Book - Manual of Human Embryology 12
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Lewis WH. XII. The development of the muscular system pp 454-522 in Keibel F. and Mall FP. Manual of Human Embryology I. (1910) J. B. Lippincott Company, Philadelphia.
XII. The Development of the Muscular System
By Warren H. Lewis, Johns Hopkins University, Baltimore, Md., U. S. A.
The entire musculature, both cross-striated and smooth, with a few exceptions that are of ectodermal origin, arises from the mesoderm. Since contractility is a fundamental property of all cells it is not surprising that the ectoderm as well as the mesoderm should give rise to cells in which this function is highly developed. The ectoderm of the optic cup undoubtedly gives rise in most vertebrates, probably in all, to ,the musculus sphincter and the musculus dilatator pupillae: Nussbaum (1900, 1902), Herzog (1902), Heerfordt (1900), Szili (1901), Lewis (1903), Collin (1903). In mammals these muscles are of smooth fibres, but in birds and reptiles of cross-striated fibres. The muscles of the glandulse sudorifera? are also of smooth muscle derived from the ectoderm: Ranvier (1889), Koelliker (1889), Stohr (1902), Heidenhain (1893).
The mesoderm, however, gives rise to the great bulk of the musculature, both smooth and cross-striated. The smooth and cross-striated muscles are not to be considered as fundamentally diif erent ; they represent different grades of development of contractile tissue or different paths of differentiation from a common fundamental form. The smooth muscle shows a lower grade of differentiation. In insects and birds, for example, portions of the intestinal tract are supplied with cross-striated muscle, while in mammals the corresponding regions are supplied by smooth muscle. Marchesini and Ferrari (1895) found that in early development smooth and striped muscles show exactly the same structure. The fact that the myotomes give origin to many of the cross-striated muscles does not distinguish this variety from the smooth muscle, inasmuch as many of the cross-striated muscles — in the head, for example — arise directly from the mesoderm quite independently of the myotomes and in a manner similar to the origin of the smooth muscle.
It has been customary to consider the voluntary musculature as being derived almost entirely from the primitive segments; yet in mammals the attempts to homologize the head muscles with those of the trunk derived from the myotomes have failed, as there are no indications of preotic segments in the head region; the head muscles develop directly from the mesoderm of the branchial arches and the dorsal eye region. The muscles of the limbs likewise arise directly from the mesoderm of the limb buds; but here uncertainty still exists as to what role migrating cells from the myotomes may play in their development.
The muscles derived directly from the myotomes — namely, the deep muscles of the back and the intrinsic thoracc-abdominal musculature — ^are to be considered as both phylogenetically and ontogenetically the oldest of the skeletal muscles. The skeletal muscles of the head and limbs are, on the other hand, of later origin, and probably not derived from the more primitive segmental musculature, but directly from the mesoderm. Until, however, we have a more complete picture of the developmental history, not only in mammals but also in the lower vertebrates, the relationships must remain obscure. The development of many of the muscles in man and mammals has never been traced, and of the remainder our knowledge is fragmentary and incomplete. It has commonly been supposed that the first differentiation of the muscles from the mesoderm takes place under the influence of the nervous system through the agency of the motor nerves, and that self -differentiation of muscles does not occur. Such a belief arose from our knowledge of the very early union of the motor nerves with the developing myotomes and muscle masses. Teratological evidence is at present conflicting.
Neumann (1891), from his studies of acephalic and amyelic monsters, concludes that the influence of the motor nerves is necessary for the differentiation of the muscular system. Leonowa (1893) and Fraser (1895) have described human monsters without brain and spinal cord in which the peripheral sensory nerves and musculature were normally developed. On the other hand, E. H. Weber (1851), Neumann (1901), and others have described cases in which absence of certain portions of the central nervous system has been accompanied by total absence of musculature which is normally supplied by the lacking nerves, although skeleton, blood-vessels, and even tendons were normally developed. Neumann would reconcile these apparent differences by assuming that muscles first arise under the influence of the nervous system, but that their nourishment and further growth during the embryonic period take place independently of the central nervous system, and not until the post-embryonic life is reached is the dependence again established. Thus, the nervous system must have developed in the early stages of embryonic life up to a certain point and undergone degeneration after differentiation of the muscular system had taken place. Herbst (1901) concludes, from the same data, that the sensory nerves, including the cells of the spinal ganglia, and not the motor nerves, are necessary to stimulate the differentiation of the muscular substance in the embryo. The wellknown fact that a muscle undergoes atrophic changes after its nerve supply has been cut off would at first sight uphold the view that the influence of the nervous system is necessary for the differentiation of contractile tissue. The study of the normal development likewise affords some evidence which might be interpreted as tending to support it, though it does not necessarily do so. In the embryos of lower vertebrates, for instance, the connection of the motor spinal nerves with the muscle plates is established just at the time when the contractile substance begins to be laid down, but in the pig embryo, according to Bardeen (1900), the musculature is differentiated to a considerable extent before the nerves establish a connection with it: Harrison (1904).
It was only by the experimental method on the lower vertebrates that this question seems to have been finally settled, especially by the brilliant work of Harrison (1904). Harrison removed the spinal cord in a series of frog embryos before the histological differentiation in either the muscular or nervous system had begun, so that from the very beginning isolation of the musculature from the nervous system was complete, and all chance for the exertion of any peculiar formation stimulus emanating from the nervous system as such was eliminated; and likewise, owing to the consequent paralysis of the muscles in question, any possible stimulus resulting from the functional activity of the muscle itself was excluded. Still the differentiation of the contractile substance took place in the normal manner, as did the grouping of fibres into individual muscles. It is not likely that the conditions in mammals and man differ from those of the frog. Thus it is seen that all the constructive processes involved in the production of the specific structure and arrangement of the muscle-fibres take place independently of stimuli from the nervous system and of the functional activity of the muscles themselves. Cross-striated muscle tissue and the indiividual muscles are thus self-differentiating.
At a later period during functional activity, as the experiments of severing the nerve to a muscle show, the muscle becomes dependent on the influence of the nervous system for its continued normal existence, either through a trophic influence or functional activiy. At how early a period in the development of the ovum this power of self -differentiation of muscular tissue begins can be but problematical. Lewis (1907) found that in the early gastrula stage of the frog, tissue in the lips of the blastopore possesses the power of independent differentiation into muscular substance. Thus it appears that muscle tissue is already predetermined in the early gastrula. It is probable that this predetermination exists much earlier, even in the egg itself. Conklin (1895) has been able to determine in ascidian eggs, even before cleavage begins, the existence of organ-forming substances, one of which, the myoplasm, that has to do with the formation of muscle tissue, is clearly recognizable and can be followed through successive stages of development into formed muscle.
Harrison's experiments likewise show that the formation of the individual muscles from the myotomes and muscle complexes takes place independently of the nervous system. It has often been assumed that this splitting up of the complex muscle masses into the individual muscles has come to pass through the active ingrowth of connective tissue, blood-vessels, and nerves. There is, however, no experimental evidence indicating that either the connective tissue, the blood-vessels, or nerves take an active part in this process, although descriptive observation might readily lead one to such a conclusion. It is more probable that the explanation lies primarily within the muscle mass itself and secondarily to the relations which the muscle masses may have to shifting skeletal elements. Harrison's experiments would also eliminate functional activity as a necessary factor in this formation of individual muscles.
