Paper - The prenatal development of the human temporomandibular joint
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Moffatt BC. The prenatal development of the human temporomandibular joint. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: .
| Online Editor |
|---|
| This historic 1957 article by Moffatt looks at the development of the human temporomandibular joint, that connecting the mandible to the skull. Pater uses Carnegie Collection embryos: 1318, 1455, 3990, 5652, 625, 6581, 6701, 6832, 858, 86, 96
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Prenatal Development Of The Human Temporomandibular Joint
Benjamin C. Moffatt, Jr.
Contributions To Embryology, No. 246
Introduction
The least dispensable synovial joint in man is the articulation between the upper and lower jaws, the temromandibular joint. This articulation is also in many asects unique, exhibiting such special anatomical char;eristics as a complete intra-articular disk formed by 3 modification of an intercepted tendon, two synovial cavities, fibrocartilaginous articular surfaces, and tootharing skeletal components. The temporomandibular nt is the only articulation which has both hinge and ding movements. This joint is a relatively new structure, existing only mammals. It appeared late in evolution when the early jaw joint was incorporated into the middle ear }aupp, 1911). What the temporomandibular joint zks in phylogenetic antiquity, however, it makes up in ??. The movements of the lower jaw in such vital functions as speech, mastication, and respiration all in place at the temporomandibular joint. If this articuion is injured during childhood or if it develops abrmally, the result will often be an extreme undervelopment of the lower jaw and face, causing severe occlusion and other dental problems, disturbed digesn and respiration, and possibly psychological complications. The temporomandibular joint has been and still is the bject of a great many clinical investigations because its intimate functional relationship with the occluhsion the teeth. There are many controversies in the literure about this and other aspects of the temporoman)ular joint. It is surprising to find, however, that no ebryological evidence has been brought to bear on these ntroversial issues. A partial explanation is the fact at a careful search has disclosed only three major urces of information on the embryology of the human nporomandibular joint. Kjellberg (1904) described the temporomandibular nt in 5 human fetuses, the smallest one having a crown—rump length of 55 mm. He observed in 3 of these specimens that some of the fiber bundles of the external pterygoid muscle pass into the articular disk of the temperomandibular joint and then continue posteriorly between the squama and the tympanic bone to attach to the head of the malleus. He concluded that the passage of the external pterygoid muscle between the temporal squama and the mandibular condyle on its way to the malleus accounts for the formation of the articular disk.
Vinogradoff (1910) studied the development of the temporomandibular joint in 9 human fetuses, beginning with a 33-mm. specimen. He concentrated on the histological changes that occur in the condyle and zygomatic process of the temporal bone during fetal life. His description of the other joint components is vague. He presents no evidence of a connection between the external pterygoid muscle and the malleus.
More recently Harpman and Woollard (1938) made a specific study of the tendon of the external pterygoid muscle; they confirmed Kjellberg's original finding that in the human fetus this muscle does contribute to the origin of the articular disk or meniscus by passing posteriorly between the squama and the condyle to the malleus.
Macalister (1955) has described some of the histological modifications that occur in the prenatal development of the temporomandibular joint.
Although these authors were limited to a small number of specimens, their descriptions do show that the embryology of the temporomandibular joint difiers in some respects from that of the more commonly studied synovial joints. Before such a comparison can be made, however, further investigation of the embryology of this joint is required. This need for detailed information led to a study of the temporomandibular joint components in human embryos and fetuses at the Department of Embryology, Carnegie Institution of Washington. The investigation was supported by a grant from the fellowship fund of the Carnegie Institution of Washington and by the Department of Anatomy, Medical College and School of Dentistry, University of Alabama. I wish to express my gratitude to Dr. George W. Corner for his generous assistance during this investigation; to Mr. Richard Grill for the accompanying photomicrographs; and to Drs. Earl O. Butcher, E. Carl Sensenig, and William L. Straus for the stimulating encouragement and interest they have shown in this project.
Anatomy of the Temporomandibular Joint in the Adult
The fully developed temporomandibular joint is a tely movable synovial joint between the condyle of 2 mandible and the articular eminence of the temral bone. A fibrous joint capsule lined with synovial sue surrounds the articular surfaces of the two bones.
Inside the joint is an intra-articular disk or meniscus which separates the condyle from the articular eminence of the temporal bone. The articular disk is attached around its entire periphery to the joint capsule. The synovial cavity is thus divided into two separate spaces: a superior joint cavity between the disk and the temporal bone; and a smaller, inferior joint cavity between the disk and the condyle of the mandible.
