Paper - Differentiation of pituicytes in the human foetus
| Embryology - 28 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Shanklin WM. Differentiation of pituicytes in the human foetus. (1940) J Anat. 74(4): 459-63. PMID 17104829
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Differentiation of Pituicytes in the Human Foetus
By William M. Shanklin
Departments of Histology, American University of Beirut and the University of Tennessee Medical Schools
Introduction
By using silver impregnation stains Retzius (1894), Berkley (1894.-), Herring (1908), and Tello (1912) showed that the parenchymatous cells of the pars nervosa of the pituitary were neuroglial in nature. More recently, Bucy (1930) published a detailed account of these cells in ox material and called them pituicytes. Gersh (1939) described the granules in these cells in the embryonic and adult material of a number of animals.
This study is based on sections of seventeen pituitaries of human foetuses ranging in age from 3 months to the new-born. Shortly after abortion each foetus was placed in refrigeration until the time of autopsy. Some of these remained in the refrigerator as long as 60 hr. without making any apparent difference in the staining of the pituicytes.
Fixation was effected by the Cajal formalin-ammonium-bromide method as described by Penfield & Cone (1937). The time of fixation ranged from 2 days to 3 weeks at room temperature, after which the tissue was washed and sectioned on the freezing microtome. The Hortega silver carbonate methods were employed, either for oligodendroglia or for microglia, as described by Penfield & Cone. The writer, like Bucy, found the silver impregnation methods that stained astrocytes were ineffective as pituicyte stains.
Three-month foetus
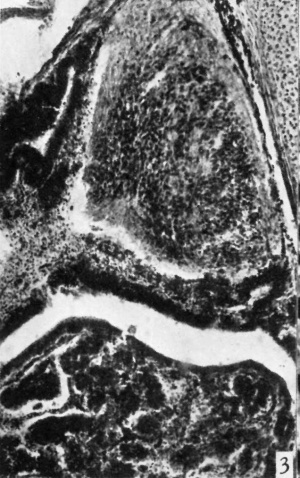
The ages of the two youngest foetuses studied were estimated by the internist at 2 months, but measurements suggest the age was nearer to 3 months. Judging by the general appearance of these pituitaries they are comparable to those shown by Tilney (1936) in his fig. 19 representing the 55 mm. human embryo. In these 3-month embryos the pars nervosa is triangular in shape with the base against the pars intermedia (Pl. I, fig. 3). The outer zone of the nervosa contains many fibres and practically no cells. The nature and origin of the fibres were not determined, but they are not processes of the pituicytes.
The central area of the pars nervosa at 3 months is filled with densely packed nuclei having very little cytoplasm. They vary in shape from round to oval and are directed along the long axis of the section. The round nuclei vary in size from 6 to 10 p., and the more elongated oval ones from 5 p. wide and 11 p. long, up to others 8 p. wide and 14 p. long. The nuclear membrane is thick and clearly seen. The darkly stained chromatin is present in granular form, there being in each nucleus from twenty to forty of these granules suspended in a distinct linin network. Nucleoli were not identified.
Four-month foetus
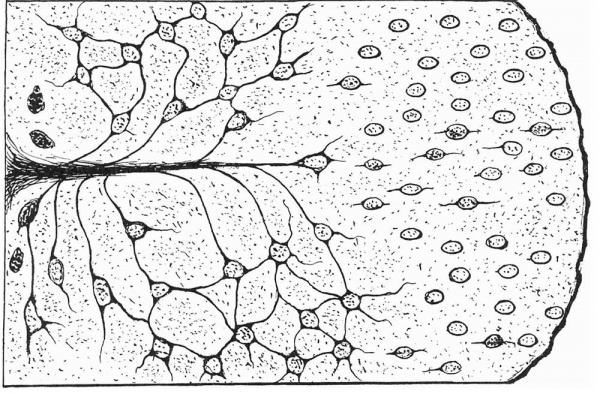
During the fourth month those cells in the pars nervosa nearest to the pars intermedia undergo a series of changes whereby more cytoplasm accumulates around the nuclei, and processes make their appearance. By the middle of the fifth month the pituicytes in that part of the pars nervosa proximal to the pars intermedia are well developed, whereas those pituicytes in the distal half are still in the earliest stages of differentiation (Text-fig. 1; Pl. I, fig. 4; Pl. II).
