Paper - Adult form of the human stapes in the light of its development
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Beaton LE. and Anson BJ. Adult form of the human stapes in the light of its development (1940) Q Bull Northwest Univ Med Sch. 14(4): 258–269. PMC3802306
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Adult Form of the Human Stapes in the Light of Its Development
Lindsay E. Beaton, M.D., and Barry J. Anson, Ph.D. (Med. Sci.)
- Contribution No. 322 from the Anatomical Laboratory of Northwestern University Medical School. An investigation conducted under the auspices of the Central Bureau of Research of the American Otological Society. Received for publication, September 29, 1940.
Introduction
In any attempt to appraise damage to the separate parts of the organ of hearing, the investigator is confronted with an exceedingly complex mechanism, the alteration of which crosses the special fields of pathology of the respiratory, circulatory, nervous and skeletal systems: as granulation tissue in the tympanic cavity ages, becoming less vascular but more fibrous, contraction of the scar may partially obliterate normal orifices or embarrass the movement of the auditory ossicles; sclerosis may result in the destruction of nerves or of special sensory ganglia; changes in the architecture of bone may have a deforming effect upon important foramina within the temporal bone or, by impingement, reduce the excursion of the ossicles. Similarly, within the stapes itself, alteration of the basal portion may fix the ossicle within the vestibular window; simple erosion may produce abnormal thinning, or actual destruction of a crus.
The present article is concerned only with the morphology of the stapes; without information upon the development and normal adult form of the stapes, pathological alteration cannot be distinguished from unimportant variation in shape and size, simple fragility from osteoporotic destruction, normal histologic replacement of cartilage from the abnormal ossification of stapedial ankylosis.[1]
In spite of the importance of the stapes, no single element in human anatomy has suffered more chronic neglect. This is a strange instance of disregard, since the stapes has great clinical importance in_ otosclerosis, passes through developmental stages of extreme interest, and is grossly the most captivating bone in the human skeleton. For all its unique fascination as the smallest essential bone and as the most exquisitely fashioned unit, this ossicle has not only been forgotten by investigators in anatomy and otology, but the descriptions of it in standard textbooks of anatomy and otology have become stylized and debased through the years; they are inaccurate and fragmentary, commonly perpetuating century-old errors. Illustrations are usually either minute or diagrammatic.
As pointed out by Dorothy Wolff, the stapes was observed very early, descriptions being presented in works which rank as classics in sixteenth century anatomy. Credit for discovery of the stapes has, at various times, been given to Giovanni Ingrassias, Matteo Realdo Colombo, Bartolommeo Eustachii, and Andreas Vesalius. Eustachius was the first to figure the ossicles, his illustrations being far superior to most of those published today. Gabriello Fallopio studied the articulations of the ossicles, and Guilio Casserio published pictures of the stapes of ten mammals. Siebenmann (1905) states that Constanzio Variolio first noted the stapedius muscle, but Capparoni (1925) gives the credit to Paolo Manfredi, who also is said to have described the channeling of the crura.
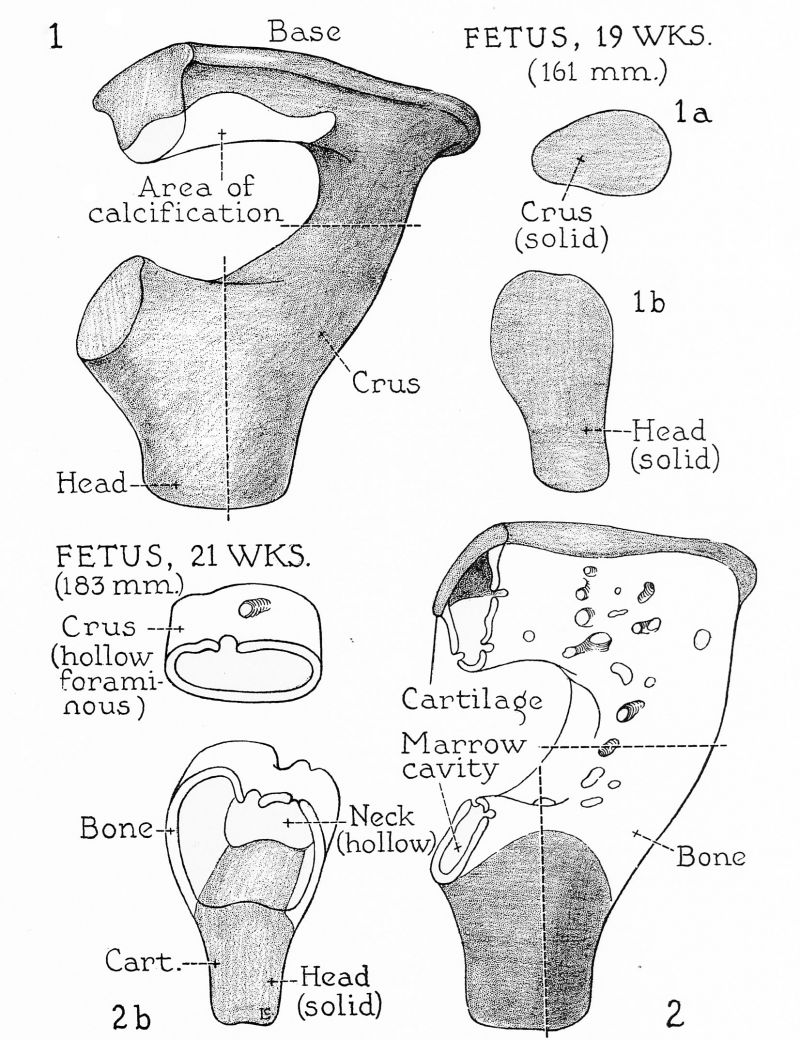
Figs. 1 and 2. Reconstructions of human stapes; semi-diagrammatic (see footnote 4) x 45. 260
With the fundamental anatomy of the stapes established, the eighteenth century writers found ready material for their textbooks, adding details gleaned from their own investigations, they presented accounts which surpass those upon which the present-day student is dependent; to the descriptions by Valsalva (1717) and Cassebohm (1734) little could be added. Thomson (1734) furnished records on dimensions, Williams (1840) on weight.[2] The process of the stapedius muscle was adequately described by Henle (1873), carefully figured by Holden (1887), Eysell (1870) and Frey (1897) gave excellent general accounts. But modern textbook descriptions have utilized only a small portion of this available information; some have wholly neglected even the obvious anatomical features; one describes the human skeleton without including the stapes, another describes the stapes without mentioning the crura, and a third states that the head of the ossicle is a ‘‘detached button of bone.’’