One of the muscles acting upon the joint is the external pterygoid muscle, which is inserted on the anterior surface of the neck of the condyle and on the anteromedial portion of the articular capsule and disk. When the mouth is widely opened or the lower jaw is protruded, this muscle pulls the condyle and disk forward, so that they slide downward along the sloping posterior surface of the articular eminence. As the mouth is closed, the external pterygoid muscle normally relaxes and the condyle and disk return to their former resting posi tion higher up on the posterior surface of the articular eminence.
The central portion of the articular disk is compressed between the condyle and the articular eminence and contains neither blood vessels nor nerves. The function of the disk is generally regarded to be to stabilize and help to lubricate the incongruent articular surfaces of the condyle and articular eminence. Some of the controversy that still continues over the temporomandibular joint centers about the anatomy and function of the articular disk. A knowledge of the embryology of this joint may, in addition to its inherent value, help resolve these controversial questions.
Prenatal Development
The development of the temporomandibular joint was studied in a series of 46 human embryos and fetuses from the Carnegie Collection, ranging from 23 to 350 mm. crown-rump length. The specimens were sectioned in the frontal, sagittal, and transverse planes as indicated in table I. In fetuses larger than 80 mm., the region of the temporomandibular joint was removed and only this part of the specimen was sectioned.
Embryonic Rudiments
The rudiments of the human temporomandibular joint make their embryonic appearance in the last two stages, xxii and xxiii, of Streeter’s (I951) developmental horizons
Developing mandible
In embryo no. 6832 (stage xxii, 25.8 mm.) the region of the future joint was recognized with great difficulty, because most of the joint components were not yet in existence and the muscles had not differentiated suffciently to serve as landmarks. Identification was established by tracing distally the masseteric and auriculotemporal nerves, which enclose the developing joint anteroposteriorly.
The mandible is the only joint component present at this stage. There is no condyle, its place being occupied by the upper end of the osseous lamella, which constitutes the mandible. Meckel's cartilage already extends from the middle-ear region to the mid-line of the future chin, where it meets the distal end of the contralateral first branchial bar. This cartilage passes posteromedially to the future temporomandibular joint and appears on the medial side of the developing mandible. No other parts of the joint can be identified in this embryo.
Origin of articular disk
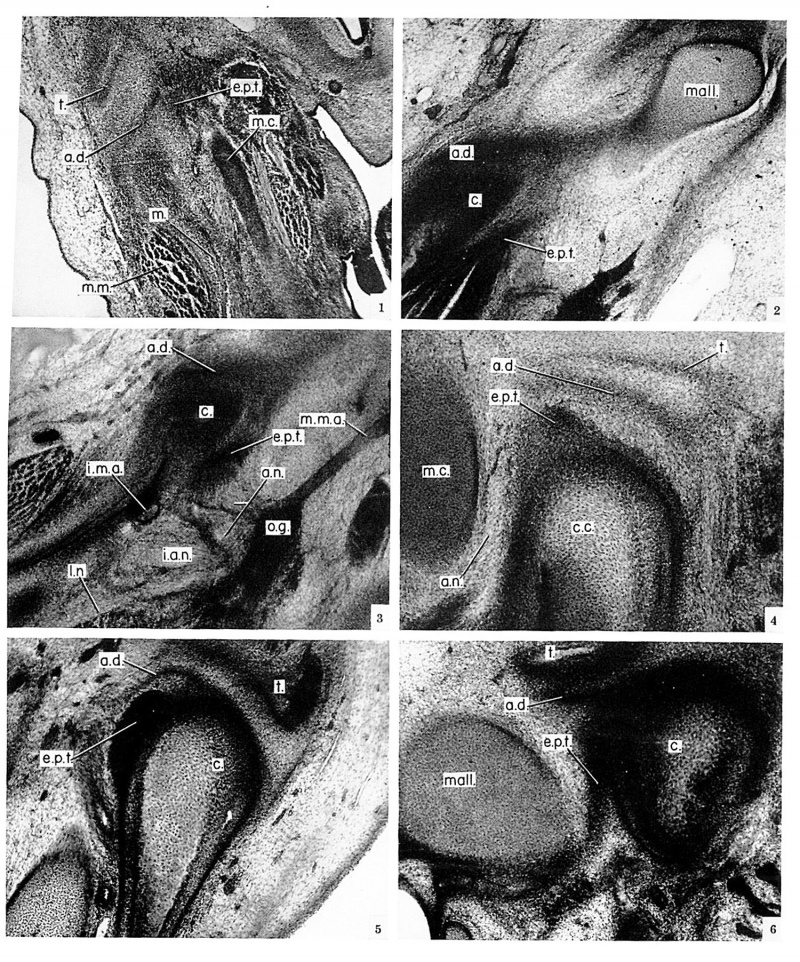
The origin of the articular disk is associated with the appearance of the external pterygoid and masseter muscles, which are recognizable in a stage-xxii embryo (no. 6701, 24 mm.). In a frontal section (fig. 1, pl. 1) through the region corresponding to the anterior portion of the future condyle, the disk is seen as a vague layer of mesenchyme extending laterally across the superior border of the external pterygoid muscle to the medial side of the masseter. The developing disk is separated from the condensation of mesenchyme which becomes the zygomatic process of the temporal bone by a large area of sparse cells, the precursor of the superior joint cavity. In contrast, the inferior joint cavity is preceded by only a very narrow zone of cells, because the articular disk, even in its early development, rests closely on the mandible. In this specimen an attachment of the external pterygoid muscle to the mandible could not be identified. The muscle, however, could be followed back to the posterior end of Meckel’s cartilage.
