Book - Text-Book of Embryology 15
| Embryology - 26 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Urogenital System
No other system in the body presents such peculiarities of development as the urogenital system. In the first place, it is exceedingly complicated on account of its many parts. It is derived from both mesoderm (mesothelium and mesenchyme) and entoderm. The urinary portion develops into a great complex of ducts for the carrying off of waste products. The genital portion in both sexes becomes highly specialized for the production and carrying off of the sexual elements. In the second place, instead of one set of urinary organs developing and persisting, three sets develop at different stages. The first set (the pronephroi) disappears in part, but leaves certain structures which are used, so to speak, in the development of the second. The second set (the mesonephroi) disappears for the most part, leaving, however, some portions which are taken up in the development of the genital organs and other portions which persist as rudimentary structures in the adult. The third set (the metanephroi or kidneys) develops in part from the second and in part is of independent origin. These conditions" afford one of the most striking examples of the repetition of the phylogenetic history by the ontogenetic, or, in other words, of von Baer's law that an individual, in its development, has a tendency to repeat its ancestral history; for the first and second sets of urinary organs in the human embryo represent systems that are permanent in the lower Vertebrates. In the third place, the ducts of the genital organs are not homologous in the two sexes. In the male the ducts (deferent duct, duct of the epididymis, efferent ductules) are derived from the second set of urinary organs; in the female they (the oviducts) are derived from other ducts which develop in the second set of urinary organs, but which are not functionally a part of the latter.
The Pronephros
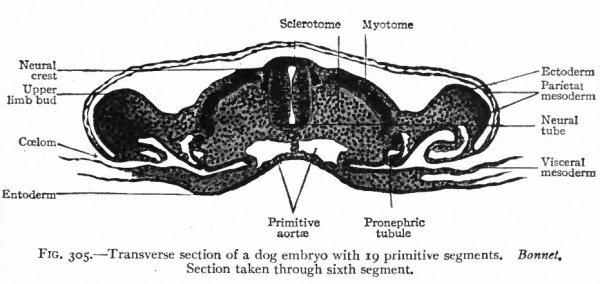
The pronephros, with the pronephric duct, is the first of the urinary organs to appear. In embryos of 2-3 mm. there are two pronephric tubules on each side, situated at the level of the heart. Although their mode of origin has not been observed in the human embryo, it is probable, judging from observations on lower Vertebrates, that they arise as evaginations of the mesothelium. The part of the mesothelium involved is that adjacent to the intermediate cell mass (Fig. 305) . (The intermediate cell mass is the portion of the mesoderm intervening between the primitive segments and the unsegmented parietal and visceral layers; p. 99.) The more cephalic of the two tubules becomes hollow and opens into the ccelom; the more caudal is merely a solid cord of cells. Neither tubule forms any connection with the pronephric duct. At each side of the root of the mesentery a small elevation, which projects into the ccelom, probably represents a rudimentary glomerulus. A glomerulus in the lower Vertebrates, where the pronephros develops to a much greater degree than in Mammals, contains tortuous vessels derived from branches of the aorta (Fig. 306). The mesonephros (p. 359), beginning to develop almost as soon as the pronephros and in the same relative position, forms a ridge which projects into the coelom. The pronephric tubules thus become embedded in the mesonephric ridge.
The pronephric duct begins to develop about the same time as the tubules. It appears as a longitudinal ridge on the outer side of the intermediate cell mass at the level of the heart and projects into the space between the mesoderm and ectoderm. The ridge is at first solid but soon acquires a lumen, and gradually extends to the caudal end of the embryo where it bends medially to open into the gut. The origin of the caudal portion of the duct is a matter of dispute. It comes in contact and fuses with the ectoderm, but whether in the higher animals the fusion is secondary or signifies a derivation from the ectoderm has not been determined. When first formed, the entire duct lies on the outer side of the intermediate cell mass, but later becomes embedded in the mesonephric ridge.
Fig. 305. Transverse section of a dog embryo with 19 primitive segments. Bonnet. Section taken through sixth segment.
The pronephric tubules are but transient structures and have no functional significance in man and the higher Vertebrates. The ducts, however, remain and become the ducts of the second set of urinary organs, the mesonephroi.
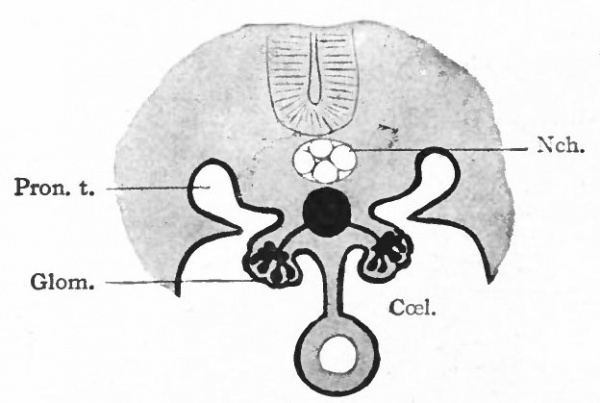
The significance of the pronephros can be understood only by reference to the conditions in the lower animals. In the latter the pronephros acquires a relatively higher degree of development than in the higher forms. The tubules are segmentally arranged and are preset in many segments of the body. They open at their outer ends into the ducts, and at their inner ends into the coelom through ciliated funnel-shaped mouths or nephrostomes. Little masses of mesoderm, containing tortuous vessels derived from branches of the aorta, form glomeruli which project into the ccelom. Waste products are removed from the blood through the agency of the glomeruli and are collected in the ccelom. They are then taken up by the pronephric tubules and carried away by the ducts. In some of the Round Worms there is not even a longitudinal duct, but the tubules open directly on the outer surface of the body. In the lowest Fishes all the tubules on each side open into a longitudinal duct which opens into the cloaca. In these lower forms of animal life the pronephroi constitute the permanent urinary apparatus. In the ascending scale the mesonephroi appear (higher Fishes, Amphibia) and assume the function of carrying off waste products. The pronephroi also develop, but to a lesser degree. Still higher in the scale (Reptiles, Birds, Mammals) the kidneys (metanephroi) appear and the mesonephroi lose their functional significance. But even in the very highest Mammals the pronephroi appear, in a very rudimentary form, in each individual in the earliest embryonic stages, thus repeating the ancestral history.
Fig. 306. Diagram of the pronephric system in an amphibian. Bonnet.
- CceL, Coelom; Glom., glomerulus, containing ramifications of a branch of the aorta; Nch., notochord; Pron. t., pronephric tubule.
The Mesonephros
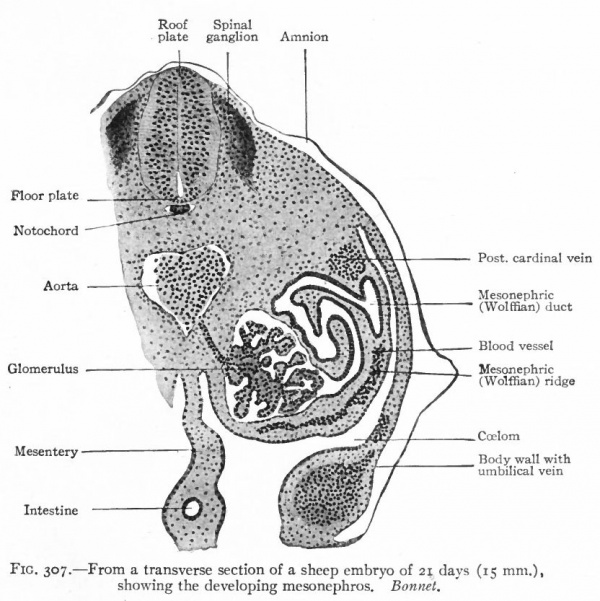
The mesonephroi, which constitute the second set of urinary organs, appear in embryos of 2.6-3.0 mm., immediately following the pronephroi. They begin to develop just caudal to the pronephric tubules and in the same relative position as the latter, that is, in the intermediate cell mass. Condensations* appear in the mesenchyme and become more or less tortuous. At their inner ends they form secondary connections with the mesothelium and at their outer ends they join the pronephric duct which now becomes the mesonephric (or Wolffian) duct. The cells acquire an epithelial character, lumina appear, and the tortuous mesenchymal condensations thus become true tubules. Their connections with the mesothelium soon disappear (Fig. 307).
- *The term " condensation " is here used to mean increased density of tissue due mainly to proliferation of cells.
Fig. 307. From a transverse section of a sheep embryo of 21 days (15 mm.) Showing the developing mesonephros. Bonnet.
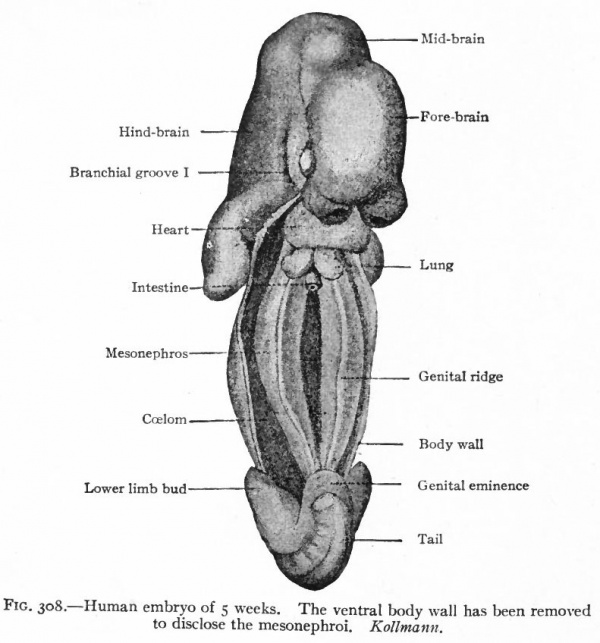
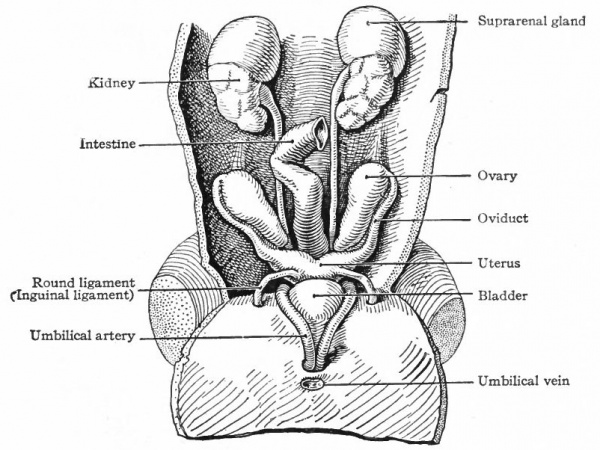
After the tubules are formed, other condensations of the mesenchyme appear near their inner ends. A branch from the aorta enters each condensation and breaks up into a number of smaller vessels which ramify inside, the entire structure thus becoming a glomerulus. Each glomerulus pushes against the corresponding tubule, the latter becoming flattened and then growing around the glomerulus. In this way the glomerulus becomes surrounded by two layers of epithelium, except at the point where the vessels enter, and the whole structure the Malpighian corpuscle resembles very closely a renal corpuscle of the adult kidney. Waste products are removed from the blood through the agency of the glomeruli and are carried to the ducts by the mesonephric tubules (Fig. 307). The tubules themselves increase in length and become much coiled. Secondary and tertiary tubules also develop and become branches of the primary. Whether these develop from condensations of the mesenchyme or as buds from the primary tubules has not been determined. Each tubule consists of two parts (1) a dilated part around the glomerulus, composed of large flat cells and forming Bowman's capsule, and (2) a narrower coiled part leading from the glomerulus to the duct and composed of smaller cuboidal cells (Fig. 307). The primary mesonephric tubules are arranged segmentally, one appearing in each segment as far back as the pelvic region. Thus the intermediate cell mass may be considered as a series of nephrotomes, corresponding to the sclerotomes and myotomes. The segmental character is soon lost, however, owing to the inequality of growth between the mesonephros and the other segmental structures, and to the development of the secondary and tertiary tubules. As stated above, the first mesonephric tubules appear immediately caudal to the pronephros From this point their formation gradually progresses in a caudal direction as far as the pelvic region. By the further development of the primary and by the addition of the secondary and tertiary tubules and the glomeruli, the mesonephros as a whole increases in size and forms a large structure which projects into the ccelom on each side of the body, forming the so-called mesonephric or Wolffian ridge. It reaches the height of its development in the human embryo about the fifth or sixth week, at which time it extends from the region of the heart to the pelvic region (Fig. 308). Each organ is attached to the dorsal body wall by a distinct mesentery which, at its cephalic end, also sends off a band to the diaphragm the diaphragmatic ligament of the mesonephros. The peritoneum is reflected over the surface of the mesonephros, and on the ventro-medial side the mesothelium becomes thickened to form the genital ridge (p. 374; Figs. 276 and 308). The mesonephric ducts are embedded in the lateral parts of the organs and extend throughout practically T their entire length. Since the ducts are identical with the pronephric ducts, they open at first into the caudal end of the gut, or cloaca (p. 355; Fig. 322). At a little later period, when the urogenital sinus is formed, they open at the junction of the latter with the bladder (Fig. 325). Still later they open into the sinus itself (p. 370) . A description of their further development is best deferred to the section on the male genital organs, since they become the genital ducts (p. 386).
Fig. 308. Human embryo of 5 weeks. The ventral body wall has been removed to disclose the mesonephroi. Kollmann.
Fig. 309. Diagram representing certain persistent portions of the mesonephros in the male (see table). Kottmann.
The mesonephroi function as urinary organs during the period of their existence in the embryos of all higher Vertebrates. Excretory products are conveyed directly to the tubules by means of the glomeruli instead of being deposited in the ccelom and then taken up by the tubules, as is the case in functional pronephroi (p. 356). The main excretory ducts are the same as in the pronephroi. Aside from the vessels in the glomeruli the mesonephroi are exceedingly vascular organs. Large and small branches of the posterior cardinal veins ramify among the tubules (Figs. 276 and 194). The blood undergoes purifying processes in its close contact with the tubules and is returned to the heart by the posterior cardinals, or, after the cephalic ends of the latter atrophy, by the subcardinals and the inferior vena cava (see p. 225; also Fig. 194, B). There is thus present a true renal portal system, similar to the hepatic portal system.
Although the mesonephroi become large functional organs during the earlier stages of development, they atrophy and disappear for the most part, coincidently with the appearance and development of the kidneys. The degeneration begins during the sixth or seventh week and goes on rapidly until, by the end of the fourth month, little remains but the ducts and a few tubules. The degenerative processes consist of (1) an ingrowth of connective tissue among the tubules, (2) atrophy of the epithelium of the tubules, and (3) atrophy of the glomeruli, The portions which remain differ in the two. sexes, and since the remnants are taken up in the formation of the male and female genital organs it seems best to discuss them more fully under those heads (pp. 383,386). The accompanying table, however, will give a clue to their fate (see also Figs. 309 and 310). A more comprehensive table will be found on p. 393.
Fig. 310. Diagram representing certain persistent portions of the mesonephros in the female (see table).
- Epo. /., Longitudinal duct of the epoophoron; Epo. t., transverse ductules of the epoophoron; O. t. a. t ostium abdominale tubae; Ovd., oviduct; X represents a small duct which, if present, leads from the epoophoron to one of the fimbriae of the oviduct.