Five-and-a-half-month foetus
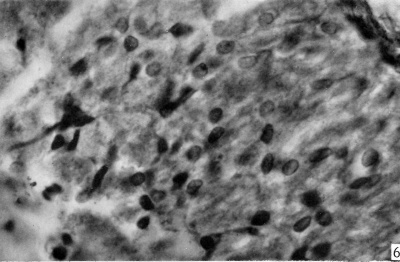
The following is a description based on sections of a 5;-month foetus. The part distal to the pars intermedia will be described first. The most striking thing observed is the similarity of the pituicytes in this portion to those observed in the 3-month foetus. The two most conspicuous elements are numerous large nuclei and abundant intercellular granules (Pl. II, fig. 6). Although the nuclei are similar to those described for the 3-month stage, many more are now oval to fusiform in shape. In general, the round nuclei are more lightly stained and the nuclear contents are clearly seen, whereas the oval nuclei are stained more darkly and the nuclear contents are mostly obscured. The more darkly stained nuclei have more abundant cytoplasm, and frequently one, or at times two, processes extending from their ends. These processes are nearly always directed towards the pars intermedia or towards the connective tissue capsule covering the outside of the pars nervosa. At this stage these rudimentary processes are unbranched. Although abundant small reddish brown intercellular granules are seen in this portion, intracellular granules are not present.
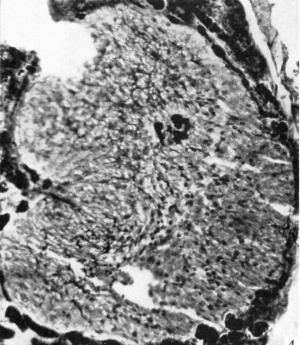
The photomicrographs of the area proximal to the pars intermedia (Pl. I, fig. 4; Pl. II, fig. 7) show the pituicytes and their processes arranged in a whorl-like formation around a large intruding trabecula of connective tissue. The pituicytes of this area are clearly stained and all are well differentiated. In these cells the nuclear membrane is distinct and more lightly stained than the deeply stained cytoplasm. The nuclei are roughly oval and the cells are stellate in shape. Each cell has from three to six processes. Those pituicytes nearest to the trabecula, or to the walls of blood-vessels, have one of their processes attached to these structures. In some cases the attached process is short and thick, in others it is long and thin. In the older stages these processes are greatly elongated. The remaining processes anastomose with those of the neighbouring pituicytes and form an intricate reticulum (Text-fig. 1; Pl. I, fig. 4; Pl. II, fig. 7).
The spaces between the reticular meshwork are filled with small, closely packed granules. Although about the same size as the granules in the distal undifferentiated part of the pars nervosa, these granules, like the nearby pituicytes, stain a distinct purple, instead of reddish brown like those in the distal portion. The relatively few granules present in the cell bodies and processes of the differentiated pituicytes at this stage increase in size and number in the older stages.
Text-fig. 1. Drawing of pituicytes of the 5.5 month human foetus, representing the poorly differentiated and the well differentiated pituicytes to each side of a large connective tissue trabecula.
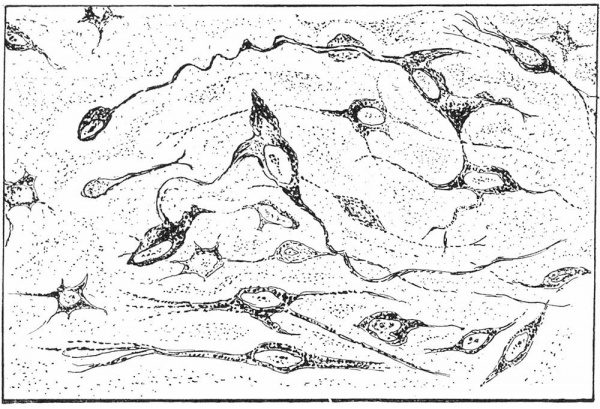
Text-fig. 2. Drawing of pituicytes of a 7.5 month human, premature new-born that lived for 2 months. Pituicytes show anastomosis between processes, granules along one of the processes, also a bulb-like ending on one of the terminals.
The following description is based chiefly on the pituitary of the new-born with a discussion of the intermediate stages. From the fifth month until the time of birth the proximal area of differentiated pituicytes increases in size at the expense of the distal portion; however, even at the time of birth the pituicytes in about one-fifth of the pars nervosa are still in the relatively undifferentiated state, like those in the distal part of the 5-month foetus.
Seven-month foetus
In the 7-month foetus many more blood-vessels and connective tissue trabeculae are present in the differentiated proximal part of the pars nervosa; furthermore, there is a corresponding increase in the number of pituicytes that have processes attached to the walls of blood-vessels and to the trabeculae. The remaining pituicyte processes still anastomose with one another. The entire field contains more fibres due to the marked increase in length of the pituicyte processes. Intracellular granules at this stage are now much more abundant and are found in the cell body and in many of the processes.
New-born foetus
In the proximal part of the new-born pars nervosa, the trabeculae of connective tissue are now abundant, but there does not appear to be a corresponding increase in the vascularity. It should be noted that there is no similar increase of the connective tissue elements in the nearby poorly differentiated distal area.