An embryo of stage XXIII (no. 86, 30 mm.) shows the further development of the articular disk. In frontal section (fig. 2, pl. 1), the upper surface of the developing condyle faces laterally. Lying against this surface is a dark band of cells which gives rise to the articular disk. The external pterygoid muscle joins the medial side of the condyle at its approximate site of attachment in the fully developed joint. The muscle does not end at this point, however. It passes posteriorly along the medial side of the condyle and joins the band of cells forming the articular disk. The cells giving rise to the articular disk and to the external pterygoid tendon continue from this point posterosuperiorly to attach to that portion of Meckel’s cartilage which becomes the malleus. This attachment of the mesenchymal layer and the external pterygoid tendon to the malleus is a constant feature, seen in all specimens up to and including a 40 mm. fetus.
- 1 The term tendon is used here in a morphological rather than a histological sense; at this stage it consists only of undifferentiated mesenchyme.
In this 30 mm embryo, membrane-bone spicules make their appearance in the zygomatic process and squama of the temporal bone. The articular disk is still separated from the temporal bone by a large area of sparse cells which are giving up their round form to become elongated. The joint capsule is not yet recognizable.
Fetal Development
Relations to adjacent structures
In a frontal section through a 37 mm. fetus, no. 1318 (fig. 3, pl. 1), the condyle is still merely a condensation of mesenchyme around the upper end of the osseous lamella forming the mandible. The articular disk and external pterygoid tendon are shown continuing posteriorly to their attachment on the malleus. The structures in immediate relation to the medial side of the temporomandibular joint are demonstrated in this section. Directly inferior to the external pterygoid muscle is the internal maxillary artery cut in cross section. The middle meningeal artery is seen in longitudinal section running superiorly between the condyle and the otic ganglion. The two portions of the auriculotemporal nerve which surround the middle meningeal artery are visible, as are also the inferior alveolar and lingual nerves.
First appearance of the condylar cartilage
No further development of the joint components was noticed until the condylar cartilage appeared in a 46mm. fetus, no. 95. The cartilage is located at the upper end of the osseous lamella, which forms the posterior border of the mandible. In sagittal section (fig. 4, pl. 1), the growth of this cartilage appears to be directed superiorly and somewhat anteriorly. The condylar cartilage has not grown sufficiently to force the disk into contact with the superior articular element. Immediately above the condyle, between it and the disk, is the external pterygoid tendon cut almost in cross section as it curves laterally to attach to the malleus. The auriculotemporal nerve has been cut obliquely, passing between Mcckel's cartilage and the posterior surface of the condyle.
The articular disk appears to be elevated from the condyle because this section is close to the external pterygoid muscle, which comes between the mesenchymal portion of the disk and the medial part of the condyle. The relationship is better understood by referring to the frontal section (fig. 5, pl. 1) of a 49 mm. fetus, no. 5652. The articular disk lies directly on the condyle except in the area occupied by the external pterygoid tendon. This frontal section illustrates several important points about the articular disk:
- The disk is associated developmentally with the mandible and the muscles of mastication, specifically the external pterygoid and the masseter.
- The major portion of the articular disk is derived from a mcsenchymal layer which, anterior to the joint, extends from the external pterygoid muscle to the masseter muscle, and, after passing over the condyle, continues posteriorly in company with an extension of the external pterygoid muscle to attach to the developing malleus.
- The posterior extension of the external pterygoid muscle does in fact contribute to the formation of the articular disk, as is shown in the other figures.
