Adipose Tissue Development
| Embryology - 6 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Adipose tissue is a form of loose connective tissue composed of adipocytes, fibroblasts, vascular endothelial cells, and some immune cells. Connective tissues in the body have a mesoderm origin, while in the head neural crest also contributes to these tissues.
There are two main forms of adipose tissue, white adipose tissue (WAT) and brown adipose tissue (BAT). White adipose tissue, is the main depot for lipid storage, while brown adipose tissue is involved in thermogenesis. We now know that adipose tissue also has important endocrine hormones secreting leptin, adiponectin, and resistin. (More? Endocrine Adipose Tissue | Endocrine other tissues)
Postnatally, a third form of "beige adipose tissue" has been identified in the mouse model.[1] This form of adipose can be induced in subcutaneous white adipose tissue as a response to cold and other thermogenic activators.[2]
In the adult tissues, White Adipose Tissue is found extensively within the hypodermis layer of the skin (integumentary). There are also sex differences in WAT distribution, related to the differences in the endocrine environment in males and females circulating androgens and estrogens.[3] (See reviews[4][5] and genital)
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Adipose Embryology | Adipose Development | Brown Adipose | White Adipose | Beige Adipose |
Development Overview
| Anatomical Region | Specific Location | Start (weeks) |
CRL (mm) | Complete (weeks) |
CRL (mm) |
|---|---|---|---|---|---|
| Head | Buccal pad | 14 | 100 | 17 | 153 |
| Cheek | 14.5 | 103 | 17 | 150 | |
| Chin | 14.5 | 103 | 17 | 150 | |
| Ocular pad | 15 | 113 | 19.5 | 170 | |
| Neck | Neck | 15 | 113 | 19.5 | 170 |
| Thorax | Anterior wall | 16 | 135 | 19.5 | 170 |
| Posterior wall | 15 | 113 | 20.5 | 190 | |
| Mammary | 14.5 | 106 | 17.5 | 156 | |
| Abdomen | Abdominal wall | 14.5 | 106 | 20.5 | 190 |
| Perirenal | 15 | 113 | 20.5 | 190 | |
| Upper limb | Shoulder | 15 | 113 | 23.5 | 216 |
| Forearm | 16 | 131 | 20.5 | 190 | |
| Arm | 16 | 131 | 20.5 | 190 | |
| Hand | 16 | 131 | l9.5 | 172 | |
| Lower limb | Gluteal | 16 | 131 | 20.5 | 190 |
| Thigh | 16.5 | 141 | 22.5 | 212 | |
| Leg | 16 | 131 | 22.5 | 212 | |
| Foot | 16 | 131 | 19.5 | 170 | |
| Table Notes - weeks are fertilization age (FA), not GA, both male and female data are combined. Table Data source[9] Links: adipose | Second Trimester | |||||
- hand - begins in the subcutis of the palm and then progresses proximally to the wrist and distally into the fingers.
- week 23 - thickened layer of subcutaneous fat covers the extremities of the limbs, as for newborn.
