Introduction
The opossums are a family of nocturnal semi-arboreal marsupials including the cuscuses, brushtail possums, and their close relatives.
Some Recent Findings
- Sequence alteration in the enhancer contributes to the heterochronic Sox9 expression in marsupial cranial neural crest[2] "Neonates of marsupial mammals are altricial at birth, because their gestation period is relatively short compared to placental mammals. Yet, as they need to travel to the teat from the birth canal, and suckle on the mother's milk, forelimbs and jaws develop significantly early. Previous studies in opossum (Monodelphis domestica), an experimental marsupial model, have revealed that cranial neural crest cells are generated significantly early compared to those in placental mammals, such as mouse, leading to an early development of jaw primordia. We have previously found that Sox9, an important neural crest-specifier gene, is expressed in the future cranial neural crest of the opossum embryonic ectoderm significantly earlier than that in mouse or quail embryos. As Sox9 is essential for neural crest formation in various vertebrates, it seems likely that the heterochronic expression of Sox9 is critical for the early cranial neural crest formation in the marsupial embryos. In this study, we show a marsupial-specific sequence in the Sox9 neural crest enhancer E3. We also reveal that the mouse E3 enhancer is activated in the cranial neural crest cells of quail embryos, that the E3 enhancer with marsupial-specific sequence is activated earlier in the Pax7-expressing neural border prior to the onset of endogenous Sox9 expression, and that a misexpression of cMyb, which is also a transcriptional activator of Pax7, in the neural border can ectopically activate the "marsupialized" enhancer. Thus, we suggest that the modification of the E3 enhancer sequence in the marsupial ancestor would have promoted the early expression of Sox9 in the neural border, facilitating the early formation of the cranial neural crest cells and the subsequent heterochronic development of the jaw primordia."
- Gene expression profiling of postnatal lung development in the marsupial gray short-tailed opossum (Monodelphis domestica) highlights conserved developmental pathways and specific characteristics during lung organogenesis[3] "After a short gestation, marsupials give birth to immature neonates with lungs that are not fully developed and in early life the neonate partially relies on gas exchange through the skin. Therefore, significant lung development occurs after birth in marsupials in contrast to eutherian mammals such as humans and mice where lung development occurs predominantly in the embryo. To explore the mechanisms of marsupial lung development in comparison to eutherians, morphological and gene expression analysis were conducted in the gray short-tailed opossum (Monodelphis domestica)."
- Embryo implantation evolved from an ancestral inflammatory attachment reaction[4] "The molecular changes that support implantation in eutherian mammals are necessary to establish pregnancy. In marsupials, pregnancy is relatively short, and although a placenta does form, it is present for only a few days before parturition. However, morphological changes in the uterus of marsupials at term mimic those that occur during implantation in humans and mice. We investigated the molecular similarity between term pregnancy in the marsupials and implantation in eutherian mammals using the gray short-tailed opossum (Monodelphis domestica) as a model. ...We demonstrate that key markers of implantation, including Heparin binding EGF-like growth factor and Mucin 1, exhibit expression and localization profiles consistent with the pattern observed during implantation in eutherian mammals. Finally, we show that there are transcriptome-wide similarities between the opossum attachment reaction and implantation in rabbits and humans. Our data suggest that the implantation reaction that occurs in eutherians is derived from an attachment reaction in the ancestral therian mammal which, in the opossum, leads directly to parturition. Finally, we argue that the ability to shift from an inflammatory attachment reaction to a noninflammatory period of pregnancy was a key innovation in eutherian mammals that allowed an extended period of intimate placentation." implantation
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Opossum Embryology | Opossum Development
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- On the prenatal initiation of T cell development in the opossum Monodelphis domestica[5] "Thymus-dependent lymphocytes (T cells) are a critical cell lineage in the adaptive immune system of all jawed vertebrates. In eutherian mammals the initiation of T cell development takes place prenatally and the offspring of many species are born relatively immuno-competent. Marsupials, in contrast, are born in a comparatively altricial state and with a less well developed immune system. As such, marsupials are valuable models for studying the peri- and postnatal initiation of immune system development in mammals. Previous results supported a lack of prenatal T cell development in a variety of marsupial species. In the gray short-tailed opossum, Monodelphis domestica, however, there was evidence that αβT cells were present on postnatal day 1 and likely initiated development prenatally. Demonstrated here is the presence of CD3ε+ lymphocytes in late-stage embryos at a site in the upper thoracic cavity, the site of an early developing thymus. CD3ε+ cells were evident as early as 48 h prior to parturition. In day 14 embryos, where there is clear organogenesis, CD3ε+ cells were only found at the site of the early thymus, consistent with no extra-thymic sites of T cell development in the opossum. These observations are the first evidence of prenatal T cell lineage commitment in any marsupial." blood | thymus