The significance of the mesonephroi, which, as well as the pronephroi, are present in the embryos of ail higher Vertebrates, can be understood only by referring to the conditions in the lower Vertebrates. In the majority of the Fishes and in the Amphibia the mesonephroi constitute the functional urinary organs of the adult and possess essentially the same structure in the embryos of higher forms. Beginning in the Reptiles and continuing up through the series of Birds and Mammals, another set of urinary organs the kidneys develops. The mesonephroi also develop in these forms, even to a high degree, thus repeating the ancestral history, but retain their original function only in the earlier embryonic stages.
The Kidney (Metanephros)
The kidneys are the third set of urinary organs to develop. They assume the function of the mesonephroi as the latter atrophy, and constitute the permanent urinary apparatus. Each kidney is derived from two separate anlagen which unite secondarily. The epithelium of the ureter, renal pelvis, and straight renal tubules (collecting tubules) is derived from the mesonephric duct by a process of evagination. The convoluted renal tubules and glomeruli are derived directly from the mesenchyme, and in this respect resemble the mesonephric tubules and glomeruli.
Fig. 311. From a reconstruction of the anlage of the kidney (metanephros) of a human embryo at the beginning of the 5th week. Schreiner.
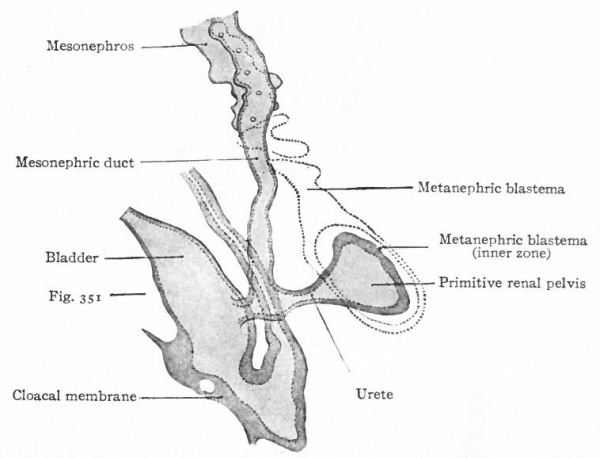
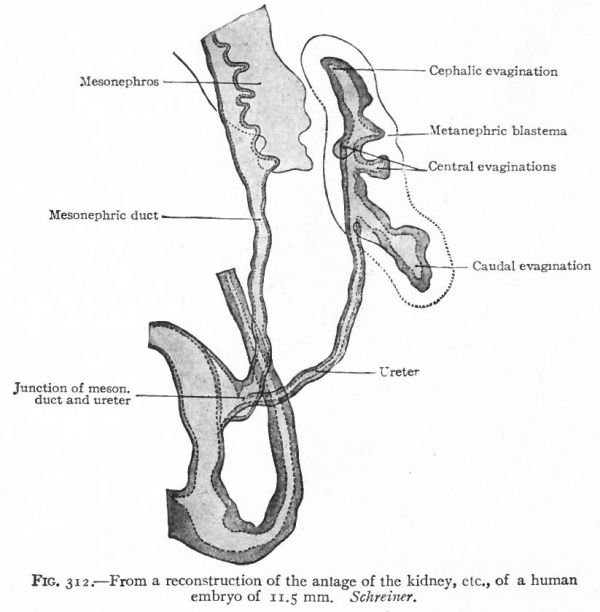
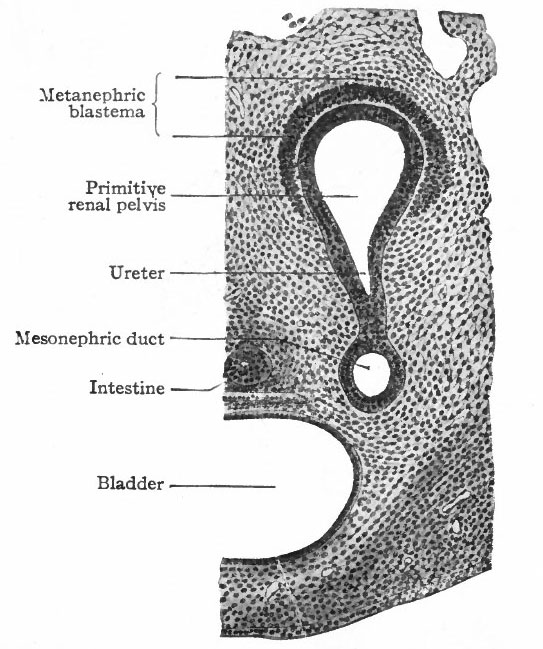
The Ureter, Renal Pelvis and Straight Renal Tubules. During the fourth week (in embryos of about 5 mm.) a small, hollow, bud-like evagination appears on the dorsal side of each mesonephric duct near its opening into the cloaca. The evagination continues to grow dorsally in the mesenchyme toward the vertebral column, and at the same time becomes differentiated into two parts, a narrow stalk and a dilated terminal portion. The stalk is the forerunner of the ureter, the dilated end is the primitive renal pelvis (Figs. 311 and 313). When the dilated end reaches the ventral side of the vertebral column it turns and grows cranially between the latter and the mesonephros. The stalk (or ureter) elongates accordingly (Fig. 312).
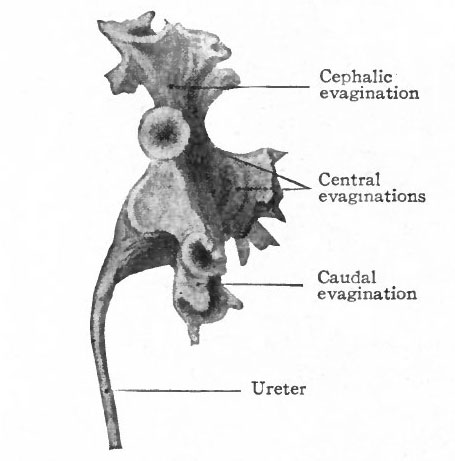
About the fifth week, four evaginations from the primitive renal pelvis appear one cephalic, one caudal and two central (Figs. 312 and 314) . These may be considered as straight renal tubules of the first order. The distal end of each then enlarges to form a sort of ampulla, and from each ampulla two other evaginations develop, forming tubules of the second order. From the ampulla of each secondary tubule two tertiary tubules grow out; and this process continues in a similar manner until twelve or thirteen divisions occur, the final divisions occurring during the fifth month. The tubules grow into the mesenchyme which surrounds the pelvis and which forms the so-called metanephric blastema, or nephro genie tissue (Fig. 313).
Fig. 312. From a reconstruction of the anlage of the kidney of a human embryo of 11.5 mm. Schreiner.
If the straight tubules were to remain in this condition, only four would open directly into the pelvis, corresponding with the four primary evaginations. In the adult, however, many hundreds open into the pelvis; consequently extensive changes of the early condition must take place. These changes are similar to the process by which the proximal ends of some of the blood vessels come to be included in the wall of the heart (p. 214). The proximal ends of the tubules become wider, the pelvis swells out, and the walls of the tubules become included in the wall of the pelvis. In certain parts of the pelvic wall this process goes on until deep bays the calyces are formed, into which a large number of tubules open. In the other parts of the wall the process does not go so far, thus leaving promontories the renal papilla upon which larger tubules or papillary ducts open. The adult renal pelvis thus consists of the primitive pelvis plus the proximal ends of the straight tubules.
Fig. 313. From a transverse section of a human embryo at the beginning of the 5th week. The plane of the section is indicated in Fig. 311. Schreiner.
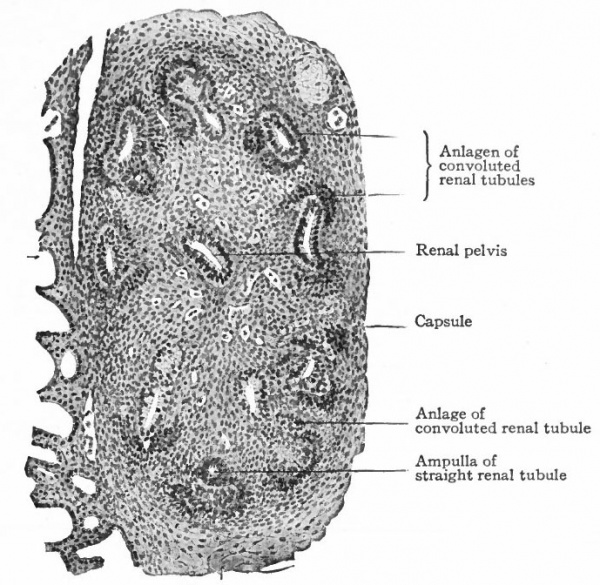
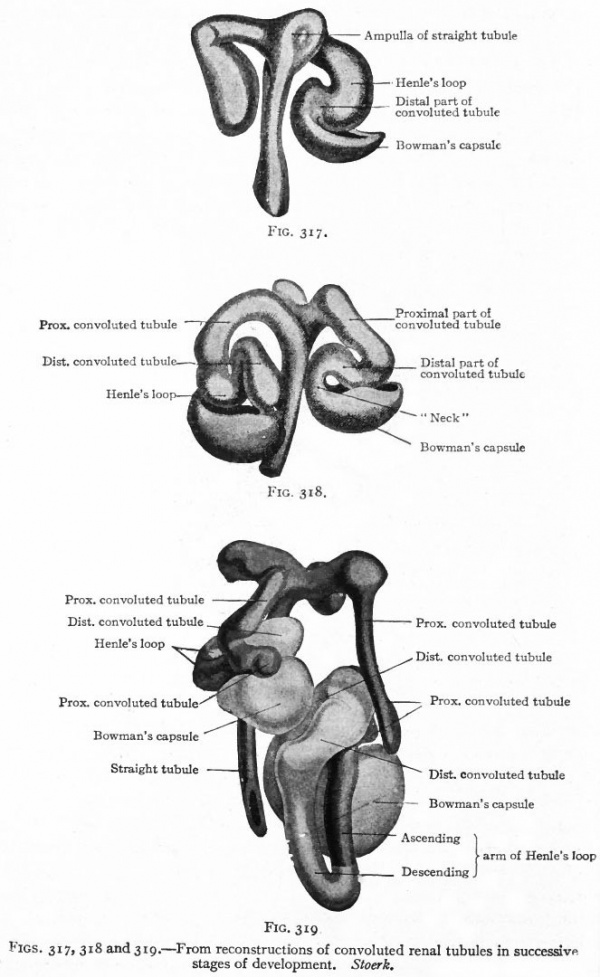
The Convoluted Renal Tubules and Glomeruli. As stated above, the metanephric blastema or nephrogenic tissue surrounds the renal pelvis and the straight tubules. It represents a condensation of the mesenchyme and is destined to give rise to the convoluted tubules and glomeruli. The cells of the blastema in the region of the ampullae of the terminal straight tubules acquire an epithelial character and become arranged in solid masses (Fig. 315). Each mass unites with an ampulla and acquires a lumen, which becomes continuous with the lumen of the straight tubule, then elongates and forms an S-shaped structure (Figs. 316 and 317). The loop of the S nearer the straight tubules elongates still more and grows toward the pelvis, parallel with the straight tubules, to form Ifenle's loop. The part between Henle's loop and the straight tubule elongates and becomes convoluted to form the proximal part of a convoluted renal tubule (second convoluted tubule) . The part between the distal end and Henle's loop elongates and becomes convoluted to form the distal part of a convoluted renal tubule (first convoluted tubule) (Figs. 318 and 319).
To avoid confusion it may be well to call attention to the fact that what has here been called the proximal part of a convoluted tubule corresponds with what is usually described as the second or distal convoluted tubule, and that the distal part of a convoluted tubule corresponds with the first or proximal convoluted tubule. In histology the distal and proximal convoluted tubules are spoken of in relation to the renal corpuscle, but in development it is more convenient to speak of the terminal part of a tubule as its distal part.
Fig. 314. From a model of the primitive renal pelvis and the evaginations which form the cephalic, central and caudal straight renal tubules of the first order. Human embryo of 4.75 months. Compare with Fig. 350. Schreiner
A glomerulus develops in connection with the extreme distal end of a convoluted tubule or, in other words, with the distal loop of the S (p. 363). There occurs here a further condensation of the mesenchyme, into which grows a branch from the renal artery. This, as the afferent vessel of the glomerulus, breaks up into several arterioles, each of which gives rise to a tuft of capillaries. These tufts are separated from one another by somewhat more mesenchymal tissue than separates the capillaries within a tuft. The tufts with the associated mesenchymal tissue constitute a glomerulus, and it is the mesenchymal septa between the tufts that give to the glomerulus its characteristic lobulated appearance. The capillaries of each tuft empty into an arteriole, and the several arterioles unite to form the efferent vessel of the glomerulus, which passes out along side of the afferent vessel. The renal tubule becomes flattened on the side next the condensation of the mesenchyme, and as the glomerulus develops, the epithelium of the tubule grows around it except at the point where the blood vessels enter and leave. Thus a double layer of epithelium comes to surround the glomerulus, the space between the two layers being the extreme distal part of the lumen of a renal tubule. The inner layer is closely applied to the surface of the glomerulus and even dips down into the latter between the tufts. The outer layer forms Bowman's capsule, the flat epithelium of which passes over into the cuboidal epithelium of the "neck" of the tubule, and this in turn is continuous with the pyramidal epithelium of the distal convoluted tubule. The entire structure is a renal corpuscle. The formation of renal corpuscles begins in embryos of 30 mm. and continues until after birth.
Fig. 315. Sagittal section of the anlage of the left kidney in a rabbit embryo of 15 days. Schreiner.
- The straight renal tubules (sections of which are shown) are embedded in the metanephric blastema. Condensations of the latter form the anlagen of the convoluted renal tubules. At the left of the figure several mesonephric tubules are shown.
Fig. 316. From a section of the kidney of a human foetus of 7 months. Schreiner.
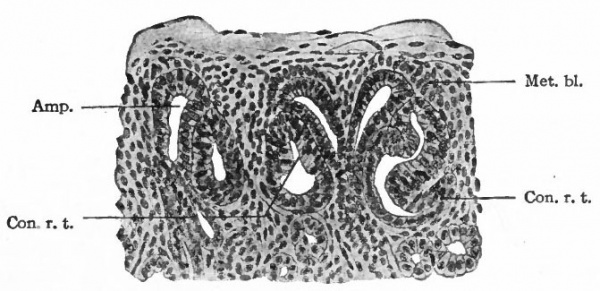
- Amp., Ampulla of a straight renal tubule; Con. r. t., anlagen of convoluted renal tubules, above and between which are two ampullae (compare Fig. 317); met. bl., metanephric blastema.
Figs. 317, 318 and 319. From reconstructions of convoluted renal tubules in successive stages of development. Stoerk.