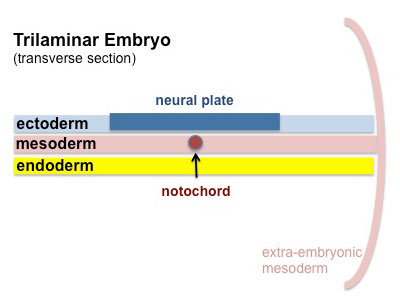
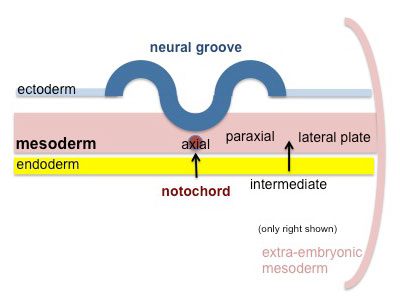
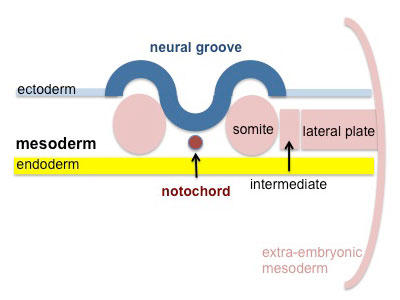
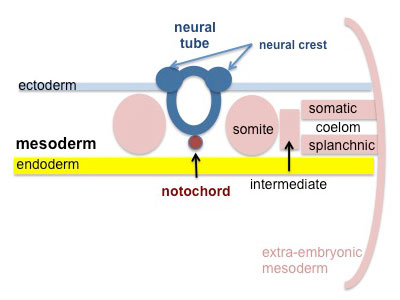
Mesoderm Development
Molecular Development
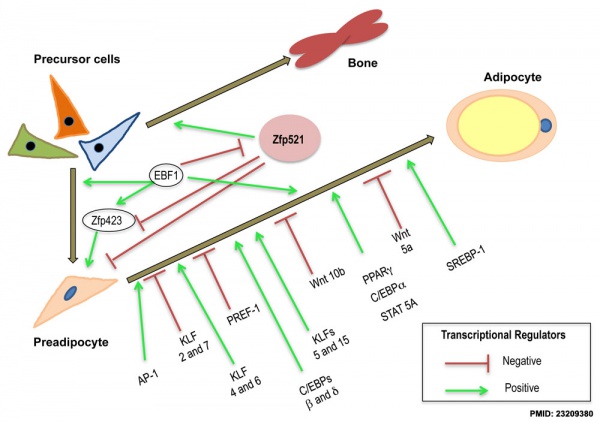
Adipocyte differentiation regulation.[7]
Notch1 signaling in adipocyte progenitor cells regulates the adipogenesis process including proliferation and differentiation of the adipocyte progenitor cells.[10]
- Links: NOTCH
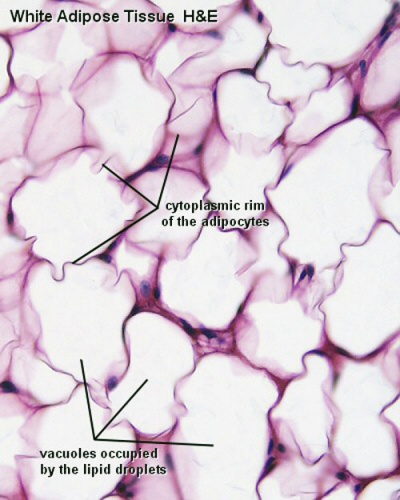
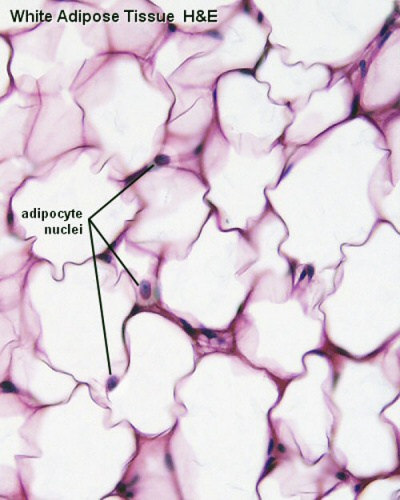
White Adipose Tissue
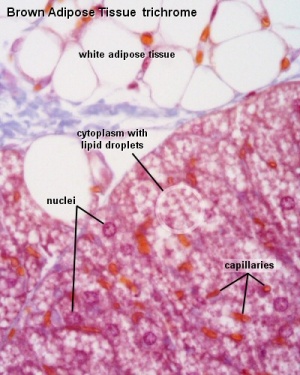
Brown Adipose Tissue
- Brown Adipose Tissue (BAT) arises from progenitor cells that also give rise to skeletal muscle,
- Brown adipocytes have numerous small lipid droplets rather than a single large one as in white adipocytes
- Elevated numbers of mitochondria
- mitochondrial expression of the nuclear gene UCP1, the uncoupler of oxidative phosphorylation responsible for non-shivering thermogenesis.