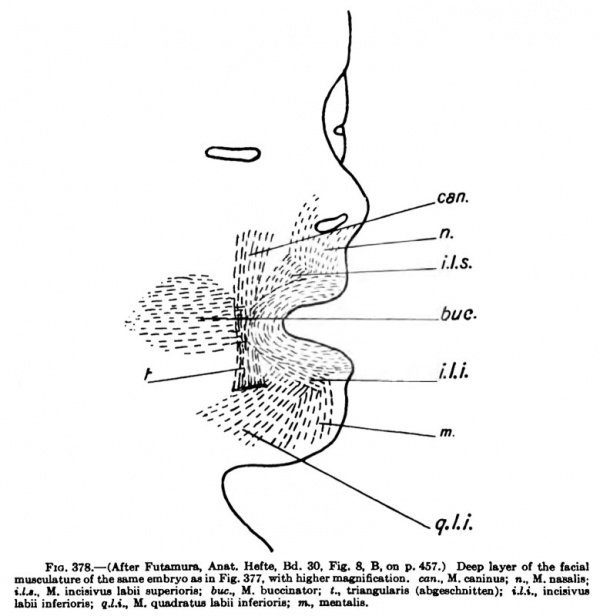
Although the nervous system does not influence muscle differentiation, the nerves, owing to their early attachment to the muscle rudiments, are in a general way indicators of the position of origin of many of the muscles, and likewise in many instances the nerv^es indicate the paths along which the developing muscles have migrated during development: Mall (1898), Nussbaum, Bardeen and Lewis (1900), Lewis (1902), Bardeen (1907), Futamura (1906), Grafenberg (1905). The muscle of the diaphragm, for example, has its origin in the region of the fourth and fifth cervical segments. The nervus phrenicus early enters the muscle mass and is carried with the muscle in its migration through the thorax. The Mm. trapezius and sternocleidomastoideus arise in the lateral occipital region as a common muscle mass, into which at a very early period the nervus accessorius extends, and as the muscle mass migrates and extends caudally the nerve is carried with it. The Mm. pectoralis major and minor arise in the cervical region and receive their nerves while in this position; then the mass migrates caudally and ventrally over the thorax. The Mm. latissimus dorsi and the serratus anterior are excellent examples of migrating muscles whose nerve supply indicates their origin in the cervical region. The M. rectus abdominis and the other muscles of the abdominal wall migrate or shift from a lateral to a ventrolateral or ventral position, carrying with them the nerves. The nervus facialis, which early enters the common facial muscle mass of the second branchial arch, is dragged about with the muscle as it spreads over the head and face and neck, and the nerve splits into its divisions parallel with the splitting of the muscle mass into its various muscles.
The nerve supply serves in part as a key to the common origin of certain groups of muscles. The nervus oculomotorius enters in the early embryo a common muscle mass which later splits into various eye muscles supplied by it. The nervus trigeminus first enters the common muscle mass in the mandibular arch which later splits into the various muscles supplied by this nerve and its branches. The lingual muscles arise from two muscle masses which are supplied by the two hypoglossal nerves. The infrahyoid muscles arise from a common mass supplied by the ramus descendens nervi hypoglossi. The M. trapezius and the M. sternocleidomastoideus arise from a single mass.
Where a muscle is supplied by nerves from two or more segments, the indication is that such muscle has had a complex origin, as the Mm. rectus abdominis, obliquus extemus and internus ; but this is not always the case, for a muscle may receive secondarily new nerves, retaining at the same time its original nerve, as the M. trapezius, which was originally supplied only by the nervus accessorius and later receives branches from the cervical plexus. The M. digastricus also is, according to Futamura (1906), at first entirely supplied by the nervus facialis, later, as the anterior belly becomes constricted off from the posterior, the former obtains its motor nerve secondarily from the nervus mylohyoideus.
The site of entry of a nerve into the muscle, as a rule, marks the region of earliest differentiation (Bardeen, 1907), and in many instances at least the distribution of the nerve within the adult muscle indicates the course of development or growth of that muscle (Nussbaum, 1894).
Histogenesis
Smooth Muscle
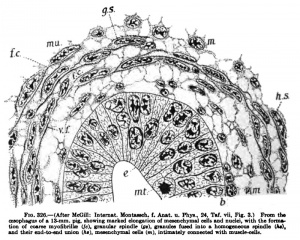
We have already noted that smooth muscle may arise either from the ectoderm or the mesoderm; the great bulk of smooth muscle, however, arises in situ from the mesoderm, either directly from the mesenchymal derivatives of the mesoderm or from embryonal connective tissue. The various stages in its histogenesis from the mesoderm have never been carefully traced in man, and the following account is based on the excellent work of McGill (1907) on the histogenesis of the smooth muscle in the alimentary canal of the pig.
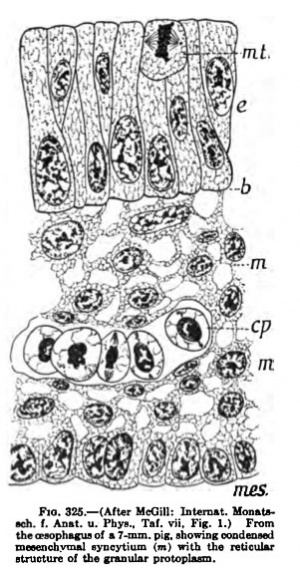
The mesenchyme which arises from the mesodermal germ layer is in the form of a syncytium, with protoplasmic continuity throughout the entire syncytial mass. ** The nuclei of the syncytium are round or oval, with distinct nuclear walls and heavy chromatin reticulum." *' The protoplasm shows a fine reticular structure, the strands of the reticulum being made up of rows of fine granules " (Fig. 325). This syncytium persists throughout intra-nterine life and even in the adult. "Separate and distinct smooth mnscle-cells or fibres do not exist at any stage of development." Thus the term "cell" as here used refers to the enlarged, thickened portion of the syncytium surrounding the nucleus. "Before muscle development begins, there is a general condensation of the mesenchyme," with multiplication of the cells, followed by marked . elongation of the nuclei in the region in which muscle is to differentiate. Not all smooth muscle develops from this primitive mesenchyme, for in later stages the muscle comes from the more developed mesenchyme or embryonal connective tissue. In the alimentary tract, for example, the longitudinal muscle appears much later than the circular and arises from the embryonal connective tissue syncytium into which the primitive mesenchyme has been transformed. "In the areas of muscle formation not all of the mesenchymal cells (or, in
later development, of the emi.itbryonal connective-tissue cells) elongate; some retain their stellate shape, with oval nuclei, and from these, in later development, mnscle-cells may arise; but they form in the main the anlage of the interstitial connective tissue."