Passage of external pterygoid tendon and disk to malleus
The actual passage of the external pterygoid tendon from the condyle to the malleus can be seen in a sagittal section (fig. 6, pl. 1) of the temporomandibular joint in a 50 —mm. fetus, no. 96. The tendon runs obliquely from the upper posterior part of the condyle to the posterior surface of Meckel’s cartilage. In adjacent sections Meckel’s cartilage changes its direction and position slightly so that the pterygoid tendon is actually attached to the lateral side of the malleus. A short segment of the articular disk extends posteriorly above the upper end of the external pterygoid tendon and parallel to the membrane bone that forms the zygomatic process of the temporal bone. This part of the disk when followed posteriorly joins the external pterygoid tendon and travels in common with it to the malleus.
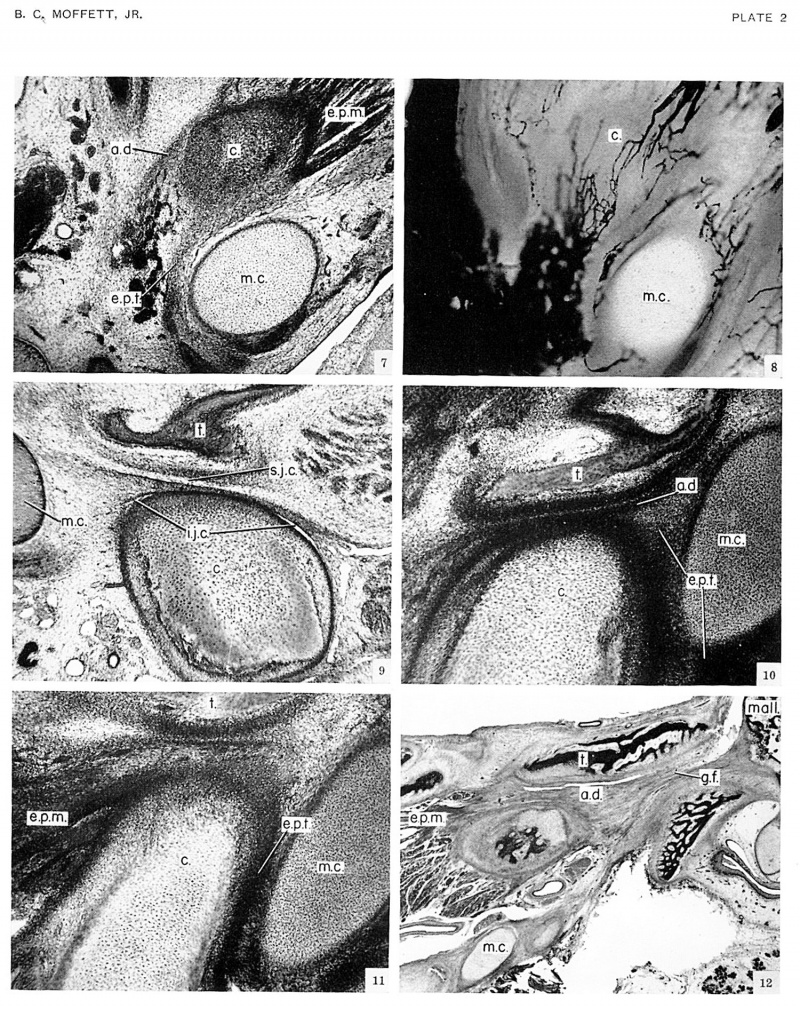
The attachment of the developing disk to Meckel’s cartilage can be seen also in superior view by looking down on a transverse or horizontal section (fig. 7, pl. 2) through the joint in a 54-mm. fetus, no. 3990. Here the tendon of the external pterygoid muscle passes over the medial side of the condyle and then turns, proceeding to the lateral side of Meckel’s cartilage, on its way to the head of the malleus. The contribution from the disk over the lateral part of the condyle is more readily visible a few sections higher, where it joins this tendon as a somewhat more deeply stained band of cells.
Development of articular blood supply
The specimen just discussed, a 54 mm. fetus, no. 3990, also illustrates certain features in the development of the blood supply to the temporomandibular joint. These features will be described here because they have a bearing on a paper soon to be published on the blood supply in the fully developed joint. In a horizontal section (fig. 7, pl. 2) through the joint, many blood vessels, mostly veins, are seen in the area enclosed on three sides by the sectioned disk, the lateral surface of the condyle, and the external pterygoid tendon lateral to Meckel's cartilage. When followed inferiorly these blood vessels are seen to run vertically against the posterolateral aspect of the condyle and mandibular ramus. At varying levels along the ramus they join the superficial temporal artery and vein, the posterior facial vein and the external carotid artery. The same vessels when traced superiorly are seen to shift slightly to a closer association with Meckel’s cartilage. They remain on the lateral side of this cartilage, gradually diminishing in number as they give off branches which surround the developing malleus and incus. They apparently terminate on the ossicles in the region of the middle ear.