The pituicytes are now considerably larger in size, as are also their nuclei, the cytoplasm, and the intracellular granules. Although many of the pituicyte processes continue to form a rich anastomosis, the processes of others are detached and their cell bodies are no longer stellate-shaped but fusiform. These free processes frequently extend beyond the microscopic field when using the oil-immersion lens. Due to the intensity of the stain very little detail can be observed concerning the finer structure of the processes. Some of them however spread out fan-wise at their free ends, giving the appearance of a brush (Pl. I, fig. 5). In this brush formation the terminal processes of the pituicytes are distinctly fibrillar, with abundant small granules which appear to be attached to the fibrils. Other processes have granules massed about them such as those shown in the photomicrograph (Pl. I, fig. 5) and the drawing (Text-fig. 2). These granules, although clumped in a definite mass, appear to be inside the cell membrane. Other pituicytes are distinctly granular but do not have fibrils.
Some of the pituicyte processes terminate in a peculiar bulb-like ending. The outer part of the bulb is darkly stained, and in the centre there is a small dark body surrounded by clear structureless material. Such a bulb is shown in the photomicrograph (Pl. I, fig. 5) and the drawing (Text-fig. 2). These bulblike endings may be remnants of pituicyte cell bodies.
At this stage intercellular granules are still present in great profusion, but they are distinctly smaller and more lightly stained than the intracellular granules.
No basophil cells have invaded the pars nervosa from the pars intermedia in any of this foetal material, although many such cells are clearly seen in the adult pituitaries stained by the Hortega silver carbonate method.
Summary
The pituitaries of seventeen human foetuses ranging in age from 3 months to the new-born were studied after staining by the Hortega silver carbonate method.
At 3 months the pituicytes are represented by nuclei that have very little cytoplasm and practically no processes. Following the third month they form three or more processes, some of which become attached to the connective tissue trabeculae or to the walls of blood-vessels, but the majority of the processes join those of nearby pitucytes and form a reticulum.
The most striking observation is the fact that the pituicytes do not all differentiate at the same time. Those near the pars intermedia differentiate first and differentiation proceeds away from this area. At the age of 5% months all of the pituicytes in the area proximal to the pars intermedia are well developed, but those in the distal part, approximately one-half of the pars nervosa, are similar to those of the 3-month foetus. Even at the time of birth, in about one-fifth of the pars nervosa the pituicytes are still in the primitive state.
The writer is greatly indebted to members of the staff of the Maternity Ward of John Gaston Hospital, Memphis, and of the Pathological Institute, for their co-operation in securing the human material; to Miss Mattie Tharp of the Department of Histology for technical assistance, and to Dr Joseph L. Scianni, Artist of the Pathological Institute for help with the illustrations. All of the above are members of the University of Tennessee Medical School Staff.
References
BERKLEY, H. J. (1894). Johns Hoplc. Hosp. Rep. 4-, 285.
BUCY, P. C. (1930). J. comp. Neurol. 50, 505.
GERSH, I. (1939). Amer. J. Anal. 64-, 407.
HERRING, P. T. (1908). Quart. J. exp. Physiol. 1, 121.
PENfiELD, W. & CONE, W. (1937). McClung’s Microscopical Technique, 2nd ed. p. 489. New York: Hoeber.
RE'rzIUs, G. ( 1894). Biol. Untersuch. N .F. 6, 1.
TELLO, F. (1912). Trab. Lab. Invest. biol. Univ. Madrid, 10, 145.
TILNEY, F. (1936). Bull. neurol. Inst. N. Y. 5, 387.
Explanation of Plates I And II
Plate I
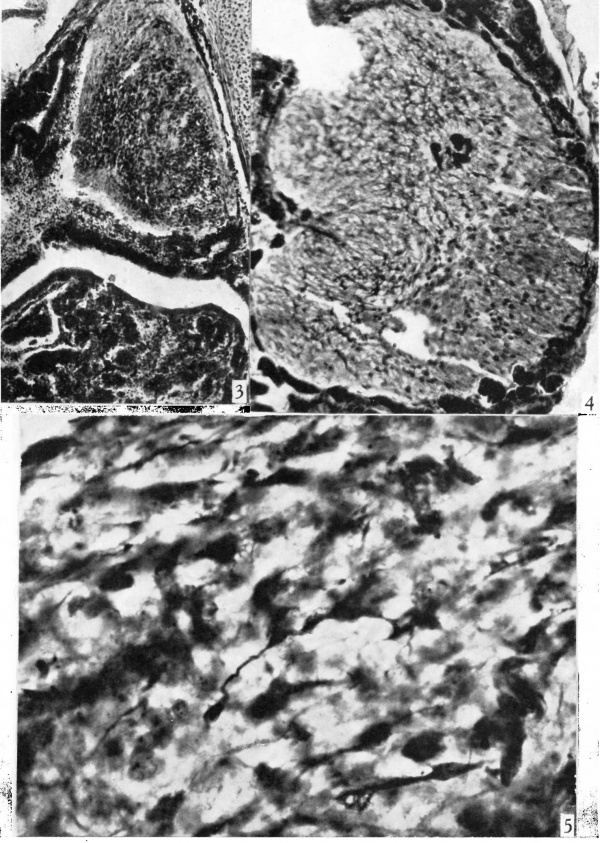
Fig. 3. Photomicrograph showing from above downward the pars nervosa, the pars intermedia, cleft, and the pars anterior of a 3-month human foetus. x 95.
