Paper - 1917 The Typical Form of the Cochlea and Its Variations
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
The Typical Form of the Cochlea and its Variations
By Henry J. Watt.
(Communicated by Prof. D. Noel Paton, F.E.S. Received October 3, 1916.)
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Reference: Watt, H. The Typical Form of the Cochlea and Its Variations. (1917) Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character (1905-1934). 1917-01-01. 89 (618):410–421
The work of this paper is based upon the photographic and descriptive material presented by Dr. A. A. Gray in his two volumes on ' The Labyrinth of Animals/ published by J. and A. Churchill, London, in 1907 and 1908.[1] I have succeeded in extracting from that impressive mass of material definite results that seem to be of some importance.
The dimensions of the cochlea measured by Dr. Gray are : (1) the diameter of the lowest whorl and (2) of the second whorl ( <c taken in a plane which passes vertically through the apex of the cochlea and the anterior margin of the round window ") ; (3) the diameter of the tube of the cochlea in front of the round window; (4) the major axis of the oval window : (5) the slant height of the cochlea (" the distance from the upper margin of the round window to the apex of the organ ") ; and (6) the number of turns of the cochlea.
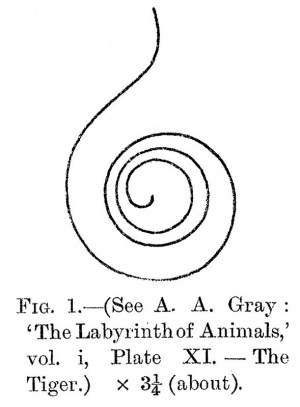
I found it desirable to add to these a measurement of the total length of the basilar membrane. That must surely represent more closely and directly than anything else the pitch-range of hearing. Fortunately, a close study of Gray's wonderful photographs showed that an approximate measurement of the length of the basilar membrane (as of the outside edge of the cochlear tube) could be got from them. The symmetrical shape of the cochlea makes it possible to measure the diameters of the successive whorls, no matter from what angle the photograph was taken. (The reader must consult Gray's pictures.) With the help of Gray's measurements of the diameters of the first and second whorls, by close attention to the consistency of these with the dimensions visible in the photograph, and by a careful comparison of the different photographs showing the cochleas of different animals from the same relative angle, I made what I think is a close approximation to the actual length of. the basilar membrane. In this way I constructed a scale drawing of the basilar membrane as seen from above on the axis of the modiolus, and I measured its length with a rotary map measurer. The reader who is not familiar with Gray's photographs will find that they are much more transparent, and that, consequently, much more detail is visible in them, than he might be inclined to expect. Fig. 1 gives, as an example, a difficult case in which Dr. Gray's photograph presents a view of the cochlea from a point on a line at right angles to the axis of the modiolus.
Doubtless there is a variable error, which probably never exceeded 2 or 3 mm., and was usually much less. And even that maximum is fairly small in comparison with most of the lengths recorded. The small error caused by the rising of the cochlea to an apex I neglected entirely. For a basilar membrane of 52.4 mm. (Eb. 12) the error neglected is not more than 0.16 mm. For Mo. 52 the error is 0.1 mm. for a length of 16*3 mm. The results obtained confirm my estimate of the reliability of the measurement of the basilar membrane.
In order to trace the connection between the dimensions of the cochlea and those of the body as a whole, I sought for a measurement which would represent this. I found that one had been recorded by E. Lydekker for most of the species represented in Gray's work, namely, the length of the head and body, not including the tail. For the aquatic mammals I took the length of the whole body. The method is rough, but no other was available, and, under the circumstances, it is probably good enough.
A. The Typical Form of the Cochlea
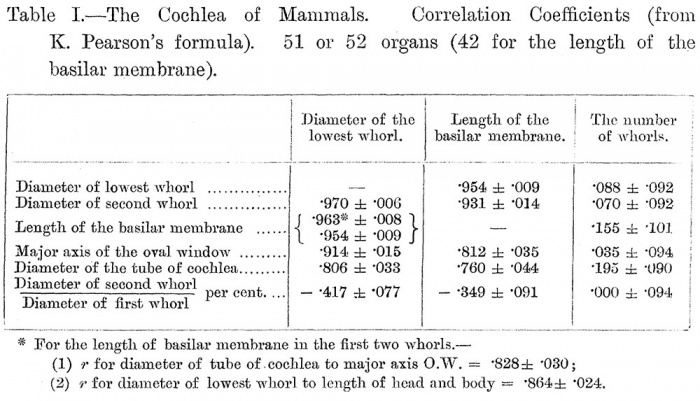
Table I gives the values of the coefficient of correlation between the various series of measurements, and of the probable error, derived with the use of Karl Pearson's formulae. I have neglected here any consideration of " the slant height of the cochlea," as that is from its definition much less a matter of the height of the cochlea than of the diameter of the lowest whorl, which is given independently.
