Uterine Gland
| Embryology - 5 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
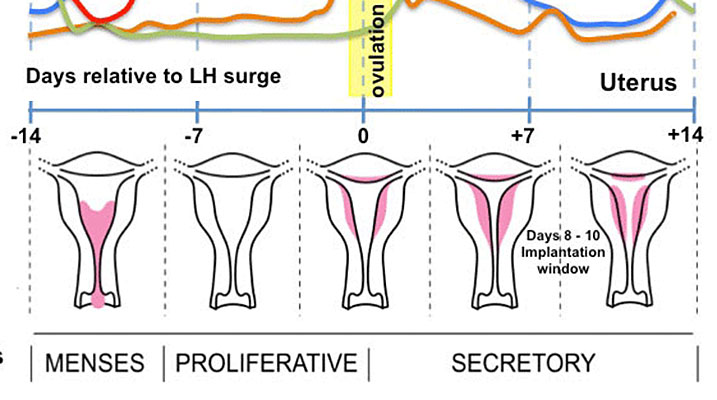
The uterine gland or endometrial gland is a simple tubular gland formed by invagination of the uterine endometrium. The uterine epithelium is described as a columnar epithelium of ciliated cells and secretory cells.
The glands extend into the underlying thick vascular stromal layer. The glands line the uterus body and change in appearance and secretion during the menstrual cycle. The glands secretions function to provide the initial nutritional support of the conceptus and may have a role in maintaining adhesion.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Uterine Gland Development | Uterine Gland |
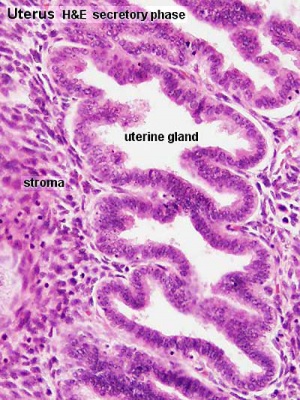
Uterine Gland Histology During the Menstrual Cycle

|
| |
| Uterine gland proliferative phase | Uterine gland secretory phase |
Menstrual Cycle Histology
The different stages of the menstrual cycle can be monitored by the cellular appearance of vaginal smears Menstrual Cycle - Histology.
A more invasive technique is dilate and curettage (DnC), which allows sampling of the functional layer of the uterine endometrium Menstrual Cycle - Histology.
Decidualization
Decidualization is the process of converting endometrial stromal cells into decimal cells and requires at least 8–10 days of hormone stimulation.
- initiated during the mid-secretory phase of the menstrual cycle
- in response to elevated progesterone levels
- acts mainly through progesterone receptor (PR) PR-A (other isoform is PR-B)
Molecular
PMID: 21546446 Prokineticin 1 (PROK1) signalling via prokineticin receptor 1 (PROKR1) regulates Dickkopf 1 (DKK1) expression, a negative regulator of canonical Wnt signaling.
- Links: Placenta - Maternal Decidua
Histiotrophic Nutrition
Term used to describe in early placenta development the intital transfer of nutrition from maternal to embryo (histiotrophic nutrition) compared to later blood-borne nutrition (hemotrophic nutrition). Histotroph is the nutritional material accumulated in spaces between the maternal and fetal tissues, derived from the maternal endometrium and the uterine glands. This nutritional material is absorbed by phagocytosis initially by blastocyst trophectoderm and then by trophoblast of the placenta.[4] in later placental development nutrition is by the exchange of blood-borne materials between the maternal and fetal circulations, hemotrophic nutrition.
References
- ↑ Kelleher AM, DeMayo FJ & Spencer TE. (2019). Uterine Glands: Developmental Biology and Functional Roles in Pregnancy. Endocr. Rev. , 40, 1424-1445. PMID: 31074826 DOI.
- ↑ Kelleher AM, Milano-Foster J, Behura SK & Spencer TE. (2018). Uterine glands coordinate on-time embryo implantation and impact endometrial decidualization for pregnancy success. Nat Commun , 9, 2435. PMID: 29934619 DOI.
- ↑ Hayashi K, Yoshioka S, Reardon SN, Rucker EB, Spencer TE, DeMayo FJ, Lydon JP & MacLean JA. (2011). WNTs in the neonatal mouse uterus: potential regulation of endometrial gland development. Biol. Reprod. , 84, 308-19. PMID: 20962251 DOI.
- ↑ Burton GJ, Watson AL, Hempstock J, Skepper JN & Jauniaux E. (2002). Uterine glands provide histiotrophic nutrition for the human fetus during the first trimester of pregnancy. J. Clin. Endocrinol. Metab. , 87, 2954-9. PMID: 12050279 DOI.
Books
- Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Walker HK, Hall WD, Hurst JW, editors. Boston: Butterworths; 1990. Birth Control | Figure 174.1 A 28-day menstrual cycle. Not all cycles are 28 days long. It is the phase before ovulation that varies in length.
- Endocrinology: An Integrated Approach. Nussey S, Whitehead S. Oxford: BIOS Scientific Publishers; 2001. Box 6.42 The human menstrual cycle | Box 6.43 Feedback control of the hypothalamo-pituitary-ovarian axis
Reviews
Gray CA, Bartol FF, Tarleton BJ, Wiley AA, Johnson GA, Bazer FW & Spencer TE. (2001). Developmental biology of uterine glands. Biol. Reprod. , 65, 1311-23. PMID: 11673245
Articles
Search Pubmed
Search Pubmed Now: Uterine Gland | Uterine Gland Secretion | Endometrial Gland
Additional Images
Terms
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 5) Embryology Uterine Gland. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Uterine_Gland
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G