Paper - On the development of the membrana tectoria with reference to its structure and attachments
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Prentiss CW. On the development of the membrana tectoria with reference to its structure and attachments. (1913) Amer. J Anat. 14(4): 425-461.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
On the Development of the Membrana Tectoria with Reference to its Structure and Attachments
C. W. Prentiss
The Anatomical Laboratory of the Northwestern University Medical School, Chicago
Forteen Figures (1913)
Introduction
For more than half a century various investigators have studied the structure of the cochlea with conflicting results. Comparatively recently Kishi ('07) and Shambaugh ('07) have championed the view originally held by Retzius ('84), that the membrana tectoria remains attached to the organ of Corti. They maintain moreover that the membrana is the^ logical structure through which sounds are transmitted to the auditory cells, and that it acts as a resonator. This function Von Helmholtz was the first to ascribe to the rods of Corti and later with Hensen to the fibers of the basilar membrane.
Hardesty ('08) denies the existence of an attachment between the membrana and the spiral organ, yet maintains that through the medium of the membrane sound vibrations are transmitted to the auditory hairs. A voluminous literature has been written deahng with the physiology of an organ the structure of which is inadequately known. To ascribe a definite function to the membrana tectoria we must first know with absolute certainty its structure and attachments. No physicist will accept as an important organ of hearing a membrane of indefinite structure and with no fixed position with reference to the spiral organ itself. For such a floating membrane, as we shall show later on, may readily change its position and relations to the auditory cells, and would certainly interfere with and interrupt the auditory function.
From the physiological standpoint it is then necessary to answer two anatomical questions before we may assign the membrana a logical role in the processes of audition: (1) Has the membrana any definite and peculiar structure w^hich may adapt it to the transmission of sound vibrations? (2) Is the membrana so attached as to be constantly in contact with the hair cells of the spiral organ? We hold that as yet these questions have received no adequate answer.
The membrana is described by nearly all of those who have investigated it, as being an elastic cuticular structure containing within its interstices a more or less fluid matrix. This cuticular membrane has been variously interpreted as formed of agglutinated ciha or hairs (Ayers '92); as lamellar (Shambaugh '07); as a reticulum (Retzius '84); as a coagulum of the endolymph (Czinner and Hammerschlag '98) ; as a fibrous feltwork embedded in a gelatinous matrix (Hardesty '08) ; as composed of fibers and cuticular layers (Held '09).
As to its attachments there is a division of opinion, some holding that it is attached to the spiral organ (Retzius '84, Coyne et Cannieu '95, ffishi '07, Shambaugh '07) while this is denied by others — more recently by Rickenbacker ('01), Hardesty ('08) and Held ('09).
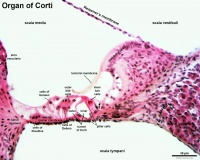
The classic figures given in textbooks of anatomy and histology (fig. 1) show it as a lamellated membrane attached to the labium vestibulare and extending outward over the internal spiral sulcus and the organ of Corti. Its outer edge thus floats free in the endolymph and the lamellae are shown parallel to the ends of the hair cells. The textbooks usually state that it takes its origin from the limbus spiralis and hence must grow by the development of new lamellae from beneath.
The conclusions of those who have worked on the development of the membrana tectoria are as contradictory as are those who have interpreted its structure. Kolliker ('61) originally described the membrana tectoria as a finely striated membrane arising from the columnar epithelial cells of the basal wall of the ductus cochlearis. Hensen ('63), Retzius ('84), Pritchard ('78), Schwalbe ('87) and others agree as to its cuticular origin. Czinner and Hammerschlag ('98) assert that it arises independently as a coagulum or concretion of the endolymph, and later becomes attached to the epithelium. Ayers ('92) maintained that the fused hairs of the auditory cells form the membrana tectoria and that their agglutinated tips later fuse to the labium vestibulare, while Bottcher ('70) asserts that it is formed from hairs arising from the epithelial cells of the cochlear duct. Coyne et Cannieu ('95) found that the membrane was attached to the organ of Corti or had been torn away from it and that it shows a lamellar or reticular structure according as it is sectioned through the axis (modiolus) of the cochlea or in a plane perpendicular to this. There are thickenings at the angles of the reticulum and these thickenings give to the membrane the striated appearance which is seen in other sections.
Fig. 1. Semi-diagrammatic representation of the organ of Corti and adjacent structures (Merkle-Henle).
Rickenbacher ('01) after studying five stages in the development of the cochlea of the guinea-pig concludes that there are two epithelial ridges (wuelste) in the floor of the cochlear duct. The inner axial ridge is the greater and its cells give rise, first, to the major part of the membrana tectoria as a cuticular secretion which increases in thickness outwardly by the addition of new lamellae. Later from these cells are formed the epithelium of the labium vestibulare, of the spiral sulcus and the inner portion of the spiral organ. The lesser or outer ridge gives rise to the outer portion of the membrana tectoria, and later forms the outer portion of the spiral organ. In the new-born guineapig the tectorial membrane loses its connection with the organ of Corti, probably owing to a dissolution of a portion of the membrane. The membrane makes its appearance before the differentiation of the organ of Corti or of the hair cells. In structure it is a cuticular reticulum which later becomes swollen, convex above and is detached from the spiral organ by the secretion of endolymph beneath it. No cilia or hairs were observed by Rickenbacher until after the differentiation of the organ of Corti, and then only the hairs of the auditory cells appeared. He does not state definitely just how the cuticular reticulum of the membrane arises nor does he account for the striated or lamellar structure which is characteristic in ordinary preparations and which he figures.
Hardesty ('08) in restudying the development of the membrana tectoria finds it first in embryos of 3 cm. as a cuticular film of appreciable thickness and decided fibrous character." Of the two epithelial thickenings in the basal epithelium the inner only takes part in forming the membrana tectoria, the outer giving rise only to the spiral organ (of Corti). Not till pigs of about 14 cm., do any preparations show evidences of differentiation of the cells of the lesser thickening into what will become the organ of Corti," says Hardesty. He thus agrees with Rickenbacher that the membrana is quite well formed before the hairs of the auditory cells appear, thus proving false the conclusions of Ayers. As he contends that the membrana does not develop over the spiral organ (though his figure 10 does not bear out this contention) Hardest}^ must account for its later position over and extending to the outer side of the hair cells. This he does by maintaining that owing to the retrogression of the cells which originally fill the spiral sulcus, there is an inward displacement of the spiral organ which thus causes the outer portion of the membrana to rest above and beyond it. Just how this can take place without a shortening of the basal membrane is not stated, nor do his measurements show conclusively that the pillar cells have actually approached the inner angle oi the cochlea a sufficient distance to warrant the change in position of the membrana tectoria. This point will be taken up in describing our own preparations. Hardesty describes an accessory tectorial membrane lying beneath the tectorial membrane proper and extending from near its outer edge to Hensen's stripe. It is composed of two sets of fibers crossing at an acute angle. In his figures these fibers form a network with diamond shaped meshes. Hardesty does not state how this accessory tectorial membrane is developed. Hensen's stripe, a line which has been described as extending lengthwise along the underside of the tectorial membrane, Hardesty regards as due to the intercrossing ends of the fibers composing the membrane. He states that its position corresponds to the line of enclasped phalanges of the pillars. Hence the stripe of Hensen should lie between the inner and outer hair cells. Hardesty believes that it has an embryological significance: "Hensen's stripe seems to be the expression of the period at which the retrogression of the epithelium began. It also represents the line along which the thick, outer edge of thickening was last attached and along which growth was last contributed to the membrane."