BAT distribution in the newborn infant:[11]
- Interscapular - mass lies in a thin diamond-shaped sheet between the shoulder blades, separated from the subcutaneous WAT by a discontinuous fibrous layer. When replete with fat it has a yellowish-brown colour; depleted it is much darker. It has a fine lobular structure.
- Neck muscles and blood vessels - many smaller masses with the main mass following the course of the internal jugular vein and common carotid artery.
- Axilla - large deposits as extensions from the neck tissue that pass under the clavicles.
- Great vessels - entering the thoracic inlet extending as fine fingers that spread out from the midline with each intercostal artery. Similar deposits lie among the internal mammary vessels. Many discrete, moderately large masses lie in the mediastinum between the oesophagus and the trachea.
- Abdomen - discrete masses accompany the aorta and lie in relation to many structures on the posterior abdominal wall such as the pancreas, autonomic ganglia and chromaffin tissue. The largest abdominal mass envelops the [[renal}} and adrenals.
Beige Adipose Tissue
A third form of "beige adipose tissue" has been identified in the mouse model.[1] This form of adipose can be induced in subcutaneous white adipose tissue as a response to cold and other thermogenic activators.[2] This conversion process has also been described as "browning".
Periaortic Arch Adipose Tissue
Periaortic arch adipose tissue (PAAT) surrounds the aortic arch, its major branches, and surrounds the pulmonary artery, the ascending aorta, and the beginning of descending aorta. PAAT originate from different lineages including neural crest[12] and functionally have roles in regulating vascular homeostasis.
In the mouse, thoracic periaortic adipose tissue is mainly BAT, while abdominal periaortic adipose tissue is both BAT and WAT.
Somite - Dermatome
The dermis and hypodermis layers of the skin.
Integumentary Hypodermis
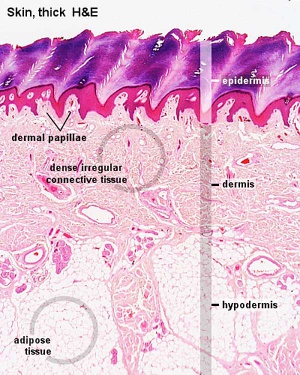
|
Histological section of skin showing the 3 main layers: epidermis, dermis and hypodermis layers. ((Stain - Haematoxylin Eosin))
Highlighted ringed regions show:
Note the junctional region between dermis and hypodermis contains macroscopically visible glands and blood vessels. |
Somatic Mesoderm
The body wall connective tissue.
Splanchnic Mesoderm
The lamina propria and submucosa layers of the gastrointestinal tract wall.
Endocrine Adipose
- Leptin - polypeptide hormone produced in adipose and many other tissues with also many different roles
- Adiponectin - regulation of energy homeostasis and glucose and lipid metabolism, as well as acting as an anti-inflammatory on the cellular vascular wall
- Resistin - (for resistance to insulin, RETN) a 108 amino acid polypeptide and the related resistin-like protein-beta (Resistin-like molecule-beta, RELMbeta) stimulate endogenous glucose production
- Links: Endocrine Adipose Tissue | Endocrine other tissues
References
- ↑ 1.0 1.1 Chan M, Lim YC, Yang J, Namwanje M, Liu L & Qiang L. (2019). Identification of a natural beige adipose depot in mice. J. Biol. Chem. , , . PMID: 30824545 DOI.
- ↑ 2.0 2.1 Tanimura K, Suzuki T, Vargas D, Shibata H & Inagaki T. (2019). Epigenetic regulation of beige adipocyte fate by histone methylation. Endocr. J. , 66, 115-125. PMID: 30606913 DOI.
- ↑ Lee MJ & Fried SK. (2017). Sex-dependent Depot Differences in Adipose Tissue Development and Function; Role of Sex Steroids. J Obes Metab Syndr , 26, 172-180. PMID: 31089514 DOI.
- ↑ Fitzgerald SJ, Janorkar AV, Barnes A & Maranon RO. (2018). A new approach to study the sex differences in adipose tissue. J. Biomed. Sci. , 25, 89. PMID: 30509250 DOI.