The Renal Pyramids and Renal Columns
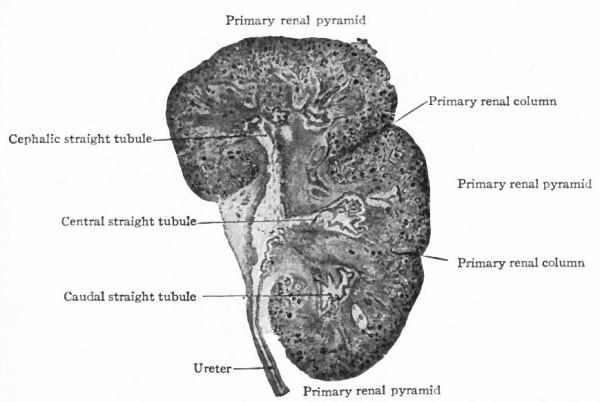
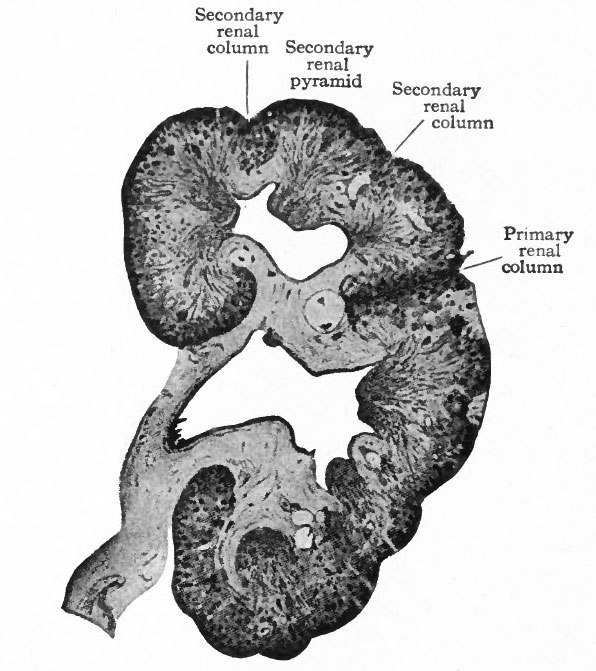
The tubules arising from the four primary evaginations of the renal pelvis together form four distinct groups or primary renal (Malpighian) pyramids one cephalic, one caudal, and two central. The central pyramids are crowded in between the end pyramids, (cephalic and caudal) and do not develop as rapidly as the latter which soon bend around toward the ureter, thus resulting in the formation of the convex side of the kidney and a depression or hilus opposite (compare Figs. 314 and 320). Between these four pyramids the mesenchyme remains for some time as rather distinct septa, forming the primary renal columns (columns of Bertini) which are marked by corresponding depressions on the surface of the kidney and extend to the renal pelvis. The four primary pyramids may be considered as lobes (Fig. 320). It should also be stated that the parts of the tubules derived from the mesenchyme form the bases of the renal pyramids. Between the groups of straight tubules derived from evaginations of the second or third order (see p. 362) there are also septa of mesenchyme which divide each primary pyramid into two or three secondary pyramids. These septa may be considered as secondary renal columns (Fig. 321). Thus the entire kidney is divided into from eight to twelve secondary pyramids. Tertiary renal columns then divide incompletely the secondary pyramids into tertiary pyramids. These are apparent on the surface of the kidney and constitute the surface tabulation, but are not clearly denned in the interior.
Fig. 320. Frontal section of the kidney of a human foetus of 3.75 months (10 cm). Hauch.
The formation of renal papillae (p. 363) corresponds to the formation of pyramids only to a certain point, for some of the tertiary pyramids appear only near the surface and consequently do not have corresponding papillae. This accounts for the fact that frequently the number of pyramids apparent on the surface does not correspond with the number of papillae. The surface lobulation is very plainly marked in kidneys up to and for a short time after birth. It then disappears and the surface becomes smooth. At the same time the connective (mesenchymal) tissue of the renal columns is largely replaced by the epithelial elements of the gland so that in the adult kidney the columns are not clearly denned.
Fig. 321. Frontal section of the kidney of a human foetus of 19 weeks (17.5 cm). Hauch.
The capsule of the kidney is derived from the mesenchyme which surroum the anlage of the organ (Fig 315). This mesenchyme is transformed into fibroi connective tissue and a small amount of smooth muscle, forming a layer which closely invests the kidney and dips into the hilus where it surrounds the blood vessels and the end of the ureter. The connective tissue and muscle of the ureter are also derived from the mesenchyme.
Cortex and Medulla
As the convoluted renal tubules develop in the metanephric blastema (p. 363), they form a cap-like mass around the group of straight tubules. This is the beginning of the renal cortex. A true cortex, however, can be spoken of only after the appearance of the glomeruli (in embryos of 30 mm.). Its peripheral boundary is the capsule, and the renal corpuscles nearest the pelvis mark its inner boundary. The mass of straight tubules forms the bulk of the medulla. It does not at this stage contain Henle's loops, the latter developing later (during the fourth month). Both cortex and medulla increase until the kidney reaches its adult size. The cortex increases relatively faster than the medulla up to the seventh year; after this the increase is practically equal. The medullary rays are probably secondary formations, being formed by groups of straight tubules which grow out into the cortex; later, ascending arms of Henle's loops are added to these groups.
Some of the glomeruli of the first generation are much larger than any found in the adult. In some of the lower Mammals these "giant" glomeruli disappear and it is probable that the same occurs in the human embryo. Some of the tubules also degenerate and disappear. The cause of these phenomena is not known.
Changes in the Position of the Kidneys. As has already been described (p. 361), the kidney buds first grow dorsally from the mesonephric ducts toward the vertebral column. They then grow cranially, with a corresponding elongation of the ureters, and in embryos of 20 mm. they lie for the most part cranial to the common iliac arteries. This migration continues until the time of birth when the cephalic ends of both kidneys reach the eleventh thoracic vertebra. When the kidneys begin to move cranially the hilus is directed caudally. Later they rotate and the hilus is turned toward the medial sagittal plane.
Since the ureter, renal pelvis and straight tubules develop from the mesonephric ducts, and since the convoluted tubules and glomeruli develop directly from the same tissue as the mesonephric tubules, namely, the mesenchyme, the renal tubules may be said to represent the third generation of urinary tubules. But no definite reason for the appearance of the third generation can be given. The atrophy of the mesonephroi would, of course, make necessary the compensatory development of new structures; but this only carries the problem a step further back, for the cause of the atrophy of the mesonephroi is not clear. In regard to this atrophy, however, there is a suggestion of a cause in the fact that in the Amphibia the mesonephroi are in part used for conveying the sexual elements, which leaves the mesonephroi less free to function as urinary organs. Possibly the loss of freedom to function leads to the development of new structures the kidneys in the higher forms (Reptiles, Birds and Mammals). In these forms the kidneys assume the urinary function after the early embryonic stages, and only the ducts and a part of the tubules of the mesonephroi persist in the male to convey the sexual elements. Thus the persistent parts of the mesonephroi assume a new function as the old one is lost. But, on the other hand, complications arise on account of the fact that in the female the sexual products are carried off by another set of ducts (the Miillerian ducts), which develop in both sexes but disappear in the male, while the mesonephroi and their ducts disappear almost entirely.
The Urinary Bladder, Urethra and Urogenital Sinus
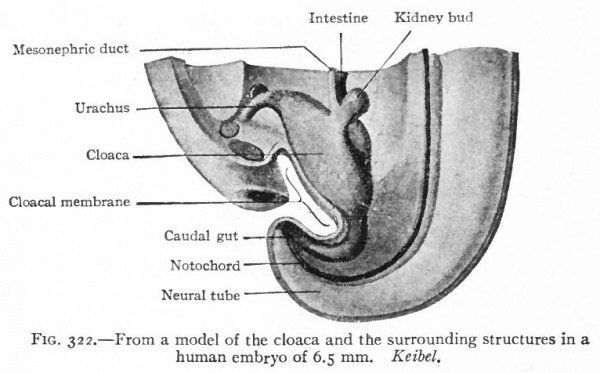
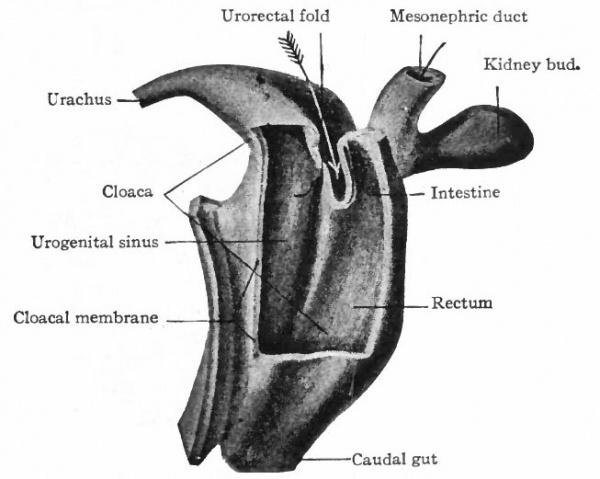
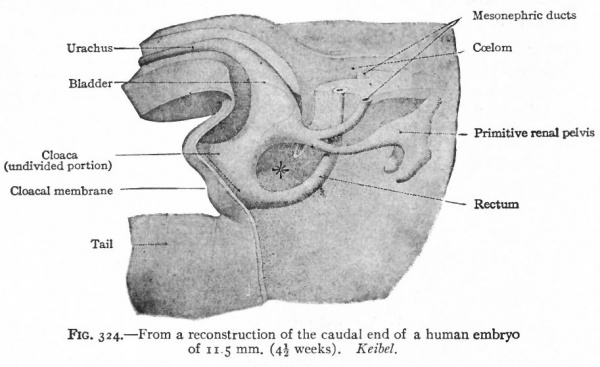
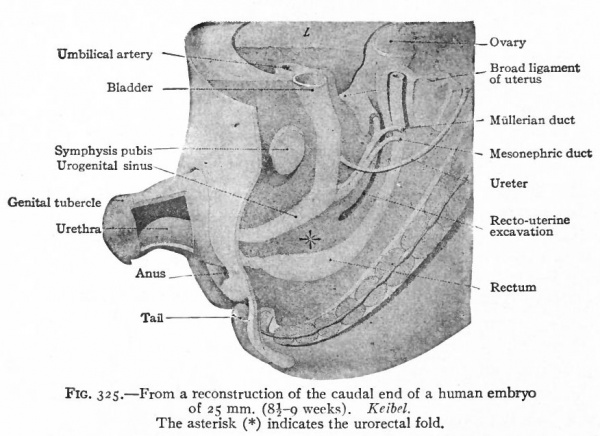
As described elsewhere, the allantois appears at an early stage as an evagination from the ventral side of the caudal end of the primitive gut (Fig. 244), grows out into the belly stalk, and finally becomes enclosed in the umbilical cord (p. 582). As the embryo develops, the proximal end of the allantois becomes elongated to form a stalk or duct which extends from the caudal end of the gut to the umbilicus (Fig. 247). The portion of the gut immediately caudal to the attachment of the allantoic duct becomes dilated to form the cloaca which at first is a blind sac, its cavity being separated from the outer surface of the embryo by the cloacal membrane (Fig. 322). The latter is composed of a layer of entoderm and a layer of ectoderm, with a thin layer of mesoderm between. The cloaca then becomes separated into two parts a larger ventral part which forms the urogenital sinus and a smaller dorsal part which forms the rectum. This Is accomplished by a fold or ridge which grows from the lateral wall into the lumen and meets and fuses with its fellow of the opposite side. The fusion begins at the cephalic end, in the angle between the allantoic duct and the gut, and gradually proceeds caudally until the separation is complete as far as the cloacal membrane. The mass of tissue forming the partition is called the urorectalfold (Fig. 323). The openings of the mesonephric ducts, which primarily were situated in the lateral cloacal wall (p. 359), are situated after the separation in the dorso-lateral wall of the urogenital sinus (compare Figs. 322, 323, 324). During the separation of the urogenital sinus from the rectum, certain changes take place in the proximal ends of the mesonephric ducts and ureters. The ends of the ducts become dilated and are gradually taken up into the wall of the sinus. This process of absorption continues until the ends of the ureters are included, with the result that the ducts and ureters open separately, the latter slightly cranial and lateral to the former. (Compare Figs. 324 and 325.) This condition is reached in embryos of 12 to 14 mm. The point at which these two sets of ducts open marks the boundary between a slightly larger cephalic part of the sinus, the anlage of the bladder, and a smaller caudal part which becomes the urethra and urogenital sinus (Fig. 325).
Fig. 322. From a model of the cloaca and the surrounding structures in a human embryo of 6.5 mm. Keibel.
After the second month the bladder becomes larger and more sac-like, and the openings of the ureters migrate farther cranially to their final position. The lumen of the bladder is at first continuous with the lumen of the allantoic duct, but the duct degenerates into a solid cord of cells, the urachus. The latter degenerates still further and finally remains only as the middle umbilical ligament. It seems quite probable that the bladder is derived almost wholly from the cloaca. A small part arises from the inclusion of the ends of the mesonephric ducts. If any part is derived from the allantoic duct, it is only the apex. After the bladder begins to enlarge, the adjacent portion of the urogenital sinus becomes slightly constricted. This marks the beginning of the urethra. In the female the constricted part represents practically the entire urethra. In the male it represents only the proximal end, the other portion developing in connection with the penis (p. 398). The urogenital sinus is narrow and tubular at its junction with the urethra; more distally it is wider and is shut off from the exterior by the cloacal membrane. After the embryo reaches a length of 16 to 17 mm., the membrane ruptures and the sinus opens on the surface.
Fig. 323. From a model of the cloacal region of a human embryo slightly older than that shown in Fig. 322. Keibel.
- The arrow points to the developing partition (uroectal fold) between the rectum and urogenital sinus. The opening of the mesonephric duct into the urogenital sinus is indicated by a small seeker.
The narrow part of the sinus is gradually taken up into the wider, resulting in the formation of a sort of -vestibule. In both sexes the urethra opens into the deeper end of the vestibule. In the male the mesonephric (seminiferous) ducts open near the external orifice. In the female the opening of the developing vagina is situated on the dorsal side near the external orifice.
Fig. 324. From a reconstruction of the caudal end of a human embryo of 11.5 mm. (4^ weeks). Keibel.
Fig. 325. From a reconstruction of the caudal end of a human embryo of 25 mm. (8i-g weeks). Keibel. The asterisk (*) indicates the urorectal fold.
The epithelium of the prostate gland is derived by evagination from the proximal part of the urethra. The first evagination appears during the third month. In the male the process continues to form a rather large gland; in the female the structure remains in a rudimentary condition. During the fourth month two evaginations arise from the urethra and develop into the bulbo-urethral (Cowper's) glands in the male, into the larger vestibular (Bartholin's) glands in the female.
Fig. 326. Transverse section through the germinal epithelium of a pig embryo of n mm. Nagel.
- The larger cells in the epithelium represent the sex cells, the smaller ones the undifferentiated mesothelial cells.
From the course of development it is seen that the epithelium of most of the bladder, of the female urethra and proximal end of the male urethra, of the prostate, of the urogenital sinus, and of the bulbo-urethral and vestibular glands is of entodermal origin. A very small part of the bladder epithelium is of mesodermal origin, since the proximal ends of the mesonephric ducts, which are mesodermal derivatives, are taken up into the wall. All the connective tissue and smooth muscle associated with these organs are derived from the mesoderm (mesenchyme) which surrounds the anlagen.
The Genital Glands
The Germinal Epithelium and Genital Ridge.