Held ('09) has made a detailed restudy of the development of the organ of Corti and the membrana tectoria in the ear of the guinea-pig, rabbit, pigeon and chick. He finds that the membrana tectoria is developed as cuticular fibers by the cells of the basal epithelium of the cochlear duct. An outer cuticular layer is first formed over the greater epithelial thickening; later growth consists in the secretion of the fibers by the cells of both thickenings, the hair cells alone taking no part in their development. Thus he holds that the membrana tectoria is developed in situ over the organ of Corti. Owing to the later elongation of its cells the organ of Corti shifts its position inward (axially) but this shifting is not extensive enough to account for the position of the membrana tectoria, which overlies and may project beyond the cells of the organ. In the adult fowl Held found that the membrana remains attached to the supporting cells of the sensory organ, but believes that in adult mammalia its attachment to the cells of the organ of Corti is lost.
Because of these contrary and diverse conclusions which the literature in regard to the development and structure of the membrana tectoria contains, and because of its importance to the physiology of audition it was determined to make a restudy of its development in pig embryos and of its structure in man. The present paper includes my work on the development only.
Methods
Experimenting with fixing fluids it was found that forinahn and Zenker's fluid preserved the membrana well but failed to bring out sharply its cuticular structure. Osmic acid of 2 per cent and Vom Rath's osmic-picric-acetic mixture was used with success in the later stages as the fixation was good and the browning of the cuticulum by the osmic acid made its structure more clear.
In the younger stages the whole head of the embryo was fixed. In the stages approaching full term the bony labyrinth was shelled out whole, and, after the stapes had been carefully removed, was immersed in the fixative two to three days. After fixation and hardening the decalcification of the older stages was completed in 80 per cent alcohol plus 5 per cent nitric acid. They were then embedded in celloidin or paraffine and cut in planes parallel and perpendicular to the modiolus of the cochleae. Preparations were thus made from pig fetuses measuring 4, 5.5, 7.5, 8.5, 13, 15, 18.5 and 20 cm. and these were compared with sections from full term fetuses. It was found that sections mounted in balsam were not favorable for a study of the membrana because its cuticular framework is of about the same refractive index as this mounting medium. Celloidin sections were therefore mounted in water, and while only temporary preparations could be made in this way, this method was of great value in determining the structure of the membrane when unstained. With sufficiently thin sections an oil immersion objective could be employed. No special staining methods were used, the browning of osmic acid being more effectual than any stain. Nuclei were demonstrated with haematoxylin and for counter stains eosin, orange G, and acid fuchsin were employed.
Description of Stages
Aside from the thickening of the basal epithehum, stages up to 4 mm. show no important changes.
Fig. 2. Section through the second turn (fig. 13) of cochlear duct from a 5.5 cm. fetus, showing basal epithelium and origin of membrana tectoria; inner or axial side to left; m.vest., membrana vestibularis; m.tect., membrana tectoria; gang.sp., spiral ganglion; t.sp., spiral tunnel. Oc. 4, Obj. 4.
5.5 cm stage
Here we have the basal epithehum composed of pseudo-stratified columnar cells (fig. 2).
The nuclei of the high columnar cells show a division into an inner and outer group wPiich correspond to the greater and lesser epithelial thickenings of Rickenbacher. A section through the cochlea of a somewhat later stage shows the topography of the spiral organ (fig. 13). The cochlear duct of the pig makes about 3.5 turns and hence a section through the modiolus shows four turns on the left and three on the right in figure 13. Figure 2 represents the basal half of the turn lettered (2) in figure 13. Of the two groups of cells which we have noted above the larger group forms the inner (axial) two-thirds of the basal wall. The nuclei of these cells are arranged in from three to six layers. They are separated from the outer group of cells by a cytoplasmic area free from nuclei. Between two cells of this area a vertical space is seen extending from the summits of the cells half way through the epithelium. This space may be due to shrinkage but represents the position of the spiral tunnel or tunnel of Corti in later stages. The outer cell group (lesser thickening of Rickenbacher) forms the outer third of the basal wall of the cochlear duct. It will eventually give rise to that part of the spiral organ which lies external to the spiral tunnel (fig. 1). From the inner epithelial cell group (greater epithelial thickening) will develop the epithelium of the labium vestibulare, of the internal spiral sulcus and that portion of the spiral organ lying internal (axial) to the tunnel. Extending over the free ends of the cells of the greater epithelial thickening may be seen a cuticular membrane which is attached between the cells by delicate threads. This cuticular membrane is the anlage of the membrana tectoria, which thus makes its appearance before the hair cells of the spiral organ are differentiated. At this stage the mesenchyma about the cochlear duct is dense and the scalae have not yet appeared. The nerve fibers of the spiral ganglion may be seen entering the epithelium internal to the organ of Corti.
8.5 cm stage
In the second turn of the cochlea at this stage (fig. 3 and fig. 13)- a space representing the spiral tunnel extends nearly through the thickness of the epithelium. The cells on each side of the tunnel are differentiating the pillars of Corti and a single inner and three outer hair cells are conspicuous. By the rapid division and elongation of the cells of the greater epithelial thickening the epithelial wall has been bent basalwards forming a concavity above and a convexity below. The concavity is the first trace of the internal spiral sulcus. The nuclei of the greater cell thickening show not so many layers as in the previous stage but the cells are longer than the pillar cells of the spiral organ. Near the inner angle of the cochlear duct the membrana tectoria has increased very Uttle in thickness and still forms a thin cuticular layer over the epithelial cells. Externally the membrana now extends beyond the outer hair cells of the spiral organ at which point a thin cuticle is just being formed. Over the spiral sulcus the growth of the membrana has been most rapid and here it is thickest. It appears to be composed of numerous parallel fibers or lamellae which are attached to the epithelium between the cells. When traced upwards away from the cells the lamellae converge and curving inwardly are continuous with the thin plate-like inner portion of the membrane which overlies the labium vestibulare.
