Endocrine - Gonad Development
| Embryology - 1 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The term gonad refers to both the female ovary and the male testis, both have their own detailed topic pages. This section of notes refers only to the development of the gonad as an endocrine organ.
Embryonically, initial endocrine development of the testis is required for development of both the internal genital tract and the external genitalia.
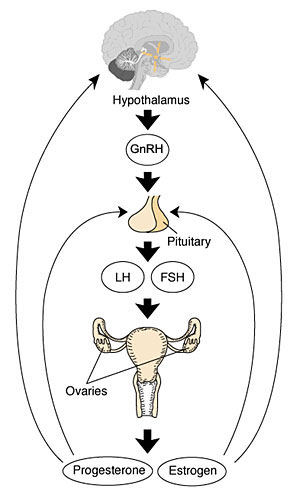
Postnatally, the gonads are part of an integrated Hypothalamus - Pituitary - Gonad (HPG) axis. Post-puberty, this axis is the key regulator of the female reproductive cycle, the menstrual cycle.
| Menstrual Cycle Links: Introduction | menstrual histology | ovary | corpus luteum | oocyte | uterus | Uterine Gland | estrous cycle | pregnancy test | ||
|
Genital Links: genital | Lecture - Medicine | Lecture - Science | Lecture Movie | Medicine - Practical | primordial germ cell | meiosis | endocrine gonad | Genital Movies | genital abnormalities | Assisted Reproductive Technology | puberty | Category:Genital
| ||||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Endocrine Gonad Development | Testosterone Embryology | Granulosa Cell Embryology | Leydig Cell Embryology | Interstitial Cell Embryology |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
HPG Axis - Endocrinology - Simplified diagram of the actions of gonadotrophins
Gonad Development
- mesoderm - mesothelium and underlying mesenchyme, primordial germ cells
- Gonadal ridge - mesothelium thickening, medial mesonephros
- Primordial Germ cells - yolk sac, to mesentery of hindgut, to genital ridge of developing kidney
Differentiation
- testis-determining factor (TDF) from Y chromosome: presence (testes), absence (ovaries)
Testis
- 8 Weeks, mesenchyme, interstitial cells (of Leydig) secrete testosterone, androstenedione
- 8 to 12 Weeks - hCG stimulates testosterone production
- Sustentacular cells - produce anti-mullerian hormone to puberty
Ovary
- X chromosome genes regulate ovary development
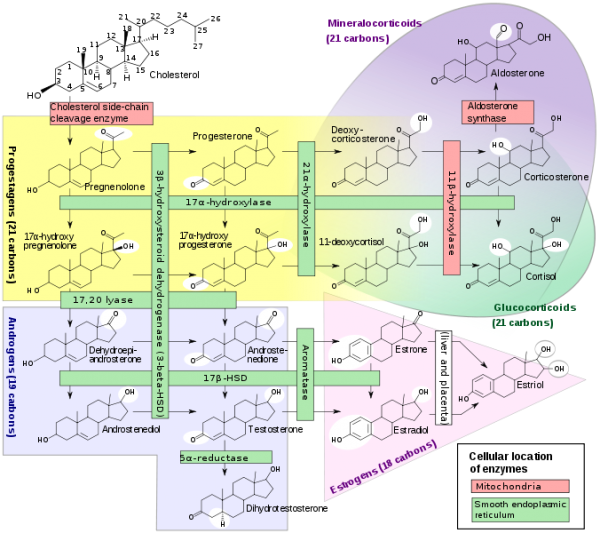
Steroidogenesis
Testosterone
Testicular Leydig cells (interstitial cells) are the main source of testosterone in males.
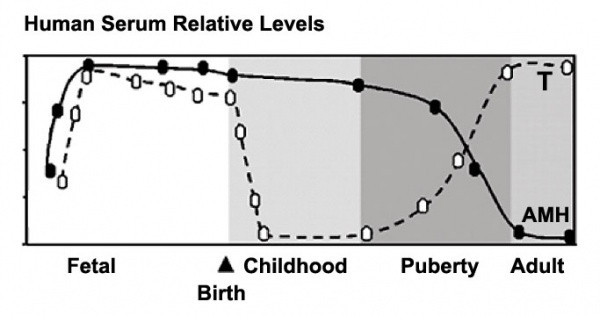
Human Male Testosterone and Anti-Müllerian Hormone (AMH) relative levels[5]
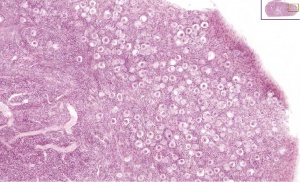
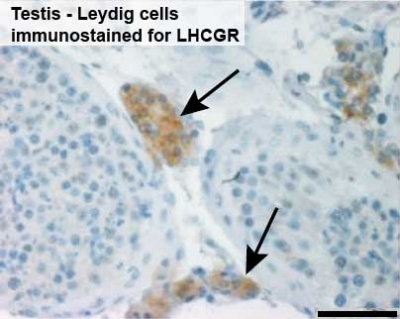
Leydig Cells
Leydig cells stained for LHCGR1[6]
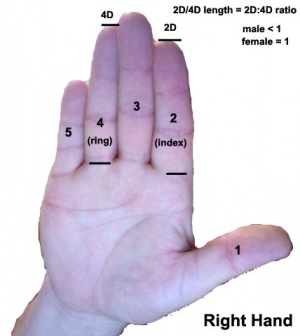
Androgen and Digit ratio (2D:4D)
The ratio of 2nd and 4th finger (D, digit) length. This ratio has been suggested to relate to high fetal testosterone concentration (males have lower 2D:4D than females) and has been shown for several species.[7] Although a study in mice has not shown the same correlation.[8] There have been some suggestions that the ratio may also be an indicator of various neurological abnormalities.