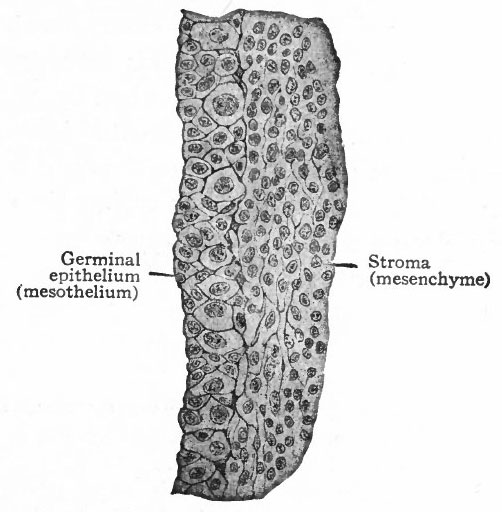
At a very early stage in the formation of the mesonephros, a narrow strip of mesothelium extending along the medial surface becomes thicker and the cells become arranged in several layers (Figs. 276 and 308). Two kinds of cells can be recognized in this (i) small cuboidal cells with cytoplasm which stains rather intensely, and (2) larger spherical cells with clearer cytoplasm and large vesicular nuclei (Fig. 326). The latter are the sex cells; and the whole epithelial (mesothelial) band is known as the germinal epithelium. The sex cells are destined to give rise to the sexual elements in the female to the ova, in the male to the spermatozoa. In the earlier stages, however, it is impossible to determine whether the sex cells will give rise to male or female elements. The differentiation of sex and the corresponding histological differentiation of the sex cells occur at a later period.
In his earlier work on the ovary and testis in Mammals, Allen has observed in very early stages (pig embryos of 6 mm., rabbit embryos of 13 days) certain large cells, with large clear nuclei, in the mesenchymal tissue of the mesentery, outside oj the genital ridge. In his investigation of the chick, Swift has discerned the sex cells at the time when the primitive streak and primitive axis are being formed. They are located in the entoderm and in the space between entoderm and ectoderm in the anterior part of the germ wall. When the mesoderm appears in this region the sex cells enter this layer, then enter the blood vessels. They are apparently amoeboid. By the blood stream they are carried to all parts of the blastoderm and embryo. Later the cells accumulate in the vicinity of the coelomic angle and finally enter the thickened mesothelium (germinal epithelium) of the genital ridge.
Beard, Eigenmann, Rabl, Woods, and others, have described sex cells, undoubtedly homologous with the early sex cells mentioned above, as occurring in various regions of the embryos of certain Fishes. These investigators also assert that the sex cells become specialized and, so to speak, segregated at a very early period of development, even at the stage of blastomere formation. Beard contends that the early differentiated sex (or germ) cells are significant in the origin of certain teratomata (see Chapter on Teratogenesis).
The cells of the germinal epithelium increase in number by mitotic division and the sex cells continue to increase in number by proliferation of their own members since there are no intermediate stages between the two types. The germinal epithelium soon becomes separated into two layers (i) a superficial layer which retains its epithelial character and contains the sex cells, and (2) a deeper layer composed of smaller cells which resemble those of the mesenchyme and which give rise to a part, at least, of the stroma of the genital glands. The elevation formed by these two layers projects into' the body cavity from the medial side of the mesonephros and constitutes the genital ridge (Fig. 308). From the superficial epithelial layer, columns or cords of cells, containing some of the sex cells, grow into the underlying tissue. This ingrowth, however, does not occur equally in all parts of the genital ridge, for three fairly distinct regions can be recognized. In the cephalic end comparatively few columns appear, but these few grow far down into the underlying tissue and constitute the rete cords. In the middle region a greater number of columns grow into the stroma, forming the sex cords. In the caudal region there are practically no columns. At first the line of demarkation between the cell columns and the stroma is not clearly defined.
The changes thus far described are common to both sexes and are completed during the fourth or fifth week. The genital ridges or anlagen of the genital glands constitute "indifferent" structures which later become differentiated into either ovaries or testicles.
Differentiation of the Genital Glands
After the fourth or fifth week, certain changes occur in the genital ridges which differ accordingly as the ridges form ovaries or testicles. While the differences are at first not particularly obvious, there are four which become clearer as the changes progress, (1) If the ridge is to become a testicle, the cells of the surface epithelium become arranged in a single layer and become flat. (2) In a developing testicle a layer of dense connective tissue grows between the surface epithelium and the sex cords, forming the tunica albuginea.
(3) In a testicle there also appears a sharper line of demarkation between the cell columns and the stroma, and the latter shows a more extensive growth.
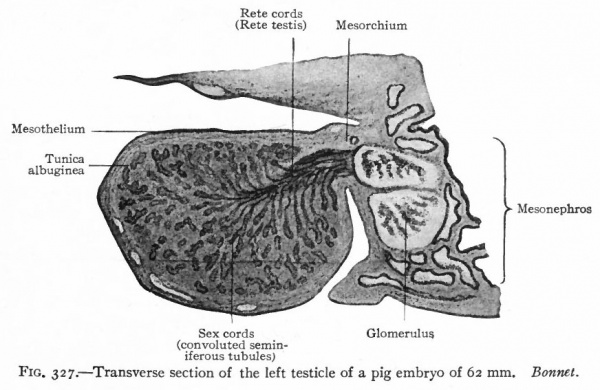
Fig. 327. Transverse section of the left testicle of a pig embryo of 62 mm. Bonnet.
(4) Another feature of the testicle is that the sex cells begin to be less conspicuous and do not increase furth2r in size, but come to resemble the other epithelial elements. The ovarian characters are to a certain extent the opposite. (i) The surface epithelium does not become flattened. (2) A layer of connective tissue, corresponding to the albuginea of the testicle, grows between the epithelium and the deeper parts, but is of a looser nature. (3) There is a less sharp line of demarkation between the cell columns and the stroma. (4) The sex cells continue to increase in size and become more conspicuous. (Compare Figs. 327 and 328.)
During these processes of development, the anlage of each genital gland becomes more or less constricted from the mesonephros and finally is attached only by a thin sheet of tissue the mesovarium in the female or the mesorchium in the male (p. 389). At the same time the anlage grows more rapidly in thickness than in length and assumes an oval shape.
Fig. 328. Longitudinal section of the ovary of a cat embryo of 94 mm. Semidiagrammatic. Coert.
The Ovary
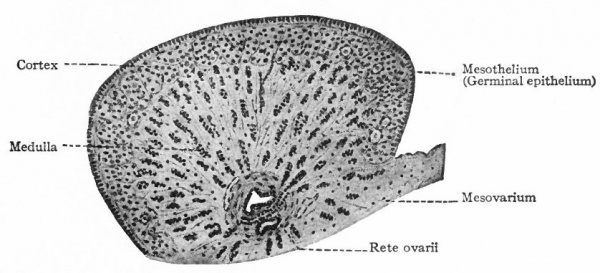
As stated above, a layer of loose connective tissue, corresponding to the albuginea of the testicle, grows in between the surface epithelium and the cell columns (sex cords) and effects a more or less complete separation. The sex cords are thus pushed farther from the surface, become more clearly marked off from the surrounding stroma and constitute the so-called medullary 'cords. The cortex of the ovary at this stage is represented only by the surface (germinal) epithelium, which is composed of several layers of cells and contains numerous sex cells in various stages of differentiation (Fig. 329). The rete cords which arise in the cranial end of the "indifferent" gland (p. 374) come to lie in what will be the hilus of the ovary. The ovary may thus be said to be composed of two parts (i) the rete anlage and (2) the stratum germinativum. The latter is subdivided by the albuginea into (a) medulla and (b) cortex.
1 . The rete cords develop into a group of anastomosing trabeculae which constitute the rete ovarii, situated in the hilus but nearer the cephalic end of the gland (Fig. 328). They are the homologues of the rete testis. The cells composing them are smaller and darker than those of the medullary cords. Sprouts grow out from the rete cords and unite with the medullary cords and the mesonephric tubules. (The same process occurs in the testicle, where the rete cords give rise to the functional rete testis and straight seminiferous tubules.) In some of the cords lumina appear and are lined with irregular epithelium. Such a condition represents the height of their development in the ovary. From this time on, they degenerate and finally disappear. The time of their disappearance varies in different individuals; they usually persist until birth, sometimes until puberty.
Fig. 329. Transverse section of the ovary of a fox embryo. Buhler in Hertwig's Handbuch. The large clear cells are the primitive ova.
Formerly it was thought that the rete cords were derived from the mesonephric tubules and entered the genital glands secondarily. More recent researches have demonstrated quite conclusively, however, that they are derivatives of the germinal epithelium and unite with the mesonephric tubules secondarily.
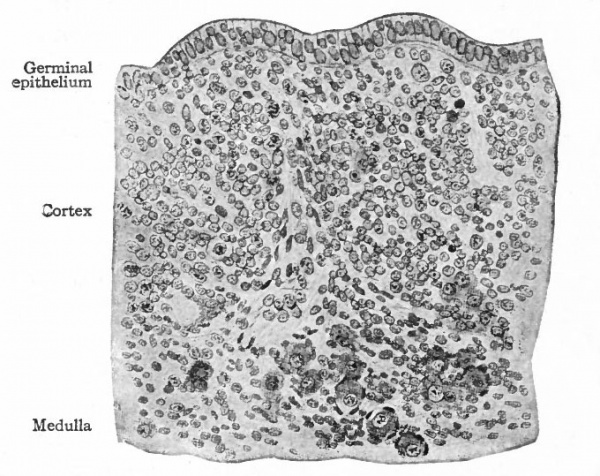
2 (a). The medullary cords are composed of small epithelial cells, contain a number of larger sex cells or primitive ova, and are surrounded by stroma (Figs. 329, 330). They are connected with the rete cords and in some places with the germinal epithelium. During foetal life they give rise to primary ovarian (Graafian) follicles; later they degenerate and finally disappear.
2(b). The cortex of the ovary, as stated above, at first consists of several layers of small, darkly staining cells, among which are many large, clearer sex cells or primitive ova (Fig. 329). From the epithelium, masses or cords of cells grow into the underlying tissue, carrying with them some of the primitive ova. These masses are known as Pfluger's egg cords. In some cases several ova are grouped together, forming egg nests (Fig. 330). The epithelial cells are the progenitors of the follicular cells and constantly undergo mitotic division. The primitive ova, on the other hand, increase in size and their nuclei show distinct intranuclear networks.
The egg cords become separated from the surface epithelium and are broken up so that in most cases a single ovum is surrounded by a single layer of epithelial cells. This constitutes a primary Graafian follicle. Rarely a follicle contains more than one ovum. In the case of the egg nests, the ova may become separated, or two or more may lie in one follicle. If two or more ova are present at first in any follicle, usually only one continues to develop and th( others either degenerate or are used as nutritive materials. In very rare cases, however, two ova may develop in a single follicle, but whether they reach maturity or not is uncertain. The formation of egg cords is usually completed before birth, but in some cases may continue for one or two years after birth. During the processes thus far described, the stroma also has been increasing, and the egg cords and follicles come to be separated by a considerable amount of connective tissue. The germinal epithelium becomes reduced to a single layer of cuboidal cells.
Fig. 330. From a section through the ovary of a human foetus of 4 months. Meyer-Ruegg, Buhler. The large cells are the primitive ova.
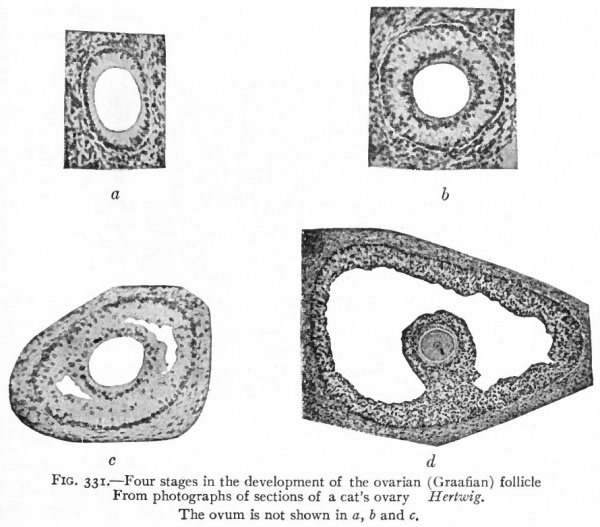
Each primary ovarian follicle, containing a primitive ovum (egg cell, sex cell) , is composed of a single layer of flat or cuboidal cells, plus a layer of stroma which gives rise to the theca folliculi. As the ovum continues to enlarge, the follicular cells become higher and arranged in a radial manner (Fig. 331, a) . By proliferation, the follicular cells come to form several layers, the innermost layer retaining the radial character and forming the zona radiata. The inner or basal ends of the cells of the zona radiata become clear to form the zona pellucida. In the latter, radial striations appear which have been described as minute channels in the cells, through which nutriment may pass to the ovum. After the follicular epithelium has become several layers thick, a fluid substance known as the liquor folliculi, and probably derived from the cells themselves, comes to lie in little pools among the cells (Fig. 33 1, b and c ) . While the follicle as a whole enlarges, these pools gradually coalesce and form a single large pool which fills the interior of the follicle (Fib. 331, d). Thus the epithelium is crowded out toward the periphery where it forms a layer several cells in thickness, known as the stratum granulosum. The ovum itself, with the zona radiata and some other surrounding cells, is also crowded off to the periphery of the follicle. The little elevation of the stratum granulosum in which the ovum is embedded is known as the cumulus ovigerus or germ hill (see Fig. i).
Fig. 331. Four stages in the development of the ovarian (Graafian) follicle. From photographs of sections of a cat's ovary Hertwig. The ovum is not shown in a, b and c.
The primary ovarian follicles at first lie rather near the surface of the ovary, but as they enlarge and as the ovary enlarges they come to lie deeper. As the follicle approaches maturity it increases greatly in size (5=*= mm.) and finally extends through the entire thickness of the cortex, its theca touching the tunica albuginea.
In speaking of the development of the follicles, it must be remembered that they develop slowly and do not reach maturity until near the age of puberty, and furthermore that one, or very few at most, reach maturity at the same time. In other words, when one follicle has reached m iturity there are all intermediate stages of development between this and the primitive follicles. When a follicle reaches maturity it ruptures at the surface of the ovary and the ovum is set free (p. 24). The ovum itself undergoes certain changes by which the somatic number of chromosomes is reduced one-half (p. 16). It then unites with the mature spermatozoon, which also contains one-half the somatic number of chromosomes, and forms the starting point, so to speak, for a new individual. At this point the processes by which an individual is carried through its life period from its beginning as a fertilized ovum to the time when it produces the next generation of mature sexual elements are ended. The developmental cycle of one generation is complete.
It has been estimated that approximately 36,000 primitive ova appear in each human ovary. Since, as a rule, only one ovum escapes from the ovary at menstrual period or between two succeeding periods, it is obvious that the vast majority of these never reach maturity. They probably degenerate, and, as matter of fact, atretic follicles may be found in an ovary at any time.
Corpus Luteum
After the rupture of the mature follicle at the surface the ovary and the escape of the ovum and liquor folliculi, blood from the rup tured vessels fills the interior of the follicle and forms a clot the corpus hcemorrhagicum. The cells of the stratum granulosum proliferate and , migrate into the clot and gradually form a mass which replaces the blood. It is held by some that the cells are derived from the theca folliculi. Whatever their origin, they become infiltrated with a fatty substance known as lutein. Trabeculae of connective tissue grow into the mass of cells, carrying small blood vessels with them. The (lutein) cells disintegrate and the products of disintegration are probably carried off by the blood, and finally the entire corpus luteum is transformed into a mass of connective tissue.
Whether the escaped ovum is fertilized or not has an influence upon the development of the corpus luteum. In case of fertilization, the corpus luteum becomes quite large, increasing in size up to the fourth month of pregnancy, and then degenerates. In case the ovum is not fertilized, the corpus luteum remains smaller. In both cases, however, the histological changes are essentially the same.
