Paper - Development of the malleus of the human ear - Illustrated in atlas series
| Embryology - 20 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hanson JR. and Anson BJ. Development of the malleus of the human ear; Illustrated in atlas series. (1962) Q Bull Northwest Univ Med Sch. 36(2): 119–137. PMID: 13904457.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Development of the malleus of the human ear - Illustrated in atlas series
Jerome R. Hanson and Barry J. Anson
1 From the Department of Anatomy of Northwestern University Medical School (Contribution No. 662) and the Department of Anatomy of the University of Wisconsin.
A study supported by grants from the Central Bureau of Research of the American Otological Society, the National Institutes of Health of the U.S. Public Health Service (B.2237.C3). The entire cost of engraving was met by the use of these funds.
The photomicrographs were taken by Homer Montague; they were labelled by Marion Mason Kohring.
Received for publication April 11, 1962.
2 Dr. Hanson served as Project Assistant under the grant from the National Institutes of Health.
3 Anson, B. J. and Bast, T. H.: Development of the Otic Capsule of the Human Ear, Quart. Bull. Northwestern Univ. M. School, 32:157-172, 1958.
Anson, B. J., and Bast, T. H.: Development of the Stapes of the Human Ear, Illustrated in Atlas Series. Quart. Bull., Northwestern Univ. M. School, 33:44-59, 1959.
Anson, B. J., and Bast, T. H.: Development of the Incus of the Human Ear. Illustrated in Atlas Series. Quart. Bull., Northwestern Univ. M. School, 33:110-119, 1959.
The photomicrographs were prepared from the following otological series in the Wisconsin collection: Fig. 1, 206; Figs. 2a, 2b and 2c, 158; Figs. 3a and 3b, 160; Fig. 4, 150; Fig. 5, 183; Figs. 6a and 6b, 30; Figs. 7a, 7b and Ze, 21: Figs. 8a and 8b, 85; Fig. 9, 296; Figs. 10a and 10b, 47; Fig. 11, 64; Fig.. 12a, 12b and 12c, 83; Figs. 13a, 13b and 13c, 120; Figs. 142, 14b and 14c, 29; Figs. lida, Loe and 1L5ec, 21; Figs. 16a and 16b, 24; Figs. 17a and 17b, 1
ee: Fig. 1, X 160; Fig. 2, X 40; oe 3, ae Fig. 4a, X 30; 46, X 100; Fig. 5a, X 30; 5b, X 100; Fig X 40; Fig. 7, X 30; Fig. 8, X 40; Fig. 9a, X 30; 9b, x 100: Fig. i0, X 40; Fig. lla, X 25; i1b, X 80: Fig. 13, X 40: Fig. 13, X 40; Fig. 14, X 40; Fig. 15, X 60; Fig. 16, X 60; Fig. 17, X 60. These are the criginal magnifications of the photomicrographs; all were reduced uniformly to foursevenths of original size in reproduction.
Draft Page only
This fourth article in an atlas series completes a study of the development of the otic capsule and the auditory ossicles in man.3
Introduction
The following commentary provides a brief introduction to the developmental steps that are described in greater detail in the legends for the figures.
The malleus becomes clearly identifiable as a condensed mass of mesenchymal cells within the first two visceral arches of a 10-mm embryo. The constituent cells differentiate from the mesenchymal to the precartilaginous stage by the seventh week; thereupon the primary relationships that establish the origin of the malleus are already lost (fig. 1). The head of the malleus and the long crus of the incus continue to be approximated, but the line of future separation is indicated by an intervening layer of mesenchymal cells. At the same time, the ossicles progress to the cartilaginous state in the 28-mm specimen (fig. 2a).
The first pharyngeal pouch invades the loose mesenchyme within the first two visceral arches to form the Eustachian tube and the tympanic cavity (fig. 2b). This entodermal evagination will invest the ossicles with a layer of mucous membrane.