At the level shown, these blood vessels give off branches which run anteriorly in the articular disk and on the ternal pterygoid tendon to anastomose with capillaries iming from the anterior surface of the joint. This capilry anastomosis, extending anteroposteriorly completely zross the articular disk, is shown in a horizontal section fig. 8, pl. 2) through a 57.25 mm. fetus, no. 858, in which re vascular system was injected with India ink. The level of section corresponds to that shown in figure 7, Id passes through the disk and pterygoid tendon nmediately above the condyle.
Growth of condyle and formation of joint cavity. As te condyle grows, its articular surface and shape undergo tarked changes. In a 30-mm. embryo, before the apearance of a condylar cartilage (fig. 2, pl. 1), the articu.r surface of the condyle faces directly laterally, almost I a vertical plane. The articular disk and the articular surface of the zygomatic process of the temporal bone are arallel to that of the condyle.
In a 49-mm. fetus, no. 5652, the condylar cartilage has taped the articular surface of the condyle into a hemi, as seen in a frontal section (fig. 5, pl. 1) through the center of the cartilage. The articular disk flattens tis contour and makes it congruous with the articular trface being formed by the squama and zygomatic rocess of the temporal bone. The plane of the articular sufaces has shifted to approximately 45° from the vertical. The joint cavities have not yet appeared in this specimen, and there is no actual contact between the two articulating elements. In five out of the six 50-mm. fetuses studied, the conyle and disk are not in contact with the temporal bone and the joint cavities are not yet formed. The sixth specimen shows an area of sparse cells between the disk of the temporal bone which appears to be developing cavity. Contact between the disk and temporal bone made in the 60 mm. fetuses. The sections in these specimens were too thick, however, to show the presence 3 joint cavities. In a 75-mm. fetus, no. 6581, sagittal section (fig. 9, pl. 2), the inferior joint cavity is present 1d the superior cavity is being formed. Development of joint capsule. The articular capsule of re temporomandibular joint is first recognizable in )-mm. fetuses in the form of faint cellular condensations along the medial and lateral sides of the joint, conecting the two skeletal components. The articular disk terges peripherally with these capsular condensations 1d thus obtains an attachment to the temporal bone 1d mandible. The joint is still not bounded anteroasteriorly by a capsule in the 75-mm. fetus. Before the irmation of the capsule posterior to the joint, the articu.r disk transfers its attachment from Meckel’s cartilage 1 the temporal bone and mandible. This transfer reilts from a narrowing of the Glaserian fissure and an aproximation of Meckel’s cartilage to the posterior part E the neck of the condyle. An early phase in the trans fer to the temporal bone is seen in a sagittal section (fig. 10, pl. 2) through the lateral portion of the temporamandibular joint in a 78.5-mm. fetus, no. 1455. The articular disk here extends posteriorly, following the contours of the zygomatic process of the temporal bone, a relation which results from the approximation of the two articular elements and from the persisting attachment of the disk to Meckel’s cartilage. As the articular disk accompanies Meckel’s cartilage through the Glaserian fissure into the middle ear, the disk comes in contact with the anterior wall of the fissure. At this point the cells of the disk are becoming attached to the temporal bone. This attachment limits the superior joint cavity posteriorly and forms the superior attachment of the developing articular capsule on the anterior lip of the Glaserian fissure posterior to the temporomandibular joint.
The inferior attachment of the disk and articular capsule on the posterior surface of the neck of the condyle results from a similar transfer involving the external pterygoid tendon, the mandible, and Meckel’s cartilage. In figure 10 (pl. 2), the external pterygoid tendon is visible on the posterior surface of the condyle and again just inferior to Meckel’s cartilage as the tendon continues toward its insertion on the malleus. Further medially in a sagittal section through the middle of the condyle in the same specimen the tendon forms an attachment to the mandible (fig. 11, pl. 2). Here the external pterygoid tendon passes through a narrow space between the mandible and Meckel’s cartilage and appears to be attaching itself to the neck of the condyle. From this point, cells are seen extending superiorly toward the attachment of the disk on the temporal bone. At this stage, however, there is not a complete capsule posterior to the joint; the development of the capsule in this region proceeds very slowly.
In a 140 mm. fetus the articular disk still passes through a narrow Glaserian fissure to be inserted on the malleus. Some of the cells are attached, however, to the anterior lip of the fissure. Meckel’s cartilage is now constricted as it passes through the fissure, but it is still continuous with the malleus. In a 130 mm. fetus, the disk ends at the Glaserian fissure and the joint is definitely enclosed posteriorly by a capsule. The joint cavities are sharply bounded by synovial tissue. Synovial villi with adipose subsynovial tissue project into the anterior and posterior ends of the cavities.