The Testicle
The processes that give rise to the "indifferent" genital glands have been described (p. 373 et seq.}. It has also been stated that there appears during the fourth or fifth week a structure that forms one of the characteristic features of the testicle. This is a layer of dense connective tissue which develops beneath the surface epithelium and constitutes the tunica albuginea (p. 375), and which separates the surface epithelium from the sex cords (Fig. 327) . The epithelium becomes reduced to a single layer of flat cells, although the cells on the tip of the gland usually remain high until after birth. Naturally this epithelium is continuous around the hilus of the testicle with the epithelium (mesothelium) of the abdominal cavity. Within the gland are the sex cords the progenitors of the convoluted seminiferous tubules, which become quite distinctly marked off from the stroma by a basement membrane. In the hilus region lie the rete cords the progenitors of the rete testis and the straight seminiferous tubules (Fig. 327) . The rete cords of the testicle are homologues of the rete cords of the ovary, and are derivatives of the germinal epithelium on the cephalic portion of the "indifferent" gland (p. 374).
Fig. 332. From a section of the testicle of a human foetus of 35 mm. Showing a developing convoluted seminiferous tubule. Meyer-Ruegg, Biihler.
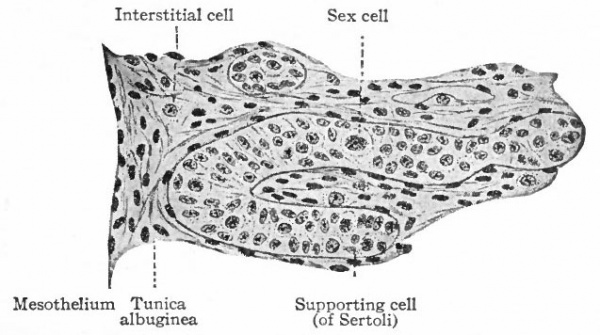
The sex cords at first are solid masses composed of several layers of cells. The latter are of two kinds, as in the ovary (i) smaller, darkly staining indifferent cells, and (2) larger, clearer sex cells (Fig. 332). The sex cells lose their clearness and come to resemble again the undifferentiated epithelial cells. They represent the spermatogonia, which correspond to the primitive ova. The spermatogonia proliferate very rapidly and become much more numerous than the epithelial cells. The sex cords become more and more coiled during development and anastomose with one another near the convex surface of the testicle. Beginning after birth and continuing up to the time of puberty, lumina appear in them by displacement of the central cells, and they thus give rise to the convoluted seminiferous tubules. The supporting cells (of Sertoli) are probably derived from the undifferentiated epithelial cells.
The details of the further development of the spermatogonia to form the the spermatozoa have been described in the Chapter on Maturation. At this point, that is, with the formation of the spermatozoon, the life cycle from a mature male sexual element in an individual to a mature male sexual element in an individual of the succeeding generation is completed.
The rete cords constitute an anastomosing network of solid cords of small, darkly staining cells, situated in the hilus region. These cords later acquire irregular lumina, which are lined with cubpidal cells, and form the rete testis. Evaginations grow out from the rete and fuse with the ends of the convoluted tubules, thus forming the straight tubules. On the other hand, outgrowths from the rete unite with the tubules in the cephalic portion of the mesonephros, so that a direct communication is established between the convoluted seminiferous tubules and theinesonephric tubules. There is thus formed the proximal part of the efferent duct system of the testicle (Fig. 327). That portion of the tunica albuginea in which the rete testis lies, becomes somewhat thickened to form the mediastinum testis.
The stroma of the testicle is derived for the most part from the mesenchyme of the "indifferent" gland or genital ridge. Probably a smaller part is derived from the germinal epithelium (see p. 374)- During development, however, the glandular elements increase more rapidly than the stroma, so that in the adult they predominate. There is a tendency for the convoluted tubules to become arranged in groups which are separated by trabeculae of connective tissue radiating from the mediastinum. The interstitial cells of the stroma are direct derivatives of the connective tissue cells (Fig. 332).
Determination of Sex
The views regarding the determination of sex are discussed in the chapter on Maturation (page 21) in connection with the question of Mendelian heredity.
The Ducts of the Genital Glands and the Atrophy of the Mesonephroi.
In the Female. Strictly speaking, the ovaries are ductless glands; for neither developmentally nor anatomically are the ducts which convey their specific secretion directly connected with them. Furthermore, these ducts are in part transformed into certain organs for the reception and retention of both kinds of sexual elements. In other words, the ducts in part become specially modified to form the vagina and uterus, of which the latter serves as an organ of maintenance for the embryos of the next generation.
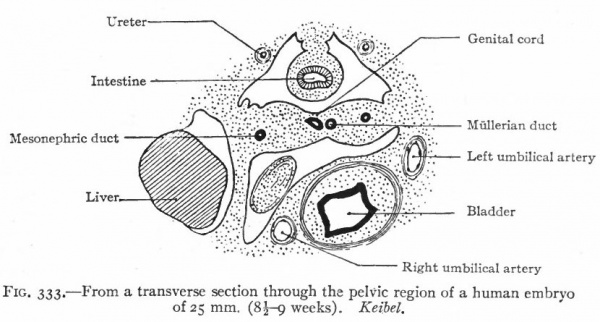
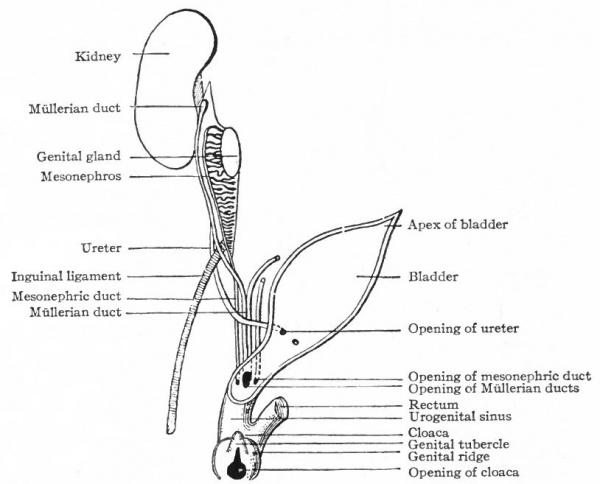
The ducts originate in connection with the mesonephroi, and are known at first as the Mullerian ducts. They appear in both sexes alike but persist only in the female. In the lower Vertebrates they are split off from the mesonephric ducts. In the higher forms, however, their mode of origin is not known with certainty, but the present evidence favors the view that they arise independently of the mesonephric ducts. They appear in human embryos of 8-14 mm. The mesothelium on the lateral surface of the cephalic end of each mesonephros becomes thickened and then invaginates or dips into the underlying mesenchyme. By proliferation of the cells at its tip, the invaginated mass grows caudally as a duct parallel with and close to the mesonephric duct. The two ducts come to be embedded in a ridge which at the cephalic end of the mesonephros is situated laterally, but toward the caudal end bends around and comes to lie ventrally. Beyond (caudal to) the mesonephros the ridge is attached to the lateral body wall, and near the urogenital sinus it meets and fuses with its fellow of the opposite side (Fig. 333).
Fig. 333. From a transverse section through the pelvic region of a human embryo of 25 mm. (8 -9 weeks). Keibel.
The two Mullerian ducts, contained in the ridges, also approach each other and fuse. The fusion begins in embryos of 25 to 28 mm. (end of second month), and about the same time they open into the dorsal side of the urogenital sinus. The relations of the Mullerian and mesonephric ducts are different in different parts of their courses. At the cephalic end the Miillerian lies dorsal to the mesonephric, but farther back it runs more laterally, then ventrally, and finally opens into the urogenital sinus on the medial side of the mesonephric duct.
The Oviduct
The single part of each Mullerian duct gives rise to the oviduct. The opening at the cephalic end remains as the ostium abdominale tuba, which from the beginning communicates directly with the abdominal cavity (ccelom) and never becomes connected with the ovary (Fig. 328). The rim of the opening sends from three to five projections into the abdominal cavity to form the primary fimbria. Secondary branches grow out from these and form the numerous fimbriae of the adult oviduct. The part of each
Fig. 334. Right half of the pelvic region of a female human foetus of 7 months. Nagel.
Mullerian duct between the fimbriated end and the fused caudal end, grows in length as the embryo develops, but not proportionately, so that in the adult the oviduct is relatively shorter than in the embryo. At first it is lined with simple cylindrical epithelium, but later the cells become cuboidal, and during the second half of f octal life acquire distinct cilia. The connective tissue and muscle of the oviduct are derived from the mesenchyme that primarily surrounds the Miillerian duct.
In connection with one of the fimbriae of the oviduct there is sometimes found a small vesicle lined with ciliated epithelium, forming the non-stalked hydatid (of Morgagni), which possibly represents the extreme cephalic end of the Miillerian duct (Fig. 342). In this case the permanent ostium of the tube would be of secondary origin.
The Uterus and Vagina
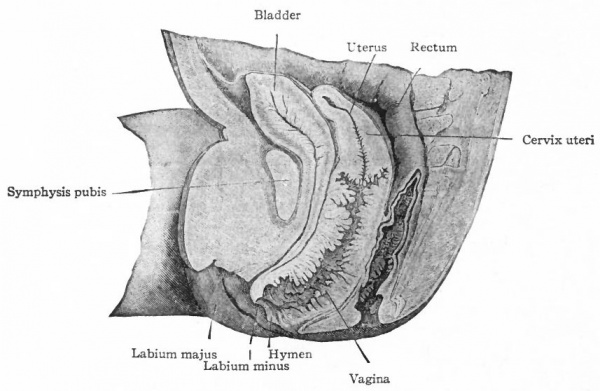
The fused caudal ends of the two Mullerian ducts form the anlage of the uterus and vagina, which is a single medial tube opening into the urogenital sinus (Fig. 325). During the third month certain histological changes bring about a differentiation between the cephalic end or uterus and the caudal end or vagina. The simple columnar epithelium of the vaginal portion changes to stratified squamous, and during the fourth month the lumen becomes closed. Near the external orifice a semicircular fold appears, which represents the hymen (Fig. 334). During the sixth month the lumen reappears by a breaking down of the central cells. The epithelium of the uterus, primarily high columnar, becomes lower and toward the end of foetal life acquires cilia. Many irregular folds appear in the mucosa of the vagina, a smaller number in the uterus (Fig. 334). Some of the folds in the uterus constitute the regular plica palmata of the cervix. The uterine glands represent evaginations from the epithelial lining. They do not begin to develop until after birth (one to five years), and their development is usually not completed until the age of puberty.
Fig.335. Transverse section through the ovary and broad ligament of a human foetus of 3 months. Nagel.
The muscle and connective tissue of the walls of the uterus and vagina are derived from the mesenchyme which surrounds the Mullerian ducts. The muscle develops relatively late (after the fourth month of foetal life).
Atrophy of the Mesonephroi
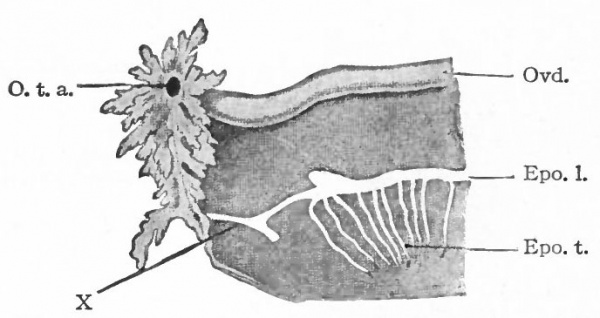
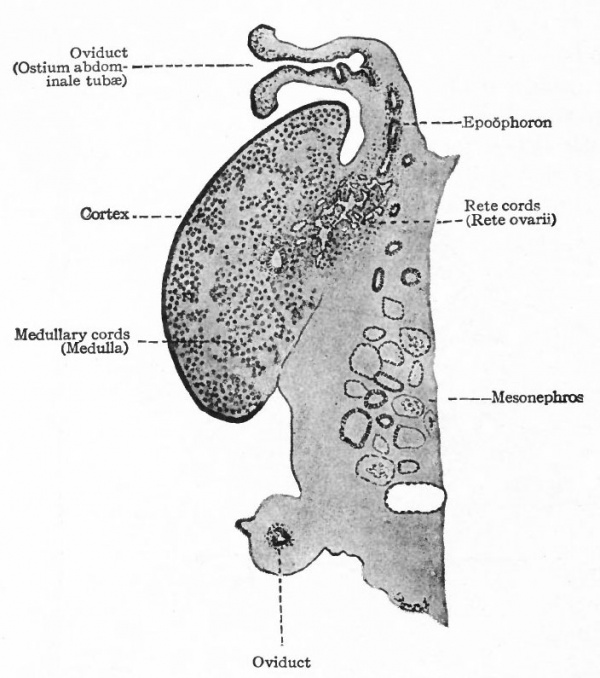
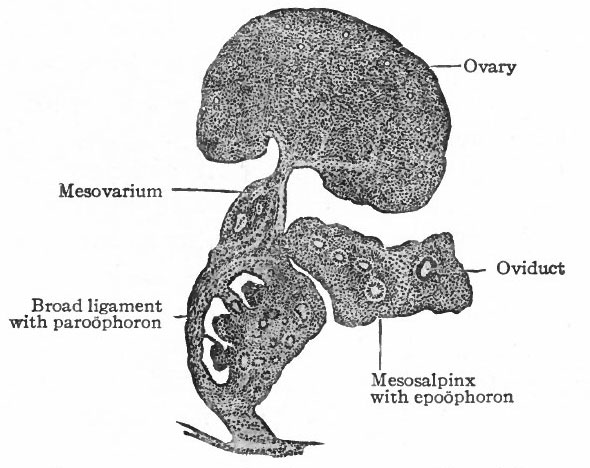
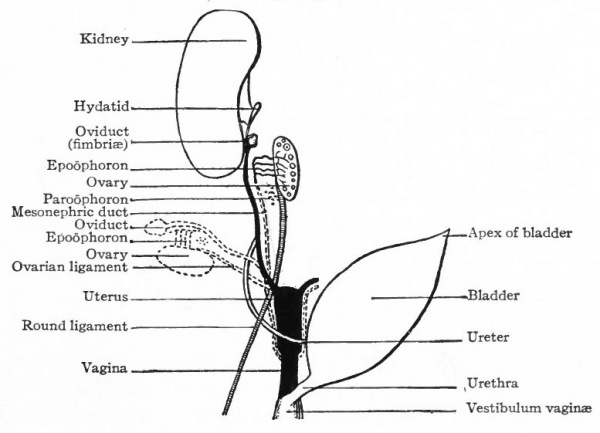
By far the greater part of each mesonephros degenerates and disappears, and the parts that do persist are rudimentary and possess no functional significance. The cephalic portion leaves ten to twenty coiled tubules which terminate blindly at one end and at the other end open into a common duct that represents the cephalic end of the mesonephric duct. These tubules constitute the epoophoron (parovarium, organ of Rosenmiiller) which comes to lie in the mesosalpinx between the oviduct and the mesovarium, and later in the mesentery between the oviduct and the ovary (Fig. 335). At the height of their development the tubules are lined with columnar, ciliated epithelium. The rete cords of the ovary (rete ovarii, p. 377) during their development unite with the tubules in the cephalic portion of the mesonephros, but later disappear. The epoophoron is homologous with the tubules of the head of the epididymis in the male.