The anterior process first appears as a spicule of membrane bone adjacent to the malleus and Meckel’s cartilage (fig. 2c). It is the only portion of the ossicles that does not exist as a cartilaginous model prior to ossification. Although the ossicles double in size between the eighth and eleventh weeks (as does Meckel’s cartilage), the anterior process grows slowly—dwarfed by the relatively large Meckel’s cartilage (fig. 3a). The ieee ossicles are still cartilaginous (fig. 3, a and b).
At the 15-week stage (before the malleus reaches maximum size), the solitary ossification center appears as a plaque of perichondral bone on the media! surface of the upper portion of the manubrium of the malleus (fig. 4, a and b). Concurrently Meckel’s cartilage is undergoing degenerative changes; the outer layers of cells flatten, preparatory to conversion into fibrous tissue of the anterior suspensory ligament (fig. 4, aand ¢).
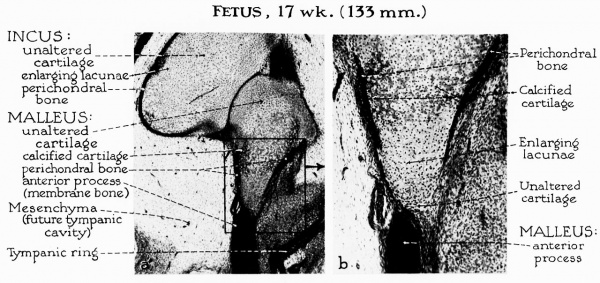
Perichondral bone spreads rapidly over the ossicle, subsequent to the enlargement of cartilage lacunae. In the fetus of 17 weeks, the process has already spread to the lateral aspect of the malleus; early
Calcification of the oe matrix has begun (fig. 5, a and b). As pericondral ossification spreads livodah the neck of the ossicle, the anterior process begins to fuse with the outer shell (fig. 6a).
After reaching its maximum |size at approximately the 17-week stage, Meckel’s cartilage rapidly degenerates into fibrous tissue (fig. 6b). As the ossicles attain adult dimensions in the fetus of 20 weeks, the once prominent Meckel’s cartilage is reduced to a relatively small accessory structure (fig. 7, a and b).
Large areas of calcified cartilage are resorbed, and endochondral bone is formed on the inner surface of the preexisting perichondral bone by a lining of osteoblasts (fig. 8, a and b). At midterm, the manubrium and lateral process are beginning to be invaded by the osteogenic process (fig. 7c).
Endochondral bone preoliferates rapidly to fill the marrow space. Spicules of calcified cartilage, scattered throughout the inner portion of the malleus, are converted to intrachondrial bone (fig. 8, a and b). Periosteum, perichondral bone, endochondral bone and endosteum, and calcified cartilage are now the constituents of the ossicle (fig. 9, a and b).
Slowly the manubrium and_ lateral process exhibit similar changes (fig. 10, a and b). However, the top of the manubrium is not covered by a perichondral shell; the endochondral bone, when eventually formed, will still be covered by a thin layer of cartilage.
Meckel’s cartilage now undergoes rapid regression; the portion within the tympanic cavity has become completely fibrous in the 26-week specimen (fig. 11, a and b). A hyalinized remnant is found in the segment that passes through the petrotympanic fissure (indicated by arrows in fig. 11b).
In the neonatal period the malleus undergoes a succession of changes in structure, which are similar to those taking place in the incus. Localized areas of bone are eroded and, after the resorption of primary osseous layers, new lamellae of bone are deposited (fig. 12, a, bandc). Random formation of secondary lamellae alters the primary pattern of ossification; this involves the perichondral as well as the endochondral layers (fig. 13, a, b and ¢).
There is wide variation in the time of appearance of these secondary ossification centers. The process of malleolar remodelling may begin during the second decade of life (fig. 14, a, b and ¢), or it may be encountered in the ossicle of the aged adult. In any event, the central marrow cavity is obliterated in the malleus, as it is in the incus. The basic pattern of perichondral and endochondral bone occurs throughout the ossicle except at the articular surface, at the site of attachment of ligaments and at the tip of the manubrium. In this last-named segment a cartilage layer covers the endochondral bone.