1. There is an absolutely certain positive correlation between the diameter of the lowest whorl, on the one hand, and, on the other hand, the diameter of the second whorl, the length of the basilar membrane in the first two whorls, the total length of the basilar membrane, the major axis of the oval window, and the diameter of the tube of the cochlea. These two last have also a high positive correlation with one another {vide Table I, Note 1). The correlation with the length of the head and body is of the same order {vide Note 2).
Table I. The Cochlea of Mammals
2. The series of correlations for the length of the basilar membrane appears in the second column of the Table. It will be seen that it is parallel in its degrees to the series appearing in the first column, but that the values of the second column, with the exception of the reciprocal pair, are all . smaller than those of the first, while the values of the probable error are, of course, larger. If the length of the basilar membrane and the diameter of the lowest whorl were perfectly correlated with one another, this difference between the two columns would mean that my measurement of the length of the basilar membrane is subject to a variable error that is probably greater than the error made by Dr. Gray in his direct measurements, but that is still not disturbing. But, as we shall see, the relative length of the basilar membrane is subject to special variations in relation to the number of whorls of the cochlea (vide below, B). These fluctuations would account for some part of the difference between the two columns. In any case, my measurements of the length of the basilar membrane may be taken as good.
3. There is no correlation at all between the number of whorls and the diameter of the lowest, or even of the second, whorl, or the major axis of the oval window, or the percentage relation of the second whorl to the first. The other two correlations, with the diameter of the tube of the cochlea and with the total length of the basilar membrane, are too small to be of any significance. There is, then, no correlation between the number of whorls and any other dimension of the cochlea.
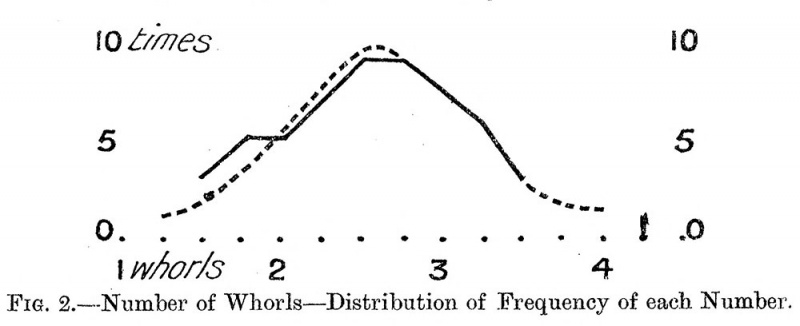
4. Fig. 2 gives the distribution of the frequency of the different numbers of whorls, i.e. there were amongst the 52 organs of mammals measured by Dr. Gray, three organs having 1|- whorls, five having If whorls, and so on. One organ had 4 J whorls. The figure shows a striking evenness of distribution about a point between 2|- and 2| whorls. The average number of whorls of all 52 organs is 2*56 whorls, or a little more than 2| turns. Thus, the average and the highest frequency agree. We may therefore put down the typical number of whorls of the mammalian cochlea as two and a half.
Fig. 2. Number of Whorls — Distribution of Frequency of each Number.
5. The absence of correlation between the number of whorls and the diameter of the second whorl is interesting. It means that the addition of a third whorl (or of more than one) does not necessitate an expansion of the second whorl to make more room for it. Generally the other whorl is merely added on where the second one stopped without any change in the other dimensions of the cochlea.
6. The two negative correlations of the " ratio of the diameter of the second whorl to that of the first " seem to indicate that the bigger the cochlea is, the greater is its rate of curvature, i.e. the smaller is the diameter of the second whorl relatively to that of the first. This correlation, especially as relating to the diameter of the first whorl, approaches towards being a practical certainty. The coefficient is almost 5 J times the amount of the probable error, 6 times yielding practical certainty of correlation.