- A new mammalian model system for thalidomide teratogenesis: Monodelphis domestica [6] "From 1957 to 1962, thalidomide caused birth defects in >10,000 children. While the drug was pulled from the market, thalidomide is currently prescribed to treat conditions including leprosy. As a result, a new generation of babies with thalidomide defects is being born in the developing world. This represents a serious problem, as the mechanisms by which thalidomide disrupts development remain unresolved. This lack of resolution is due, in part, to the absence of an appropriate mammalian model for thalidomide teratogenesis. We test the hypothesis that opossum (Monodelphis domestica) is well suited to model human thalidomide defects. Results suggest that opossum embryos exposed to thalidomide display a range of phenotypes (e.g., heart, craniofacial, limb defects) and penetrance similar to humans. Furthermore, all opossums with thalidomide defects exhibit vascular disruptions. Results therefore support the hypotheses that opossums make a good mammalian model for thalidomide teratogenesis, and that thalidomide can severely disrupt angiogenesis in mammals." thalidomide
- Comparative gene expression analyses reveal heterochrony for Sox9 expression in the cranial neural crest during marsupial development[7] "Compared to placental mammals, marsupials have short gestation period, and their neonates are relatively immature. Despite these features, marsupial neonates must travel from the birth canal to the teat, suckle and digest milk to complete development. Thus, certain organs and tissues of marsupial neonates, such as forelimbs to crawl and jaw elements to suckle, must develop early. Previous reports showed that cranial neural crest (CNC) cells, as the source of ectomesenchyme of jaw elements, are generated significantly early in gray short-tailed opossum (Monodelphis domestica) compared to other amniote models, such as mouse. In this study, we examined the expression of genes known to be important for neural crest formation, such as BMP2/BMP4 (neural crest inducer), Pax7 (neural border specifier), Snail1 and Sox9/Sox10 (neural crest specifier) in Monodelphis domestica, and compared the expression patterns with those in mouse, chicken, and gecko embryos. Among those genes, the expression of Sox9 was turned on early and broadly in the premigratory CNC cells, and persisted in the ectomesenchyme of the cranial anlagen in opossum embryos. In contrast, Sox9 expression diminished in the CNC cells of other animals at the early phase of migration. Comparison of the onset of Pax7 and Sox9 expression revealed that Sox9 expression in the prospective CNC was earlier and broader than Pax7 expression in opossum, suggesting that the sequence of border specification and neural crest specification is altered."
- Developmental origins of precocial forelimbs in marsupial neonates[8] "Marsupial mammals are born in an embryonic state, as compared with their eutherian counterparts, yet certain features are accelerated. The most conspicuous of these features are the precocial forelimbs, which the newborns use to climb unaided from the opening of the birth canal to the teat. The developmental mechanisms that produce this acceleration are unknown. Here we show that heterochronic and heterotopic changes early in limb development contribute to forelimb acceleration. Using Tbx5 and Tbx4 as fore- and hindlimb field markers, respectively, we have found that, compared with mouse, both limb fields arise notably early during opossum development."
|
Taxon
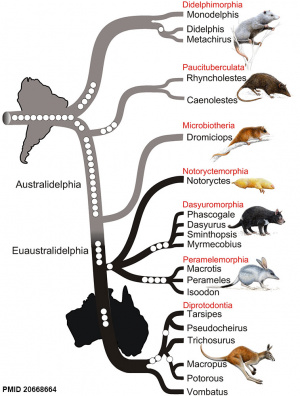
Phylogenetic tree of marsupialsNilsson MA, Churakov G, Sommer M, Tran NV, Zemann A, Brosius J & Schmitz J. (2010). Tracking marsupial evolution using archaic genomic retroposon insertions.
PLoS Biol. , 8, e1000436. PMID:
20668664 DOI.
Taxonomy ID:
Genbank common name: opossum
Rank: species
Genetic code: Translation table 1 (Standard)
Mitochondrial genetic code: Translation table 2 (Vertebrate Mitochondrial)
Lineage ( full )
- Links: Taxonomy Browser Phalangeridae
| Marsupial - Opossum Taxon List
|
| Taxon Name
|
Common Name
|
| Chacodelphys formosa
|
Chacoan pygmy opossum
|
| Gracilinanus marica
|
northern gracile opossum
|
| Marmosops juninensis
|
Junin slender opossum
|
| Marmosops ojastii
|
Ojasti's slender mouse opossum
|
| Marmosops ocellatus
|
spectacled slender opossum
|
| Marmosops creightoni
|
Voss' slender opossum
|
| Marmosops bishopi
|
Bishop's slender opossum
|
| Marmosops fuscatus
|
dusky slender opossum
|
| Cryptonanus guahybae
|
Guahiba gracile opossum
|
| Monodelphis palliolata
|
hooded red-sided opossum
|
| Monodelphis gardneri
|
Gardner's short-tailed opossum
|
| Caenolestes convelatus
|
northern shew opossum
|
| Caenolestes sangay
|
Sangay shrew opossum
|
| Marmosops paulensis
|
Brazilian slender opossum
|
| Philander opossum
|
fuscogriseus subspecies
|
| Cryptosporidium sp.