- ↑ Newell-Fugate AE. (2017). The role of sex steroids in white adipose tissue adipocyte function. Reproduction , 153, R133-R149. PMID: 28115579 DOI.
- ↑ Sebo ZL, Jeffery E, Holtrup B & Rodeheffer MS. (2018). A mesodermal fate map for adipose tissue. Development , , . PMID: 30045918 DOI.
- ↑ 7.0 7.1 Stephens JM. (2012). The fat controller: adipocyte development. PLoS Biol. , 10, e1001436. PMID: 23209380 DOI.
- ↑ Cannon B & Nedergaard J. (2004). Brown adipose tissue: function and physiological significance. Physiol. Rev. , 84, 277-359. PMID: 14715917 DOI.
- ↑ Poissonnet CM, LaVelle M & Burdi AR. (1988). Growth and development of adipose tissue. J. Pediatr. , 113, 1-9. PMID: 3290412
- ↑ Shan T, Liu J, Wu W, Xu Z & Wang Y. (2017). Roles of Notch Signaling in Adipocyte Progenitor Cells and Mature Adipocytes. J. Cell. Physiol. , 232, 1258-1261. PMID: 27869309 DOI.
- ↑ Aherne W & Hull D. (1966). Brown adipose tissue and heat production in the newborn infant. J Pathol Bacteriol , 91, 223-34. PMID: 5941392 DOI.
- ↑ Fu M, Xu L, Chen X, Han W, Ruan C, Li J, Cai C, Ye M & Gao P. (2019). Neural Crest Cells Differentiate Into Brown Adipocytes and Contribute to Periaortic Arch Adipose Tissue Formation. Arterioscler. Thromb. Vasc. Biol. , 39, 1629-1644. PMID: 31189430 DOI.
Reviews
Montanari T, Pošćić N & Colitti M. (2017). Factors involved in white-to-brown adipose tissue conversion and in thermogenesis: a review. Obes Rev , 18, 495-513. PMID: 28187240 DOI.
Newell-Fugate AE. (2017). The role of sex steroids in white adipose tissue adipocyte function. Reproduction , 153, R133-R149. PMID: 28115579 DOI.
Tews D & Wabitsch M. (2011). Renaissance of brown adipose tissue. Horm Res Paediatr , 75, 231-9. PMID: 21372557 DOI.
Schulz TJ & Tseng YH. (2009). Emerging role of bone morphogenetic proteins in adipogenesis and energy metabolism. Cytokine Growth Factor Rev. , 20, 523-31. PMID: 19896888 DOI.
Forhead AJ & Fowden AL. (2009). The hungry fetus? Role of leptin as a nutritional signal before birth. J. Physiol. (Lond.) , 587, 1145-52. PMID: 19188249 DOI.
Billon N, Monteiro MC & Dani C. (2008). Developmental origin of adipocytes: new insights into a pending question. Biol. Cell , 100, 563-75. PMID: 18793119 DOI.
Cannon B & Nedergaard J. (2004). Brown adipose tissue: function and physiological significance. Physiol. Rev. , 84, 277-359. PMID: 14715917 DOI.
Articles
Billon N, Kolde R, Reimand J, Monteiro MC, Kull M, Peterson H, Tretyakov K, Adler P, Wdziekonski B, Vilo J & Dani C. (2010). Comprehensive transcriptome analysis of mouse embryonic stem cell adipogenesis unravels new processes of adipocyte development. Genome Biol. , 11, R80. PMID: 20678241 DOI.
Billon N, Iannarelli P, Monteiro MC, Glavieux-Pardanaud C, Richardson WD, Kessaris N, Dani C & Dupin E. (2007). The generation of adipocytes by the neural crest. Development , 134, 2283-92. PMID: 17507398 DOI.
Search PubMed
Search Pubmed: adipose Development
Additional Images
Terms
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 6) Embryology Adipose Tissue Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Adipose_Tissue_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G