The amount of remodelling is sometimes widespread (fig. 15, a, b and c). It is manifest in the cecurrence of tertiary lamellae and accompanying disruption of the primary pattern. In some cases the malleus, as well as the incus, is virtually undisturbed by the remodelling process (fig. 16, a and b). In such instances they could be mistaken for the ossicles of a young individual.
This condition may persist in the malleus of individuals in the sixth and seventh decades of life (fig. 17, a and b). This means that histological structure is not wholly dependent upon age. The presence of cartilage in the zone beneath the periosteal layer may be taken as furtherevidence of the fetal character of the malleus.
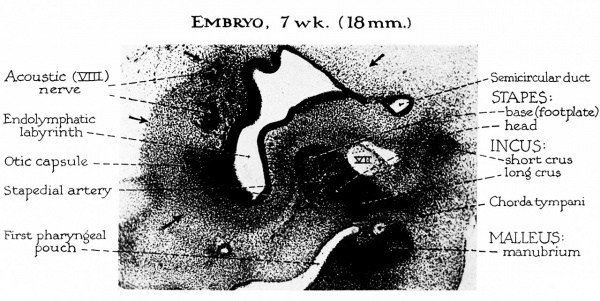
Fig. 1. The malleus Embryo 7 week 18 mm, like the incus and stapes, develops from a mesenchymal blastema located within the first two branchial arches. In the earliest stages of differentiation (approximately 10 mm) the malleus is an indistinctly outlined component of this cellular blastema.
As the primordial tissue of the ossicles differentiates into precartilage, then cartilage, the malleus becomes histologically demarcated from the incus. In an embryo of seven weeks, the enlargement of mesenchymal cells within the ossicles indicates early precartilaginous cellular development. The chorda tumpani serves to separate the manubrium of the malleus from the long crus of the incus (as the nerve passes toward the first branchial arch). At this stage the malleus has already lost its primary connection to that portion of the second visceral bar which will become the styloid process. Secondary continuity of the ossicular chain is established as the long crus of the incus approximates the portion of the stapedial ring destined to become the head of the stapes. At a higher level (not shown) Meckel’s cartilage is continuous with the head of the malleus, which, in turn, approximates the body of the incus. The precartilaginous tissue of the developing otic capsule (outline indicated by arrows) is differentiating around the membranous labyrinth.
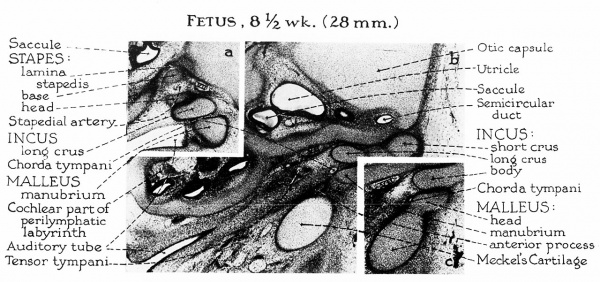
Fig. 2. Embryo 8.5 week (28 mm) a, At this level the ‘‘stapes”’ (still shaped like a ring rather than a stirrup) presses against the lateral wall of the cartilaginous otic capsule to form the lamina stapedis. The long crus of the incus approximates the future capital part of the stapes but is separated from the manubrium by the chorda tympani. The loose mesenchyme is being invaded by the first parhyngeal pouch, which eventually will line the tympanic cavity and envelop the ossicles. The anterior process of malleus appears for the first time as a spicule of membrane bone situated medialto Meckel’s cartilage and anterior to the manubrium. [tis the only portion of the ossicles which is not formed in cartilage.
b, The ossicles are fully formed in cartilage between the eighth and ninth{weeks of fetal life. A dense zone of mesenchymal cells renders the two ossicles distinguishable from each other.
c, Meckel’s cartilage is broadly continuous with the head of the malleus.