Fig. 3. Section through the second spiral of the cochlear duct from a fetus 8.5 cm. long, showing the basal half of the cochlear duct and a portion of the scala tympani; h.c, hair cells of spiral organ; i.ep.c, inner epithelial thickening; o.ep.c, outer epithelial thickening; sc.tymp., scale tympani; other lettering as in figure 2. Oc. 4, Obj. 4, t. 1. 160.
The appearance of the tectorial membrane at this stage has been explained correctly, we believe, by Hardesty ('08). After a cuticular layer has been formed as in figure 2, the cells internal to the sulcus spiralis secrete very slowly or cease altogether. The other cells which are forming the membrana continue to secrete actively. At the same time these cells by growth and multiplication increase the width of the basal epithehum, carrying the spiral organ outwards. Thus the distance from the inner angle of the cochlear duct to the spiral tunnel is increased. In the second spiral of the 5.5 cm. stage as in figure 2 this distance is 140 ju- In the 8.5 cm. stage the same distance is about 280 mCells near the pillars of Corti which are secreting the membrane may thus be carried outward approximately 140 m, while that part of the membrane first formed does not grow. As the socalled 'lamellae' are secreted at the ends of the cells and the cells are shifted outward as the lamellae lengthen, naturally the bases of the lamellae will also be carried outward while their tips remain stationary. The inward trend of the lamellae from base to tip is thus satisfactorily accounted for.
It may be well to emphasize here the fact that the greater epithelial thickening gives rise not only to the epithelium of the labium vestibulare and of the spiral sulcus but also to the inner axial half of the spiral organ, including the inner supporting cells, and possibly the inner hair cells and inner pillars. This is in agreement with the results of Coyne et Cannieu ('95) and Rickenbacher ('01) Van der Stricht and Held ('09). Hardesty states that the lesser thickening is the first indication of the differentiation of the organ of Corti while the cells of the greater give origin to the tectorial membrane .... and the low indifferent cells lining the spiral sulcus." This misinterpretation is important as it partly accounts for his later statement that the memhrana is not derived from the cells of the spiral organ.
The next question to decide is the true structure of the membrana tectoria. Is the membrane composed of lamellae or hairs or fibers or is it a reticulum? If it is formed at the ends of the cells just how is it developed there? These points were decided by a study of later stages, the cochleae of a 13 cm. fetus proving most favorable material. In the various cochleae which were examined it was found that differentiation begins in the basal turn and is much less advanced in the upper turns. Thus in the 5.5 cm. stage the membrana tectoria was not yet developed in the upper turn though it had appeared in the second turn. The upper spiral of the 8.5 cm, stage was only slightly .advanced in development beyond that of the basal turn of the previous stage.
13 cm stage
The upper coil in this stage showed but little more differentiation of the tectorial membrane than figure 3. In the second spiral, however, a marked difference may be seen (fig. 4). A fibrous basement membrane is stretched beneath the epithelium, extending between the limbus spiralis and the spiral ligament. The epithelium itself shows a greater thickening in the region of the future sulcus spiralis, the cells being more elongate and clearer. At the axial side they show but one row of nuclei. At X these cells are sharply marked off from the epithelial cells of the labium vestibulare, the latter cells forming the so-called teeth of Huschke. The sulcus spiralis is somewhat deeper than in the preceding stage and the outer supporting cells of the spiral organ are longer and more sharply differentiated from the single layer of cubical cells external to them. The membrana tectoria is larger and extends from the inner angle of the cochlea duct to well beyond the spiral organ externally.
Fig. 4. Section through the basal portion of the second turn of the cochlear duct from a 13 cm. fetus; m.bas., membrana basilaris; limb.sp., spiral limbus. X shows point at which later labial teeth appear separating labium from the sulcus; * marks portion of field similar to that shown in figure 5. Oc. 4, Obj . 4, t. 1. 190.
Over the labium vestibulare it forms a thin nearly structureless cuticular layer which becomes thicker and shows lamellae over the labial teeth. In this region it is detached from some of the epithelial cells, a condition due to shrinkage.
In the region of the future sulcus spiralis and over the inner portion of the spiral organ the membrana tectoria appears composed of delicate parallel plates which have the appearance of hairs or fibers in section, and have so been interpreted by some investigators. These plates may often be traced between the cells or about their ends. They are separated by spaces which correspond frequently to the width of the cells at the surface of the epithelium. As one follows the plates away from the epithelium the spaces become smaller and the plates or lamellae approach each other until the membrana has the appearance of a solid structure with fine parallel striations; striations which, as we have seen, converge towards the inner angle of the cochlear duct. The relation of the plates or lamellae to the cells lining the future sulcus spiralis is shown in figure 5. Using an oil immersion objective the lines were seen as sharply as in a diagram, many passing between the cells and thus taking their origin as an inter-cellular secretion. The thicker lines undoubtedly represent two plates agglutinated.
Thus far we have shown, conclusively it seems to us, that the membrana tectoria takes its origin partly from cells which in the adult line the spiral sulcus and partly from the inner supporting cells of the spiral organ; and that the cuticular plates are not hke hairs or cilia in their development, as they may be traced between the cells. The next question is whether the outer cells of the spiral organ takes part in the formation of the membrana. Figure 6 shows the relation of the membrana tectoria to the cells of the spiral organ. At this stage the membrana is composed of a thin cuticular plate attached between the ends of the cells by what are apparently delicate threads. Internally (axially) the membrane is thicker and shows converging striae. Externally the membrana extends well beyond the cells of the spiral organ. The hairs of the two outer auditory cells were apparently attached to the outer surface wall of the membrana. Except in so far as they are enclosed therein the hairs have nothing to do with the development of the tectorial membrane.
Fig. 5. Portion of basal epithelium indicated by * in figure 4, showing the structural relation of the membrana tectoria to the columnar cells; m.tect., membrana tectoria; X, threadlike lamellae originating between two epithelial cells; col.ep., columnar cells of inner group; m.bas., basilar membrane. Oc. 2, 2 mm. Obj., t. 1. 160.
Fig. 6. Drawing showing the membrana tectoria developing over the hair cells of the spiral organ (marked h.c. in fig. 4); i.h.c, inner hair cell; o.h.c, outer hair cell; t.sp., spiral tunnel. Ob. 4, 2mm. Obj.