Fig. 4. Photomicrograph through the pars nervosa of a 55-month human foetus showing the differentiated pituicytes in the part proximal to the pars intermedia and the poorly differentiated pituicytes in the distal portion. Note a single clump of blood-vessels deep in the nervosa. x 175.
Fig. 5. Photomicrograph of field corresponding in part to the drawing in Text-fig. 2. Note cells of various shapes, anastomosis, end bulb, and the brush-like ending of one of the pituicyte processes. x 725.
| Fig. 3. | Fig. 4. | Fig. 5. |
|---|---|---|
|
|

|
| Fig. 3. Photomicrograph showing from above downward the pars nervosa, the pars intermedia, cleft, and the pars anterior of a 3-month human foetus. x 95. | Fig. 4. Photomicrograph through the pars nervosa of a 55-month human foetus showing the differentiated pituicytes in the part proximal to the pars intermedia and the poorly differentiated pituicytes in the distal portion. Note a single clump of blood-vessels deep in the nervosa. x 175. | Fig. 5. Photomicrograph of field corresponding in part to the drawing in Text-fig. 2. Note cells of various shapes, anastomosis, end bulb, and the brush-like ending of one of the pituicyte processes. x 725. |
Plate II
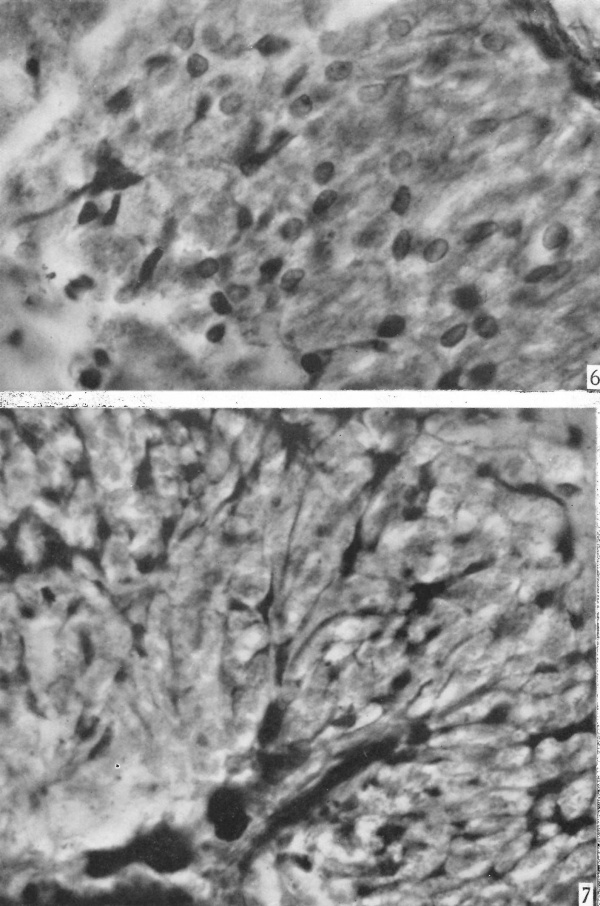
Fig. 6. Photomicrograph of same section shown in P1. I, fig. 4 showing details of cells and intercellular granules in the zone distal to the pars intermedia. x 725.
Fig. 7. Photomicrograph demonstrating details of the proximal area of the same section shown in P1. I, fig. 4. Note the prominent trabecula, the accompanying blood-vessels stained black, the - processes attached to the septum, and processes anastomosing with one another to form a reticulum. Due to lack of focal depth the reticulum is not adequately represented. x 725.
| Fig. 6. | Fig. 7. |
|---|---|
|

|
| Fig. 6. Photomicrograph of same section shown in P1. I, fig. 4 showing details of cells and intercellular granules in the zone distal to the pars intermedia. x 725. | Fig. 7. Photomicrograph demonstrating details of the proximal area of the same section shown in P1. I, fig. 4. Note the prominent trabecula, the accompanying blood-vessels stained black, the - processes attached to the septum, and processes anastomosing with one another to form a reticulum. Due to lack of focal depth the reticulum is not adequately represented. x 725. |
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Cite this page: Hill, M.A. (2024, April 28) Embryology Paper - Differentiation of pituicytes in the human foetus. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Differentiation_of_pituicytes_in_the_human_foetus
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G