Minor variations in the form of the stapes, all within range of the normal, are readily distinguishable from marked aberrancies reported in the literature. The ankylosing deformities of otosclerosis are common pathological features; fracture is a possibility, though a remote one (Drury, 1925); caries is known to occur in chronic middle ear suppuration (Dickie, 1929) and may result in fragmentation (Keen, 1927), especially in otitis media due to the exanthemata (Fraser, 1927). Syphilis and tuber culosis may also affect the ossicle (Griinberg, 1917).
Absence of the stapes has been reported (Hartmann, 1887; Horbst and Sauser, 1937), as has also that of the malleus (Buch, 1880). Cruveilhier (1834) reports an atrophied stapes, but this may have been merely a smaller specimen than those he was accustomed to find. Accessory ossicles are reported by Cassebohm (1734) and Politzer (1909).
Stapes with only one crus are described by Comparetti (Meckel, 1825), Politzer (1909), Horbst and Sauser (1937); it would seem that these are merely stapes in which the two crura have become fused by ossification of the obturator membrane, by a process such as has been described by Luschka (1867) and Cruveilhier (1834). Exostoses are not uncommon (fig. 11, posterior crus, at arrow). Other anomalies are listed by Politzer: cartilaginous footplates, abnormally large footplate or crura or head; but these may come well within the limits of anatomic variation.
The abnormalities which are not ascribable to actual disease probably accrue from excessive bone formation, increased bone resorption, or from developmental arrests or mishaps; Horbst and Sauser would put many anomalies in this latter class.
Variation in stapedial morphology is best understood when considered in the light of developmental history. This history is a remarkable one: formed in bone by the middle of intrauterine life, ‘‘adult’’ dimensions are already established; then through rapid histological remodelling, adult form is attained before the fetus enters its seventh month of development. Some variability in shape and size is discernible even in the ossicles of late fetuses and of infants. Only minor alterations are made during the course of the individual’s lifetime; no other fully formed bone in the human body passes through so many years of ‘adult’? existence.
In the embryo of 40 mm crown-rump length, while the stapes is composed of cartilage throughout, its form suggests that of a stirrup; a center of ossification has appeared in the base of the stapes at the 161 mm. stage (fig. 1); the crura (fig. la) and the base are still composed of cartilage, are solid cylinders.[3]
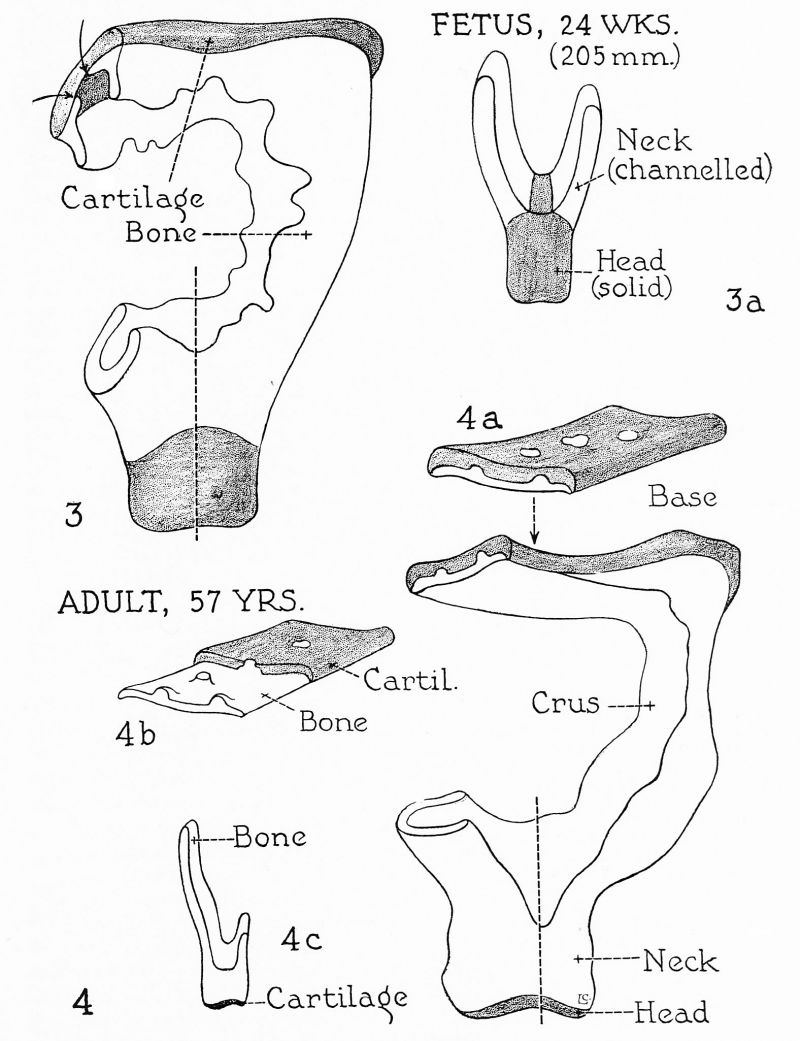
Figs. 3 and 4. Reconstruction of human stapes; semi-diagrammatic. x 45. 262
The stapes exhibits no regular increase in size after birth. While it is longer in the infant than in the fetus at term, in the child and in three specimens from adults (18, 57 and 70 years) its length is actually less than it is in the infant. Therefore, once adult dimensions have been attained in the fetus of 21 weeks, considerable individual variation may be expected in all later stages. Once definitive form is established in late fetal life, alteration proceeds at a slow pace, producing ultimately a stapes which is thinner than that of the fetus at term.