7. In this connection reference may be made to the only other description applied to the cochlea by Dr. Gray, namely, the distinction of the " flat " from the " sharp " cochlea. In the latter the whorls seem to be piled on the top of one another ; in the former the second whorl lies more or less in the coil of the first. This difference is connected with the ratio of the diameter of the second whorl to that of the first. If an arbitrary numerical value be given to Dr. Gray's degrees of flatness and sharpness: very sharp or sharp and convex, 6 ; sharp but concave, 5 ; sharp, 4 ; medium, 3 ; flat, 2 ; very fiat, 1 : then there is a correlation of ~j-0'482 + 0*071 between these values and the ratio of the diameter of the second whorl to that of the first. This correlation is a practical certainty, though not quite decided (i.e. > -fO'50). The sharp cochlea has the second whorl relatively wide. A better basis of correlation would probably have been a measurement of the real height of the cochlea, i.e. the vertical distance from the level of the under edge of the lowest whorl to the apex of the organ.
We may sum up the data and conclusions thus far by saying that the cochlea is built according to a constant plan, of which the scale alone varies from case to case. This scale shows a decidedly high correlation with the size of the organism as a whole. A change of scale will obviously alter all the dimensions recorded except the number of whorls. But even that number, when it varies independently, does not alter the other dimensions of the cochlea. The only other variant thus far detected is the rate of curvature of the spiral, which is greater in the bigger scale organs.
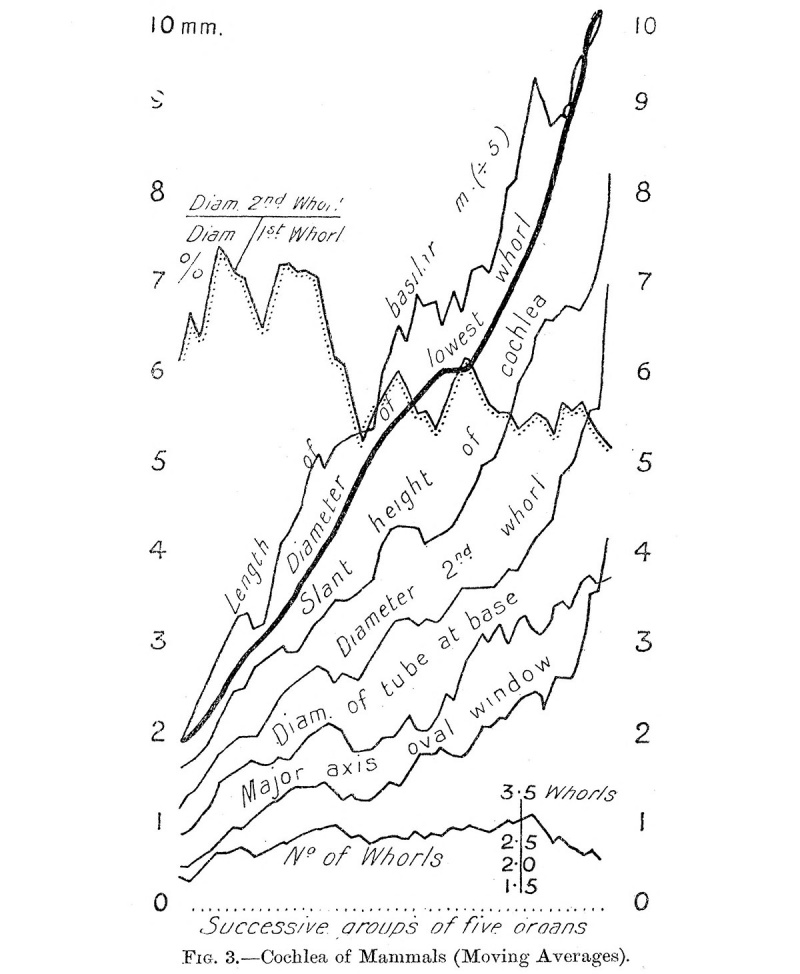
Fig. 3 shows graphically the relation between the various dimensions of the cochlea on the principle of moving averages. The serial arrangement of the organs follows the increase of the diameter of the lowest whorl. The five lowest values were averaged ; then the lowest was omitted and the sixth value in order of size was taken up instead, and the resulting group of five values was averaged ; and so on, through the whole series.
8. For the cochlea of birds Grav gives the measurements of 17 organs of different species. The coefficients of correlation between the three series of measurements given, the major axis of the oval window, the diameter of the tube of the cochlea, and the length of the tube (i.e. including the lagena), appear in Table II. The values are not so high as those of Table I, but there is a clear correlation between the major axis of the oval window and the length of the tube of the cochlea.