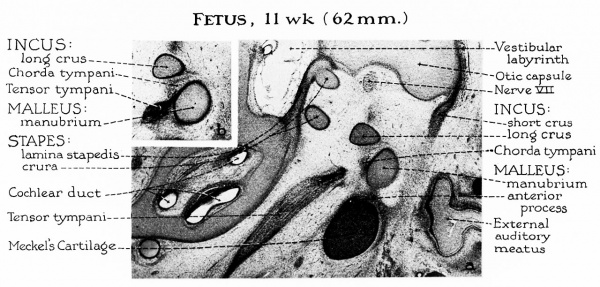
Fig. 3. Fetus, 11 wk (62 mm) a, In less than a 3-week period, the ossicles have doubled in size. However, they remain embedded in the loose mesenchyme of the tympanic cavity. Meckel’s cartilage continues to grow at a rate comparable to that of the malleus; as a result, in the fetus of 11 weeks the anterior process is dwarfed by this relatively large cartilaginous bar. The lamina stapedis is distinguishable from the remainder of the otic capsule. The otic (endolymphatic) labyrinth is formed, but the future periotic (perilymphatic) labyrinth is represented by reticular tissue (except when an early space forecasts the vestibule).
b, The newly formed tensor tympani muscle inserts into the manubrium of the malleus below the level of the chorda tympani, and below that of the lateral process (not shown). This projection is a cartilaginous outgrowth from the upper portion of the lateral aspect of the manubrium.,
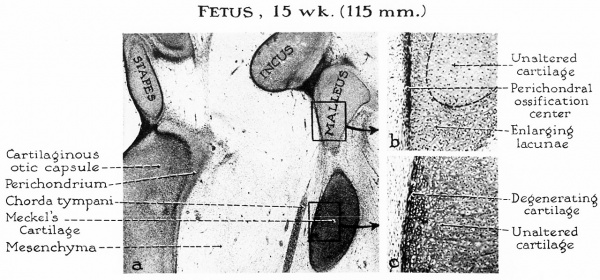
Fig. 4. a, In the 15-week fetus (115 mm) the malleus begins to ossify from a single center located medially, nee the neck of the ossicles. The incus, at this level, is almost completely surrounded by a perichondral ayer.
Although the maximum growth of Meckel’s cartilage is not attained until the 17-or-18-week stage, the cartilage at the periphery of the bar shows early evidence of degeneration in the stage here illustrated.
b, Detailed examination (of the area blocked in a) reveals a layer of osteoblasts which will lay down a plaque of perichondral bone. This will spread rapidly over the ossicle. Calcification of cartilage matrix and primary resorption will ensue prior to actual endochondral ossification.
c, The cells near the periphery of Meckel’s cartilage (in the rectangular portion of a) flatten and assume an arrangement parallel to the surface of the bar. This stage in differentiation precedes their conversion into the fibrous tissue of the anterior ligament of the malleus.
Fig. 5. a, By the 17-week stage, perichondral ossification has spread to the lateral aspect of the malleus, accompanied by widespread enlargement of cartilage cells and early calcification of the matrix. Endochondral bone-formation follows beneath the perichondral shell. Exceptions will occur at the articular surface, in areas of ligamentous attachment and at the distal extremity of the manubrium. Each of these will retain a thin, cartilaginous layer rather than a periosteal shell.
b, The area shown at higher magnification reveals that the anterior process of the malleus is formed in membrane bone. The process is in close proximity to the neck of the malleus but separated therefrom by fibrous tissue. Fusion will take place when ossification has been completed in the adjacent portion of the malleus.
File:HansonAnson1962 fig06.jpg
Fig. 6. a, The osteogenic process spreads quickly through the malleus. It is characterized by calcification of the cartilage matrix, resorption of calcified cartilage, formation of bone marrow and development of perichondral bone. The formation of the latter produces an appreciably thickened osseous shell prior to the deposition of endochondral bone. Now that the entire neck of the malleus is being ossified, the antervor process will soon fuse with the perichondral shell to complete the gross anatomy of the ossicle.
b, Anterior to the head of the malleus,'Meckle’s cartilage is being converted into jibrous tissue that will become the anterior ligament of the malleus. Cartilage lacunae enlarge, much as they do prior to ossification, but rather than develop into osteoblasts, the peripheral cells flatten and become fibroblasts which form the collagenic fibers of the ligament. Retrogressive change occurs from the outside to the center in the conversion of Meckel’s cartilage to the anterior suspensory ligament of the malleus. The incudomalleolar articulation is indicated by the arrow.