"As the elongation of mesenchymal and connective-tissue nuclei continues, in the formation of muscle tissue, there is an increase in the amount of protoplasm surrounding each nucleus. The perinuclear protoplasmic masses also elongate, corresponding to the nuclei, so that the cells change from stellate to spindleshaped," without, however, losing the protoplasmic bridges which unite the entire mass into a syncytium; in fact, the bridges become larger in places. During the earliest stages the muscles increase in size by additions from and transforniation of the mesenchymal cells. Mitosis is abundant in the mesenchyme and rare in the developing muscle. During the second period, in which the muscles are differentiating by the rapid formation of myofibrills and increase in the size of the elongated nuclei, there is little formation of new muscle tissue. Still later there is a second period of muscle growth in the circular layer of the cesophagus, which continues until the adult form is reached. "This increase is apparently due to two factors, — first, differentiation of the embryonal connective tissue both at the margins of the already formed muscle layer and also apparently of that lying between the muscle elements, and second, by the mitotic division of the already formed muscle nuclei,"
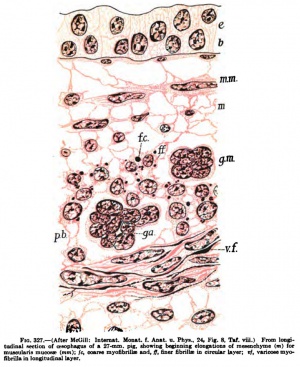
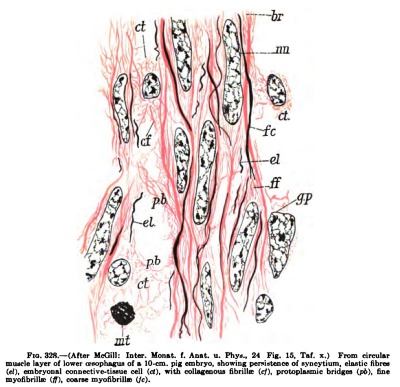
"Immediately following the process of elongation of the mesenchyme, or in later stages of the embryonal connective tissue, the myoflbrillje are fonned in the protoplasm of the elongating cells or nuclear masses" (Fig. 326). "There are two kinds of myofibrilliE, coarse and fine." "The coarse myofibrillte are the first to develop." "The protoplasm of the stellate mesenchyme appears to contain a granular reticulum," and, as the cells elongate to form muscle, the granules increase in number and take a more intense stain than the ordinary protoplasm. "As the elongation continues, the granular fibrils of the protoplasmic reticulum are stretched out more and more, and finally appear as more or less distinct longitudinal strlations" {Fig. 327). "The protoplasm of the cell body appears to be made up largely of these irregular, longitudinal rows of granules, instead of the finemeshed protoplasmic reticulum." "However, at the margins of the cell, in the protoplasmic processes connecting it with neighboring cells and also around the nucleus and between the myofibrilliB, more or less ordinary granular protoplasm remains." These granular fibrillre represent the rudiments of the myofibrillfe and occasionally branch and anastomose with each other. The granular myofibrillae enlarge at certain points, usually near the nuclei; here the granules become coarser, are closely packed together, and form spindle-shaped structures which taper off at each end into myofibrillaB composed of a single row of granules. *'In some cases the end of the spindle appears to break up into several branches of fine granular fibrils, which may anastomose with neighboring fibrillae. * * For the most part, however, the spindles are joined by the intermediate fibrillae into long varicose fibrils which pass through several cells, extending parallel to the elongated nuclei." The granular stage of the myofibrillae does not persist long, however," as soon condensation and fusion of the granules both in the spindles and in the anastomosing fibrillae produce apparently solid varicose smooth fibrillae. These smooth varicose fibrillae may be of great length, extending half-way around the oesophagus, for example, and passing through several cells or nuclear territories. **Soon there may be several running through each cell, causing a marked longitudinal striation. " ' ' Between the spindles, the fibrillae are at first slender, but they gradually become thicker, so that the fibrillae become of a uniform, coarse caliber" (Fig. 328). Here and there finer fibrillae do occur, but they are usually granular in structure and are probably merely stages in the formation of the coarse smooth myofibrillae. "
"In later embryos the granular myofibrillae and spindles are absent or inconspicuous, though smooth, varicose myofibrillae are not infrequent. " "In the late fetus or adult the coarse myofibrillae are sometimes few in number or altogether absent." The fine myofibrillae, to all appearances homogeneous, are always present.
In the second period of the formation of smooth muscle from the embryonal connective tissue the development of the myofibrillae in this embryonal connective tissue seems to follow a different course from that observed in the younger embryos. The presence of the large number of collagenous fibres makes the process difficult to follow. "The fine myofibrillae appear to arise directly as such, without passing through a granular stage," and the coarse myofibrillae arise by increase in the caliber or by fusion of several of the fine myofibrillae into a compact bundle. These coarse myofibrillae differ from those seen in the earlier stages; they are even larger in caliber and present no spindles or varicosities, but in favorable preparations are seen in places to fray out at the ends into fine homogeneous fibrillae indicating that they are merely bundles of fine myofibrillae. In the late fetus and in adult life there is usually a decrease in the number of coarse fibrillae, with a corresponding increase in the number of fine myofibrillae, probably from the splitting of the coarse fibrillae. And "where there are few or no coarse myofibrillae, and at the same time a rapid increase in the number of fine myofibrilltt," it would seem that the latter increase by longitudinal division. In the later stages, then, the fine myofibrillie appear while the coarse fibrillie decrease in number. The fine myofibrillie arise by splitting of the coarse fibrilla or in later stages by new formation in the protoplasm. They become uniformly scattered through the protoplasm, while the coarse fibrilUe come to lie in more or less compact bundles, often near the surface. In later stages myofibrillie cease to develop as new structures, but increase through longitudinal splitting of the fibrillie already present.
Fig. 328.— (After McGill: Inter. Honat. f. Anmt. u. Phyg., Z4 Fig. 15, Tat. x.) From drcular miucle layer of lower cPKiphagua of b lO-cm, pig embryo, ahoning per^isleoce of Hyacytium. elaetic librea (el}, embryoDal connectlve-tiMU* Mil (el), witJi collagencnu Ebrille iefj, protoplMinic bridges (pft), fine myofibrillc (If), coarse myofibrillie ijc).
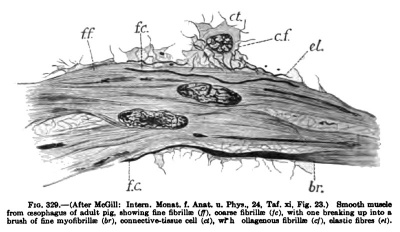
At a certain stage in development collagenous fibrillie begin to form throughout the mesenehjine and also in the smooth-muscle syncytium, and in a single cell both myofibrillje. and collagenous fibrillie frequently diflferentiate side by side (Fig. 329). The presence of collagenous fibrillae in the later stages of the smooth muscle indicates the origin at this time of muscle from the embryonal connectivetissue sjTicytium in which these fibrilte have already developed. This intimate relation of the collagenoas fibrillse with smooth muscle may continue even in adult muscle and the collagenous fibrillffi often run from the protoplasm of one musclecell into neighboring cells, thus binding the tissue firmly together. ' ' In development, protoplasmic connections between the muscle-cells and the connective-tissue cells are easily demonstrated." Through the connecting bandsof protoplasm, myofibrilliemay enter "the protoplasm of the connective-tissue cells or collagenous fibrillse make their way into the muscle-cells." "In later development, most of the collagenous fibrillas are crowded out of the protoplasm of the muscle-cells into the intercellular spaces."