The base of the stapes, similarly, is longer in the infant than in any of the other specimens studied: the width is highly variable, bearing no constant relation to increase in age. In regard to form and to histologic structure, however, progressive change is strikingly evidenced. In the specimen from the fetus at term, the infant of 3 months and the child of 3 years, the cartilage along the margin of the base is rolled backward toward the crural or tympanic aspect, most prominently on the two extremities. In the adults, this feature of peripheral lipping is less pronounced. The base of the stapes, once definitive ‘‘adult’’ form is attained in the late fetal stage, remains throughout life a bilaminar plate (fig. 4). Cartilage progressively gives way to bone. In the fetus at term, in the infant and in the child, the base is thick; cartilage, predominating, not only covers the vestibular aspect completely, but also appears on the opposite or tympanic surface of the base in the form of small patches of chondrial tissue; in adult stages ossification has advanced to a point at which osseous tissue presses through the vestibular lamina, to appear on its medial surface as islets of bone (figs. 4a and 4b). In the adult of 18 years these are small and isolated, in the adult of 57 years some have coalesced to produce a larger area of bone, while in the adult 70 years old cartilage occupies approximately half the surface. Concurrently, the entire base becomes thinner. In the 183 mm. fetus the crura and the lateral part of the base have become osseous, but they are hollowed and contain vascular tissue, (figs. 2 and 2a). The osseous portion of the stapes is now a modified ring, foraminous on its internal aspect; cartilage remains as a basal plate and a capital cylinder (figs. 2 and 2b).[4] Gradually excavated on their internal surface, the base becomes a flattish plate, the crura guttered columns and the neck a hollow cylinder (figs. 3 and 3a). As cartilage is reduced in thickness at the base and the head, bone advances to line the internal surface of each of these cartilaginous plates (figs. 3 and 3a, at arrows).
Cartilage remains at the capital extremity as an articular lamina only (fig. 4c). In general, the crura become thinner as age advances, and, just as they are implanted into the base by less wide expansions in older persons, so they usually enclose less of a circle when seen in cross section. This change would indicate that resorption has acted not only to thin the crura but also to erode their free margins and to expose more of their internal surface.
In the fetus at term, cartilage remains only on the articular surface of the head, the capital and the cervical portion otherwise being osseous. Not only has the neck been hollowed, but, in some specimens, the superior wall has been made perforate; such puncturing is even more prominent in ceriain of the infantile and adult series. In the adult of 18 years the greater part of the superior surface of the neck is wanting, the crura joining near the head—the capital end of the anterior crus remains as a bridge of bone; the medial margin of the head, as disjoined prongs. In the adult of 70 years the posterior wall of the head is deficient, the antero-superior wall widely perforate and the head less broadly connected with the neck than in any other example in the present group. Oddly, the neck of the stapes from the adult of 57 years exhibits no perforations, merely extensive hollowing. The shape of the articular surface varies considerably in specimens from persons of different ages.
It may then be said, in summary, that the true stapedial (stirrup-like) form is suggested in the cartilaginous ossicle of the 40 mm. embryo, definitely established in that of the 135 mm. fetus. At 161 mm., and with but slight growth during the period, ossification is initiated in the tissue situated on the tympanic aspect of the base; spreading therefrom, in the 183 mm. stage, bone has replaced all cartilage save that on the medial aspect of the base and the lateral surface of the head; its distribution at this stage is such that bone is continuous around the circumference of the intercrural space; it is an osseous ring uninterrupted by epiphysial cartilage, and resembles a diaphysis split longitudinally (see preceding article). Thus left without the customary mechanism of lengthening and expansion in the middle of intrauterine life—when long bones generally are still destined to grow for twenty years—the stapes has attained adult dimensions when the remainder of the body is still fetal. Fitting into a fenestral orifice of an otic capsule which is equally static, the ligamentous, muscular and articular relations are well established.[5] Such slight transformations as do occur are the result of exceedingly slow histological rearrangement.