The caudal portion of the mesonephros leaves a few tubular remnants which come to lie in the broad ligament near the hilus of the ovary. These constitute the paroophoron which is homologous with the paradidymis in the male (Fig. 335). They may disappear before birth or may persist through life.
The mesonephric duct also leaves certain remnants which are situated (i) in the broad ligament, (2) in the lateral wall of the uterus, (3) in the lateral wall of the vagina, and (4) in the tissue lateral to the external genital opening. These remnants are known as the canals of Gartner, and they naturally lie in the course of the duct in the embryo. All the rudimentary structures derived from the mesonephroi and their ducts are extremely variable.
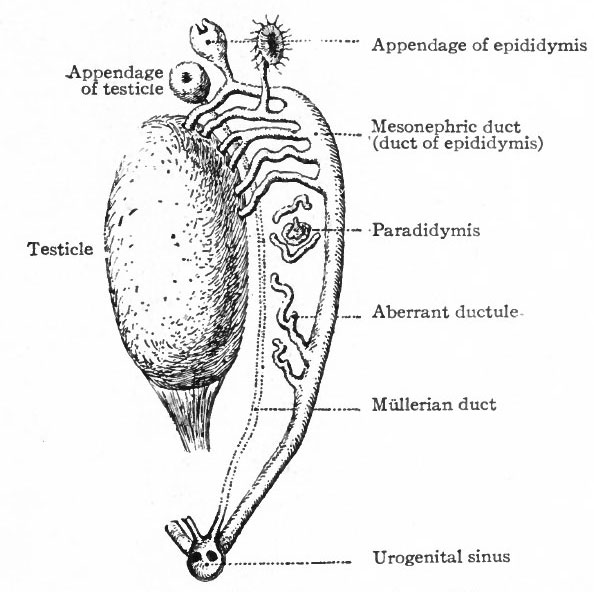
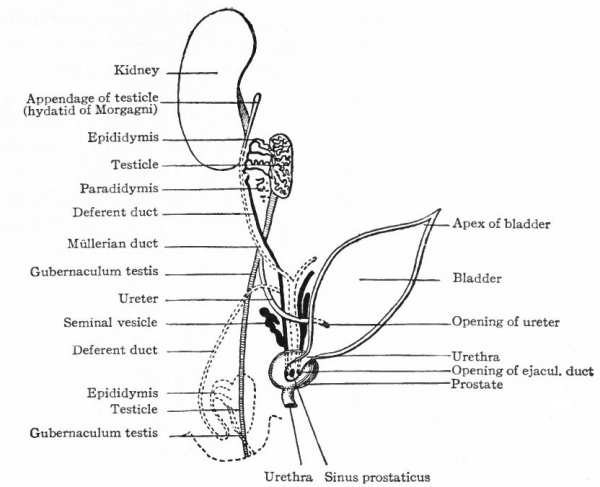
In the Male. In the male all the efferent ducts of the genital glands, except the rete testis, are derived from the mesonephroi and their ducts. As described earlier in this chapter (p. 381) , the rete testis acquires a connection with some of the tubules in the cephalic end of the mesonephros and with the sex cords or anlagen of the convoluted and straight seminiferous tubules (see Fig. 327). This establishes a communication between the seminiferous tubules and the tubules of the mesonephros. Those mesonephric tubules with which the rete testis unites persist as the efferent ductules (or vasa eff erentia) . The latter form a set of coiled ducts which are situated in the head of the epididymis and which open into the cephalic part of the mesonephric duct (Fig. 309). They are homologous with the epoophoron in the female.
The next succeeding portion of the mesonephric duct becomes the duct of the epididymis which in its tortuous course constitutes the bulk of the body and tail of the epididymis and passes over into the caudal portion of the mesonephric duct. The latter portion becomes the deferent duct (vas def erens) . The caudal end of the deferent duct forms the ejaculatory duct which opens into the urogenital sinus. The seminal vesicles appear during the third month as lateral evaginations from the ejaculatory ducts.
The portions of the mesonephros not involved in the formation of the duct system of the testicle atrophy and for the most part disappear. They leave certain tubules, however, which persist as rudimentary structures connected with the testicle. In the cephalic end, some of the tubules persist in part and come to lie among the efferent ductules, being either attached to the latter or unconnected, and forming the appendage of the epididymis. The caudal part of the mesonephros leaves a few tubules which come to lie near the head of the epididymis and form the paradidymis (or organ of Giraldes), the tubules of which are lined with columnar, ciliated epithelium. Near the transition from the duct of the epididymis to the deferent duct there is almost invariably a tubule (sometimes branched) which also represents a remnant of the mesonephros and is known as the aberrant ductule. It usually opens into the duct of the epididymis, but may lie free in the tissue around it (Fig. 309).
Atrophy of the Mullerian Ducts
These ducts persist in the female and become the oviducts, uterus and vagina; in the male they degenerate and disappear almost entirely. The degeneration begins about the time they open into the urogenital sinus (embryos of 25 to 28 mm.) ; by the time the embryo reaches a length of 60 mm. only the extreme cephalic end and the caudal third remain, and at 90 mm. the entire duct is gone except the extreme ends. The cephalic end persists as the appendix testis (or hydatid of Morgagni) (Figs. 309, 341). The caudal end persists as the utriculus prostaticus (uterus masculinus).
Fig. 336. Urogenital organs in a human embryo of 17 mm. (6 weeks). Kollmann's Alias.
Changes in the Positions of the Genital Glands and the Development of their Ligaments.
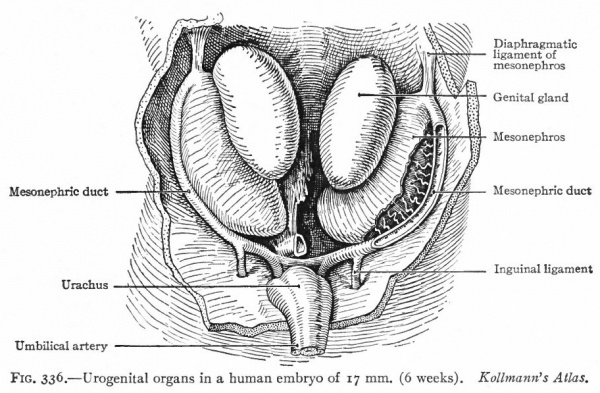
During the early stages of development the genital glands testicles or ovaries are situated far forward in the abdominal cavity. During the eighth week they lie opposite the lumbar vertebrae. During the succeeding months, up to the time of birth, they gradually move caudally to the positions they occupy in the adult. This migration is brought about, to some extent at least, by the influence of certain bands of tissue which are primarily like mesenteries. As the mesonephros develops and projects into the body cavity it comes to be attached along the dorsal body wall, lateral to the dorsal mesentery, by a sheet of tissue which is called the mesonephric mesentery. Cranial to the mesonephros, this mesentery is continued as the diaphragmatic ligament of the mesonephros, which as the name indicates, is attached to the diaphragm; caudally it is continued to the inguinal region as the inguinal ligament of the mesonephros (Fig. 336). The genital gland lies on the medial side of the mesonephros and is attached to the latter by a sort of mesentery which becomes the mesovarium in the female or the mesorchium in the male. The cephalic portions of the ducts (Miillerian and mesonephric) lie close together in a ridge on the lateral surface of the mesonephros; as they pass caudally they extend around to the ventral surface of the mesonephros and approach the medial line, and finally, in the pelvic region, the two ridges meet and fuse, forming the socalled genital cord (Fig. 333). The genital cord thus contains the mesonephric and Miillerian ducts, the latter fusing to form a single tube (the anlage of the uterus and vagina, p. 385). It also contains the umbilical arteries.
Fig. 337. From a dissection of the pelvic region of a male human foetus of 21 cm. Kollmann's Atlas.
Fig. 338. From a dissection of the scrotal region of a human foetus of 25 cm. Kollmann's Atlas.
Fig. 339. From a dissection of the pelvic region of a female human foetus of 7.5 cm. Kollmann's Atlas.
Such a condition is found in embryos of about eight weeks. From this time on, the processes of development follow divergent lines in the two sexes, the differences becoming more marked from month to month. Certain structures persist and othersdisappear, according to the sex. The mesenteries and ligaments undergo metamorphoses and the genital glands migrate caudally.
Descent of the Testicles
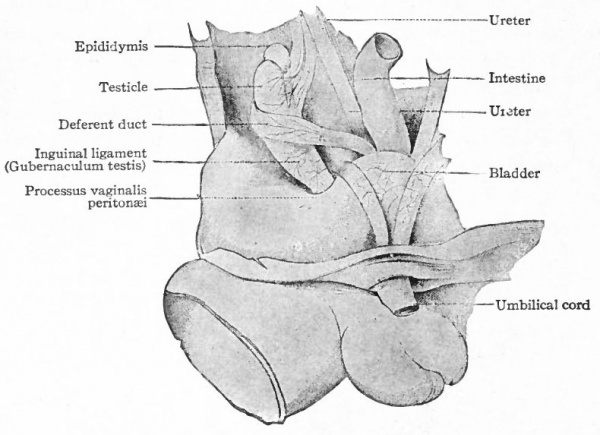
As the mesonephros atrophies, its mesentery and the mesentery of the testicle are combined to form a single band of tissue which, of course, is continuous with the inguinal ligament. The latter now becomes the so-called gub.ernaculum testis (Hunteri), a strong band or cord composed of connective tissue and smooth muscle. Its cephalic end is attached to the epididymis; its caudal end pierces the body wall in the inguinal region and is attached to the corium of the skin (Fig. 337). It plays an important part in the descent of the testicle. The descent is brought about through the principle of unequal growth. As the body grows in length, the gubernaculum grows much less rapidly and, since the caudal end of the latter is fixed, the natural result is the drawing downward of the testicle. This takes place gradually, and at the end of the third month the testicle lies in the false pelvis; at the end of the sixth month close to the body wall at the inguinal ring.
Fig. 340. Diagrammatic representation of the urogenital organs in the "indifferent" stage. Hertwig.
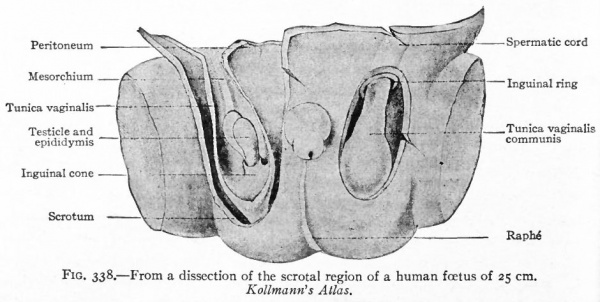
During the third month a second factor in the descent of the testicle appears. This is an evagination of the peritoneum at the point where the gubernaculum pierces the body wall. The evagination at first is a shallow depression, known as the processus vaginalis peritonei, but continues to burrow through the body wall and causes an elevation in the skin which is destined to become one side of the scrotum (see p. 396) . The opening of the peritoneal sac into the body cavity is the inguinal ring. In its descent the testicle passes through the inguinal ring and comes to lie in the elevation in the skin or scrotum (ninth month) . Whether its passage into the scrotum is the result of a traction by the gubernaculum is not certain. The inguinal ring then closes by apposition of its walls and the testicle lies in a closed sac which has been pinched off, so to speak, from the body cavity (Fig. 338).
Fig. 341. Diagram of the development of the male genital organs from the "indifferent" anlagen. Hertwig.
Fig. 342. Diagram of the development of the female genital organs from the "indifferent" anlagen. Hertwig. These diagrams should be compared with Fig. 340. The dotted lines represent the organs in the relative positions they occupy in the adult (with the exception of the Mullerian duct in the male and the mesonephric duct in the female, which ducts disappear for the most part).
Since the testicle is invested by peritoneum from the beginning of its development, it must be understood that in its passage into the scrotum it passes along under the peritoneum. Consequently when it reaches the scrotum it is surrounded by a double layer of peritoneum, the tunica vaginalis propria.
The descent of the testicle also produces marked changes in the course of the deferent duct. Primarily the (mesonephric) duct extends cranially from the urogenital sinus in a longitudinal direction. But as the testicle migrates, the cephalic end of the duct is drawn caudally so that in the adult the deferent duct extends cranially from the scrotum to the ventral side of the urinary bladder and then bends caudally again to open into the urethra.
Descent of the Ovaries
The ovaries undergo a change of position corresponding to the descent of the testicles, although the change is not so extensive. Primarily the Miillerian and mesonephric ducts lie in a ridge on the surface of the mesonephros (p. 383). As the mesonephros and its duct atrophy, the Miillerian duct (oviduct) comes to lie in a fold, the mesosalpinx, which is attached to the mesovarium (Fig. 335) . At the same time the mesovarium becomes directly continuous with and really a part of the inguinal ligament. The latter corresponds, of course, to the gubernaculum testis, and plays a role in the descent of the ovaries. It may be conveniently divided into three parts, (i) a cephalic part which is attached to the hilus of the ovary, (2) a middle part which extends from the ovary to the uterus, forming the ovarian ligament, and (3) a caudal part which extends from the uterus to the inguinal region, forming the round ligament of the uterus (Fig. 339). The round ligament pierces the body wall and is attached to the corium of the skin. At the point where it passes through the body wall there is a slight evagination of the peritoneum, the diverticulum of Nuck, which corresponds to the processus vaginalis peritonei in the male.
The ovaries gradually migrate caudally from their original position into the false pelvis (third month) and thence into the true pelvis (at birth) . Obviously no traction can be exerted upon them by the round ligament (or caudal part of the inguinal ligament) , since the latter extends from the uterus to the inguinal region. Their descent into the pelvic seems to be due to the unequal growth of the ovarian ligaments, or in other words, to the fact that the ovarian ligaments grow proportionally less than the surrounding parts. During their descent the ovaries become embedded in the broad ligaments of the uterus, which represent further development of the peritoneal folds of the genital cord. In this way the mesovarium becomes merged with the broad ligament.
On pages 390 and 391 are three diagrammatic representations of the changes that take place in the genital systems of the two sexes. Fig. 340 represents the "indifferent" stage in which all the embryonic structures are present; Fig. 341 represents the changes that occur in the male; Fig. 342 represents the changes that occur in the female. A careful study of the diagrams will assist the student materially in understanding the processes of development which have been described in the preceding paragraphs.
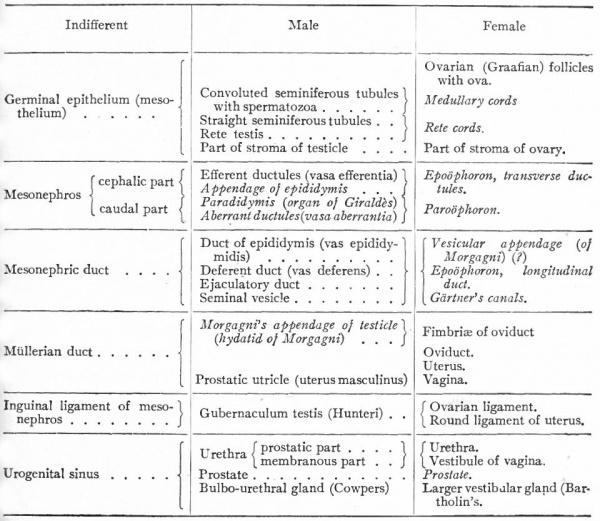
Below is a table that is meant to set forth briefly the various structures which belong to the internal genital organs in the two sexes, and which are derived from the structures in the "indifferent" stage. The words in italics are the names of structures that persist in a rudimentary form.