Table II The Cochlea of Birds
For the reptiles and amphibians Gray gives the measurements of only four or five organs. One of these— the black pointed teguixin — is clearly of the avian type. This group is distinguished, as Gray says, only by a shorter cochlear tube. It is too small to be treated statistically.
B. The Variations in the Form of the Cochlea
Having thus shown that the mammalian cochlea is an organ of a definite type, we may enquire whether there is any regularity in the way in which the cochleas of different species deviate from the type. In order to ascertain this I divided each measurement of each organ given by Gray (with the exception of " the slant height of the cochlea ") and my estimate of the length of the basilar membrane by the diameter of the lowest whorl of that organ. The resulting values I may call the cochlear indexes of the organ.
Fig. 3. Cochlea of Mammals (Moving Averages).
9. It may be of interest to state the average index build of the mammalian cochlea ; in relation to the diameter of the lowest whorl of the cochlea the proportions are : —
- Second whorl . 0.61
- Diameter of tube in front of round window 0.44
- Major axis oval window 0.32
- Length of basilar membrane 5.65
- Number of whorls (absolute) 2.5 +
In Table III the correlations between the different cochlear indexes are given. The following conclusions may be drawn :—
Table III. Cochlear Indexes
10. There is a high correlation between the number of whorls and the index of the basilar membrane, but not between the former and any other cochlear index. This confirms the conclusion of (5) that variation in. the number of whorls from the typical number two and a half occurs without any alteration in the basal plan of the cochlea ; extra turns are added on at the top of the cochlea by simple continuation of the whorl, or if the turns are fewer than usual, the whorl simply stops short at the required point. The cochlea does not grow by accretions at the base as a shell does, but by accretions at the apex ; if it grew in the former way, all the dimensions of the cochlea would be correlated highly and positively with the number of its whorls, which is not the case; there is not even any correlation between the number of turns and the index of the length of the basilar membrane contained in the first two whorls (vide note to Table III).
11. There is also a- positive- correlation— practically certain, if not quite decided— between the index of the basilar membrane and the index of the second whorl. This evidently means that the basilar membrane may be increased by an increase in the diameter of the second whorl as well as by the addition of further whorls to the number that was more remotely inherited.
This result may seem to contradict that reached above (6), that there is a fair sign of a negative correlation between the absolute length of the basilar membrane or the diameter of the lowest whorl and the index of the second whorl (diameter of the second whorl divided by that of the lowest whorl). But there is really no contradiction. For there is no correlation between the absolute length of the basilar membrane and the index of the basilar membrane. Therefore there may well be an increased rate of curvature for absolutely long basilar membranes (large cochleas) and a decreased rate for relatively long ones.
12. When the index of the second whorl varies, the index of the diameter of the tube of the cochlea varies in the same direction. This is a practically certain and almost decided correlation. We might well expect a cochlear tube that is thicker than usual to coil less willingly, as it were, and so to give a wide second whorl. If the other dimensions of the cochlea, including the number of whorls, then remained typical, the basilar membrane would be longer for that organ than it usually is for any organ.
13. The correlation between the index of the basilar membrane and that of the diameter of the cochlear tube at its base is low and uncertain, being only three times the probable error. This would confirm the inferences made in (11) and (12) that in these connections the diameter of the cochlear tube, not the basilar membrane, is the leading variant.
14. This is further supported by the correlation indicated between the indexes of the major axis of the oval window and of the cochlear tube. There is no correlation between the former and the index of the basilar membrane.
We may sum. up again by saying that there are two sources of change in the length of the basilar membrane. The chief one is its own absolute increase in length, which appears in a greater number of whorls than usual. The other is the relative increase in the diameter of the tube of the cochlea. There are no other internal variations in the dimensions of the cochlea than these.