|
apicomplexans
|
|
|
| Didelphis pernigra
|
Andean white-eared opossum
|
| Caluromys derbianus
|
Derby's woolly opossum
|
| Monodelphis umbristriata
|
Red three-striped opossum
|
| Monodelphis scalops
|
Long-nosed short-tailed opossum
|
| Monodelphis sorex
|
Southern red-sided opossum
|
| Monodelphis glirina
|
Amazonian red-sided opossum
|
| Thylamys tatei
|
Tate's fat-tailed mouse opossum
|
| Thylamys sponsorius
|
Argentine fat-tailed mouse opossum
|
| Cryptosporidium sp.
|
oposapicomplexans
|
| Micoureus constantiae
|
White-bellied woolly mouse opossum
|
| Marmosa tyleriana
|
Tyleria mouse opossum
|
| Marmosa xerophila
|
Guajira mouse opossum
|
| Monodelphis americana
|
Northern three-striped opossum
|
| Trichosurus cunninghami
|
Mountain brushtail opossum
|
| Monodelphis iheringi
|
Ihering's three-striped opossum
|
| Caenolestes caniventer
|
grey-bellied shrew opossum
|
| Marmosops invictus
|
Panama slendeer opossum
|
| Didelphis imperfecta
|
Guianan white-eared opossum
|
| Thylamys velutinus
|
Dwarf fat-tailed mouse opossum
|
| Marmosops handleyi
|
Handley's slender mouse opossum
|
| Monodelphis osgoodi
|
Osgood's short-tailed opossum
|
| Hyladelphys kalinowskii
|
Kalinowski's opossum
|
| Gracilinanus aceramarcae
|
Aceramarca gracile mouse opossum
|
| Gracilinanus emiliae
|
Emilia's gracile mouse opossum
|
| Thylamys cinderella
|
Cinderella fat-tailed opossum
|
| Micoureus paraguayanus
|
Tate's wolly mouse opossum
|
| Tlacuatzin canescens
|
grayish mouse opossum
|
| Philander frenatus
|
Southeastern four-eyed oppossum
|
| Marmosops incanus
|
gray slender mouse opossum
|
| Monodelphis theresa
|
Theresa's short-tailed opossum
|
| Marmosa mexicana
|
Mexican mouse opossum
|
| Marmosa andersoni
|
Anderson's mouse opossum
|
| Monodelphis kunsi
|
Pygmy short-tailed Opossum
|
| Monodelphis brevicaudata
|
Red-legged short-tailed Opossum
|
| Thylamys pusillus
|
Common fat-tailed mouse opossum
|
| Thylamys macrura
|
long-tailed fat-tailed opossum
|
| Thylamys elegans
|
elegant fat-tailed opossum
|
| Gracilinanus agilis
|
Agile Gracile Mouse Opossum
|
| Thylamys pallidior
|
pallid fat-tailed opossum
|
| Monodelphis emiliae
|
Emilia's short-tailed opossum
|
| Marmosops parvidens
|
delicate slender mouse opossum
|
| Marmosops noctivagus
|
white-bellied slender mouse opossum
|
| Marmosa murina
|
murine mouse opossum
|
| Marmosa lepida
|
little rufous mouse opossum
|
| Lestodelphys halli
|
Patagonian opossum
|
| Gracilinanus microtarsus
|
Brazilian gracile mouse opossum
|
| Caluromysiops irrupta
|
black-shouldered opossum
|
| Gnathostoma sp.
|
from P.