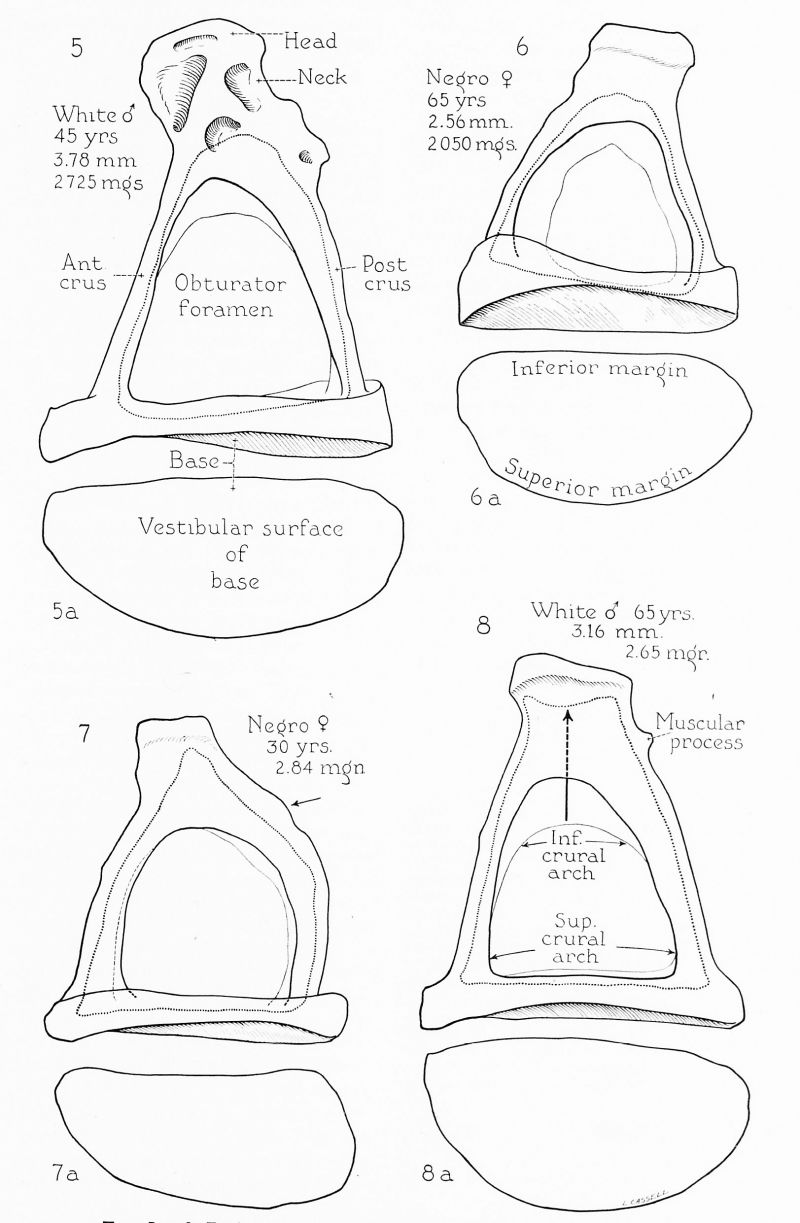
Figs. 5 to 8. Excised specimens of human stapes (see footnote 4). x 20.
Variations are to be expected in the anatomy of the stapes of older persons, since they have been observed in reconstructions of the ossicle from advanced fetus, newborn and infant: the neck may be excavated or entire; the tubercle prominent or wanting, the medial ends of the crura narrow or broad at their point of continuity with the base; the length of the crura or width of the base may be slightly less in the stapes of an infant than in that of late fetus. All of these differences are within normal range of anatomic variation, being regularly encountered in ossicles from adults between the ages of twenty-five and seventy-five years.[6]
The Stapes
The stapes (Latin, stirrup) is well described by its name. It consists of a flat base (basis) or footplate, roughly elliptical in shape, to the extremities of which are attached the two limbs (crura). These crura converge to join at a constriction, termed the neck (collum); the neck is continuous with a terminal articular portion, the head (caput). The crura form an arch, and thus bound, with the base as a third boundary, the obturator foramen of the stapes; the foramen is partially closed by a membrane. The crura, head and neck and base are channelled. The anterior crus is usually shorter, less curved, and more slender than the posterior. The head is hollowed for the reception of the lenticular process of the incus, thus forming the incudostapedial articulation.
Base
The base of the stapes is that part of the ossicle which closes the fenestra vestibuli (vestibular or oval window); it is bound down by an annular ligament to the _ fenestral rim. It is partly osseous, partly cartilaginous, a fact which may account for the early statement that the footplate is pierced by many small holes (Palfin, 1731).