The External Genital Organs
In addition to the internal organs of generation, to which the description has thus far been confined, certain other structures appear on the outside of the body to form the external genitalia. In the case of these also there is an " indifferent" stage from which the courses of development diverge in the two sexes.
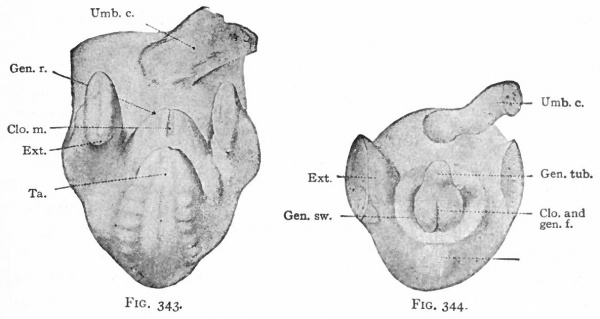
During the sixth week a depression appearing on the ventral surface of the caudal end of the body indicates the position of the cloacal membrane (p. 370). This becomes surrounded by a slight elevation, produced by the thickening of the mesoderm which is known as the genital ridge (Fig. 343). The cephalic side of the ridge becomes raised still farther above the surface, forming a distinct protrusion, the genital tubercle. The tubercle continues to increase in size, and the distal end forms a knob-like enlargement. Along the ventral (or rather caudal) side a groove appears, which extends distally as far as the base of the enlarged end. The ridges along the sides of the groove increase in size and form the genital folds. In the meantime a second pair of elevations appears lateral to the genital folds to form the genital swellings (Fig. 344).
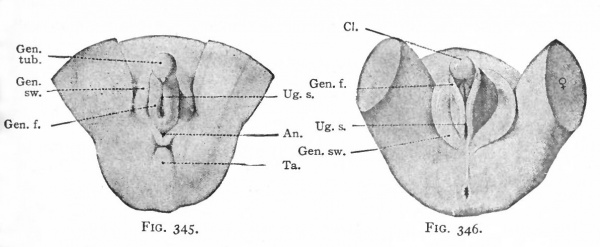
After the cloacal membrane ruptures, a single opening is produced which leads from the exterior into the cloaca. This opening is then separated by the further growth of the urorectal fold (p. 370) into the opening of the urogenital tract and the anal opening. The caudal part of the fold then enlarges to form the perineal body, which serves to push the anus farther away from the genital ridges. The latter, together with the genital tubercle and swellings, all of which lie in the immediate vicinity of the urogenital opening, constitute the anlagen of the external genital organs (Fig. 345). These at this time are in the "indifferent" stage, from which development proceeds in one of two directions, accordingly as the embryo is a male or a female. Up to the fourth month there is little difference between the structures in the two sexes. After this the differences become more and more obvious.
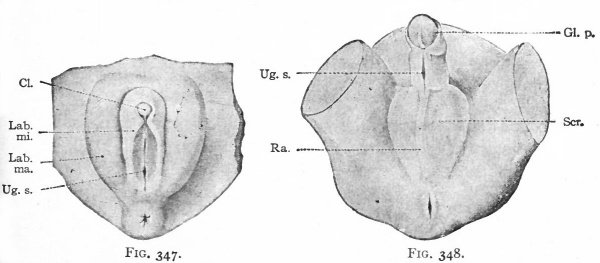
In the female the changes in the originally "indifferent" structures are comparatively slight. The genital tubercle grows slowly and becomes the clitoris. The enlarged extremity becomes more clearly marked off from the other part to form the glans clitoridis. The skin covering the glans is converted by a process of folding into a sort of prepuce. The genital folds, which bound the opening of the urogenital tract, become elongated and form the labia minora. The opening of the urogenital tract is the vestibulum vagina. The genital swellings enlarge still more than the genital folds, by a deposition of a considerable mass of fat in the mesenchyme, and become the labia majora. The latter are the structures (mentioned on p. 390) which mark the points at which the inguinal ligaments of the mesonephroi pierce the body wall, and are homologous with the scrotum in the male (Figs. 346 and 347).
In the male the "indifferent" anlagen undergo more extensive changes than in the female. The genital tubercle continues to grow more rapidly and forms the penis, which is homologous with the clitoris. The enlarged extremity becomes the glans penis, and an extensive folding of the skin over the glans forms the prepuce. The groove on the caudal or lower side of the tubercle elongates as the latter elongates and becomes deeper. Finally the ridge (or genital fold) on each side of the groove meets and fuses with its fellow of the opposite side, thus enclosing within the penis a canal the penile portion of the urethra. The groove is primarily continuous with the opening of the urogenital tract, and as the fusion takes place the penile portion forms a direct continuation of the internal (membranous and prostatic) portion of the urethra. The genital swellings also fuse and form the scrotum, the line of fusion in the medial line becoming the raphe (Fig. 348) . Primarily the inguinal ligaments of the mesonephroi are attached to the corium of the skin in the genital swellings, and as the testicles descend they pass through the inguinal ring into the scrotum. In a sense the scrotum represents an evagination of the body wall
Figs. 343-348. Stages in the development of the external genital organs. Kollmann's Atlas.
- indifferent " stage embryo of 17 mm.; Fig. 344, " indifferent " stage embryo of 23 mm ; g. 345, " indifferent " stage embryo of 29 mm. (beginning of 3d month) ; Fig. 346, female embryo of 70 mm. (n weeks); Fig. 347, female embryo of 150 mm. (16 weeks); Fig. 348, male embryo of 145 mm. (16 weeks).
- An., Anus; CL, clitoris; Clo.and gen. /., cloaca and genital folds; Cl. m., cloacal membrane; Ext. y lower extremity; Gen. /., genital folds; Gen. r., genital ridge; Gen. sw., genital swelling; Gen. tub., genital tubercle; Gl. p., glans penis; Lab. ma., labium majus; Lab. mi., labiura minus; Ra., raphe of scrotum; Scr., scrotum; Ta., tail; Ug. s., urogenital sinus; Umb. c, umbilical cord.
The Development of the Suprarenal Glands
Although the suprarenal glands do not logically come under the head of the urogenital system, being neither functionally nor developmentally a part of the latter, it is most convenient to consider them in this chapter.
In Mammals including man these glands are composed of two parts which can be differentiated histologically and topographically the cortex and medulla. The cortex is composed of trabeculae and spheroidal masses of cells which do not have a strong affinity for the ordinary cytoplasmic stains am which contain granules of a fat-like substance known as lipoid granules. Th( medulla is composed of irregularly arranged sympathetic ganglion cells am other granular cells which, after treatment with chrome salts, acquire a pea brownish color. The brown cells are known as chromaffin (or phaeochrome) cells and their granules as chromaffin (or phaeochrome) granules. As cort( and medulla are distinct anatomically, they are also distinct developmentally, being derived from two distinct and different parent tissues which unite secondarily. Furthermore, it is an interesting fact that in the lower Vertebrates (Fishes) the two parts remain permanently separate; that in the ascending scale of animal life (Amphibia, Reptiles, Birds) they become more closely associated; and that finally (in Mammals) they unite to form a single glandular structure. In Mammals the phylogenetic history is repeated with remarkable precision during the development of an individual : The two parts arise separately, come closer together, and finally unite.
Fig. 349. Section of a sympathetic ganglion in the coeliac region of a frog (Rana esculenta) Showing differentiating phaeochrome cells. Giacomini.
The Cortical Substance
The cortex is of mesothelial (mesodermal) origin. In embryos of five to six mm. the mesothelium at the level of the cephalic third of the mesonephros proliferates and sends buds or sprouts into the mesenchyme at each side of the root of the dorsal mesentery. These sprouts soon lose their connection with the parent mesothelium and unite with one another to form a rather compact mass of epithelial-like cells ventro-lateral to the aorta (Fig. 276). Frequently the two masses fuse across the medial line ventral to the aorta. They constitute the anlagen of the cortical substance of the two suprarenal glands. From the fact that in the lower forms they remain separate from the medullary substance and lie between the urinary organs, they are known as the inter renal organs.
Fig. 350. From a transverse section of a 40 mm. pig embryo. Showing the growth of the medullary substance into the cortical substance of the suprarenal gland. The vessel in the center of the figure is the aorta. Wiesel.
The Medullary Substance
A little later than the appearance of the cortical anlage, the cells of some of the developing sympathetic ganglia become differentiated into two types (i) the so-called sympathoblasts which develop into sympathetic ganglion cells, and (2) phaochromoblasts which are destined to give rise to the phaeochome or chromafiin cells (Fig. 349). Hence the chromaffin cells are derivatives of the ectoderm, since the ganglia are of ectodermal origin.
They soon become more or less separated from the ganglia, migrate to the region of the cortical anlagen and then penetrate the latter in cord-like masses (Fig. 350). Finally these masses unite in the interior of the cortical substance to form a single compact mass (Fig. 351) . Along with the phaeochrome masses, sympathoblasts also are carried in and give rise to the sympathetic ganglion cells within the gland. The two types of cells together constitute the medullary substance. In the lower forms the phaeochrome masses remain separate from the cortical substance and are known as the suprarenal organs. In Mammals the two sets of organs (interrenal and suprarenal) unite to form the suprarenal gland.
Fig. 351. Section of the suprarenal gland of a 119 mm pig embryo. Cor., Cortex; Cor.*, some cortical substance in the center of the gland; Med., medulla. Wiesel.
At the time when the mesonephros is fully developed, the cortical substance forms a small oval body near its cephalic end. During the union of the cortex and medulla and the atrophy of the mesonephros, the suprarenal gland becomes more closely associated with the cephalic end of the kidney, and by the middle of the third month has practically reached its adult position. During the third month and the first half of the fourth month the glands increase in size and become relatively large structures, larger in fact than the kidneys. From the fourth month on, they grow proportionately less than the neighboring organs, and by the sixth month are about half as large as the kidneys. At birth the ratio of their weight to that of the kidneys is about 1:3; in the adult about 1:128.
While perhaps in a normal course of development all the anlagen are united in the adult suprarenal gland, it is not unusual to find accessory structures in various places. Some of these consist of cortical tissue only and are usually found in or near the capsule of the gland. Others may consist of both cortical and medullary substances, and are found in the vicinity of or embedded in the kidneys, in the retroperitoneal tissue near the kidneys, in the walls of neighboring blood vessels, or associated with the internal genital organs in the rete testis or epididymis, or in the broad ligament. These accessory structures may arise independently of the main gland, or they may be portions of the main gland which were separated during the union of the different anlagen of the latter and were carried away in the descent of the genital glands.
In addition to the chromamn tissue which enters into the formation of the main gland or of accessory glands, there are other small masses of this tissue which remain permanently associated with some of the prevertebral and peripheral sympathetic ganglia.
Recent researches have shown that the Carotid Skein (glomus caroticum, intercarotid ganglion, carotid gland), which formerly was believed to be a derivative of the epithelial lining of one of the branchial grooves, is of sympathetic origin and that the cells acquire the characteristic chromaffin reaction. These facts indicate that it is closely allied with the medullary substance of the suprarenal gland.
Anomalies
The Kidneys
Rarely is there congenital absence of both kidneys. More often there is a high degree of aplasia in both organs in otherwise well-developed children. In either case death necessarily soon follows. Not infrequently one kidney, usually the left, is poorly developed or absent and a compensatory enlargement of the other exists. Such malformations are due to deficient development of the organs, but the causes underlying the deficient development are obscure.
One of the most common malformations is the abnormal position of one or both kidneys (ectopia of the kidneys). Usually they occupy a position lower than the normal in the abdominal cavity, which indicates that they have failed, during development, to migrate forward to the normal limit (see p. 369). Very rarely one or both organs migrate beyond the normal limit, in which case they occupy positions cranial to the normal.
Not infrequently the lower ends of the two kidneys are fused across the medial line, giving rise to the so-called "horseshoe kidney." Two renal pelves and ureters are usually present. Occasionally the fusion is so extensive that a single flat mass is formed. This occupies a medial position or lies at either side of the medial line, and may be situated at the normal level or lower. The renal pelvis may be single or double, with one or two ureters. In cases of double ureters and pelves it seems most likely that the anlagen of the kidneys have fused secondarily, that is, after the evagination from the mesonephric ducts (p. 361) . In cases where the pelvis and ureter are single, the fusion may have occurred secondarily, although there is the possibility that only a single anlage appeared.
Occasionally in children and even in adults the kidneys show a distinct lobulation. This is due to the persistence of the lobulation that normally exists in the foetus (p. 367).
The kidney may be more or less movable owing to laxity of the surrounding tissue, or it may Refloating, in which case it has a distinct mesentery. These cases should be distinguished from those in which similar conditions have been acquired, usually as the result of trauma.
Congenital cysts of the kidney are not uncommon. They vary in size and number, sometimes being so numerous that they crowd out the greater part of the renal tissue. Rarely they are so large and numerous that the affected organ fills a large part of the abdominal cavity, resulting in serious or even fatal disturbances of the functions of other organs. There are three views concerning the origin of these cysts, (i) They may be the result of dilatation of certain renal tubules derived from the nephrogenic tissue, which failed to unite with the straight tubules (p. 363). (2) Inflammation in the medulla of the foetal kidney may effect a closure of the lumina of some of the tubules, with subsequent dilatation of the portions (tubules. or renal corpuscles) that are cut off from communication with the renal pelvis. (3) Normally some of the renal corpuscles and tubules degenerate (p. 369), and the,cysts may arise as dilatations of incompletely degenerated corpuscles or tubules or both. While these views appear reasonable, none of them has been proven. All three views express possibilities, and there is no good reason for believing that any one of them expresses the only possibility.
The Ureters
The renal pelvis is sometimes absent, the calyces uniting to form two or more tubes which in turn unite to form the ureter. This probably is the result of abnormal branching of the ureter during development and the failure of the ends of the branches to become dilated. Occasionally the ureter is double or triple throughout the whole or a part of its length. The most reasonable explanation of two or three complete ureters on either side is that two or three separate evaginations arose from the mesonephric duct (p. 361.) Where the tube is double in only a part of its length, an abnormal branching of the single original evagination is indicated. Atresia of one or both ureters is occasionally met with. This probably represents a secondary constriction after the ureter is formed since both ex-aginations are hollow from the beginning (p. 391), but the cause of the constriction is not understood. The atresia results in dilatation of the portion of the ureter on the side toward the kidney.
Abnormal situations of the openings are sometimes seen, the explanation of which is to be found in the relations of these tubes to the mesonephric ducts, to the cloaca, and to the Miillerian ducts. In the male the ureters may open into the seminal vesicles, the prostatic urethra, or the rectum. If one recalls that the ureter arises as an evagination from the mesonephric duct near the opening of the latter into the cloaca (p. 361), that the cloaca becomes separated into a dorsal part (the rectum) and a ventral part (the urogenital sinus) (p. 370), and that the proximal end of the mesonephric duct is so far taken up into the wall of the urogenital sinus (or bladder) that the ureter opens separately (p. 370), it is readily seen that any interference with these normal processes of development will result in abnormal opening of the ureter. If the ureter does not become separated from the mesonephric duct, it will open into the deferent duct (vas deferens), the latter being the proximal part of the mesonephric duct. And since the seminal vesicle is an outgrowth from the proximal end of the mesonephric duct, the opening of the ureter is likely to be associated with the vesicle^ If the separation between the ureter and mesonephric duct is complete, but the opening of the ureter does not migrate cranially on the wall of the bladder, the opening comes to lie in the wall of the prostatic urethra. If the wall (urorectal fold) separating the urogenital sinus and rectum is situated too far dorsally, the opening of the ureter comes to be in the wall of the rectum. (Consult Figs. 322, 323, 324, 325.)
