gradient in transcriptional activity that leads to the expression of a secreted ligand (Folded gastrulation) and a transmembrane protein (T48). Together with the GPCR Mist, which is expressed in the mesoderm, and the GPCR Smog, which is expressed uniformly, these signals activate heterotrimeric G-protein and small Rho-family G-protein signaling to promote apical contractility and changes in cell and tissue shape. A notable feature of this signaling pathway is its intricate organization in both space and time. At the cellular level, signaling components and the cytoskeleton exhibit striking polarity, not only along the apical-basal cell axis, but also within the apical domain. Furthermore, gene expression controls a highly choreographed chain of events, the dynamics of which are critical for primordium invagination; it does not simply throw the cytoskeletal "on" switch." fly
- Sticking, steering, squeezing and shearing: cell movements driven by heterotypic mechanical forces[3] "During development, the immune response and cancer, cells of different types interact mechanically. Here we review how such heterotypic mechanical interactions enable cell movements. We begin by analyzing the heterotypic forces that single cells use to adhere and squeeze through tight barriers, as in the case of leucocyte extravasation and cancer metastasis. We next focus on the different mechanisms by which adjacent tissues influence each other's movements, with particular emphasis on dragging forces during dorsal closure in Drosophila and shearing forces during gastrulation in zebrafish. Finally, we discuss the mechanotransduction feedback loops that enable different cell types to steer each other's migration during development and cancer. We illustrate these migration modes focusing on the combination of attractive and repulsive cues during co-migration of neural crest cells and placodes in Xenopus, and of fibroblasts and cancer cells during invasion. Throughout the review, we discuss the nature of the heterotypic contact, which may involve both homophilic and heterophilic interactions between adhesion receptors."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Morphodynamics
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Friction forces position the neural anlage[4] During embryonic development, "mechanical forces are essential for cellular rearrangements driving tissue morphogenesis. Here, we show that in the early zebrafish embryo, friction forces are generated at the interface between anterior axial mesoderm (prechordal plate, ppl) progenitors migrating towards the animal pole and neurectoderm progenitors moving in the opposite direction towards the vegetal pole of the embryo. These friction forces lead to global rearrangement of cells within the neurectoderm and determine the position of the neural anlage. Using a combination of experiments and simulations, we show that this process depends on hydrodynamic coupling between neurectoderm and ppl as a result of E-cadherin-mediated adhesion between those tissues. Our data thus establish the emergence of friction forces at the interface between moving tissues as a critical force-generating process shaping the embryo."
- Review - Tissue morphodynamics shaping the early mouse embryo[5] "Generation of the elongated vertebrate body plan from the initially radially symmetrical embryo requires comprehensive changes to tissue form. These shape changes are generated by specific underlying cell behaviors, coordinated in time and space. Major principles and also specifics are emerging, from studies in many model systems, of the cell and physical biology of how region-specific cell behaviors produce regional tissue morphogenesis, and how these, in turn, are integrated at the level of the embryo. New technical approaches have made it possible more recently, to examine the morphogenesis of the mouse embryo in depth, and to elucidate the underlying cellular mechanisms. This review focuses on recent advances in understanding the cellular basis for the early fundamental events that establish the basic form of the embryo."
- Mechanically patterning the embryonic airway epithelium[6] "Collections of cells must be patterned spatially during embryonic development to generate the intricate architectures of mature tissues. In several cases, including the formation of the branched airways of the lung, reciprocal signaling between an epithelium and its surrounding mesenchyme helps generate these spatial patterns. Several molecular signals are thought to interact via reaction-diffusion kinetics to create distinct biochemical patterns, which act as molecular precursors to actual, physical patterns of biological structure and function. Here, however, we show that purely physical mechanisms can drive spatial patterning within embryonic epithelia. Specifically, we find that a growth-induced physical instability defines the relative locations of branches within the developing murine airway epithelium in the absence of mesenchyme. The dominant wavelength of this instability determines the branching pattern and is controlled by epithelial growth rates. These data suggest that physical mechanisms can create the biological patterns that underlie tissue morphogenesis in the embryo."
