Adipose Tissue Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 7: | Line 7: | ||
Connective tissues in the body have a mesoderm origin, while in the head neural crest also contributes to these tissues. | Connective tissues in the body have a mesoderm origin, while in the head neural crest also contributes to these tissues. | ||
This topic is also covered in musculoskeletal ( | This topic is also covered in musculoskeletal ({{tendon}}), integumentary ([[Integumentary Development]]) and endocrine development (Adipose Tissue). | ||
Blood is a liquid connective tissue (More? | Blood is a liquid connective tissue (More? {{blood}}). | ||
| Line 24: | Line 24: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''A mesodermal fate map for adipose tissue'''{{#pmid:30045918|PMID30045918}} "The embryonic origin of distinct fat depots and the role for ontogeny in specifying the functional differences among adipocyte lineages between and within depots is unclear. Using a Cre/Lox-based strategy to track the fate of major mesodermal subcompartments in mice we present evidence that fewer than 50% of interscapular brown adipocytes are derived from progenitors of the central dermomyotome. Furthermore, we demonstrate that depot-specific adipocyte lineages spatially diverge as early as gastrulation and that perigonadal adipocytes arise from separate mesodermal subcompartments in males and females. Last, we show adipocyte precursors (APs) of distinct lineages within the same depot exhibit indistinguishable responses to a high fat diet, indicating ontogenetic differences between APs do not necessarily correspond to functional differences in this context. Altogether, these findings shed light on adipose tissue patterning and suggest the behavior of adipocyte lineage cells is not strictly determined by developmental history." | ||
* '''Brown adipose tissue: function and physiological significance''' | |||
* '''The fat controller: adipocyte development'''{{#pmid:23209380|PMID23209380}} "Obesity is a condition characterized by excess adipose tissue that results from positive energy balance and is the most common metabolic disorder in the industrialized world. ... Adipocytes are not created from other adipocytes, but they arise from precursor cells. In the last two decades, scientists have discovered the function of many proteins that influence the ability of precursor cells to become adipocytes. If the expansion of the adipose tissue is the problem, it seems logical that adipocyte development inhibitors could be a viable anti-obesity therapeutic. However, factors that block adipocyte development and limit adipocyte expansion also impair metabolic health. This notion may be counterintuitive, but several lines of evidence support the idea that blocking adipocyte development is unhealthy. For this reason it is clear that we need a better understanding of adipocyte development." | |||
* '''Brown adipose tissue: function and physiological significance'''{{#pmid:14715917|PMID14715917}} "The function of brown adipose tissue is to transfer energy from food into heat; physiologically, both the heat produced and the resulting decrease in metabolic efficiency can be of significance. ... The development of brown adipose tissue with its characteristic protein, uncoupling protein-1 (UCP1), was probably determinative for the evolutionary success of mammals, as its thermogenesis enhances neonatal survival and allows for active life even in cold surroundings." | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 46: | Line 49: | ||
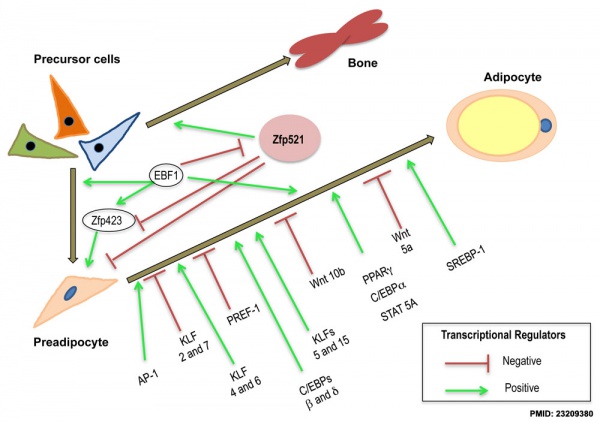
[[File:Adipocyte differentiation regulation 01.jpg|600px]] | [[File:Adipocyte differentiation regulation 01.jpg|600px]] | ||
Adipocyte differentiation regulation. | Adipocyte differentiation regulation.{{#pmid:23209380|PMID23209380}} | ||
[[Developmental Signals - Notch|Notch1]] signaling in adipocyte progenitor cells regulates the adipogenesis process including proliferation and differentiation of the adipocyte progenitor cells. | [[Developmental Signals - Notch|Notch1]] signaling in adipocyte progenitor cells regulates the adipogenesis process including proliferation and differentiation of the adipocyte progenitor cells.{{#pmid:27869309|PMID27869309}} | ||
| Line 107: | Line 110: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:21372557}} | |||
{{#pmid:19896888}} | |||
{{#pmid:19188249}} | |||
{{#pmid:18793119}} | |||
{{#pmid:14715917}} | |||
===Articles=== | ===Articles=== | ||
{{#pmid:20678241}} | |||
{{#pmid:17507398}} | |||
===Search PubMed=== | ===Search PubMed=== | ||
| Line 135: | Line 141: | ||
{{Footer}} | {{Footer}} | ||
[[Category:Adipose Tissue]] | [[Category:Adipose Tissue]][[Category:Mesoderm]] | ||
Revision as of 15:57, 30 July 2018
| Embryology - 10 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Draft Page- notice removed when completed.