The attachments of the disk to the temporal bone and condyle are shown in sagittal section through the joint of a 220-mm. fetus, no. 625 (fig. 12, pl. 2). This section passes through the medial part of the condyle. The external pterygoid muscle is seen merging with the anterior part of the disk. The Glaserian fissure is still open.
Meckel’s Cartilage
Before the first components of the temporomandibular joint became visible in a stage-xxii embryo, Meckel’s cartilage extended all the way from the base of the skull to the future chin, this relation being maintained during the entire development of the temporomandibular joint. The cartilage serves, therefore, as a supporting framework around which the mandible develops, and as a strut which holds the lower jaw in place until the temporomandibular joint has formed. It plays a direct role in the formation of the temporomandibular joint through its association with the external pterygoid tendon and the mesenchymal portion of the articular disk. Meckel’s cartilage also carries blood vessels through the Glaserian fissure which vascularize the developing malleus and incus. These vessels can be seen in 57- to 180-mm. fetuses.
At 220 mm., when the temporomandibular joint can be considered developed, Meckel's cartilage still extends through the Glaserian fissure. The portion of Meckel’s cartilage extending between the mandible and the skull, however, is very thin, and in a 270 mm. fetus it becomes recognizable as the sphenomandibular ligament. K.jellberg (1904) presents an interesting historical summary of information on this and other derivatives of Meckel’s cartilage. At 270 mm. the superior attachment of the sphenomandibular ligament appears to be on the medial end of the temporal bone, the sphenoid bone being directly adjacent to this point. The Glaserian fissure is practically closed at this stage. In the 350-mm. fetus, the superior attachment of the sphenomandibular ligament is on the sphenoid bone just lateral and posterior to the foramen spinosum.
Skeletal Components
The mandibular fossa develops its final contours only during postnatal life, with the appearance of the articular eminence of the temporal bone. A slight concavity present in the 180 mrn. and larger fetuses is due only to a slight prominence or projection at the posterior border of the fossa.
During its development the condylar cartilage shows a peculiarity which has been described by Vinogradoff (1910). In the 140-mm. fetus and on through the 350mm. fetus, peglike processes, which Vinogradoff likens to “crampons,” occur at first in the lateral portion of the condyle. Then they appear also on the medial side, and finally all over the articular surface of the condylar cartilage. Vinogradoff says that these “crampons” serve to anchor the fibrous articular surfaces in the temporomandibular joint to the condyle and to the temporal bone.
Discussion
Time of Development
The first significant fact brought out by this study is the relatively late embryonic development of the temporomandibular joint as compared with the larger joints of the extremities. During the last stage, xxiii, of embryonic development, all the major components of the elbow, hip, and knee joints “are present in a form and arrangement closely resembling the adult" (Gardner, 1932). In this same embryonic stage, however, the temporomandibular joint is still rudimentary in comparison with the fully developed joint; the condylar cartilage, joint cavities, synovial tissue, and articular capsule have not yet appeared, and the skeletal elements——mandible and temporal bone-—are not in contact. All the components of the temporomandibular joint except the articular eminence of the temporal bone are present in the 220-mm. fetus. The joint does not assume its typical adult contours, however, until the articular eminence has completed its development during the twelfth year of age (Humphreys, 1932). Aarrcuma D151;
The development of the articular disk is worthy of discussion in order to correct some common misstatements about this structure. The articular disk of the temporomandibular joint should not be regarded as forming from tissue which is caught or pinched between the mandible and the temporal bone by upward growth of the condyle, as Keith (19.18) states. This erroneous con cept can be traced to Vinogradoff (1910) and to other investigators who did not study embryos young enough to show the early development of the disk. Actually, the articular disk can be identified as a mesenchymal and muscular derivative of the first branchial arch, which appears earlier than the condyle, long before the mandible comes in contact with the temporal bone. If the disk were to develop as stated by Keith, disturbances in its formation would undoubtedly result in specific congenital anomalies which have in fact never been reported. The only known developmental anomaly of the temporomandibular joint is the absence of one or both condyle— a very rare condition.
The development of the temporomandibular intraarticular disk resembles certain processes seen in other joints and yet in other respects appears to be a unique phenomenon. For instance, a small medial portion of the disk is derived from the posterior extension (referred to as a tendon) of the external pterygoid muscle. This passage of a muscle tendon through a synovial joint is similar to the situation described in the human shoulder joint by Gardner and Gray (1953), in which the tendon of the long head of the biceps brachii muscle develops in situ in the extrablastemal tissue of the glenohumeral joint. These situations differ, however, in that the external pterygoid tendon becomes incorporated into the disk and later in fetal life loses its attachment to the malleus. Development here does not proceed rapidly from the primordial state to a form and arrangement characteristic of the adult. The disk passes through the .ntermediate stages described here and by other investigators. The temporomandibular joint does truly seem :0 have a developmental stage in which it resembles the normal structure of lower forms where the jaw joint oc:urs between the os quadratum and os articulare. Its relatively late appearance as a new structure in evolution supports this conclusion, which as far as is known applies to no other joint.