- Mechanical control of notochord morphogenesis by extra-embryonic tissues in mouse embryos[7] "Here, we show that in mouse embryos, the expansion of the amniotic cavity (AC), which is formed between embryonic and extraembryonic tissues, provides the mechanical forces required for a type of morphogenetic movement of the notochord known as convergent extension (CE) in which the cells converge to the midline and the tissue elongates along the antero-posterior (AP) axis. The notochord is stretched along the AP axis, and the expansion of the AC is required for CE. Both mathematical modeling and physical simulation showed that a rectangular morphology of the early notochord caused the application of anisotropic force along the AP axis to the notochord through the isotropic expansion of the AC. AC expansion acts upstream of planar cell polarity (PCP) signaling, which regulates CE movement. Our results highlight the importance of extraembryonic tissues as a source of the forces that control the morphogenesis of embryos." notochord
- Apical constriction initiates new bud formation during monopodial branching of the embryonic chicken lung[8] "Branching morphogenesis sculpts the airway epithelium of the lung into a tree-like structure to conduct air and promote gas exchange after birth. In the avian lung, a series of buds emerges from the dorsal surface of the primary bronchus via monopodial branching to form the conducting airways; anatomically, these buds are similar to those formed by domain branching in the mammalian lung. Here, we show that monopodial branching is initiated by apical constriction of the airway epithelium, and not by differential cell proliferation, using computational modeling and quantitative imaging of embryonic chicken lung explants."
|
References
- ↑ 1.0 1.1 Martin AC. (2020). The Physical Mechanisms of Drosophila Gastrulation: Mesoderm and Endoderm Invagination. Genetics , 214, 543-560. PMID: 32132154 DOI.
- ↑ Bogolyubova I & Bogolyubov D. (2020). Heterochromatin Morphodynamics in Late Oogenesis and Early Embryogenesis of Mammals. Cells , 9, . PMID: 32575486 DOI.
- ↑ Labernadie A & Trepat X. (2018). Sticking, steering, squeezing and shearing: cell movements driven by heterotypic mechanical forces. Curr. Opin. Cell Biol. , 54, 57-65. PMID: 29719271 DOI.
- ↑ Smutny M, Ákos Z, Grigolon S, Shamipour S, Ruprecht V, Čapek D, Behrndt M, Papusheva E, Tada M, Hof B, Vicsek T, Salbreux G & Heisenberg CP. (2017). Friction forces position the neural anlage. Nat. Cell Biol. , 19, 306-317. PMID: 28346437 DOI.
- ↑ Sutherland AE. (2016). Tissue morphodynamics shaping the early mouse embryo. Semin. Cell Dev. Biol. , 55, 89-98. PMID: 26820524 DOI.
- ↑ Varner VD, Gleghorn JP, Miller E, Radisky DC & Nelson CM. (2015). Mechanically patterning the embryonic airway epithelium. Proc. Natl. Acad. Sci. U.S.A. , 112, 9230-5. PMID: 26170292 DOI.
- ↑ Imuta Y, Koyama H, Shi D, Eiraku M, Fujimori T & Sasaki H. (2014). Mechanical control of notochord morphogenesis by extra-embryonic tissues in mouse embryos. Mech. Dev. , 132, 44-58. PMID: 24509350 DOI.
- ↑ Kim HY, Varner VD & Nelson CM. (2013). Apical constriction initiates new bud formation during monopodial branching of the embryonic chicken lung. Development , 140, 3146-55. PMID: 23824575 DOI.
Textbooks
Reviews
Goodwin K & Nelson CM. (2020). Branching morphogenesis. Development , 147, . PMID: 32444428 DOI.
Heyn R, Makabe S & Motta PM. (2001). Ultrastructural morphodynamics of human Sertoli cells during testicular differentiation. Ital J Anat Embryol , 106, 163-71. PMID: 11732573
Articles
Dimchev G, Amiri B, Humphries AC, Schaks M, Dimchev V, Stradal TEB, Faix J, Krause M, Way M, Falcke M & Rottner K. (2020). Lamellipodin tunes cell migration by stabilizing protrusions and promoting adhesion formation. J. Cell. Sci. , 133, . PMID: 32094266 DOI.
Rienzi L, Cimadomo D, Delgado A, Minasi MG, Fabozzi G, Gallego RD, Stoppa M, Bellver J, Giancani A, Esbert M, Capalbo A, Remohì J, Greco E, Ubaldi FM & Meseguer M. (2019). Time of morulation and trophectoderm quality are predictors of a live birth after euploid blastocyst transfer: a multicenter study. Fertil. Steril. , 112, 1080-1093.e1. PMID: 31843084 DOI.
Search PubMed
Search Pubmed: Embryo Morphodynamics | Morphodynamics
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 25) Embryology Developmental Mechanism - Morphodynamics. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Mechanism_-_Morphodynamics
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G