In the walls of the large blood-vessels elastic fibres may arise in the margins of the muscle-cells and are only with difificulty distinguished from the coarse myofibrillje.
« fioa fibrillc Uf). c bnish 01 lioe myoiiDniiK (or), cxHia«tive-lis>ue coll (d>, "In the region of muscle formation some of the embryonal connective-tissue cells do not elongate or form myofibrillfe, but persist as the interstitial connective-tissue cells. The connective tissue, therefore, does not invade the muscle from without, but arises in situ." "After the layers of smooth muscle are established, the tissue increases in amount in two ways: first, by a continuation of the process of transformation of mesenchyme (or later of embryonal connective tissue) into smooth muscle at the surface of the muscle layer, or from the transformation of the interstitial cells. This process predominates in earlier embryos. Second, the nuclei of the already formed smooth muscle multiply by mitosis, especially in more advanced fetal stages."
Cross-striated Muscle
In the mammals and man all the cross-striated muscle arises from the mesoderm, either directly from the mesoderm or its mesenchyinal derivatives, as in the head, or indirectly from the primitive segments, as with the deep muscles of the back and the thoracc-abdominal musculature. The main problem of its histogenesis has been to determine if the adult multinucleated muscle-cell arises from a single cell whose nucleus divides many times or from the fusion of several cells. Each view has had many supporters since the time of Schwann, who held the view of end-to-end fusion, and even to-day the problem is unsettled. Bardeen (1900), in his studies on the pig embryo, takes the view that each muscle-cell arises from a single myoblast which elongates and the nuclei increase within it by direct division. Godlewski (1902), working on the rabbit embryo, found that several myoblasts fuse to form an adult fibre and only rarely do the myoblasts remain single even in the mvotomes.
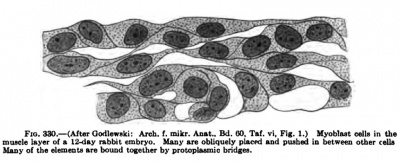
Fig. 330. (After Godlewski: Arch. iDuiele liiyer q[ s 12-day rsbbit embryo. Many of the elemenU are bonded logelher
In the myotomes, muscle differentiation progresses from the anterior to the posterior end of the myotome series. At first the long axes of the myoblasts are at right angles to the sagittal plane of the body, but as these epithelial cells of the myotome differentiate into spindle-shaped myoblasts, their long axes become parallel to the long axes of the body. The pointed ends of the myoblasts, according to Godlewski (1902), push in between other cells and anastomose to form a syncytium, and only rarely are single cells found extending the full length of the myotome with only one nucleus (Fig. 330). In this syncytium differentiation proceeds and most muscle-fibres arise from several cells which have thus fused into the syncytium. Cell division goes on in the syncytium, the daughter cells retaining often protoplasmic connections. The syncytium may extend from one myotome to another. Thus, according to the view of Godlewski, cross-striated muscle like the smooth muscle forms a syncytium, and distinct and separate cells or fibres rarely are to be found.
This view is somewhat different from that of Maurer, who found that cell borders at certain stages become invisible, and concluded that the nuclei divided without cell division. Godlewski holds that cell division goes on hand in hand with nuclear division in the earliest stages of muscle formation, the cells retaining protoplasmic continuity.
The muscle-forming cells are at first cylindrical and epitheliallike. As the myoblasts increase in size they are found to contain many small round granules scattered throughout the cell (Fig. 330). These granules soon become arranged in rows, first in the central part of the protoplasm. They then migrate peripheralwards (Godlewski), forming the rudimentary fibrillae. As the fibrillae develop the granules lie closer and closer together by increasing in number, rather than size, until finally a continuous thread is formed. Here again is seen a striking similarity between the smooth and cross-striated muscle development in the formation of the fibrill®. Wliether these granules actually fuse or are only pressed together Godlewski could not determine. The granular chains, as well as the continuous fibrillse which arise from them, in later stages pass through several cell territories, and as the fibrillae rapidly increase in length they soon extend the entire length of a myotome. The granules between the fibrillae become rarer and rarer as the fibrillse increase in number.
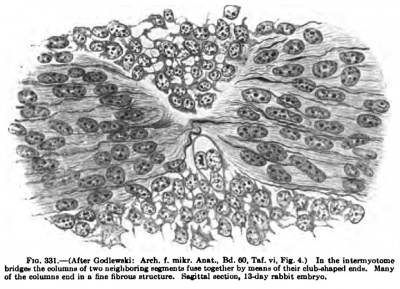
The fibrillae become arranged more and more parallel to the axis of the cells and are grouped about the nuclei. The fibrillae extend through several cells from one myotome septum to another. The rich connections between the cells often make the myotome one complete syncytial mass. The fibrillae are gathered together into columns, which become spear or spindle shaped towards the myosepta, and often fuse with the colunms in the next myotome through the protoplasmic bridges connecting the myotomes (Fig. 331). The club-like ends of the fibrillae columns are often seen to be composed of many fine fibrillae (Fig. 331). In later stages some of the columns may pass through several myosepta, independent of cell or myotome boundaries ; many of them, however, end at the myosepta in club-like thickening, often with tufts of fine fibrillae, the primitive fibre components.
The simple fibrillae soon begin to show a segmentation into two differently staining substances. Do the segments correspond with the original granules which may not have completely fused into the thread, or are they entirely new structures f The deeply staining segment corresponds with the Q anisotropic band of the adult fibre and the other with the I isotropic striation. Wagner, Bardeen, and others have also observed that the fibrillae at first show no cross-striation.
The histogenetic process during this first period is essentially the same in muscles which arise independently of the myotomes. This new formation of muscle from the mesoderm Godlewski found was best observed on the surface of developing muscles. The mesodermal cells elongate into spindle-shaped myoblasts, rich in protoplasm and containing many granules. The myoblasts arrange themselves with their long axes parallel and unite into a syncytium by their processes. The nuclei divide by mitosis, but the daughter cells often remain connected together. The fibrillte form in a similar manner as described above in the myotomes.
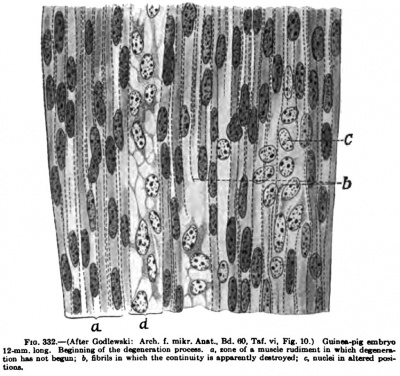
The second period begins with the physiological degeneration which has been described by S. Mayer (1886), Barfurth (1887), Bataillon(1891, 1892), Schaffer(1893), Bardeen(1900), Godlewski (1902). This account is based on the description of Godlewski (1902). In certain regions of the developing muscles the fibrillar columns are seen to break up and disappear, the nuclei become irregular, and the direction of the long axis, which imtil now has been parallel to the long axis of the fibre, is altered so that the nuclei are often diagonal or crosswise. The protoplasm becpmes more homogeneous and gathers about the nuclei in stellate masses'. The nuclei become pale and poor in chromatin, but often retain the power of division for a long time. In some forms of degeneration the muscle-cells become vacuolated (Fig. 332). Many of the degenerating muscle-fibres disappear. After the stage of degeneration new muscle-fibres are formed by longitudinal splitting of the normal ones already present.