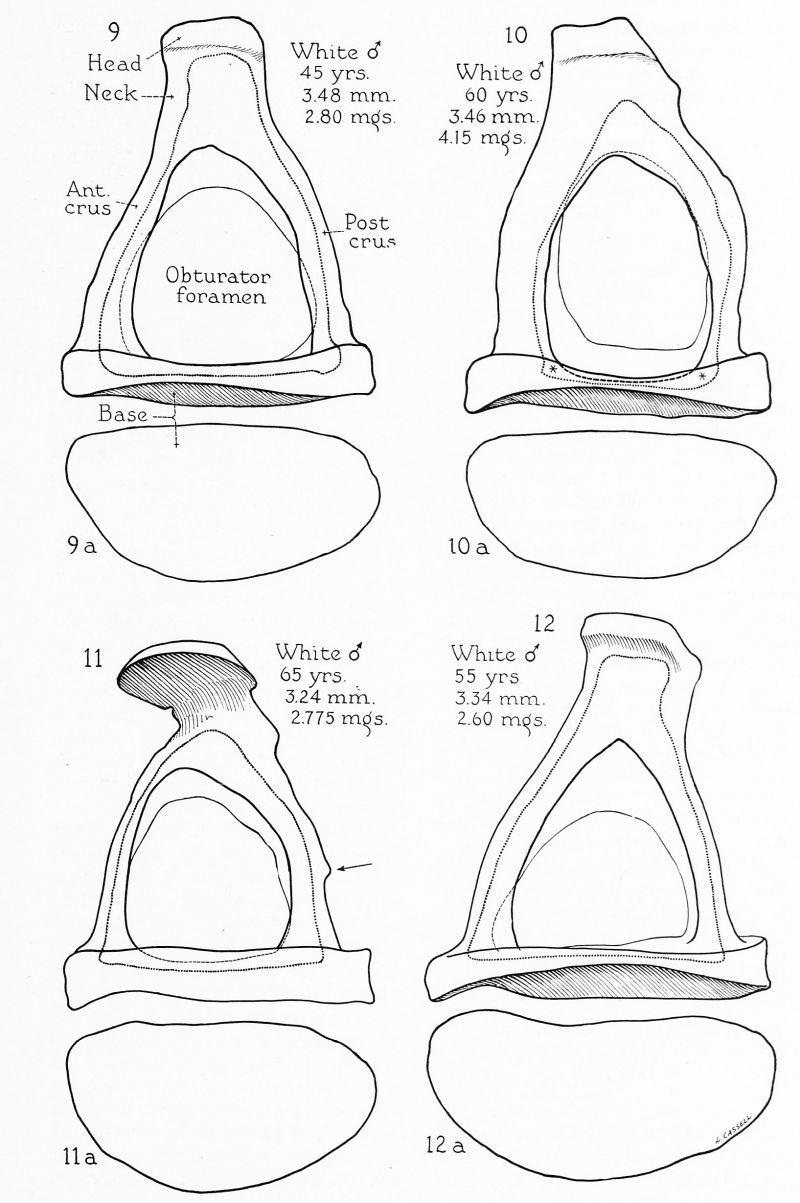
The base, as viewed from the medial or tympanic surface, varies widely in outline. Although usually described as oval, elliptical or bean-shaped, such forms are not common. The customary shape is one in which the superior surface is generously convex, the inferior surface or edge almost straight, the anterior and posterior borders rounded, with the anterior tending to be a little more pointed than the posterior (fig. 6 a). This morphological character was well described by Boehmer (1751), Cassebohm (1734). Uncommonly the inferior edge is concave, to produce a reinform base (fig. 12 a). On the other hand, the inferior edge may be convex, the whole outline oval (fig. lla). Frequently the angle between the anterior and superior margins is sharp, owing to straightness in the anterior part of the superior margin (figs. 8 a, 9 a); this type, figured by Bierkowski (1827), was thought by Eysell (1870) to be typical. Size variations make for all gradations between a short, wide base (fig. 11 a) and a long, narrow, base (figs. 5 a and 7 a). Generally the anterior end is either slightly larger than the posterior (figs. 8 a and 9 a), or the two ends are equal (fig. 11 a). Occasionally the anterior end is pointed (fig. 7 a); Urbanschitsch (1876) thought this to be a regular feature.
The vestibular surface of the base has been found, in the current series, to be slightly convex (fig. 9); this has been regarded as a regular feature since the time of Cassebohm. Rarely the base is flat (fig. 11). No example of basal concavity on the vestibular surface has been encountered. At times the vestibular surface at the anterior end is flat or weakly convex, the posterior end strongly convex (fig. 5), a form regarded as typical by Eysell (1870).
Figs. 9 To 12. Excised specimens of human stapes. x 20. 266
The tympanic surface of the base is gently concave; concavity is in part due to the presence of an encircling rim. Cassebohm first described this circumferential lipping. In the specimens currently studied this marginal elevation has been found in some specimens to be complete (fig. 11), and in others mergent with the edges of each crus (fig. 8). Since in young specimens continuity of basal lip and crural edge is always encountered, its occurrence in adult specimens may be regarded as retention of a fetal character. The crura usually originate from points nearer the inferior than the superior margin of the base; the opposite arrangement has not been observed in the current group of specimens.
On the tympanic surface of the footplate is found an inconstant ridge, the crista stapedis, which passes between the crura (fig. 10 at asterisks). It is present in less than one third of the specimens, and even then is often incomplete; it may be absent anteriorly, posteriorly, or in the midportion; it may be merely a continuation of the superior or inferior edge of either crus (fig. 10), arise in the hollow between the two edges, or exhibit any combination of these origins. It supposedly represents the line of attachment of the obturator membrane. It is reported that at times a groove may replace the crest (Lizars, 1844).