Connective tissues in the body have a mesoderm origin, while in the head neural crest also contributes to these tissues.
This topic is also covered in musculoskeletal (tendon), integumentary (Integumentary Development) and endocrine development (Adipose Tissue).
Blood is a liquid connective tissue (More? blood).
- Loose and dense connective tissue
- Reticular connective tissue
- Adipose Tissue
- Mesenchymal connective tissue
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Adipose Embryology <pubmed limit=5>Adipose Embryology</pubmed>
<pubmed limit=5>Brown Embryology</pubmed> |
Molecular Development
Adipocyte differentiation regulation.[2]
Notch1 signaling in adipocyte progenitor cells regulates the adipogenesis process including proliferation and differentiation of the adipocyte progenitor cells.[4]
- Links: NOTCH
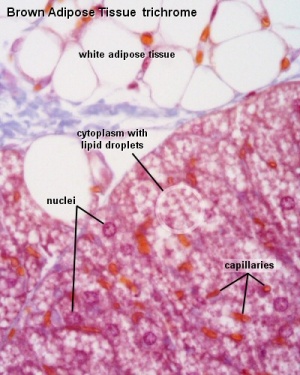
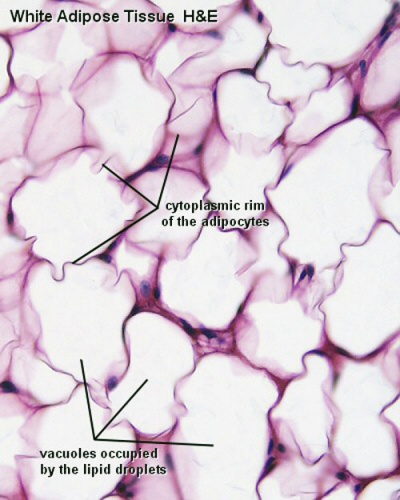
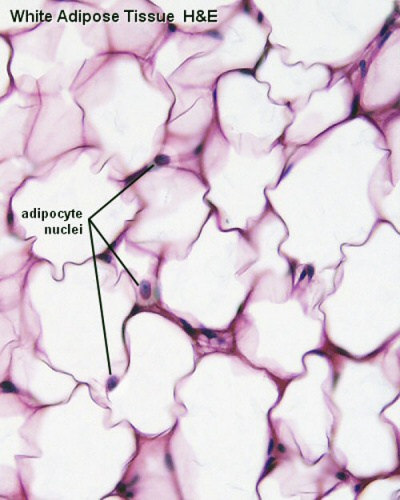
White Adipose
Brown Adipose
- Brown Adipose Tissue (BAT) arises from progenitor cells that also give rise to skeletal muscle,
- Brown adipocytes have numerous small lipid droplets rather than a single large one as in white adipocytes
- Elevated numbers of mitochondria
- mitochondrial expression of the nuclear gene UCP1, the uncoupler of oxidative phosphorylation responsible for non-shivering thermogenesis.