The fibrillse multiply by longitudinal splitting and becomfe grouped into the columns in such a manner that similar cross striations (Q and I) in the same muscle-fibre lie in the same planes. Parallel with this process new striations appear, first the Z and later the M zones. In a ten-weeks hiunan embryo the crossstriation is already completely developed.
Both Bardeen and Godlewski noticed in early stages that during mitosis the protoplasmic granules increase in number. At this time the cell bodies usually split during mitosis, but occasionally more than one nucleus is found in a cell. In later stages amitotic division occurs, and fibres come to have many nuclei, both medial and peripheral. Godlewski believes that the fibrillte play an active part in the wandering of the nuclei to the periphery. The inner nuclei are present in the earliest phases of development and later wander to the periphery of the musele-fibre. Mitosis occurs in both inner and outer nuclei.
During the development of the myotomie muscles new myoblasts seem only to arise through division of the myoblasts already present in the myotomes, and there is probably no direct transformation of mesenchyme cells into myoblasts. But with those muscles arising directly from the mesenchyme there is for a period a continuous transformation of mesenchyme cells into myoblasts, as in the muscles of the limbs and head. This process early ceases, and new muscle-fibres then arise only by cleavage of those present.
Fig. 332.— (After Godleiralii: Arch. t. mikr. AnBt., Bd. flO. Tal. vi, Fig. 10.) Giunw-pig embryo 12 mm long. Beginning of the deecaeiscian procesa. a, lone of & miude rudiment in which deseocmtion baa Dot begun; b, fibrils in which the continuity in appuvotly deatroycd; c, nusld in altered prni
According to Maccallum (1898), who counted the fibres of the sartorius muscle in man at various ages, muscle-fibres cease to multiply in fetuses from 130 to 170 mm. in length, and henceforth the muscles increase in size by enlargement of the individual fibres. Meek (1898, 1899) observed in several mammals (rat, cat, and sheep) that the fibres decrease in number soon after birth. The lucky fibres get into better relations with the nutritive supply, etc., while the unfortunate ones are squeezed out and so degenerate (a survival of the fittest). The increase in the size of the muscle after birth is dependent on increase in the size of the individual fibres. Morpurgo (1898) was unable to find this decrease in the number of the fibres, but did observe that multiplication ceases a short time after birth and muscles increased in size by increase in the size of the fibres.
The Segmentation of the Mesoderm
The development of the voluntary skeletal musculature may be said to begin with the segmentation of the dorsal division of the tnmk mesoderm into the primitive segments. The musculature arising from the primitive segments is both phylogenetically and ontogenetically the oldest in the body.
It is uncertain which segment is the first one to form, Keibel claiming that in mammals, and Maurer that in all vertebrates, the most anterior one is the first to become segmented off from the mesoderm. Paterson (1907) has shown, by experiments on chick embryos, that the first segment to form is the most anterior one and that segmentation progresses posteriorly. This probably holds true for all vertebrates.
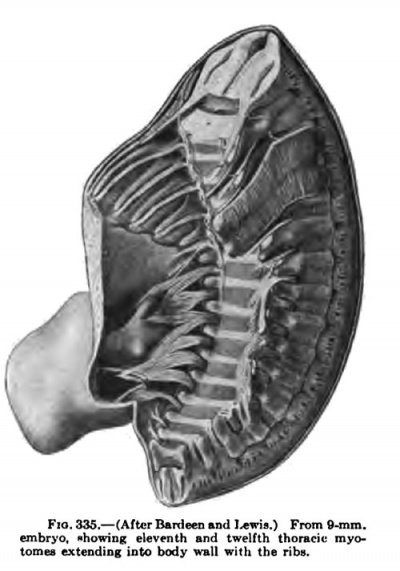
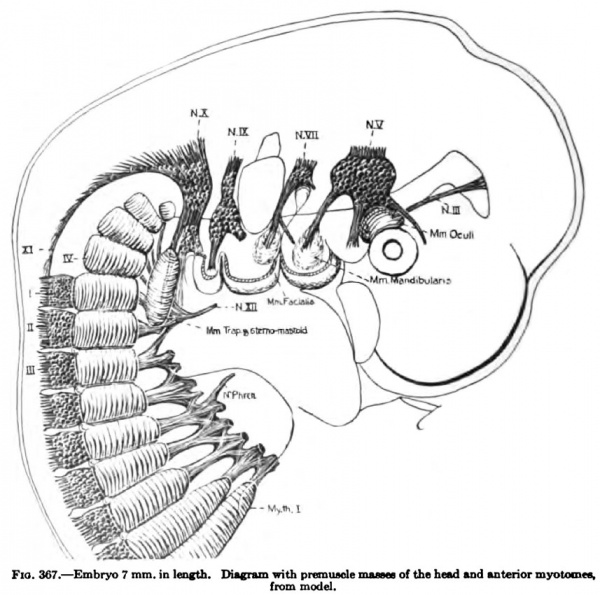
The segmentation of the mesoderm begins in embryos between 1.17 mm (Frassi) and 1.38 mm (Kroemer-Pfannenstiel) in length. In the latter embryo there are 5 or 6 pairs of primitive segments and in the former none. There is perhaps some variation in the time of the first appearance of the segments, for in an embryo 1.54 mm. in length (Spec, Gle) there are no primitive segments, while in an embryo 1.6 mm in length (Unger-Keibel) there are about 9 pairs. In an embryo of 2 mm in length (Mall, 391) there are 8 to 9 pairs of primitive segments which are still connected with the lateral mesoderm. Segmentation progresses rapidly in a caudal direction. In embryos 2 to 3 mm in length the number of segments ranges from about 10 to 20 pairs; in embryos 3 to 4 mm. in length, from 20 to 30 pairs; 4 to 5 mm, 30 to 35 pairs; 5 to 6 or 7 mm, 35 to 40 pairs. From 38 to 40 pairs of segments are formed, 3 to 5 occipital, 8 cervical, 12 thoracic, 5 lumbar, 5 sacral, and about 5 coccygeal. In embryos of 7 to 9 mm in length this segmentation attains its highest development (Figs. 367, 336, 368, 335, 334). The segments soon lose their individual identity as such, and in embryos from 10.5 to 12 mm in length the myotomes fuse to form a continuous column (Fig. 337). This fusion progresses in an anteroposterior direction.
The Differentiation of the Primitive Segments
Just as there is a progressive segmentation of the mesoderm in an anteroposterior direction, so we find there is a progressive differentiation of these primitive segments in the same direction. Thus, in embryos of 4 to 5 mm. in length the anterior segments show quite advanced differentiation while the posterior ones still retain the more primitive conditions.