14 cm stage
In describing a 14 cm. stage Hardesty says: Up to this stage, the membrane never overlaps the lesser thickening and in confirmation of the statement of Rickenhacher it must be said that at no stage is there good reason to assume that the cells giving rise to the organ of Corti ever have anything to do with its development."^ Rickenbacher in his summary states that Die Cortische membran ist somit doppelten Ursprungs: Die innere Zone ist die primare welche von grossen Epithelialwulst abgeschieden wird. Die schmale Randzone ist eine sekundiire Bildung, welche an dem kleinen EpitheUalwulst abgesondert wird." As Rickenbacher states that the outer portion of the spiral organ is developed from the lesser 'Epithelial-wulst' and the inner portion from a part of the greater 'Epithelialwulst,' the above quotation is not in accord with Hardesty's statement. Rickenbacher's figures (11, 12, 13) show the membrana developing over the cells of the spiral organ and attached to the hairs, so also do the figures of Held ('09) and even in Hardesty's figure 10 the membrana is shown projecting beyond the outer pillar cells and attached to the inner cells of the spiral organ. On dissecting away the membrana at this stage it was found that the thin platelike zone, overlying the spiral organ, and extending beyond it was no artifact due to coagulation, but a definite structure of the same appearance and continuous with the rest of the membrane. The total width of this membrana was found to be equal approximately to that of the membrana in the same turn of a 18.5 cm. stage.
In our descriptions we have referred heretofore to the structures composing the membrana tectoria as 'lamellae or fibers.' (Italics mine) From the manner in which these are attached to the cells and from horizontal sections it will be seen that such terms can not rightly be applied to them. Sections of the membrana cut through the cochlea perpendicular to the modiolus or axis show their true significance. In such a section the organ of Corti (spiral organ) and the limbus are cut at right angles to the long axes of their cells, the line of section being indicated by x in figure 5. That portion of the membrana extending over the sulcus is seen cut perpendicular to the fibers or lamellae (fig. 7).
Its structure is that of a reticulum. The meshes are composed of delicate cuticular walls and at their angles are triangular or rectangular thickenings. The walls of the network are sharply defined and in unstained preparations appear highly refractive and clear. This structure can not be due to the effects of fixing reagents upon a gelatinous substance for in this case the lines of strain would not be as definite and would have a grayish, granular appearance instead of being clear and refractive as is the case with this cuticulum of the membrana tectoria. The spaces enclosed by the network correspond in form and size to cross sections of the epithelial cells and where "the membrana approaches the epithelium may be seen to correspond to the ends of the cells. The structure of the membrane is thus neither lamellar nor reticular but 'cellular' in the sense that honeycomb is cellular. The cuticular portion of the membrane corresponds to the waxen cells and these chambers are closed during development by the ends of the epithelial cells. There is this difference in the comparison that while the 'cells' of a honeycomb are nearly straight and of the same diameter throughout, the chambers in the membrana taper as we go from the epitheUum and curve toward the inner angle of the cochlear duct and are probably irregular in length and arrangement. My conception of the structure of the membrana based upon the preparations already described is shown diagrammatically in figure 8. The reticular structure is shown at the bases of the chambers, the thickenings at the angles of the meshes extend lengthwise of these chambers and when seen in side view, as in axial sections, they give the membrane the fibrous or striated appearance which has been so frequently described. This appearance was rightly interpreted by Coyne et Cannieu ('95). In a vertical section usually more than one layer of cuticular chambers may be seen and hence the striations appear numerous, indistinct and close together. Few investigators have made horizontal sections of the cochlea and in the adult and in late fetal stages such sections are difficult to obtain. Hardesty shows a section (fig. 9) in which at a cross sections of the 'fibers' are seen, and he has drawn a reticulum with thickenings at the angles. He states that the fibers seem to anastomose and appear to be connected with each other by fine collateral filaments but attributes this appearance to shrinkage and coagulation. On pages 161 to 162 he states that The membrana is not a lamellated structure. Ever since 1869 when Bottcher teased portions of it and found them to contain fibers the fibrous structure of the membrane has been conceded by all who have studied it with reference to its structure. Sections in different planes, as made by Coyne and Cannieu ('85) ['95] and here (Fig. 9) indicate clearly its fibrous structure." ^
It is certain that Shambaugh did not concede its fibrous structure, as he states that it is lamellated. On page 132 Hardesty states: "Lowenberg ('64) thought that the membrane consisted of layers one above the other; Gottstein ('72) pictured it as structureless, and many others after these have failed to comprehend its character." Rickenbacher does not figure any very definite structure nor does he account for its development. As to Coyne et Cannieu ('95), in describing the sections mentioned by Hardesty as confirming the fibrous character of the membrana, they say (p. 286) :
Cette membrane offre I'aspect d'un reseau, dont les travees serient constituees par une substance amorphe, claire et transparente. Ces travees circonscrivent des cavites polygonales diminuant d'epaisseur a mesure qu'on s'eloigne de I'organe de Corti pour se rapprocher de la protuberance de Huschke. Les cloisons de ces cavites se reunissent au niveau des angles du reseau et forment, en ce point, des espaississments sur toute la longueur de leurs bords de reunion. Ces espaississments sur des coupes radiales de la membrane se montrent sous I'aspect des stries dont nous avons dej^ parle.
- Italics mine.
Fig. 7. Membrana tectoria sectioned in a plane perpendicular to the axis of the cochlea, thus cutting across the 'fibers.' The drawing shows its reticular structure with thickenings at the angles of the meshes. From a fetus of 15 cm.; ep., epithelium. Oc. 2, 2 mm. Obj., t. 1. 160.
Fig. 8. Diagram showing structure of the membrana tectoria as proved by figures 4 to 7; r., reticulum, as seen in horizontal sections; I., 'lamellae' or 'fibers,' seen in axial sections.
Coyne et Cannieu thus are in agreement with my interpretation of the structure and on page 280 state definitely that the membrana is not composed of fibrils imbedded in a homogeneous matrix. Hardesty could not demonstrate by special stains the presence of a matrix which would hold the fibers together. His conclusions are based apparently on surface tiews of the membrana in which he saw an apparent fibrillar structure. The ends of the fibers w^hich one may see on the under side of the membrana may be interpreted also as the thickenings at the angles of the reticulum shown by Coyne et Cannieu and myself and as drawn by Hardesty himself in figure 9. It is improbable that this structure can be due to shrinkage and coagulation, for the walls of the meshes are sharply defined, clear and refractive, the size of the meshes corresponds to the size of the cells in transverse section and the network may be seen attached between cells of the spiral organ when studied in serial sections.
The accessory tectorial membrane which Hardesty describes as composed of two sets of obliquely crossing fibers he figures as a reticulum with 'diamond' shaped meshes. Its probable structure is that of a reticulum and it may be explained as a thin layer of the membrana tectoria which was left adherent to the spiral organ and later was torn away. It probably represents the reticular membrane or lamina reticularis of the spiral organ which Coyne et Cannieu interpret as a portion of the membrana tectoria which has remained attached to the cells of the spiral organ. Horizontal sections also explain why the membrana, or portions of it, have been described by some as ha\'ing a reticular structure. Held ('09) figures the membrana as arising from the ends of the supporting cells of the basal epithelium in the form of parallel fibers. Yet he does not show these fibers as continuous with the cytoplasm of the cells, like the hairs of the auditory cells, nor did he study horizontal sections through the membrana.