Development Overview
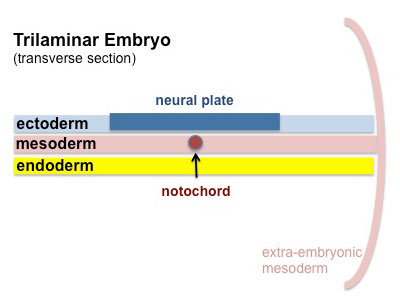
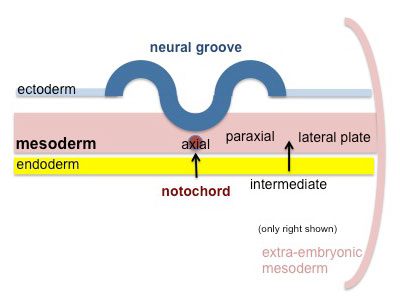
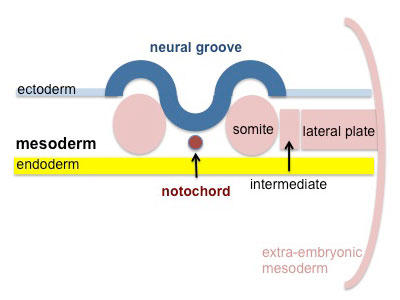
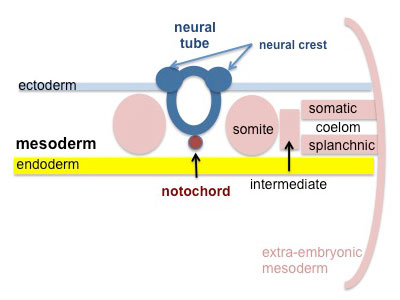
Mesoderm Development
Somite - Dermatome
The dermis and hypodermis layers of the skin.
Somatic Mesoderm
The body wall connective tissue.
Splanchnic Mesoderm
The lamina propria and submucosa layers of the gastrointestinal tract wall.
References
- ↑ Sebo ZL, Jeffery E, Holtrup B & Rodeheffer MS. (2018). A mesodermal fate map for adipose tissue. Development , , . PMID: 30045918 DOI.
- ↑ 2.0 2.1 Stephens JM. (2012). The fat controller: adipocyte development. PLoS Biol. , 10, e1001436. PMID: 23209380 DOI.
- ↑ Cannon B & Nedergaard J. (2004). Brown adipose tissue: function and physiological significance. Physiol. Rev. , 84, 277-359. PMID: 14715917 DOI.
- ↑ Shan T, Liu J, Wu W, Xu Z & Wang Y. (2017). Roles of Notch Signaling in Adipocyte Progenitor Cells and Mature Adipocytes. J. Cell. Physiol. , 232, 1258-1261. PMID: 27869309 DOI.
Reviews
Tews D & Wabitsch M. (2011). Renaissance of brown adipose tissue. Horm Res Paediatr , 75, 231-9. PMID: 21372557 DOI.
Schulz TJ & Tseng YH. (2009). Emerging role of bone morphogenetic proteins in adipogenesis and energy metabolism. Cytokine Growth Factor Rev. , 20, 523-31. PMID: 19896888 DOI.
Forhead AJ & Fowden AL. (2009). The hungry fetus? Role of leptin as a nutritional signal before birth. J. Physiol. (Lond.) , 587, 1145-52. PMID: 19188249 DOI.
Billon N, Monteiro MC & Dani C. (2008). Developmental origin of adipocytes: new insights into a pending question. Biol. Cell , 100, 563-75. PMID: 18793119 DOI.
Cannon B & Nedergaard J. (2004). Brown adipose tissue: function and physiological significance. Physiol. Rev. , 84, 277-359. PMID: 14715917 DOI.
Articles
Billon N, Kolde R, Reimand J, Monteiro MC, Kull M, Peterson H, Tretyakov K, Adler P, Wdziekonski B, Vilo J & Dani C. (2010). Comprehensive transcriptome analysis of mouse embryonic stem cell adipogenesis unravels new processes of adipocyte development. Genome Biol. , 11, R80. PMID: 20678241 DOI.
Billon N, Iannarelli P, Monteiro MC, Glavieux-Pardanaud C, Richardson WD, Kessaris N, Dani C & Dupin E. (2007). The generation of adipocytes by the neural crest. Development , 134, 2283-92. PMID: 17507398 DOI.
Search PubMed
Search Pubmed: adipose Development
Additional Images
Terms
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 10) Embryology Adipose Tissue Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Adipose_Tissue_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G