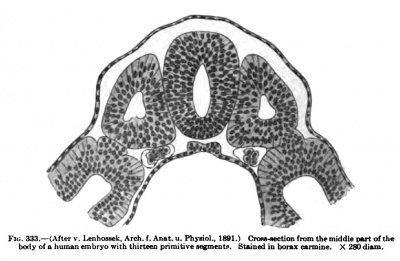
Fig. 333. Cross-section of the middle part of the human body with 13 primitive segments. Stained in borax carmine. x290 diem. After v. Lenhossek. Arch, f. Anat. u. Physiol. (1891)
The differentiation of each segment follows a common plan progressing in a cranio-caudal and a dorso-ventral direction. The primitive segment is at first cubical, with simple epithelial walls surrounding an empty cavity or myoccel. In sections the dorsal, lateral, medial, and ventral walls are more or less clearly to be recognized (Fig. 333). The first change which takes place is the migration of cells from the walls into the cavity, especially from the medial and ventrnl walls. Later, as the myoccel becomes filled with these cells, this nidimeat, with much of the medial and ventral walls, migrates medially towards the chorda and neural tube, forming the rudiment of the sclerotome. Following the migration of the sclerotome the dorsal wall grows ventrally along the medial surface of the outer lamella to form the medial lamella of what has now become the myotome. With the growth of this medial lamella to the ventral edge of the outer lamella, with which it imites, the myoccel becomes reduced to a narrow cleft between the two lamella?. The myotome now consists of a two-layered quadrilateral body, with the lateral or cutis plate, the medial or muscle plate, dorsal and ventral edges, and anterior and posterior edges which are in contact with the preceding and succeeding myotomes (Fig. 334). In a threeweeks embryo this condition is found in the anterior segments while the more posterior ones still show the first stages of the primitive segments.
In an embryo 4.9 mm in lengtli, for example (Ingalls, 1907 ) , the second coccygeal myotome is just separating from the caudal mesoderm. In the anterior coccygeal myotome the myoccpl is filled with sclerotome cells arising from its walls. In the sacral region these cells, together with tlie medial and ventral walls, are pushing towards the chorda and neural tube, while in the anterior sacral region the cranial part of the medial wall is broken through and it together with the sclerotome cells of the myoccel are migrating towards the chorda and neural tube. In the lumbar region the medial and ventral walls, together with the myoctel cells, have migrated toward the chorda and neural tube, leaving a wide opening into the myoccel, the intervertebral cleft. At the dorsal edge of the mj-otome the inner lamella {muscle-plate) is beginning to grow ventralwards along the medial surface of the outer lamella. In the thoracic region this inner lamella has grown well towards the ventral edge of the outer lamella, which edge has rolled medially, and from this free edge sclerotome cells are still migrating. With the ventral growth of the inner lamella and its union with the edges of the outer lamella, the opening into the myocrel is gradually reduced in size, but persists towards the caudal end of the ventral border as the intervertebral cleft. In this region later lie the spinal nerves and segmental blood-vessels. The outer lamella during this process has extended further ventrally and also increased in thickness, its cylindrical epithelial cells remaining for the most part with their long axis at right angles to the cleft-like myoccel. The medial lamella as it grows ventrally increases in thickness; its cells become spindle shaped, with their long axis parallel to the dorsal edge of the myotome. The cells of the dorsal edge, however, retain longer their primitive epithelial character and form the vegetative centre for the dorsal extension of the myotome. In the anterior thoracic region cells from the medial surface of lateral lamella separate from it to join the medial or muscle-plate. The ventral edge of the myotome is formed by the bending medially of the ventral edge of the lateral plate, which has elongated ventrally during the formation of the myotome. Ingalls also finds in this 4.9-mm. embryo that in both sacral and lumbar regions the ventral edge is not very sharply marked off from the surrounding mesenchyme. In the lumbar region cells appear to be migrating from the edge of the myotome into the limb bud. In the cervical region the migration of cells into the arm bud is even more marked, and from some of the myotomes distinct epithelial buds project towards the arm bud from the lateral lamella near its ventral border. The fourth, fifth, and sixth cervical myotomes also show migrating cells from their ventral edges.
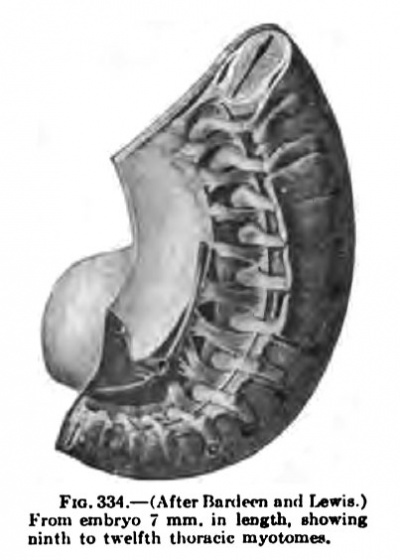
Fig. 334. From embryo 7 mm in length, showing the ninth to twelfth thoracic myotomes. (After Bardeen and Lewis)
The cells of the medial or muscle lamella are gradually transformed into elongated spindle cells extending in an anteroposterior direction from one end of the myotome to the other. These cells later form muscle-cells.
The fate of the lateral or cutis lamella is still in dispute, even in the lower vertebrates. Most observers agree that the lateral or cutis lamella gives rise to both muscle and connective-tissue forming cells (Kollmann, Kastner, Fischel). Ingalls found muscle forming cells, but could not determine in the 4.9 mm embryo studied by him whether the lateral lamella gives rise to connective tissue cells or not. Bardeen (1900), in his studies on both pig and man, concluded that the lateral lamella, except for the degeneration of some of its cells, gives rise only to muscle-cells.
The ventral edge or portion of the myotome gives rise to the ventrolateral trunk and neck musculature. The median or muscle lamella is entirely transformed into muscle-fibres. By longitudinal fusion and splitting of the myotomes arise the deep back muscles of the trunk and neck. The deepest layers, however, probably retain more or less of the primitive segmental arrangement (Bardeen).
The Muscles of the Trunk
The intrinsic muscles of the trunk are derived directly from the myotomes. By the intrinsic muscles I mean, first, the deep muscles of the back, — namely, those beneath the musculi serratus posterior superior and inferior. In the adult the deep muscles, especially in the lumbar and thoracic regions, are encased in the fascia lumhodorsalis. Thus, the muscles from the trunk to the shoulder-gii-dle are excluded from this group, and, as will be shown below, probably do not come from the myotomes. The second group includes the ventrolateral muscles of the thorax and abdomen,— namely, the Mm. serratus posterior superior and inferior, intercostales, obliquus abdominis internus and externus, rectus and transversus abdominis, and quadratus lumhorum. In the third group are the pre-vertebral muscles, the Mm. longus capitis and colli, and rectus capitis anterior.