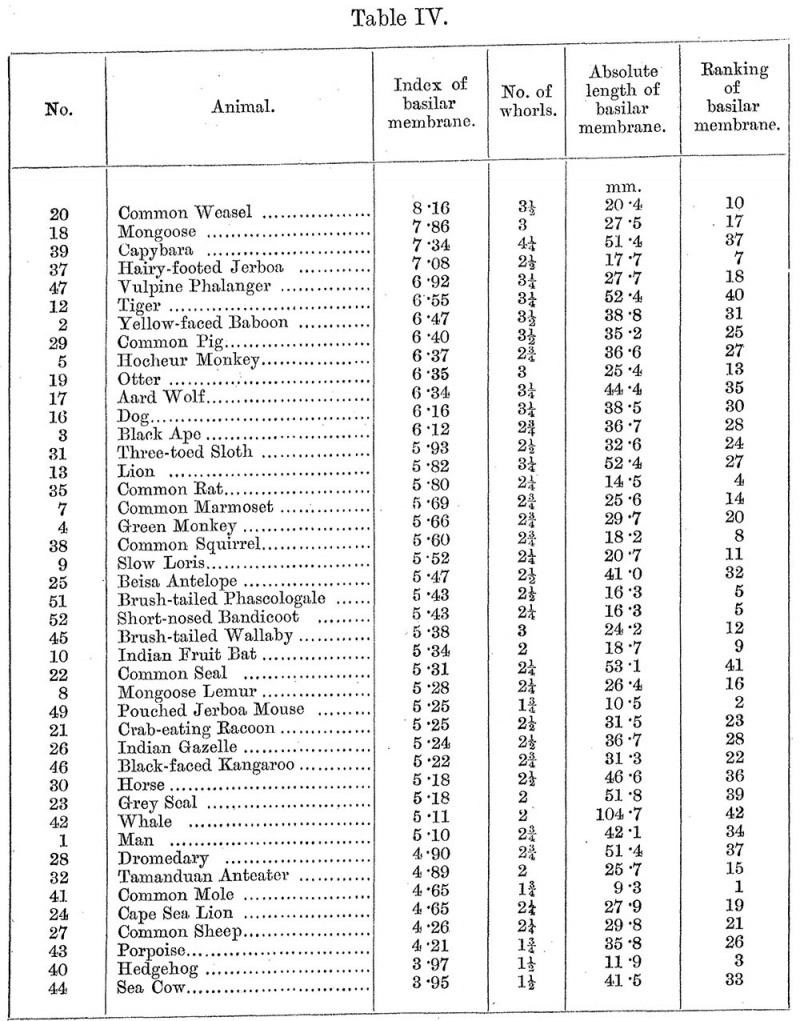
15. In Table IY will be found the ranking of the mammals according to the index of the basilar membrane. The column marked " No." gives the order of succession in which the organs measured are given in Dr. Gray's two volumes, but Dr. Gray himself numbered only the somewhat smaller number of photographs in his books. The absolute length of the basilar membrane is my estimate from Gray’s photographs. For the other measurements upon which this paper is based, see Gray's two volumes.
Table IV.
This ranking can hardly be taken strictly as it stands, but it will be valid on broader lines. Half the number of organs measured have an index of five and a fraction. An index of over 6*5 may safely be held to be unusually large, an index of below 4*5 unusually small. I should divide the whole group into two parts at the, index of 5*5 ; that gives two rather characteristic groups, in the first of which are many carnivora and some much hunted animals, while the lower group consists mainly of herbivorous, insectivorous, and aquatic creatures.
It is of interest to note how low in the scale man stands. He is associated with the horse, the whale, the dromedary, the mole, the sheep, the hedgehog, and the sea-cow. Whatever an increased basilar membrane means functionally for hearing, whether it be a greater sensitivity or a greater pitch-range of hearing, or both, man can hardly claim to have any high power therein, if we may judge from the general impression of the power given by his associates in this Table. Amongst Dr. Gray's photographs there are depicted many more regular and beautiful organs than man's. Music may require a great mind and a great soul, but it can hardly presuppose a very fine receptor organ.
[16. In Table IV the positive correlation between the index of the basilar membrane and the number of whorls (cf. Table III) may be seen at a glance. But minor deviations from it are also apparent ; for example, one might have expected the capybara with 4.25 turns to have the highest basilar index. And it is striking that the next organ on the list has only 2.5 turns. Obviously some other variant must have caused these " short-time oscillations," so to speak. The measurements given for the capybara's organ suggest a likely cause. The photograph of that organ shows a high rate of curvature, the relation of the diameter of the second whorl to that of the first being only 64 per cent. So I reckoned a short-time fluctuation in relation to rate of curvature with the help of moving averages (groups of five organs) and the modifications of Karl Pearson's formula used in connection therewith, and found it to be —0.474 + 0.083. That is very nearly a decided correlation. And it indicates that an increased rate of curvature calls for more whorls, a wider second whorl for fewer whorls, than would generally be required by the functional demands summed together in the index of the basilar membrane.