In describing the fully developed temporomandibular joint, the term articular disk is frequently limited to the fibrous, hard, glistening portion of the intra—articular tissue. This definition excludes a considerable quantity of less-dense connective tissue, vascularized and innervated, which connects the fibrous portion of the disk on all sides to the joint capsule. Developmentally, there is no basis for this distinction. During fetal life the entire disk is vascularized. Only after the central portion of the disk is compressed between the condyle and temporal bone does this portion become avascular. In the fully developed joint the peripheral portions of the disk still retain their blood and nerve supply, the distribution of which will be described in later papers. The complete vascularization of the disk seen in the fetus indicates that both the vascular and avascular portions in the fully developed joint should be included in the definition of the articular disk.
Mechanical Theories of Joint Formation
The formation of intra-articular disks is explained by several investigators as resulting from the absence of pressure exerted on the undiflerentiated mesenchyme between the developing bones. Whillis (1940) says that relief of pressure on the joint disk in the ulnar-triquetral joint by the ulnar styloid process results in a non-chondrified disk which becomes stretched and incorporated in the medial ligament of the wrist joint. He cites the clavicle as another example by saying that the centers of ossification that appear in the early development of the clavicle lead to a slowing in the growth of the precartilage. This relief of pressure on the r esenchyme removes the stimulus for chondrification, d the disks become modified to form the articular dis of the sternoclavicular and aeromioclavicular joints Prentiss (1918) had already proposed this explanatio in connection with the development of the knee and tr' iporomandibular joints. The embryonic human knee joint contains at first a complete meniscus separating thr femur from the tibia. Prentiss says that at birth th central portion of this meniscus or disk has disappeared, undoubtedly because of pressure exerted on the dint by powerful muscular pull across the joint. When in postnatal life great pressure is brought to bear on this joint by standing, etc., the disk continues its retrograde changes until only semilunar cartilages remain.
Prentiss explains the development and also the pathology of the temporomandibular articular disk by this concept of the eflect of pressure. He says that eruption of the teeth and, earlier, the presence of the large alveolar processes of the jaws prevent the condyles from pressing against the temporal articular surfaces. The disk, therefore, is not obliterated by pressure. The finding reported here that the condyle and disk are separated from the temporal bone during the early development of this joint favors this hypothesis. An observation familiar to anatomists and dentists is mentioned by Prentiss: with the loss of the teeth and resorption of the alveolar processes, the disk is subjected to increased pressure, and it degenerates, disappearing partially or almost completely.
Another hypothesis that interprets joint formation on a mechanical basis is presented by Carey (1922). He postulated that joints are the mechanical resultants of the opposed actions of accelerated growth centers of the segmental skeleton; the contours of the opposed surfaces forming a joint are dependent upon the intensity of the force of interstitial growth on the cross-sectional area of the segments forming the joint and upon the resistances to the growth of each skeletal segment. As an example Carey cites the hip joint, where the head of the femur advances farther into the acetabulum because the femur has a greater longitudinal growing force than the acetabulum.
Both these suppositions—that pressure determines the fate of an articular disk, and that it establishes the contours of the joint surfaces—are compatible with the development of the temporomandibular joint. They must remain hypothetical, however, until experimental evidence determines whether a cause-and-effect relation exists between these conditions in the embryo as it appears to do in pathological changes of the temporomandibular joint.
Joint Interzones
Considerable attention has been given by many investigators (see Gardner, 1950) to the question, Are articular structures derived from a blastemal interzone? Much evidence has been offered from the study of the joints of the extremities to indicate that either the blastema or the mesenchyme, or both, may be the source of articular structures. The temporomandibular joint consists of membrane-bone skeletal components that are not connected by a blastemal interzone. A eondylar growth-cartilage appears later in one of the skeletal components, but there is no indication in this joint of a continuous blastema which chondrifies and becomes segmented. The interzone appears to be mescnchymal, and through it the external pterygoid tendon passes to contribute to the formation of the articular disk, which as stated before is a derivative of the first branchial arch. The late evolutionary development of the temporomandibular joint, and the fact that its skeletal components are membrane—bone elements of the skull and first branchial arch, result in a developmental pattern which differs from that seen in the joints of the extremities.