The Deep Muscles of the Back
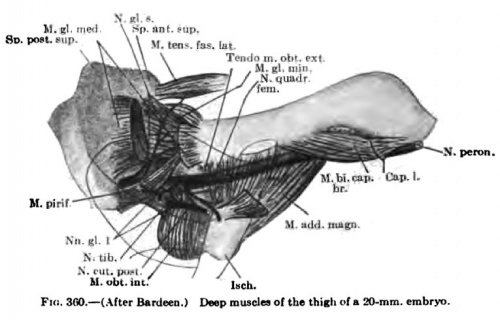
The deep muscles of the back arise from the myotome column which results from the fusion of the myotomes and the disappearance of the myosepta. This fusion, as we have seen, progresses m an anteroposte,om« «tendm« into body v^n wi.h the rib«. rior direction, and already in a 9-mm. embryo traces of segmentation, at least superficially, are beginning to disappear in the cervical and thoracic regions (Figs. 335, 336). This process is complete in embryos of 11 to 12 mm. in length (Fig. 337). The occipital myotonies, at least the caudal ones, are probably fused into this myotome column (Figs. 367, 368). Superficially all traces of segmentation are lost, but the deeper portion of the myotomes lying in contact with the vertebral column probably retains in man the primitive segmentation, as described by Bardeen (1900), in the pig. Thus, the Mm. interspinales, rotatores breves, levatores costarum, and intertransversarii probably retain throughout intra-uterine and extra-uterine life the primitive segmental arrangement of the myotomes.
Fig. 335. (AttcrB^rcleemmdl^wiB.) Fn>mB-mm. _, , embryo, »homng; eleventh and twelfth thoraric myo
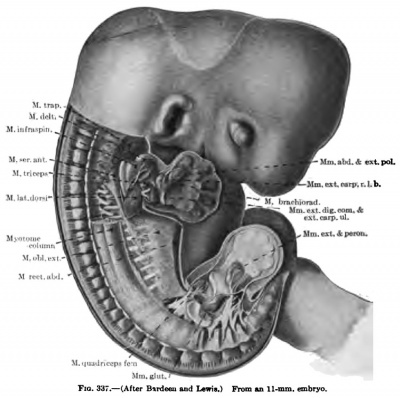
By longitudinal and tangential splitting of the myotome column the various muscles of the back arise, and the subsequent segmentation of muscles arising from the myotome column is a secondary one and need bear no relation to the primitive segmentation. This process of splitting has already began in an embryo 11 mm. in length in tiie cervical and thoracic regions, even wlnle traces of segmentation are still visible at the caudal end of the myotome column (Fig. 337).
The superficial portion of the myotome column seems at first to split longitudinally into two main divisions, — a dorsal one which gives rise to the spinalis and longissimus groups, and a ventrolateral one which gives rise to the ilioccstalis group of muscles (Fig. 337). In the cenical region this process is more complex than in tlie thoracic region, while in the lumbar region the more primitive conditions of the myotome column persist to adult life.
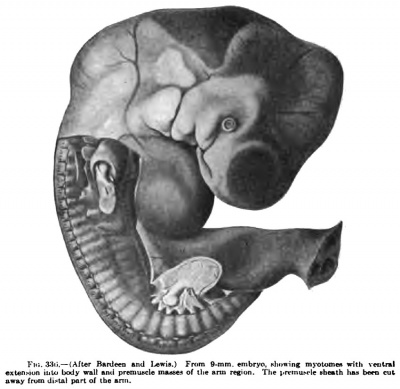
Fig. 336. AIter Bonteea find I.eRii.) Fnim Sjnm. cmbryo showing m}-oUilucii irith veulrml exteiiMon iulu body mil arul premuncle muHS of llie uno ngion. Tlie pmiiu>rlt ihtatli liu ben cut sway frum dl<ul part of tbe snii.
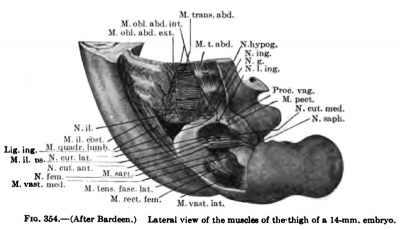
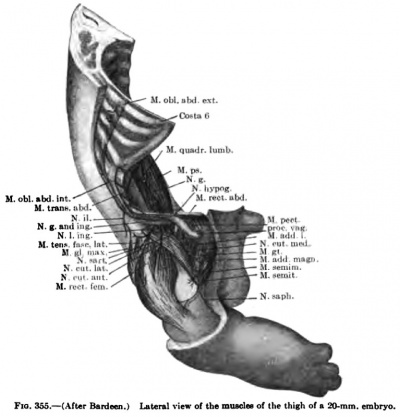
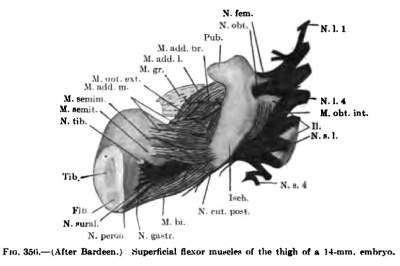
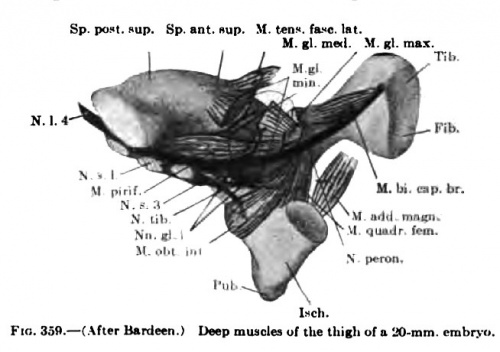
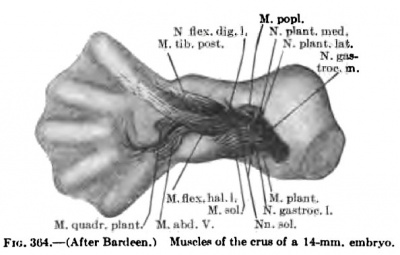
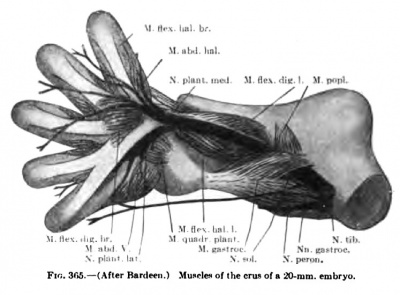
The details in the development of the deep back muscles have never been studied. I have observed that in a 14-mm embryo practically all the deep muscles of the back are to be recognized without great difficulty except the suboccipital muscles. At this stage the Mm. rectus capitis posterior minor and major and the obliquus capitis inferior form a more or less continuous sheet. Through the different attachments of various portions of this muscle sheet one can recognize the position of the individual muscles. In a 20-mm. embryo these muscles are distinct and easily recognized.
The development of the fascia lumbodorsalis proceeds parallel with the development of the myotome column and its muscle derivatives. In an 11 mm embryo this fascia is already present in the upper thoracic and lower cervical regions, enclosing the muscles in a distinct sheath which extends from the spinous processes into the lateral region of the neck and thorax. This fascia remains an important landmark between the true myotome muscles and those which subsequently migrate into the more superficial region, such as the Mm. trapezius, rhomboideus, and latissimus dorsi, and in part also the Mm. serratus posterior superior and inferior.
The growth of the dorsal muscles towards the mid-dorsal line is dependent on the extension and formation of the vertebral arches.