File:HansonAnson1962 fig07.jpg
Fig. 7. a, Maximum size of the ossicles is precociously attained in fetuses of 20 to 21 weeks. Endochondral shell is being formed on the inner surface of the previously formed perichondral shell in the head of the ossicle. Osteoblasts cover scattered islands of calcified cartilage; while they are being converted into intrachondrial bone, they serve as bases for the deposition of endochondral bone. Meckel’s cartilage lies medial to the head of the tympanic ring. Cartilage has undergone rapid regression.
b, The anterior process of the malleus, after existing as a separate entity for 12 weeks, has reached its maximum length by 21 weeks. It is applied to the neck of the ossicle just above the level of the lateral process.
Whereas the long crus of the incus is acquiring endochondral bone internal to the investing layer of perichondral bone, the osteogenic process is just being initiated in the manubrium at the level of insertion of the tensor tympani muscle.
File:HansonAnson1962 fig08.jpg
Fig. 8. Sections through comparable levels of the head of the malleus, in separate specimens, demonstrate the proliferation of the endochondral layer during a 3%-week period.
a, As osteoblasts deposit endochondral bone beneath the perichondral shell, some cartilage becomes converted to intrachondrial bone. Osteogenesis of fetal-type bone (i.e., without lamellae) will eventually obliterate the marrow space by encroachment of the endochondral layer.
b, The proliferation of bone greatly reduces the marrow space by the twenty-fifth week of fetal life. Similar changes occur in the incus. The anterior process is molded to the malleus by periosteal bone formation, thus strengthening the fusion-area of bone of dissimilar developmental patterns (7.e., of membranous and perichondral ossification),
Fig. 9. a, In the fetus of approximately 22 weeks, ossification has spread from the primary center into the head and the upper portion of the manubrium of the malleus. At the neck the endochondral layer has become a prominent constituent. The compact bone of the anterior process is still distinguishable from the periosteal bone.
b, Magnification (of the blocked area) reveals detailed histology of ossification at the level of the anterior process. The zones of osteogenic development in the rapidly maturing malleus are, from outside inward; periosteum; osteoblasts; periosteal layer of bone; endochondral layer of bone; marrow cavity; cartilage in the stage of resorption. Within the circumferential periosteal shell the original cartilage cells enlarge, the matrix calcifies (prior to resorption) and some marrow spaces are formed, Endochondral ossification gradually fills in the central cavity.
Fig. 10. a, Ossification of the lateral process takes place at a relatively slow rate. Soon after midterm (24-w22% fetus), exrtilage d2generation and replacement by osseous tissue finally begin. Here, as in the remainder of the ossicle, formation of perichondral bone precedes resorption of cartilage and deposition of endochondral bone (except medially at the insertion of the tensor tympani muscle). Intrachondrial bone is formed from spicules that have not been resorbed, As a result of deposition on these spicules, the marrow spaces will be reduced in size and number.
The developing tympanic membrane is now clearly trilaminar, consisting of the following layers: membranous (inner); fibrous (middle); cuticular (outer). The inner layer is composed of the mucous membrane of the enlarging tympanic cavity (formed from the first pharyngeal pouch). As the cavity enlarges to replace the loose mesenchyme, the membrane lines the walls and invests the ossicles.
b, Toward the tip of the manubrium, the cartilaginous changes that precede ossification are just beginning. In this area there will be no perichondral shell; endochondral bone will be covered by cartilage.
Fig. 11.a, In the 10-week period following the maximum growth of Meckel’s cartilage, it regresses from a prominent structure in the primitive tympanic cavity to a less conspicuous supporting ligament. The proximal portion is entirely fibrous tn nature; however, the segment between the tympanic ring laterally, and petrous bone medially, contains a remnant of the cartilaginous precursor.
b, As demonstrated at higher magnification (of the area blocked in a), the remnant of Meckel’s cartilage assumes a hyaline nature. The border of the remnant ts marked by arrows.