To sum up the development of the membrana previous to fetuses of 15 cm., we may say that it is a cuticular organ with a definite though irregularly chambered structure which is secreted between, and at the ends of the cells composing the basal epithelium of the cochlea. Both the greater and lesser epithelial thickenings take part in its development, its outer zone arising between the cells of the spiral organ. It appears first near the inner angle of the cochlea over the labium vestibulare but growth in thickness here soon ceases. Next it develops rapidly over the cells which later line the spiral sulcus and form the inner supporting cells of the spiral organ. Finally, in later stages {yet to be described), it grows rapidly over the spiral organ. From a study of my preparations it ivas not possible to demonstrate distinct fibers imbedded in a matrix nor are there grounds for believing that hairs or cilia take part in its developynent.
18.5 cm stage
The later stages in the development of the cochlea show the further growth of the membrana over the spiral organ, its attachment to the latter, and the metamorphosis of the high columnar cells of the inner cell group to form the lining of the spiral sulcus. We have seen in earlier stages that differentiation of the cochlear duct is much more advanced in the basal coil than in the apical. This difference is very marked in a fetus of 18.5 cm. In figure 14 the microphotograph shows sections of three turns on each side. The scalae are both large in the basal turn but in the upper turns the scala tympani is still small. The coagulated endolymph more or less completely fills the scala vestibuli. It will be seen when compared with the 13 cm. stage that the membrana has continued to grow rapidly over the spiral organ in the two upper turns but its growth has ceased and it has remained small in the basal turn. Three stages in the development of the spiral sulcus and organ are seen. In the upper turn (fig. 14, 3 and fig. 9) the epithelial cells just external to the teeth of the labium vestibulare have become lower, free from the membrana and tend to form a simple epithelium. The space left between the membrana and the shortening cells is the spiral sulcus.
Fig. 9. Section of 3d (upper) spiral of cochlear duct of the 18.5 cm. stage; i.h.c, inner hair cells; o.h.c, outer hair cells; lab.vest., labium vestibulare; limb.sp., spiral limbus; m.bas., membrana basilaris; m.tect., membrana tectoria; m.vest., membrana vestibularis; n.coch., cochlear nerve; sc.tymp., scala tympani; t.sp., spiral tunnel. Oc. 4, Obj. 4, t. 1. 190.
The cells remaining between the spiral sulcus and the pillars of Corti still form a very high pseudostratified epithelium. In the middle turn (fig. 14, 2) the cells lining the spiral sulcus are of the low columnar type with one or two rows of nuclei while the cells internal to the pillars are but little higher than the pillars themselves. Finally, in the lower turn {1) the cells lining the spiral sulcus are of the cubical type, in a single layer, and the remaining columnar cells persist as the internal supporting cells of the spiral organ. It seems probable that large numbers of the cells of the greater epithelial thickening degenerate, liquefy and disappear; those remaining flatten out and form the simple epithelium of the spiral sulcus.
In the apical turn, by comparing with figure 4 it will be seen that the membrana is but little thicker over the labium vestibulare, is much thicker over the inner cell group and thickest over the spiral organ where, in the 13 cm, stage, it was just beginning to develop. It extends far beyond the outer supporting cells. In the second turn the membrana is not so thick over the spiral organ but still extends beyond the outer hair cells. In the basal turn the membrana is thickest over the spiral sulcus and extends as far as the outer hair cells. In the two upper turns the membrana seems to be firmly attached to the cells of the greater epithelial thickening and to those of the spiral organ except along its outer zone where it show^s sighs of having shrunken and pulled away. In the basal turn the outer half was free but this also showed the effects of shrinkage and distortion.
Hardesty has suggested that there is a displacement of the spiral organ when the spiral sulcus is developed, thus accounting for the position of the membrana over the spiral organ in the later stages of its development.
There are a number of facts which make this hypothesis untenable: (1) Sections and dissections of the 13 to 15 cm. stages show that the membrana is developed over the cells of the spiral organ. (2) In the 18.5 cm. stage the membrana projects further beyond the spiral organ in the apical turn where the differentiation of the spiral sulcus has only just begun, and least in the lower turn where the spiral sulcus is fully developed. (3) The distance of the pillar cells from the inner angle of the cochlea (the only definite points which may be taken for comparative measurements) is about the same in the 13 cm. and 18.5 cm. stages. (4) The total width of the thickened portion of the membrana is about one-fourth greater in the 18.5 cm. stage than in the 13 cm. showing that growth has taken place along its outer border. This growth must have been supplied by the cells of the spiral organ. (5) To show that displacement takes place Hardesty measured the floor of the spiral sulcus and compared with the width of the inner cell group. There are no definite points which may be taken for measuring the floor of the spiral sulcus, and in measuring the width of the inner cell group one is including cells which form part of the spiral organ. No accurate comparison can thus be made. The distance between the inner angle of the cochlea and the pillar cells, two definite points, may be measured with considerable accuracy and shows no important change in the position of the spiral organ from the 13 cm. to the 18.5 cm. stage, nor later in the new-born animal. (6) As the basal membrane does not shorten, the displacement theory must assume that dead passive structures like the pillars actively move inward over the surface of the basal membrane.
One argument which Hardesty uses to prove that inward. displacement of the spiral organ has occurred is that the 'fibers' of the membrana when traced from' its upper and outer border curve outward, downward, and then inward as though they had been pulled inward by the migration of the spiral organ. This inward curvature of the fibers is only found in the upper turns of the cochlea where the membrana is of greatest width and thickness. It may easily be accounted for. In the stages up to 16 cm. the cells of the spiral organ slant outward but as the width of the basal membrane is rapidly increasing the inclination of the chambers, hence of the 'fibers' is downward and outward. When the membrane begins to develop actively over the spiral organ the basal epithelium has attained its maximum width but as the cells are directed outward the inclination of the chambers will now be inward. When the spiral sulcus is developed by the degeneration of its cells, the outer cells of the spiral organ elongate and straighten somewhat so that they are no longer directed outward. This shifting, which is relatively slight and not enough to account for the displacement of the membrana, would nevertheless increase the inward tr^nd of its chambers. My observations are supported by those of Held ('09) on the guinea-pig and rabbit.
In taking measurements of the 18.5 cm. stage the marked changes found are : (1) The increase in thickness of the membrana tectoria; (2) The increased distance from the inner angle of the cochlea to the labial teeth. The outer cells of the labium have grown rapidl}^ outward beneatli the membrana thus pushing the ends of its chambers outward. The result is that in this region the chambers come to He parallel to the surface of the labium and give the membrana a lamellated appearance which is especially marked in the lower turns of the cochlea. The membrana may be divided into zones at this stage: (1) A thin structureless zone over the inner portion of the labium vestibulare; (2) A thicker second zone of flattened horizontal chambers over the outer portion of the labium vestibulare ; (3) A still thicker third zone of chambers curving downward and outward unattached over the spiral sulcus; (4) An outer zone, thickest in the upper turns with chambers trending downward, outward then inward, largely attached to the cells of the spiral organ and probably normally wholly thus attached.