Summary
- The temporomandibular joint develops from the membranous portion of the temporal bone; from skeletal, muscular, and mesenchymal derivatives of the first branchial arch; and from a mesenchymal interzone.
- The rudiments of the temporomandibular joint appear in the last stages of embryonic development. The joint passes slowly through a developmental stage in which it resembles the normal structure of lower forms where the jaw joint occurs between the os quadratum and os articulate. Its development is essentially complete in the 220 mm fetus.
- The articular disk develops early from muscular and mesenchymal derivatives of the first branchial arch. It consists mainly of a mesenchymal layer which passes between the external pterygoid and masseter muscles and then extends posteriorly over the developing condyle to attach to the part of Meckel’s cartilage that becomes the malleus. A small medial segment of the disk is formed by the tendon of the external pterygoid muscle, which is also attached to the malleus.
- The articular disk maintains its attachment to the malleus until fetal development of the joint is completed. It then takes part in the formation of the posterior joint capsule by becoming attached to the anterior lip of the Glaserian fissure and to the mandible.
- Meckel’s cartilage plays an important role in the development of the temporomandibular joint by (a) forming a framework around which the mandible develops; (b) acting as a strut which holds the lower jaw in place until the temporomandibular joint is formed; (c) providing an attachment for the developing articular disk.
- Developmental differences between the temporo-mandibular joint and the joints of the extremities are described, and mechanical theories of joint formation are
discussed.
Literature Cited
Carey EI. Direct observations on the transformation of the mesenchyme in the thigh of the pig embryo (Sus scrofa), with especial reference to the genesis of the thigh muscles, of the knee- and hip-joints, and of the primary bone of the femur. (1922) J Morphol. 37: 1-77.
GARDNER, E. 1950. Physiology of movable joints. Physiol. Rcv., vol. 30, pp. 127-176.
- 1952. The anatomy of joints. Amer. Acad. of Orthopaed. Surgeons Instructional Course Lectures, vol. 9, pp. 149-170.
- and D. I. GRAY. 1953. Prenatal development of the human shoulder and acromioclavicular joints. Amer. Jour. Anat., vol. 92, pp. 219-276.
CAUPP, E. 1911. Bcitriige zur Kenntnis des Unterkiefers der Wirbcltiere. Anat. Anz., vol. 39, pp. 609-666.
Harpman A., and H. H. Woollard 1938. The tendon of the lateral pterygoid muscle. Jour. Anat., vol. 73, pp. 112-115.
Humphreys, H. 1932. Age changes in the temporomandibular joint and their importance in orthodontics. Internat. Jour. Orthotlontia Oral Surgery and Radiography, vol. 18, pp. 809-815.
Keith, A. 1948. Human embryology and morphology. 6th ed. 690 pp. Baltimore.
Kolliker, K. 1904. Beitriige zur Entwicklungsgeschichte des Kiefergelenks. Gegenbaurs Morphol. Iahrb., vol. 32, pp. 159I84.
McCallister, A. D. 1955. The development of the human temporomandibular joint. A microscopic study. Australian Jour. Dent., vol. 59, pp. 21-27.
Prentiss, H. I. 1918. A preliminary report upon the temporomandibular articulation in the human type. Dent. Cosmos, vol. 60, pp. 505-512.
Streeter GL. Developmental Horizons In Human Embryos Description Or Age Groups XIX, XX, XXI, XXII, And XXIII, Being The Fifth Issue Of A Survey Of The Carnegie Collection. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: 167-196.
VINOGRADOFF, A. 1910. Développemcnt (le l'articulation temporo-maxillairc chcz l'homme dans la période intrautérine. Intcrnat. Monatsschr. E. Anat. u. Physiol., vol. 27, 1319- 490-523
Whillis J. The development of synovial joints. (1940) J Anat. 74(Pt 2): 277-283. PMID: 17104813
Plates
Abbreviations Used In Plates
art. d., articular disk
a. 71., auriculotemporal nerve
c., condyle
5'. c., condylar cartilage
c. p. 121., external pterygoid muscle 9. p. t., external pterygoid tendon g. f., Glaserian fissure
m., mandible maI1., malleus
1'. (1. 11., inferior alveolar nerve
i. ta, inferior joint cavity
2'. m. (1., internal maxillary artery 1. 12., lingual nerve
m. c., Meckel’s cartilage
m. m., masseter muscle
m. m. 11., middle meningeal artery 0. g., otie ganglion
5. j. c., superior joint cavity
1., temporal bone
Plate 1
Plate 2
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The prenatal development of the human temporomandibular joint. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_prenatal_development_of_the_human_temporomandibular_joint
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G