The average circumference of the base is 7.45 mm., with extremes of 6.48 and 8.30; the average length is 2.99 mm. (extremes, 2.64 and 3.36), the average width is 1.41 mm. (extremes, 1.08 and 1.66).
Crura
Arising from the extremities of the base, the crura join laterally to complete the crural arch. The shape of the arch is usually ovoid (figs. 5 and 8); rarely it is pointed (fig. 12), quite often almost circular (fig. 7).
Each crus is channelled, even in fetal specimens; the hollowing is on the internal surface. By virtue of this peculiar construction each crus has a superior and an inferior edge. The two superior edges meet at the capital end, as do the two inferior edges, so that, in reality, two arches are formed. The hollowed portion then continues into the neck, and sometimes even into the head; in specimens with tall basal lips the base itself lends the appearance of being similarly excavated (fig. 10).
The meeting of the crura to form the stapedial arch gives the stapes its distinctive shape; depending on crural length and curvature, the shape of the obturator foramen is altered. The crura vary widely in size, from slender stems (fig. 5) to sturdy structures (fig. 10). Almost without exception the anterior crus is the slenderer; rarely is it the bulkier of the two (fig. 10), usually the straighter (hence the term, crus rectilineum); the posterior crus is curved (crus curvilineum). Only Hyrtl (1873) disagrees with this otherwise universally accepted description; exceptional specimens do occur in which the posterior crus is straighter than the anterior (fig. 12). Because of the difference in degree of curvature, the anterior crus (head to base) is 3.62 mm., that of the posterior crus, 3.73 mm. (average length). So regularly does this difference obtain that presentday writers treat it as dogma; but Portal (1804) early questioned the validity of the notion. Actually, rare specimens are encountered in which the anterior crus is the longer of the pair (fig. 12), frequent specimens in which the crura are approximately equal in length and curvature (fig. 9).
The plane of the crura may meet that of the base at a right angle, or at an angle acute either superiorly or inferiorly. The in situ appearance would lead one to expect that an inferior angulation is the rule: however, this appearance is due in part to the inclination of the vestibular window; the excised specimen often shows a true superior inclination. In addition to this angulation, the crura are often slightly concave on the side toward which they incline.
The crura are subject to the formation of exostoses, as is any other part of the ossicle (see posterior crus, fig. 11 at arrow); when posterior crural, an exostosis may be mistaken for the muscular spine for the attachment of the stapedius muscle. The posterior crus occasionally possesses an additional feature of note, namely, a kneelike thickening at the junction of its lateral and middle thirds (fig. 10), first described by Holden (1887).
Obturator foramen
The obturator foramen owes its variatiom in shape to variation in the length, curvature and position of the two crura. Thus the foramen, and hence the stapes, may be long (fig. 5) or short (fig. 6). If the crura are of equal length and similar curvature, the foramen and stapes have the general form of an isosceles triangle (fig. 9). The usual shape (dependent on common differences in curvature) departs from the isosceles form (fig. 8). If the crura meet at an acute angle a pointed arch and foramen result (fig. 12), while if they meet obtusely, the arch is rounded (fig. 7). At times the arch is recurved, with the top of the arch inclining posteriorly instead of anteriorly; this is due to a strong curvature in the lateral part of the posterior crus (fig. 7); it is the mirror image of the type with anterior inclination (fig. 8). Howden (1914) would have it that, since the crura are less curved in the child, the foramen is more liable to be triangular in the young, becoming ovate with increasing age. Actually, the foramen is oval in younger specimens, as a partial retention of the original annular shape of the ossicle (Anson, Karabin, Martin, 1939).
As has been observed by many authors, the superior crural arch is generally somewhat greater in circumference than the inferior (figs. 8 and 12); rarely this size relation is reversed (fig. 6). The difference that usually exists between circumferences of the superior and inferior crural arches is due to the extent to which each was eroded in process of conversion from hollow cylinders to channelled members (fig. 2 and 3). The anterior arch is generally higher than the posterior, and is likely to show a more acute angle than the posterior (figs. 8 and 12). At times the crural edges are almost parallel (fig. 7). The crural edges may end without expansion on the base, may expand to join the peripheral rim, or may be continued into the crists stapedis. Rarely the two superior or the two inferior edges may be so tall at the basal extremity as to continue across the corresponding margins of the base in the manner in which they do in the fetal or infantile stapes (fig. 10; see also Anson, Martin and Karabin, 1938). The space of the obturator foramen then appears to be invading the tympanic surface of the base deeply, suggesting the regular extension of the same space into the capital part of the ossicle. Of course, when viewed in the natural condition, these osseous hollowings are masked by the presence of mucous membrane.[7]
Neck
The neck is the constricted area between the articular head and the point of origin of the crura. Although, sometimes, an indefinite area, it is generally recognized as a special subdivision by American, English and French anatomists; it is not so recognized in most of the German textbooks. In the specimens studied by the present authors it is usually definable (figs. 5, 6, 8 and 11); exceptionally no neck can be defined, the crura blending insensibly with the head (fig. 7).
The neck is subjected to early bone resorption, a process which, seemingly, continues slowly through life. The external configuration of the neck is strikingly variable; extensive external pitting produces foramina, bony bridges (fig. 5), or marked thinning (fig. 7); resorption may be minimal, the neck remaining as a smooth osseous structure (fig. 9).