The sections of the 18.5 cm. stage thus show that the membrana tectoria has developed rapidly over the spiral organ especially in the upper turns of the cochlea; that the membrana is attached to the cells of the spiral organ in the upper coils and shows shrinkage and distortion in the lower; that the inner cells of the greater epithelial thickening degenerate or persist as the lining of the spiral sulcus while the outer cells of this group form the inner supporting cells of the spiral organ. Finally there is no evidence of an inward shifting of the spiral organ sufficient to account for the position of the membrana at this stage assuming (which we do not) that it is not developed from the spiral organ and that there is a necessity for such a displacement.
The development of the structures arising from the basal epithelium of the cochlea is practically complete at 18.5 cm., but a number of cochleae were studied from the full-term fetus. The structure of the membrana at this stage has been figured by Shambaugh ('07) and Hardesty ('08) both of whom found attachments between the cells of the spiral organ and the membrana. These attachments are regarded as normal by many investigators, as due to coagulation and shrinkage by others. There is shown in figure 10 one of the many cases which occurred in in}preparations showing attachment to the outer supporting cells.
The membrana is undeniably shrunken and partly pulled away from the spiral organ, but the hairs of the outer auditory cells are firmly imbedded in the membrana. The under surface of the membrana shows a thickening, st.H., which according to Shambaugh ('08) corresponds to Hensen's stripe and represents the inner line of its attachment to the inner supporting cells. In other sections the membrana was firmly attached to the inner supporting cells as well as to the hairs. In all of my preparations at full-term the membrana was badly shrunken. Frequently also the organ of Corti was distorted, being pulled inward by the attached membrana.
Fig. 10. Section through the apical turn of the cochlea at about full term, showing outer auditory hairs imbedded in the membrana tectoria; ep.s.sp., epithelium of spiral sulcus; i.h.c, inner hair cells; i.^il., inner pillar; m.has., basal membrane; m.tect., membrana tectoria; lab.vest., labium vestibulare; limb.sp., limbus spiralis; n.coch., cochlear nerve; o.h.c, outer hair cell; sc.tymp., scala tympani; s.sp., sulcus spiralis. Oc. 4, Obj. 4, t. 1. 190.
The attachment of the membrana to the spiral organ I regard as normal for the following reasons:
- In development the membrana is normally so attached.
- The auditory hairs when attached could be traced into the membrana, even though the latter was badly shrunken.
- The shrinking membrane frequently exerts such a pull upon the spiral organ as to distort it.
- Physiologically and anatomically it is the condition we should logically expect if the menibrana is functional in transmitting sound waves to the auditory hairs.
- Were the membrana merely floating in contact with the hairs and unattached to them or their supporting cells it would not retain its position constantly and would thus interfere with the auditory function.
In describing the structures of the cochlea the apex is regarded as above, the base as below and the membrana tectoria as lying over the spiral organ. As a matter of fact when in its normal position the apex of the cochlea is directed cephalad and ventrad. This may be well seen in a dissection of the brain and cochlea of the pig (fig. 11).
Fig. 11. Dissection of the head of a pig fetus to show the position of the brain and cochlea. X 4.
When the pig's snout is directed downward, as in feeding, the base of the cochlea would be above, the apex below. The same would be true of the human cochlea when the head is bent forward. The membrana tectoria would then he beneath the spiral organ and as it is slightly heavier than the endolymph and very flexible it would naturally sink downward and away from the spiral organ assuming that it was not attached to the cells of the latter. This would be all the more apt to occur when the membrana is subjected to the heavy jars incident to active movements, running and jumping, and should interfere with hearing. As we know, such interference does not occur.
The arguments raised by Hardesty against any normal attachment of the membrana, save to the labium vestibulare, are :
- In dissecting the fresh membranous labyrinth to expose the membrana its outer portion could be seen floating free along its entire extent.
- In the majority of sections it is entirely free from the spiral organ and when attached such attachments are filamentous and may be explained as abrasions of the under surface or coagulations of precipitated albumins.
- From the process of its development it seems probable that the membrane is free from the underlying structures, and as its outer zone acquires its position over the spiral organ by displacement, one must assume that any attachment which exists between the membrana and the spiral organ must have developed secondarily.
Hardesty's first argument bears little weight because in describing his method of studying the fresh membrana tectoria he states that it was necessary to crush the bony labyrinth with a hammer and that the disturbances caused by his dissection caused the membrana to float free from the labium vestibulare an attachment which is never entirely ruptured in carefully fixed sections. A method which would destroy the strong attachment to the labium would certainly set free the more delicate attachments to the spiral organ.
Moreover, dissections which were made by using more favorable methods did not seem to support Hardesty's observations. As to the attachments seen in sections being artifacts it is sufficient to say that I have traced the hairs into the membrana in many cases and that attachments to the inner supporting cells and to the outer hair cells are so strong as to distort the spiral organ during the shrinkage of the membrana. The very fact that the membrana shrinks shows that its normal position has been disturbed. We may as logically assume that it was attached and in many cases has shrunken away as to assume that it floated just parallel to the surface of the spiral organ and has become pressed down upon and attached to it by coagulations.
We may assume this even more logically for we hold, and our preparations and dissections and the observations of Held {'09) prove absolutely that the membrana is attached to the epithelial structures of the spiral organ in late fetal stages. There is no necessity for, and my preparations afford no proof of, an inward shifting of the spiral organ and a consequent displacement of the membrana. It is therefore unnecessary to assume with Hardesty and Von Ebner ('02) that any attachment between membrana and spiral organ must be of secondary development.
While these arguments against the existence of an attachment between the membrana and spiral organ may be readily answered it is none the less true that a complete attachment to the cells of the spiral organ, such as exists in the fetus, has never been demonstrated in the adult organ. Nor, to my knowledge, has it been explained why the membrana should detach itself so readily from the spiral organ yet always retain its attachment to the labium vestibulare. First, as to the reason the complete attachment may be demonstrated in the early fetus and not in the adult: This is probably because the attachment is more firm in the fetus and because the basal epithelium and the basal membrane are less rigid in the fetus and tend to shrink pari passu with the membrana. In the adult or even the new-born young the tissues are less watery, more rigid and more resistant to reagents. The basilar membrane is attached to the bony labyrinth, now strongly ossified. The membrana alone shrinks to any great extent and as a result is more or less completely torn away.