|
| Chironectes minimus
|
water opossum
|
| Marmosops dorothea
|
Dorothy's slender mouse opossum
|
| Marmosa robinsoni
|
Robinson's mouse opossum
|
| Didelphis aurita
|
big-eared opossum
|
| Caluromys philander
|
bare-tailed woolly opossum
|
| Lestoros inca
|
Incan shrew opossum
|
| Philander opossum
|
Philander opossum
|
| Monodelphis adusta
|
sepia short-tailed opossum
|
| Metachirus nudicaudatus
|
brown four-eyed opossum
|
| Marmosops impavidus
|
Andean slender mouse opossum
|
| Micoureus regina
|
Short-furred woolly mouse opossum
|
| Micoureus demerarae
|
long-furred woolly mouse opossum
|
| Marmosa rubra
|
red mouse opossum
|
| Didelphis albiventris
|
white-eared opossum
|
| Glironia venusta
|
bushy-tailed opossum
|
| Caluromys lanatus
|
western woolly opossum
|
| Lutreolina crassicaudata
|
thick-tailed opossum
|
| Caenolestes fuliginosus
|
silky shrew opossum
|
| Rhyncholestes raphanurus
|
Chilean shrew opossum
|
| Monodelphis domestica
|
gray short-tailed opossum
|
| Philander opossum
|
gray four-eyed opossum
|
| Didelphis marsupialis
|
southern opossum
|
| Didelphis virginiana
|
North American opossum
|
|
Development Overview
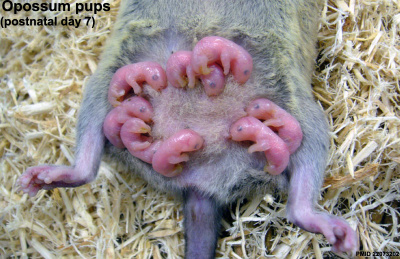
Opossum and day 7 pups[1]
System Development
The marsupial neonate at birth has a variation between the degree of development of different systems.[9]
- well-developed - digestive, respiratory and circulatory system.
- not well-developed - retains fetal excretory system with a fully functional mesonephric kidney and undifferentiated gonads and genitalia.
Oocyte
Ovarian Follicle Development
Marsupial eggs are enclosed by a series of layers:[10]
- zona pellucida, three zona proteins (ZPA, ZPB, ZPC)
- an additional extracellular matrix coat that lines the zona pellucida also occurs in some species.
- mucoid coat
- outer shell coat.
- Links: oocyte
Spermatozoa
- Links: spermatozoa
References
- ↑ 1.0 1.1 Wheaton BJ, Callaway JK, Ek CJ, Dziegielewska KM & Saunders NR. (2011). Spontaneous development of full weight-supported stepping after complete spinal cord transection in the neonatal opossum, Monodelphis domestica. PLoS ONE , 6, e26826. PMID: 22073202 DOI.
- ↑ Wakamatsu Y & Suzuki K. (2019). Sequence alteration in the enhancer contributes to the heterochronic Sox9 expression in marsupial cranial neural crest. Dev. Biol. , 456, 31-39. PMID: 31430446 DOI.
- ↑ Modepalli V, Kumar A, Sharp JA, Saunders NR, Nicholas KR & Lefèvre C. (2018). Gene expression profiling of postnatal lung development in the marsupial gray short-tailed opossum (Monodelphis domestica) highlights conserved developmental pathways and specific characteristics during lung organogenesis. BMC Genomics , 19, 732. PMID: 30290757 DOI.
- ↑ Griffith OW, Chavan AR, Protopapas S, Maziarz J, Romero R & Wagner GP. (2017). Embryo implantation evolved from an ancestral inflammatory attachment reaction. Proc. Natl. Acad. Sci. U.S.A. , 114, E6566-E6575. PMID: 28747528 DOI.
- ↑ Hansen VL & Miller RD. (2017). On the prenatal initiation of T cell development in the opossum Monodelphis domestica. J. Anat. , 230, 596-600. PMID: 28052333 DOI.
- ↑ Sorensen D, Sackett A, Urban DJ, Maier J, Vargesson N & Sears KE. (2017). A new mammalian model system for thalidomide teratogenesis: Monodelphis domestica. Reprod. Toxicol. , 70, 126-132. PMID: 28130151 DOI.
- ↑ Wakamatsu Y, Nomura T, Osumi N & Suzuki K. (2014). Comparative gene expression analyses reveal heterochrony for Sox9 expression in the cranial neural crest during marsupial development. Evol. Dev. , 16, 197-206. PMID: 24934187 DOI.
- ↑ Keyte AL & Smith KK. (2010). Developmental origins of precocial forelimbs in marsupial neonates. Development , 137, 4283-94. PMID: 21098569 DOI.
- ↑ Renfree MB, Pask AJ & Shaw G. (2001). Sex down under: the differentiation of sexual dimorphisms during marsupial development. Reprod. Fertil. Dev. , 13, 679-90. PMID: 11999321
- ↑ Selwood L. (2000). Marsupial egg and embryo coats. Cells Tissues Organs (Print) , 166, 208-19. PMID: 10729728 DOI.
Reviews
Articles
Books
Search PubMed
Search PubMed: Dromiciops gliroides | Opossum development | marsupial development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 24) Embryology Opossum Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Opossum_Development
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G