Since the hollowing of the crura usually extends into the capital subdivision of the stapes, the neck in some specimens is a hollow shell (figs. 8 and 12). Less frequently, when the neck is but slightly excavated internally it remains a solid cylinder (fig. 6). Internal excavation seems to proceed quite independently of external.
Muscular Process
The stapedius muscle is attached to the neck of the ossicle in over three-fourths of the cases. In other instances it is inserted into the head or into the ‘“‘shoulder”’ of the posterior crus (fig. 7, at arrow). The area of attachment may be smooth (fig. 9), a mere roughening (fig. 7), or a spine of fair prominence (figs. 5 and 8). The spine may be situated on the neck (figs. 5, 6, 8, 10 and 11), or on the head (fig. 12). A depression at the point of attachment has been described by Hildebrandt (1803) and Lauth (1835), but does not occur in the present author’s series. The spine may be present in the young (Anson, Karabin and Martin, 1939) and in many lower animals (Henle, 1887). It is, therefore, probably not an ossification within the tendon, but a true traction-process. This spinous elevation is very rarely figured (see Holden, 1887).
Head
The head is perhaps the most variable portion of the ossicle. Variation is due to the amount of bone resorption, the presence or absence of the processus muscularis, differences in the depth and outline of the fovea for reception of the lenticular process of the incus. The head may be very small (fig. 7), large (figs. 8 and 11), or of intermediate size (fig. 9). External erosion may extend into the head (fig. 5) as may, also, internal hollowing (fig. 8). The presence of a processus muscularis affects the margin of the posterior surface (fig. 12). The depression for reception of the lenticular process may be foveate, grooved, triangular, or quadrilateral. Erosion of the neck, if extreme, converts the head into a flat plate of bone, connected by only a thin cylinder or plate with the remainder of the ossicle (fig. 11). In the absence of sculpturing, the head is a cylindrical column, completely attached to the crural arch (fig. 6).
The head of the stapes usually meets the crura at a slight angle, being tilted superiorly. Inferior tilting is uncommon. In many cases the planes of head and crura are in a straight line. The head is usually turned anteriorly to a slight degree (fig. 5), rarely posteriorly (fig. 12); sometimes the head meets the crural junction without curve (fig. 8).
Stapes as a Whole - Dimensions
It will readily be seen by inspection of the selected examples that there is great variation in stapedial size (compare heights, figs. 5 and 6). These differences are not dependent entirely on size-differences of the individual. The average height of the stapes is 3.26 mm., with extremes of 2.50 and 3.78 mm.
There is also great variation in weight; the average weight is 2.860 mgms., with extremes of 2.050 and 4.350 mgms. There is no regular correlation between age and weight. Bone resorption, of course, affects weight; stapes with areas of resorption are relatively light (compare weights, figs. 10 and 11). Erosion of this type sometimes occurs early, having been observed in the term fetus (Anson, Karabin and Martin, 1939, figs. 1 and 13).
Footnotes
- ↑ Subsequent studies will include a consideration of the normal and pathological form of incus and malleus.
- ↑ Urbantschitsch (1876) made the first accurate measurements of the stapes, Eitelberg (1884) the first careful weighings.
- ↑ The illustrations of the stapes from the 161 mm and 183 mm fetuses are based upon wax-plate reconstructions already described and figured (Anson, Karabin and Martin, 1938: 161 mm, figs. 9, 28 and 29; 183 mm, figs. 12, 31 to 33) as is also that of the stapes from the 57-year adult (Anson, Karabin and Martin, 1939, figs. 12, 26 to 28; that of the stapes in the more advanced fetus (205 or 210 mm.) is based upon an examination of the sections.
In the major figures (1 to 4) the superior aspect of the stapes is shown; a portion of each reconstruction is removed in order to demonstrate, on the cut surfaces, the form of the crus and the base. In the supplementary figures (la, 1b, etc.) further sectional views are shown, and along the lines indicated in the more inclusive pictures. In figures 4a and 4b the lamination of the base is illustrated: in figure 4a by simple cut, in figure 4b by removing part of the layer which represents the osseous lamina (cf. fig. 20 and 21 in Anson, Karabin and Martin, 1939).
The original drawings of the excised stapes (figs. 5 to 12) were prepared at a magnification of 50 diameters by the use of an Edinger projection apparatus. The specimens had been freed of soft tissues by mild maceration, then dehydrated in alcohols. In the major figures, except figure 6, the superior aspect of the stapes is shown; in the supplementary figures (5a, 6a, etc.) the vestibular aspect of the base is pictured. Depth of hollowing is indicated by dotted lines (to point of arrow in figure 8). - ↑ In the preceding article in this issue of the Quarterly Bulletin the development of the stapes is more fully discussed.z
- ↑ The semicircular canals, the cochlea, and the capsule as a whole are approximately as large in the midterm fetus as they will ever be (Bast, 1932; also Anson and Wilson, 1939).
- ↑ The present description is based upon a study of 77 excised stapes.
- ↑ The obturator membrane is a fold of the tympanic mucous membrane; draped over the stapes, it occludes part of the obturator foramen. It may be absent, or may be only partial. Its existence was denied by Valsalva (1717) but the structure was described and figured by Cassebohm (1734), Duverny (1748), and Muller (1749), and rarely since. By virtue of the fact that the mucous membrane is draped over the stapes from both sides, the obturator membrane is a double fold, as was first pointed out by Gruber (1891). The membrane soon disappears except across the obturator foramen, where it usually finds attachment to the edges of the crura, and to the crista stapedis; at times it arises in the depths of the crural channels.