Why the membrana should always lose its connection with the spiral organ and not its attachment to the labium vestibulare is explained by its structure. Over the labium it is an almost solid cuticular structure and the few chambers in this region are flattened and contain little fluid. Over the spiral sulcus the membraiia is composed of chambers, filled with fluid and open at their lower ends, while over the spiral organ these ends are assumed to be closed by the ends of the epithelial cells. The action of most fixing reagents and alcohol is to take water from the membrana. This would cause the open chambers to shrink, narrow, and so suddenly diminish the width and length of the membrana.- As the membrana has the form of a spiral the shrinkage of the outer portion of the membrana throughout its whole length would tend to draw it toward the labium and away from the spiral organ, as it would diminish the diameter of the spiral. The effect would be most marked in the larger basal turns and it is there that the membrana is almost invariably torn away from the spiral organ even in late fetal stages. This alone would account for the detachment of the membrana from the spiral organ in most fixed preparations. Over the spiral organ, assuming that the membrana is attached, the chambers would be closed by the ends of the epithelial cells. Upon the action of fixing reagents or alcohol, the withdrawal of water must take place chiefly about the ends of the chambers as their cuticular walls are not permeable. The result would be the shrinkage of the chambers and their separation from the cells. Even after the membrana is freed from its attachments Hardesty has shown that it shrinks very badly during the process of dehydration and clearing, and I have noted the same. The shrinkage of the membrana may also be aided by.normal tension in pulling the membrane away from the spiral organ. It is very possible that such tension exists especially in the lower turns of the cochlea, as held by Kishi ('07).
The Function of the Membrana Tectoria
It is not my intention here to go into a detailed account of the physiology of audition but simply to emphasize certain anatomical facts which have a bearing upon the transmission of the sound waves. Recent investigators all agree that the hair cells form the perceptive end organ of the cochlea and that the tectorial membrane is probably the medium through which the sound waves are transmitted to the hairs of the auditory cells. Arguments against the old theory which regarded the basilar membrane as a resonator are many :
- The structure of the basilar membrane, clothed as it is by several layers of cells, precludes its responding to delicate stimuli (VonEbner'02).
- In the basal coil it is thick and rigid or may be replaced by a plate of bone though in this region the spiral organ is normally developed (Shambaugh '07).
- Hardesty has shown that the basilar membrane is merely a flattened tendon, the fiber bundles of which are closely bound together and thus could not vibrate separately.
- The pillars are also rigidly united, and it is probable that the functions of the basilar membrane and of the pillars in conjunction with the lamina reticularis is to give rigiditj^ to the auditory cells in order that their hairs may respond more readily to sound vibrations.
- The inner pillars do not always rest upon the basilar membrane but upon the edge of the labium tympanicum (Shambaugh, Hardesty).
- Sound waves entering the perilymph would affect the basilar membrane more strongly from the side of the scala tympani yet to do this would have to pass up and down the entire length of the spiral. The amplitude of the vibrations would be lessened by this and th^re' would be interference between the weaves going up in the scala vestibuli and the waves descending in the scala tympani.
The objections raised against the basilar membrane do not apply to membrana, and there are many points in its favor:
- The membrana is an exceeding delicate, chambered cuticular membrane, flexible yet elastic and of a specific gravity only slightly greater than that of the endolymph.
- It is co-extensive with the spiral organ while the basilar membrane is not.
- It lies on that side of the spiral organ at which sound waves would first enter the cochlea b}^ way of the scala vestibuli.
- It is attached along its inner edge to the labium vestibulare, stretches over the spiral sulcus and overlies the spiral organ in contact with and probably attached to its cells.
- It is narrow and thin in the basal coil and becomes wider and thicker as it approaches the apex. Measurements of the functional zone of the membrana taken from its outer border to the labial teeth, show that the sectional area of the membrana in the apical turn is from thirty to forty times that in the basal turn. Owing to the shrinkage of the membrane such measurements can be only approximate. Figure 12 shows the relative size of the membrana as seen in sections of the first, second, third and fourth turns.
Fig. 12. Sections through the membrana tectoria of a full term fetus showing its relative size in the four turns.
From Hardesty's measurements of fresh and fixed preparations he found that the greatest width (first or apical turn) was about five times that of the minimal width (tip of basal turn) while the thickness in these same turns was as 6 : 1.
The membrana tectoria is thus to be regarded as a spiral cuticular band of dehcate chambered structure, which becomes gradually thicker and wider from base to apex. This band is attached by its flattened inner edge to the labium vestibulare, spans the spiral sulcus and its outer portion is so attached between the cells of the spiral organ that the ends of the cells close the chambers and the auditory hairs project into them. It may be that the membrana is stretched with some tension between the labium and the spiral organ, the tension being greater at the base of the spiral. Whether a membrane of such structure and attachments may act as a whole or as a resonator, must be left to physicists to decide. There seems to be little doubt, however, that the membrana tectoria is the structure through which sound waves are transmitted to the auditory cells, and that it is in every way better adapted to this function than the basilar membrane. As it is thin, narrow and perhaps under tension in the basal turns and it has been shown that notes of high pitch are perceived here, it is probable that the membrana of the basal turns responds only to the sound waves of greatest frequency. While the apical turn, which receives notes of low pitch would respond only to waves of low frequency. It does not seem possible that any cuticular chamber could alone respond sympathetically to a given note but rather that a portion of the membrana, of nearly the same breadth and thickness, vibrates as a whole.
Summary
- The membrana arises as a thin cuticular plate which is first developed over the free ends of the columnar cells which form the greater (inner) epithelial thickening of the basal cochlear wall.
- As it is present in fetuses of 5 cm., before the development of the hair cells in the spiral organ, it cannot be regarded as developed from these hairs.
- The greater epithelial thickening gives rise to the epithelium of the labium vestibulare, to the lining of the spiral sulcus and to the inner half of the spiral organ (inner supporting cells, and probably to the inner hair cells and inner pillars). The lesser epithelial thickening forms the external portion of the spiral organ.
- The membrana grows in thickness by the secretion of a cuticulum formed between the ends of the epithelial cells, rapidly at first over the cells of the greater epithelial thickening (5 to 13 cm. stages), later over the cells of the lesser epithehal thickening.
- In sections through the axis of the cochlea the membrana has a striated or lamellated appearance. The striae curve outward and downward from the labium vestibulare where the membrane remains thin. In sections perpendicular to the lamellae the structures of the membrana is that of a reticulum with thickenings at the angles of the meshes. It is therefore neither lamellar nor reticular but a chambered structure or 'honeycomb' of hollow tapering cuticular tubes or chambers normally filled with a fluid resembling the endolymph. The bases of chambersduring development rest between the ends of the epithelial cells.
