Chorionic villus sampling: Difference between revisions
No edit summary |
|||
| Line 38: | Line 38: | ||
{{Template:Prenatal Diagnosis terms}} | {{Template:Prenatal Diagnosis terms}} | ||
==External Links== | |||
{{External Links}} | |||
* '''WHO''' - Oladapo OT. Amniocentesis and chorionic villus sampling for prenatal diagnosis: RHL commentary (last revised: 1 April 2009). [http://apps.who.int/rhl/pregnancy_childbirth/fetal_disorders/prenatal_diagnosis/CD003252_Oladapot_com/en/index.html The WHO Reproductive Health Library]; Geneva: World Health Organization. | |||
{{Template:Glossary}} | {{Template:Glossary}} | ||
{{Template:Footer}} | {{Template:Footer}} | ||
[[Category:Abnormal Development]] [[Category:Genetic Abnormalities]] [[Category:Human Embryo]] [[Category:Human Fetus]] | [[Category:Abnormal Development]] [[Category:Genetic Abnormalities]] [[Category:Human Embryo]] [[Category:Human Fetus]] | ||
Revision as of 15:39, 18 August 2011
Introduction
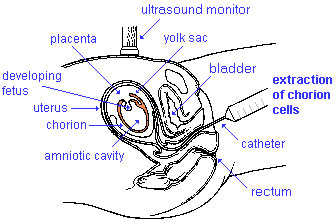
Chorionic Villus Sampling test is done in the 10th to 12th week after the first day of the mother's last menstrual period. The test is done by looking at cells taken from the chorionic membrane or placenta. No anaesthetic is required, and a test result is usually available in two to three weeks.
When the test is carried out by an obstetrician experienced in the technique, the risk of miscarriage related to the test is about 2 %. (Modified from: Checking your baby's health before birth. State Health Publication Number (PA) 94-090)
Potential disadvantages include maternal cell contamination, placental mosaicism and failure to obtain an adequate specimen. This may result in the need for a repeat procedure or amniocentesis.
Some Recent Findings
|
References
Reviews
<pubmed>21504867</pubmed> <pubmed>21413037</pubmed> <pubmed>21297473</pubmed>
Articles
Search PubMed
Search PubMed: Chorionic villus sampling
Search Entrez: Search All Databases - CVS
- ART - Assisted Reproductive Technology a general term to describe all the clinical techniques used to aid fertility.
- blastomere biopsy - An ART preimplantation genetic diagnosis technique carried out at cleavage stage (day 3), excluding poor quality embryos, detects chromosomal abnormalities of both maternal and paternal origin. May not detect cellular mosaicism in the embryo.
- blastocyst biopsy - An ART preimplantation genetic diagnosis technique carried out at blastocyst stage (day 4-5), removes several trophoblast (trophoderm) cells, detects chromosomal abnormalities of both maternal and paternal origin and may detect cellular mosaicism.
- cell-free fetal deoxyribonucleic acid - (cfDNA) refers to fetal DNA circulating and isolated from the plasma portion of maternal blood. Can be performed from GA 10 weeks as a first-tier test or as a second-tier test, with women with increased probability on combined first trimester screening offered cfDNA or diagnostic testing.
- false negative rate - The proportion of pregnancies that will test negative given that the congenital anomaly is present.
- false positive rate - The proportion of pregnancies that will test positive given that the congenital anomaly is absent.
- free β human chorionic gonadotrophin - beta-hCG subunit of hCG used as a diagnostic marker for: early detection of pregnancy, Trisomy 21, spontaneous abortion, ectopic pregnancy, hydatidiform mole or choriocarcinoma.
- multiples of the median - (MoM) A multiple of the median is a measure of how far an individual test result deviates from the median and is used to report the results of medical screening tests, particularly where the results of the individual tests are highly variable.
- negative predictive value - The probability that a congenital anomaly is absent given that the prenatal screening test is negative.
- Non-Invasive Prenatal Testing - (NIPT) could refer to ultrasound or other imaging techniques, but more frequently used to describe analysis of cell-free fetal DNA circulating in maternal blood.
- polar body biopsy - (PB biopsy) An ART preimplantation genetic diagnosis technique that removes either the first or second polar body from the zygote. As these are generated by oocyte meiosis they detects chromosomal abnormalities only on the female genetics.
- positive predictive value - The probability that a congenital anomaly is present given that the prenatal screening test is positive.
- pre-implantation genetic diagnosis - (PGD, pre-implantation genetic screening) a diagnostic procedure for embryos produced through Assisted Reproductive Technology (ART, in vitro fertilisation, IVF) for genetic diseases that would generate developmental abnormalities or serious postnatal diseases.
- prenatal screening sensitivity - (detection rate) The probability of testing positive on a prenatal screening test if the congenital anomaly is present.
- prenatal screening specificity - The probability of testing negative on a prenatal screening test if the congenital anomaly is absent.
- quadruple test (maternal serum testing of a-fetoprotein Template:AFP, free B-hCG or total hCG, unconjugated estriol, and inhibin A) is a fetal chromosomal anomaly test usually carried out later in pregnancy (GA 14 to 20 weeks).
- single nucleotide polymorphisms - (SNPs) the variation in a single DNA nucleotide that occurs at a specific position in the genome.
- triple test - (maternal serum testing of a-fetoprotein Template:AFP, free B-hCG or total hCG, and unconjugated estriol) is a fetal chromosomal anomaly test usually carried out later in pregnancy (GA 14 to 20 weeks).
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- WHO - Oladapo OT. Amniocentesis and chorionic villus sampling for prenatal diagnosis: RHL commentary (last revised: 1 April 2009). The WHO Reproductive Health Library; Geneva: World Health Organization.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 15) Embryology Chorionic villus sampling. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Chorionic_villus_sampling
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G