References Cited
Anson BJ. Karabin JE. and Martin J. Stapes, fissula ante fenestram and associated structures in man: I. From embryo of seven weeks to that of twenty-one weeks (1938) Arch. Otolaryng. 28: 676-697.
Anson BJ. Karabin JE. and Martin J. Stapes, fissula ante fenestram and associated structures in man: II. From Fetus at Term to Adult of Seventy (1938) Arch. Otolaryng. 28: 676-697.
Anson, B. J., and Wilson, J. G. Structure of the Petrous Portion of the Temporal Bone with Special Reference to the Tissues in the Fissular Region, Arch. Otolaryng., 1939, 30;922-942.
Bast, T. H. Development of the Otic Capsule: I. Resorption of the Cartilage in the Canal Portion of the Otic Capsule in Human Fetuses and Its Relation to the Growth of the Semicircular canals, Arch. Otolaryng., 1932, 16;19-38.
Bierkowski, L. J. von Anatomische-chirurgische Abbildungen. Berlin, 1927.
Boehmer, P. A. Institutiones Osteologicae, L75I;
Buck, A. H. Diagnosis and Treatment of Ear Diseases, 1880.
Capparoni, P. Gli studi di Paolo Manfredi sull’ anatomia degli ossicini dell’ orecchio. Il Valsalva Rivista Mensile di Oto-RinoLaringojatria. 1925, 1;195-198.
Cassebohm, J. F. Tractatus Quatuor de Aure Humana, 1734.
Cruveilhier, J. Anatomie Descriptive, Vol. III, 1834.
Dickie, J. K. M. in Jackson and Coates’, The Nose, Throat, and Ear and Their Diseases, 1929.
Drury, D. W. Fractures of the Malleus, Laryngoscope, 1925, 35;345-353.
Duverny, M. A Treatise of the Ear, 1748.
Eitelberg, A. Resultate der Wagungen menschlicher Gehodrknochelchen, Monatsschrift fur Ohrenheilkunde, 1884, 18; 80-87.
Eysell, A. Beitrage zur Anatomie des Steigbugels und seiner Verbindungen, Archiv fiir Ohrenheilkunde, 1870, 5; 237-249.
Fraser, J. S. Turner’s Diseases of the Nose, Throat and Ear, 1927.
Frey, E. Beitrage zur Anatomie des Steigbiigels. Dissertation, K6nigsberg, 1897.
Gruber, J. A Textbook of the Diseases of the Ear. Trans. from the German by E. Low and C. Jewell, 1891.
Griinberg, K. In Manasse, Handbuch der Pathologischen Anatomie des Menschliches Ohres, 1917.
Hartmann, A. The Diseases of the Ear and Their Treatment. Trans. from the German by J. Erskine, 1887.
Henle, J. Handbuch der Systematischen Anatomie des Menschen, 1873.
Hildebrandt, F. Lehrbuch der Anatomie des Menschen, Bad. III. 1803.
Holden, L. Holden’s Human Osteology, 1887.
Horbst, L. and Sauser, G. Stapesmissbildung. Archiv fiir Ohren, Nasen- und Kehlkopfheilkunde, 1937, 143;48-51.
Howden, R. in Cunningham’s Textbook of Anatomy, ed. by A. Robinson. 1914.
Hyrtl, J. Lehrbuch der Anatomie des Menschen, 1873.
Keen, J. A. The ossicles in middle-ear suppuration and their comparative size in children and adults, Journal of Laryngology and Otology, 1927, 42: 174-180.
Lauth, C. A. Neues Handbuch der praktischen Anatomie, Bd. I, 1835.
Lizars, A. J. Elements of Anatomy, 1844.
Luschka, H. von. Die Anatomie des Menschen, Bd. III, pt. 2, 1867.
Meckel, J. F. Manuel d’Anatomie, Trans. by A. J. L. Jourdan and G. Breschet, Vol. III, 1825.
Miller, G. W. Kupfer Tafeln welche die Knochen des ganzen menschlichen Korpers vorstellen, 1749.
Palfin, J. Anatomie Chirurgicale, Vol. I, 1734.
Politzer, A. A Textbook of the Diseases of the Ear, Trans. by M. J. Ballin and C. L. Heller, 1909.
Portal, A. Cours d’Anatomie Médicale. Vol. 4, 1804.
Siebenmann, F. Bardeleben’s Handbuch der Anatomie des Menschen, Bd. 5, pt. 2, 1905.
Thomson, G. The Anatomy of the Human Bones, 1734.
Urbantschitsch, V. Zur Anatomie der Gehdrknéchelchen des Menschen, Archiv fir Ohrenheilkunde, 1876, 11;1-10.
Valsalva, A. M. De Aure Humana Tractatus, L717.
Williams, J. Treatise on the Ear, 1840.
Wolff, D. A Brief History of the Early Development of the Anatomy of the Ear, Laryngoscope, 1934, 44;1-28.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - Adult form of the human stapes in the light of its development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Adult_form_of_the_human_stapes_in_the_light_of_its_development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G