This result might have been deduced from the results stated in Park A. For if extra whorls are added on at the apex without alteration of the other dimensions, the extra amount of basilar membrane required in a cochlea with a wide second whorl will go into fewer turns than in a cochlea with a narrow second whorl. But it is well to derive it independently, for it clarifies the functional significance of the basilar index, showing it to be independent of the other variable features, and helps towards a reasoned understanding of the architectonic of the cochlea. — Added December, 1916.]
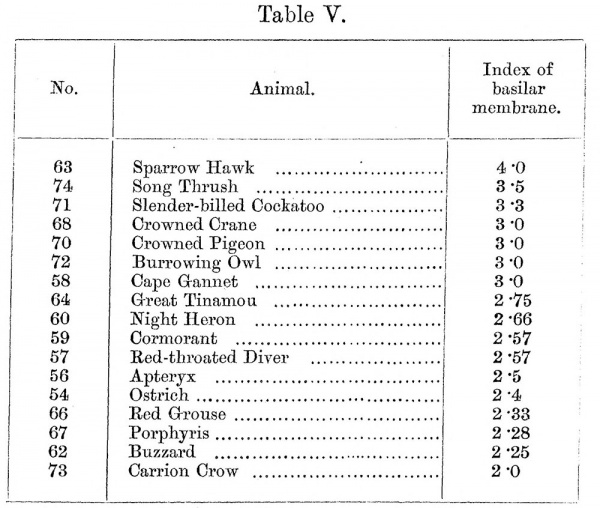
17. Table V gives the ranking for the birds, if an index is taken from the highest correlation— the length of the tube of the cochlea divided by the major axis of the oval window. Here, again, a carnivore heads the list, followed appropriately by the song thrush. By this method of calculation the ducked-billed platypus, which has a cochlea of approximately avian type, shows an index of only 2*0— the lowest in the Table.
Table V.
C. General Remarks
How do these results modify the generally accepted notions regarding the mode of action of the cochlea ? The chief point is that they clear up our ideas. The cochlea, it may be said, has usually been vaguely apprehended as a curious organ, and, like the sensations it yields, rather unique in its nature. But just as a renewed psychological analysis of the sensations of sound brings them into clear accord with the structure of the sensations of the other senses,[2] so this study brings the cochlea morphologically into line with the eye. Each is an organ in which certain main relationships hold universally, with minor variations for special purposes. The cochlea is perhaps more dependent upon the gross bulk of the organism than is the eye.
Little is yet known directly about the auditory functions of the different animals. But the series given under " Animal " in Table IV seems more acceptable as an indication of auditory power than that formed by increase in the absolute length of the basilar membrane. We should hardly expect the finest hearing in the whale, the seal, (the tiger), the 'grey seal, the dromedary, (the capybara), the horse, (the aard-wolf, man ?), and the sea-cow which is the order of decreasing cochlear dimensions. And the close correlation of cochlear sizes with gross bulk makes any such assumption highly untenable. If this he granted, then it would seem that the absolute length of the basilar membrane (and, therewith, the scale of dimensions of the cochlea as a whole) is neither relatively, nor perhaps at all, important for efficiency of hearing. It is rather the index of the basilar membrane that counts, its length relatively to the size of the organ as a whole. This conclusion is supported by the fact elucidated above (10) that the chief internal variation of cochlear structure is the number of whorls or the relative length of the basilar membrane.
But if we thus abrogate the importance of the absolute length of the basilar membrane, may we not also sacrifice the absolute breadth of it as well, and within limits the absolute number of its transverse fibres ? These limits seem inevitable, in so far as pitch discrimination could hardly be refined much beyond the subdivision given by the transverse fibres ; but in a larger organ it need not approach even within some distance of that sub-division. I have elsewhere (op. cit.) tried to prove that the most important property of the basilar membrane in its longitudinal aspect is its elasticity ; and that a functional or psychical hearing, in all respects similar or parallel to our own, in so far as its sensory integrations are concerned, might be got from any absolute length of basilar membrane. The perfection of its efficiency would depend only upon the elasticity of the membrane and its relative length. The results of this paper bear well with this theory. On the other hand, it must be noted that these results are not directly incompatible with the hypotheses of any of the chief physiological theories of hearing.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Cite this page: Hill, M.A. (2024, April 26) Embryology Paper - 1917 The Typical Form of the Cochlea and Its Variations. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_1917_The_Typical_Form_of_the_Cochlea_and_Its_Variations
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G