Beyond the area shown in the section, the distal extremity of Meckel’s cartilage is being surrounded by membrane bone of the developing mandible. There the cartilaginous remnant will eventually be incor porated into the osseous mandible and thus lose its identity.
Fig. 12. a, In the 10-week infant the neck of the malleus is composed of dense endochondral and perichondral bone, and irregularly distributed areas of intrachondrial bone. There are no new lamellae of bone and no areas of resorption of fetal bone at this level. The anterior process has become fused to the remainder of the ossicle, and the perichondral bone of the proximal segment of the manubrium is indistinguishable from the membrane bone in the anterior process.
b, The small, persistent marrow cavity in the head of the malleus is reduced further by bone formation. However, before this process is completed, there is evidence of resorption of the original bone and of concurrent formation of secondary lamellae,
c, At the level of the tensor tympani muscle the manubrium is composed of endochondral bone with interspersed secondary lamellae. The outer, perichondral layer has undergone localized erosion. Spicules of ossified cartilage remain as islands of intrachondrial bone.
Fig. 13. a, More striking destruction of the primary osseous matrix of the malleus is evidenced in the 7-month specimen. Large vascular channels in the endochondral bone from eroded areas have resulted from removal of the primary matrix and the deposition of secondary lamellae within the excavations,
b, More extensive excavation and rebuilding is occurring at the transverse level of the lateral process. The erosion of perichondral matrix with deposition of lamellae stands as evidence that the ossicle is undergoing a rebuilding process. The line of demarcation between the endochondral and perichondral layers of bone is distinct in the zones of alteration of the pri mordial pattern.
c, The tip of the manubrium is made up of irregular patterns of endochondral bone without a peripheral shell of perichondral bone, Erosion involves the outer cartilage layer, as well as the centrally located endochondral bone.
Fig. 14. a, In some specimens of malleus in young adults, remodelling is less striking than in infantile stages. Both the malleus and incus are dense skeletal elements lacking a central marrow cavity. Thus their pattern differs strikingly from that of the stapes.
b, The manubrium at the insertion of the tensor tympani is equally dense except in areas of lesser, secondary excavation. The layer of perichondral bone is thin or absent.
c, At the distal extremity of the manubrium a perichondral layer is lacking. This is regularly the case whether the specimen be prenatal or postnatal.
Fig. 15. The process of rebuilding is identical in the malleus and the incus. It is accomplished by the removal of bone in certain areas and production of new bone where thus excavated. In the incus the process was observed as early as the twenty-fifth week; in the malleus it was first noticed in the 10week infant.
a, The extensive remodelling in the head of the malleus in this 37-year-old adult suggests a long-term course of histological rebuilding.
b and c, Similar formation of secondary and tertiary lamellae has taken place at the level of the tensor tympani and at the tip of the manubrium, indicating that the process is widespread through the ossicle,
Fig. 16. a, Remodeling takes place to a minimal degree at the level of the anterior process. This is the level of the ossification center, revealing that the primordial formation of bone does not determine predilection in regional reconstruction of the ossicle.
b, The head of the malleus in an adult of 38 years displays much less rebuilding than that of the preceding specimen (37 years); fewer lamellae of new bone are formed in the areas of excavation, with lesser disruption of the primary pattern.
Fig. 17. a, The malleus of a 62-year-old adult is almost devoid of erosive action and new bone-formation at the level of the anterior process. The undisturbed osseous matrix would suggest the histological structure of an ossicle in a very young specimen rather than that of an adult. Thus remodelling is not an essential accompaniment of the aging process.
b, The head of the malleus is the site of minimal new bone-formation. Cartilage persists laterally between the endochondral and perichondral components. Primary erosion medially has caused removal of the perichondral layer of bone. Again, it is demonstrated that remodelling may be initiated in the head of the malleus, rather than in the area where perichondral bone was first deposited.
Cite this page: Hill, M.A. (2024, April 20) Embryology Paper - Development of the malleus of the human ear - Illustrated in atlas series. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Development_of_the_malleus_of_the_human_ear_-_Illustrated_in_atlas_series
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G