To measure (2D:4D) - using your right hand palm up, measure the index finger (2) and ring finger (4) length from palm to tip. Dividing the index finger by the ring finger gives the 2D:4D ratio, average women ratio is 1, average men is 0.98.
Anti-Mullerian Hormone
Estrogen
| The estradiol (estrogen, oestrogen) hormone is a steroid sex hormone expressed in both male and female.
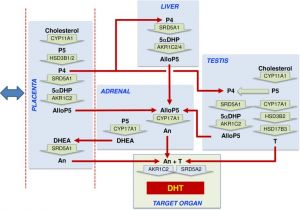
estrogenic activity in human placental extracts was due to the presence of at least three compounds: estriol, estrone, and 17β-estradiol. In the female, this hormone together with progesterone regulate changes that occur each menstrual cycle. During female development the fetal adrenal gland cortex synthesises DHEA (and DHEA/S), an oestrogen precursor (see image Fetal adrenal gland steroidogenesis), converted by the placenta into estrogen compounds; estriol, estrone, and 17β-estradiol. During puberty, ovarian estrogen production is responsible for development of the secondary feminine sex characteristics. In the male, Leydig cells produce estrogen into the rete testis fluid at variable levels in different species. During male embryonic development exposure to high levels of estrogen can lead to genital abnormalities. |
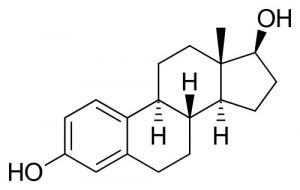
Estradiol |
Progesterone
| The progesterone (progestin) hormone is produced by the granulosa cells of the ovarian follicles at different levels during the menstrual cycle and at high levels by the luteal cells (P4) of the corpus luteum.
|
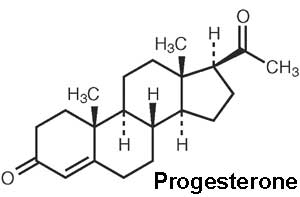
Progesterone molecular structure |
Genital Links: genital | Lecture - Medicine | Lecture - Science | Lecture Movie | Medicine - Practical | primordial germ cell | meiosis | endocrine gonad | Genital Movies | genital abnormalities | Assisted Reproductive Technology | puberty | Category:Genital
| ||||
|
Adult Histology
References
- ↑ O'Shaughnessy PJ, Antignac JP, Le Bizec B, Morvan ML, Svechnikov K, Söder O, Savchuk I, Monteiro A, Soffientini U, Johnston ZC, Bellingham M, Hough D, Walker N, Filis P & Fowler PA. (2019). Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. , 17, e3000002. PMID: 30763313 DOI.
- ↑ Della Torre S, Rando G, Meda C, Ciana P, Ottobrini L & Maggi A. (2018). Transcriptional activity of oestrogen receptors in the course of embryo development. J. Endocrinol. , 238, 165-176. PMID: 30012715 DOI.
- ↑ Li X, Wang Z, Jiang Z, Guo J, Zhang Y, Li C, Chung J, Folmer J, Liu J, Lian Q, Ge R, Zirkin BR & Chen H. (2016). Regulation of seminiferous tubule-associated stem Leydig cells in adult rat testes. Proc. Natl. Acad. Sci. U.S.A. , 113, 2666-71. PMID: 26929346 DOI.
- ↑ Lombardo MV, Ashwin E, Auyeung B, Chakrabarti B, Taylor K, Hackett G, Bullmore ET & Baron-Cohen S. (2012). Fetal testosterone influences sexually dimorphic gray matter in the human brain. J. Neurosci. , 32, 674-80. PMID: 22238103 DOI.
- ↑ Rey R. (2005). Anti-Müllerian hormone in disorders of sex determination and differentiation. Arq Bras Endocrinol Metabol , 49, 26-36. PMID: 16544032 DOI.
- ↑ Kossack N, Simoni M, Richter-Unruh A, Themmen AP & Gromoll J. (2008). Mutations in a novel, cryptic exon of the luteinizing hormone/chorionic gonadotropin receptor gene cause male pseudohermaphroditism. PLoS Med. , 5, e88. PMID: 18433292 DOI.
- ↑ McIntyre MH. (2006). The use of digit ratios as markers for perinatal androgen action. Reprod. Biol. Endocrinol. , 4, 10. PMID: 16504142 DOI.
- ↑ Yan RH, Bunning M, Wahlsten D & Hurd PL. (2009). Digit ratio (2Dratio4D) differences between 20 strains of inbred mice. PLoS ONE , 4, e5801. PMID: 19495421 DOI.
- ↑ Butenandt A. Neure Ergebnisse auf dem Gebiet der Sexualhormone. Wien Klin Wochenschr. 1934;47:936.
- ↑ Slotta KH, Ruschig H, Fels E. Ȕber der Hormon aus dem Corpus-luteum. Ber Chem Ges. 1934;67:1270.
- ↑ Hartmann M, Wettstein A. Ein krystallisiertes Hormon aus Corpus-luteum. Helv Chim Acta. 1934;17:878.
- ↑ Allen WM & Wintersteiner O. (1934). CRYSTALLINE PROGESTIN. Science , 80, 190-1. PMID: 17817057 DOI.
Reviews
Articles
Search PubMed
Search Pubmed: endocrine gonad development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Andrology Australia - Androgen deficiency PDF | Childhood and adolescence. Examination of male genitals and secondary sexual characteristics PDF
Additional Images
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 1) Embryology Endocrine - Gonad Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Gonad_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G