In the female the ureters may open into the urethra, the vagina, or the uterus. The explanation of the opening into the urethra is the same as in the male (see preceding paragraph). The opening into the genital tract is probably to be explained on the ground that the ureters fail to migrate cranially along the wall of the urogenital sinus to the bladder, and as the fused ends of the Mtillerian ducts enlarge to form the uterus and vagina, the openings of the ureters are taken up into their walls.
The Bladder
Absence of the bladder is very rare. Abnormal smallness, due to imperfect dilatation of the urogenital sinus (p. 371), is not infrequent.
The urachus, which represents the portion of the allantoic duct between the bladder and the umbilicus (p. 371), not infrequently persists as a whole or in part, giving rise to certain anomalous conditions in the region of the middle umbilical ligament. The urachus may persist as a complete tube, lined with epithelium, thus forming a means by which urine can escape at the umbilicus. This condition is usually associated with obstruction of the urethra and is known as uracho-vesical fistula. The urachus may degenerate in part, leaving disconnected portions which frequently become dilated to form cysts.
Vesical fissure, the most serious malformation of the bladder, is associated with fissure of the lower abdominal wall. The edges of the cleft in the bladder are continuous with those of the cleft in abdominal wall, the integument being continuous with the lining of the bladder. In some cases the bladder is everted through the cleft, and the cleft may even be so extensive as to involve the external and internal genital organs. Vesical fissure is much more common in the male than in the female. No very satisfactory explanation of this malformation has yet been given. It is in some way connected with imperfect formation of the ventral abdominal wall resulting from influences acting at a very early period of development.
The Urethra
The urethra in both sexes may be abnormally small or abnormally large or partly occluded, owing to faulty development of the urogenital sinus. In the male the penile portion also may be malformed, being represented merely by a furrow on the lower side of the penis. This condition, known as hypospadias, is due to the incomplete fusion or lack of fusion between the genital folds along the lower side of the genital tubercle (p. 394). In extreme cases the defect may involve the scrotum and extend back as far as the prostate gland, the two halves of the scrotum being separated. Epispadias, in which the urethral cleft extends along the upper side of the penis (or the clitoris) is rare, and is usually associated with vesico-abdominal fissure. Its mode of origin is not understood.
The Testicles
One of the most common malformations affecting the male genital glands is the condition known as chryptorchism, in which the glands, instead of descending into the scrotum, are retained within the abdominal cavity. One or both testicles may be affected. They may occupy their original position far forward in the abdominal cavity or may be situated near the inguinal canal, or may lie at some intermediate point. The malposition is due to a failure in the normal descent into the scrotum (p. 389). The cause of the failure is obscure. Not infrequently the ectopic testicles atrophy or fail to develop properly at puberty.
Congenital absence of one or both testicles is rare. More frequently the gland or efferent system of ducts is defective in part, owing to imperfect development. In case of absence of the testicles the individual is small and poorly developed ; when the glands are imperfectly developed the individual is effeminate.
Cysts which are sometimes met with in the epididymis are possibly due t( dilatation of incompletely degenerated portions of the mesonephric tubules or Mullerian ducts. Teratoid tumors and chorio-epitheliomata are occasionally found in the testicle. For a further discussion of these see chapter on Teratogenesis (XX).
The Ovaries
Congenital absence of both ovaries is rare; defective development of one is more common. Either anomaly may occur with or without defects in the other genital organs. Occasionally the ovaries remain rudimentary, their function as egg-producing organs never being assumed. Malpositions, due to partial or complete failure in the normal descent into the pelvis (p. 392), are not infrequent. Sometimes, on the other hand, they descend to the inguinal canal and may even pass through the latter into the labia majora.
Ovarian cysts occur frequently. Some of these (follicular cysts) may arise during, postnatal life as dilatations of Graafian follicles. Others probably arise during foetal life in the same manner. Certain other forms of ovarian tumors, known as cystadenomata, are possibly to be considered as derivatives of the epithelium of the medullary cords which in normal cases disappear entirely (p. 377; also Fig. 328). A discussion of the origin of teratoid tumors of the ovary will be found in the chapter on Teratogenesis (XX).
The Oviducts, Uterus and Vagina
Absence of the oviducts is usually associated with malformations of other parts of the genital tract. On the other hand, normal oviducts may be present in conjunction with defective uterus and vagina. Atresia may occur at the uterine or fimbriated end, or at any intermediate point.
The majority of the malformations of the uterus and vagina can be attributed to defective processes of development in the caudal ends of the Miillerian ducts. It will be remembered that the caudal ends of these ducts normally fuse to form a single medial tube which opens into the urogenital sinus, and which constitutes the anlage of the uterus and vagina (p. 385; Fig. 325). It is obvious that any defect in this fusion will result in some degree of duplicity in the two organs in question. The fusion may be almost complete, the resulting abnormality being merely a small pocket which forms, at each side of the fundus, a continuation of the cavity of the uterus. There may be a greater degree of imperfection in the fusion, resulting in a partial division of the uterus into two horns bicornuate uterus. The wall between the two Mullerian ducts may remain patent in the entire uterine portion of the tract, thus giving rise to a bipartite uterus. If the wall between the ducts remains intact throughout both uterine and vaginal portions, the result is a complete division of the uterovaginal tract uterus didelphys. Occasionally the uterine portion of one Mullerian duct may fail to develop properly and becomes a solid cord, resulting in an unicornuate uterus.
Not infrequently the uterus remains rudimentary infantile uterus. This anomaly is usually accompanied by stenosis of the vagina. Stenosis or other defects in the vagina may occur, however, when the uterus is normal. In rare instances the hymen is absent; in other cases it closes the entrance to the vagina a condition known as imperforate hymen.
Malformations of the uterus and vagina resulting from persistence of the cloaca and atresia of the anus are mentioned on page 326.
Hermaphroditism
--Mark Hill 13:22, 17 January 2011 (EST) Note this is a historic term, now replaced with "intersex".
This condition implies a combination of the male and female sexual organs in one individual, accompanied by a blending ot the general characteristics of the two sexes When such an individual possesses both ovary and testicle, the condition is known as true hermaphroditism ; when the individual possesses ovaries or testicles, the condition is known as false hermaphroditism.
True Hermaphroditism
The presence of both ovary and testicle in one individual is one of the rarest anomalies in man. Furthermore, one or both of the organs are sexually immature. Three forms can be recognized (Klebs) :
1. Lateral hermaphroditism, in which an ovary is present on one side and a testicle on the other;
2. Unilateral hermaphroditism, in which both ovary and testicle are present on one side, either ovary or testicle, or neither, on the other side;
3. Bilateral hermaphroditism, in which both ovary and testicle are present on both sides.
In all these cases the general character of the body is of an intermediate type, sometimes tending toward the male, sometimes toward the female. The external genitalia are also of an intermediate type, with hypospadias, small penis, separate scrotal halves, and small vaginal orifice. The uterus usually shows some degree of duplicity.
False Hermaphroditism
In this type of hermaphroditism, in which either ovaries or testicles are present in an individual with mixed general sexual characteristics, two varieties can be recognized:
- Masculine false hermaphroditism, the more common, in which testicles are present but the external genitalia and general character of the body approximate the female;
- Feminine false hermaphroditism, in which ovaries are present but otherwise male characteristics predominate.
The causes underlying the origin of hermaphroditism are among the most obscure in teratogenesis. It is well known that up to the fourth or fifth week the anlagen of the sexual glands are histologically "indifferent," and later become differentiated into ovaries or testicles (p. 375). Since the secondary sexual characteristics are dependent upon the development of the primary, they also are brought out later. If the " indifferent " glands give rise to both ovaries and testicles, true hermaphroditism is the result; if they give rise to either ovaries or testicles but the external genitalia and general characteristics develop in the opposite direction, false hermaphroditism is the result. Thus the hermaphroditic condition is potentially present in every individual during the earlier stages of development; the most remarkable fact is that it is not more common.
- Next: Integumentary
References for Further Study
ADAMI, J. G.: The Principles of Pathology. Vol. I, 1908.
AICHEL, O.: Vergleichende Entwickelungsgeschichte und Stammesgeschichte der Nebennieren. Arch.f. mik Anat., Bd. LVI, 1900.
Allen BM. The embryonic development of the ovary and testis of the mammals. (1904) J. Anat. 3(2): 89-154.
BEARD, J.: The Germ-cells of Pristiurus. Anat. Anz., Bd. XXI, 1902.
BEARD, J.: The Morphological Continuity of the Germ Cells in Raja batis. Anat. Am., Bd. XVIII, 1900.
BREMER, J. L. : The Interrelation of the Mesonephros, Kidney and Placenta in different Classes of Mammals. Am. Jour, of Anat., Vol. XIX, 1916.
BONNET, R.: Lehrbuch der Entwickelungsgeschichte. Berlin, 1907.
Corner GW. On the origin of the corpus luteum of the sow from both granulosa and theca interna. (1919) Amer. J Anat. 26(1): 117-183.
EGGERTH, A. H.: On the Anlage of the Bulbo-urethral (Cowper's) and Major Vestibular (Bartholin's) Glands in the Human Embryo. Anat. Record, Vol. IX, 1915.
EIGENMANN, C. H.: On the Precocious Segregation of the Sex-cells of Micrometrus aggregatus. Jour, of MorphoL, Vol. V, 1891.
FELIX, W.: Entwickelungsgeschichte des Excretions-systems. Ergebnisse der Anat. u. Entunck., Bd. XIII, 1903.
FELLX, W., and BUHLER, A.: Die Entwickelung der Harn- und Geschlechtsorgane. In Hertwig's Handbuch d. vergleich. u. experiment. Entwickelungslehre der Wirbeltiere, Bd. III. Teil I, 1904.
GAGE, S. P.: A Three Weeks' Human Embryo, with Especial Reference to the Brain and Nephric System. Am. Jour, of Anat., Vol. IV, 1905.
GERHARDT, U.: Zur Entwickelung der bleibenden Nieren. Arch. f. mik. Anat., Bd. LVII, 1901
HERTWIG, O. : Lehrbuch der Entwickelungsgeschichte des Menschen und der Wirbeltiere. Jena, 1906.
Hill EC. Development and vascularization of the testis. (1906) Amer. J Anat. 6: 390-360.
HUBER, G. C. : On the Development and Shape of the Uriniferous Tubules of Certain of the Higher Mammals. Am. Jour, of Anat., Vol. IV, SuppL, 1905.
KED3EL, F.: Zur Entwickelungsgeschichte des menschlichen Urogenitalapparatus. Arch.f. Anat. u. Physiol., Anat. Abth., 1896
KINGSBURY, B. F.: The Morphogenesis of the Mammalian Ovary: Felis domestica. Am. Jour, of Anat., Vol. XV, 1913.
KOHN, A.: Das chromaffine Gewebe. Ergebnisse der Anat. u. Enlwick., Bd. XII, 1903.
KOLLMAN, J. Lehrbuch der Entwickelungsgeschichte des Menschen. Jena, 1898.
KOLLMAN, J.: Handatlas der Entwickelungsgeschichte des Menschen. Jena, 1907, Bd. II.
MARCHAND, F.: Missbildimgen. In Eulenburg's Real-Encyclopddie der gesammten HeUkunde, Bd. XV, 1897.
McMuRRiCH, J. P.: JThe Development of the Human Body. Philadelphia, 1919.
MINOT, C. S.: Laboratory Text-book of Embryology. Philadelphia, 1903.
MORGAN, T. H.: ,The Cause of Gynandromorphism in Insects. Am. Naturalist, Vol. XLI, 1907.
NAGEL, W.: Ueber die Entwickelung des Urogenitalsystems des Menschen. Arch. f. Mik. Anat., Bd. XXXIV, 1889.
NAGEL, W.: Ueber die Entwickelung der Urethra und des Dammes beim Menschen. Arch.f. mik. Anat., Bd. XL, 1892.
NAGEL, W.: Ueber die Entwickelung des Uterus und der Vagina beim Menschen. Arch.f. mik. Anat., Bd. XXXVII, 1891.
PIERSOL, G. A.: Teratology. In Wood's Reference Handbook of the Medical Sciences, Vol. VII, 1904.
POHLMAN, A. G.: The Development of the Cloaca in Human Embryos. Am. Jour, of Anat., Vol. XII, 1911.
POLL, H.: Die Entwickelung der Nebennierensysteme. In Hertwig's Handbuch der vergleich. u. experiment. Entwickelungslehre der Wirbeltiere, Bd. Ill, Teil I, 1905.
POLL, H.: Die Entwickelung der Nebennierensysteme. In Hertwig's Handbuch der vergleich. u. experiment. Entwickelungslehre der Wirbeltiere, Bd. Ill, Teil I, 1905.
RABL, C.: Ueber die Entwickelung des Urogenitalsystems der Selachier. Morphol. Jahrbuch, Bd. XXIV, 1896. Theorie des Mesoderms. Ueber die erste Entwickelung der Keimdruse. Morphol. Jahrbuch, Bd. XXIV, 1896.
SCHREINER, H. E.: Ueber die Entwickelung der Amniotenniere. Zeitschr. /. wissensch. Zoologie, Bd. LXXI, 1902.
SOULIE, A.: Sur le mechanisme de la migration des testicules. Comp. Rend, de la Soc. de Biol., Paris, Ser. 10, T. II, 1895.
SOULIE, A. : Recherches sur le developpement des capsules surrenales chez les vertebres superieurs. Jour. de. V Anat. et de la Physiol., T. XXXIX, 1903.
STOERK, O.: Beitrag zur Kenntnis des Aufbaus der menschlichen Niere. Anat. Hefte, Bd. XXIII, 1904.
SWIFT, C. H.: Origin and Early History of the Primordial Germ-cells in the Chick. Am. Jour, of Anat., Vol. XV, 1914.
SWIFT, C. H.: Origin of the Definitive Sex-cells in the Female Chick and their Relation to the Primordial Germ-cells. Am. Jour, of Anat., Vol. XVIII, 1915.
SWIFT, C. H.: Origin of the Sex-cords and Definitive Spermatogonia in the Male Chick. Am. Jour, of Anat., Vol. XX, 1916.
TANDLER, J.: Ueber Vornieren-Rudimente beim menschlichen Embryo. Anat. Hefte, Bd. XXVIII, 1905.
TAUSSIG, F. J. : The Development of the Hymen. Am. Jour, of Anat., Vol. VIII, 1908.
WIESEL, J.: Ueber die Entwickelung der Nebennieren des Schweins, besonders der Marksubstanz. Anat. Hefte, Bd. XVI, 1900.
WINIWARTER, H.: Recherches sur 1'ovogenese et 1'organogenese de 1'ovaire des Mammiferes. Arch, de Biol., T. XVII, 1900.
WATSON, E. M.: The Development of the Seminal Vesicles in Man^ Am. Jour, of Anat., Vol. XXIV, 1918.
WOODS, F. W.: Origin and Migration of the Germ-cells in Acanthias. Am. Jour, of Anat., Vol. I, No. 3, 1902.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 26) Embryology Book - Text-Book of Embryology 15. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_15
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G