- The thickenings at the angles of the meshes of the reticulum extend lengthwise along the whole extent of the tubes or chambers and in sections through the axis give the membrane its striated appearance, the striae having been variously interpreted as hairs, cilia, fibers and lamellae.
- As the basal epithelium increases its width its cells are carried outward, away from the modiolus. This carries the bases of the growing cuticular chambers outward also, though their tips remain stationary. The result is the inward inclination of the chambers as they are followed from base to tip.
- The chambered structure of the membrana explains the 'border-plexus' of Lowenberg, the accessory tectorial membrane observed by Hardesty, and the 'reticular structure' of the membrana described by various investigators.
- In fetuses of 18.5 cm., the membrana in the upper turns of the cochlea projects outward beyond the spiral organ and is firmly attached to the cells of both the spiral organ and of the greater epithelial thickening. In this turn the spiral sulcus has not yet fully formed and the distance from the inner angle of the cochlea to the pillars is fully as great as in the preceding stage. Thus the position of the membrana cannot be ascribed to an inward shifting of the spiral organ, but is due to its rapid development from the cells of the spiral organ.
- The attachment of the membrana to the spiral organ was proved not only by sections but by dissections of both fresh and fixed cochleae.
- Between stages of 15 and 25 cm., the inner cells of the greater epithelial thickening change from a high pseudostratified columnar type to that of a simple cubical epithelium. These cells lose their attachments to the membrana and the space which as high columnar cells they occupied, becomes the spiral sulcus. The change is brought about by the degeneration of many of the cells and the transformation of those remaining.
- In sections of the cochlea at full term the membrana was found attached to the inner supporting cells of the spiral organ and to the outer hair cells as well as to the labium vestibulare. This attachment is regarded as normal because it was indicated by dissection of fresh cochleae; because in development it is so attached; because the attached membrane when shrinking under the action of reagents exerts such a pull upon the spiral organ as to distort it. Lastly, because physiologically and anatomically it is the condition which we should expect to find if the membrana is functional in transmitting sound waves to the auditory hairs.
- Although usually described as lying above, the normal position of the membrana may be directly beneath the spiral organ. As it is slightly heavier than the endolymph if unattached it would float free especially when actively moved or jarred. This would interfere seriously with the function of the organ.
- Reasons why the membrana detaches itself from the spiral organ more readily than from the labium vestibulare are as follows : (a) The outer portion of the membrana being chambered shrinks much more than the inner zone which is a solid cuticular plate; (b) Shrinkage of the outer zone affects not only the width but the length of the membrana; (c) Being a spiral structure, the decrease in length decreases the diameter of the turns thus drawing the membrana inward. This would tend to separate it from its outer attachment to the cells of the spiral organ.
- The arguments against regarding the basilar membrane as a medium for transmitting sound waves to the hair cells, do not hold for the membrana tectoria.
- The membrana tectoria is a delicate chambered cuticular structure, co-extensive with the spiral organ. It is attached by its inner zone to the labium vestibulare by its outer zone between the cells of the spiral organ thus bridging over the spiral sulcus. Its sectional area at base and at apex is as 1 : 40 approximately. As the hairs of the auditory cells project directly into the chambers of the membrana, vibrations of the membrana Would be directly transmitted to them.
- As the membrana is much thinner and narrower in the basal turns than in the apical region it is probable that different portions of it respond to sounds of different pitch. In this sense it may act as a resonator.
Bibliography
Ayeks, H. 1891 Die membrana tectoria - was sie ist, und die membrana basilaris — was sie verrichtet. Anat. Anz., Bd. 6.
1898 On the membrana basilaris, the membrana tectoria and the nerve endings in the human ear. Zool. Bull., vol. 1, no. 6.
BoTTCHER, A. 1870 Ueber Entwicklungsgeschichte und Bau des Gehorlabyrinthes. Verhandl. der Kaiserl. Leop. Karol-deutschen Akad. der Naturforseher, Bd. 35.
Corti, A. 1851 Reserches sur I'organe de I'ouie des mammiferes. Zeitschr f. wiss Zoologie, Bd. 3.
CoYXE, P., ET Cannieu, A. 1895 Contribution a I'etude de la membran de Corti. Jour, de'l'anatomie et de la physiol., Ann. 31.
CziNNER, H. I. UND Hammerschlag, V. 1898 Beitrag zur Entwicklungsgeschichte der Corti'schen membran. Archiv f. Ohrenheilk., Bd. 44.
Von Ebner, V. 1902 In KoUiker's Handbuch der Gewebelehre des Menschen, Bd. 3, 2d Halfte.
GoTTSTEiN, J. 1872 Ueber den feineren Bau und die Entwicklung der Gehorschnecke der Menschen und der Saiiger. Archiv f. mikr. Anat., Bd. 8.
Hardesty, I. 1908 The nature of the tectorial membrane and its probable role in the anatomy of hearing. Amer. Jour. Anat., vol. 8.
Held, H. 1909 Der feinerer Bau der Ohrlabyrinthes der Wirbelthiere. II. Abhandl. d. Sachs. Ges. d. Wiss. Math.-Phys. Kl. Bd. 31, pp. 191-293, 18 Taf.
Vox Helmholtz, H. L. T. 1896 Die Lohre von den Tonempfindungen. Ausgabe 5, Braunschweig.
KisHi, K. 1907 Cortische Membran und Tonempfindungstheorie. Pfliiger's Archiv, Bd. 116.
Kolliker, a. 1801 Entwicklungsgeschichte des Menschen und der hoheren Tiere, Leipsig.
LowENBERG, B. 1864 Beitrage zur Anatomie der Schnecke. Archiv f. Ohronheilk., Bd. 1.
Retzius, G. 1884 Das Gehororgan der Wirbeltiere. Biologische Untersuchungen., Bd. 1, Stockholm.
RiCKENBACHER, O. 1901 Untersuchungen liber die cmbryonale JXIembrana tectoria der Meerschweinchen. Anat. Hefte, Abth. 1, Bd. 16.
ScHWALBE, G. 1887 Lehrbuch der Anatomie der Sinnesorgane.
Shambaugh, G. E. 1907 A restudy of the minute anatomy of the structures in the cochlea, etc. Amer. Jour. Anat., vol. 7, no. 2.
Plate 1
Explanation of Figures
Fig. 13 Microphotograph of an axial section through the cochlea of a 7.5 cm. fetus. The numerals 1, 2, 3, indicate the turns of the spiral corresponding with those similarly numbered in figure 14. X 20.
Fig. 14 Microphotograph of a section through the modiolus of a cochlea from an 18.5 cm. fetus. Turns of cochlea numbered on right as in figure 13. X 20.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - On the development of the membrana tectoria with reference to its structure and attachments. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_On_the_development_of_the_membrana_tectoria_with_reference_to_its_structure_and_attachments
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G