The Thoracic-Abdominal Muscles
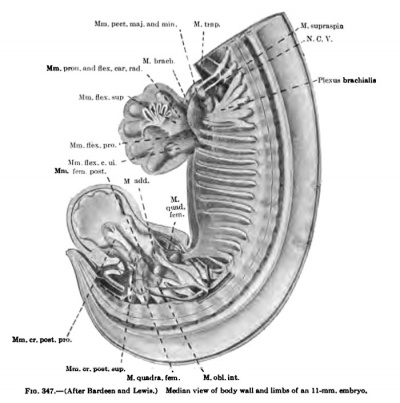
The thoracc-abdominal muscles arise through the ventral extension of the thoracic myotomes into the body wall. The myotomes begin to grow into the body wall with the development and extension of the ribs. Even in a 7 mm embryo, before the myotomes are fused into a continuous column, this process has already begun (Fig. 367). In a 9 mm embryo the myotome processes extend farther ventrally than do the ribs (Figs. 335, 336). As the myotomes extend into the body wall, they lie partially between the ribs and partially lateral to them, where they fuse into a continuous lateral sheet which extends beyond the tips of the ribs, ending ventrally in a continuous column, the rectus rudiment, formed by the fusion of the entire thickness of the myotome processes (Fig. 336). The myotome processes also fuse over the medial surface of the ribs to form a continuous medial sheet, which is continuous with this rectus rudiment lying ventral to the tips of the ribs. The ribs and this ventrolateral musculature gradually grow farther and farther into the body wall and finally reach the mid-ventral region. Meanwhile, long before this occurs, while the ventral edge of this invading skeletal and musculature wall occupies the extreme lateral position along the sides of the embryo, the muscles begin to differentiate (Figs. 337, 347). Through longitudinal splitting off of the muscle column formed by the fusion of the ventral ends of the myotome processes arises the M. rectus abdominis. Through tangential splitting of the lateral sheet arises the M. obliquus abdominis externus and more dorsal to this the Mm. serratus posterior superior and inferior. The medial sheet gives rise to the Mm. obliqnus abdominis intemus and transversus abdominis. The portions of the myotome processes remaining between the ribs give rise to the Mm. intereostales extemi and interni. They are already clearly differentiated in an 11-mm. embryo. The Mm. intereostales apparently always retain the primitive segmentation of the rayotoraes. The lines transversa; of the rectus are often spoken of as representing the primitive myosepta and the muscle bellies between as primitive myotome segments. In the fusion of the ventral ends of the myotomes to form the rectus, I have been imable in early stages to recognize the myosepta, and it may be that the segmentation of the rectus is a secondary process. The segmentation of the M. obliquus abdominis extemus is for the most part secondary, except as pointed out by Bardeen (1900), traces of segmentation remain where it lies in direct contact with the intercostales extemi throughout its period of development. The Mm. obliquus abdominis intemus and transversus abdominis do not retain any traces of the primitive segmentation.
Fig. 337. (After Bardeen and Lewis) From a 11 mm embryo.
Fig. 338. (After Bardeen and Lewis)
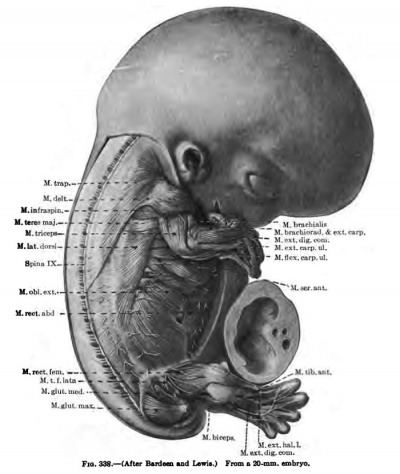
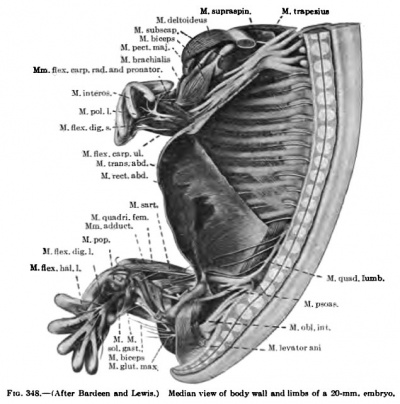
In 14 mm and 16 mm {Fig. 349) embryos the abdominal muscles are clearly differentiated and present much the adult form, although they still occupy a more lateral position, and even in a 20 mm embryo the two recti are widely separated (Figs. 338, 339, 348).
There are no observations that 1 am aware of that explain the origin of the Unese semicircularis (Douglas).
The Subvertebral Muscles
The subvertebral muscles of the cervical regiou are formed by the growth of the myotomes on to the ventral surfaces of the vertebral bodies and subsequent fusion of these myotome processes into a continuous colunm to form the Mm. longus colli and capitis. The M. iliopsoas is not derived in this manner, but extends upwards from the femoral region and belongs to the nmsculature of the leg.
The Muscles of the Perineum
Popowsky (1899) has given the best account of the development of the muscles of the perineum. They arise from the musculus sphincter cloaca?, a skin muscle which is present in a 2-months embryo (Fig. 340). During the third month of fetal life it divides into a M. sphincter ani extemus and a M. sphincter sinus urogenitalis (Fig. 341). This division is dependent upon the separation of the simple single cloaca! opening into two openings. The M. sphincter ani extemus alters very little during the later development, bat the M. sphincter urogenitalis undergoes manychanges, giving rise as it does to perineal muscles. During the fourth to fifth month the M. sphincter urogenitalis gives rise first to the M. ischiocavernosus, bulboeavemosus, and M. sphincter urethra membranaceic (Figs. 342, 343, 344, 345). The M. transversus perinei are the last ones to develop out of the peripheral and lateral part of the M. bulbocavemosus.
The M. levator ani arises from a quite different source and only secondarily comes into relation with the M. sphincter ani and the perineal muscles. The M. levator ani arises in connection with the M. coccygeus and gradually descends to the rectum, bladder, prostate, and vagina and thus comes into contact and intimate union with the muscles of this region. The difference in the nerve supply between the perineal muscles and the M. levator ani indicates their different origin.
The Ventrolateral Muscles of the Neck
Under this heading are included three groups of muscles: (1) the infrahyoid and diaphragm group, (2) the scaleni group, and (3) the levator scapulae and serratus anterior. The Mm. trapezius and stemocleidomastoideus arise in the occipital region and migrate to the shoulder-girdle, and are described with the muscles of the shoulder-girdle. The myotomes of the cervical region do not appear to extend very far ventrolateralwards into the neck region, as do the thoracic myotomes into the intercostal spaces (Fig. 367). This ventral extension of the thoracic myotomes is closely associated with the origin and development of the ribs. In the cervical region the only apparent ventral extension of the myotomes is between the transverse processes onto the ventral surface of the vertebral column to form the rudiments for the subvertebral muscles. There occurs, however, in the cervical region of very young embryos, 7 mm. in length, closely packed mesenchyme or premuscle tissue in the region where later these three groups of neck muscles differentiate. The origin of this premuscle tissue is uncertain. It may arise entirely from the mesoderm of this region or the myotomes may contribute cells to it, but there is no direct proof of this nor of any distinct myotome buds.
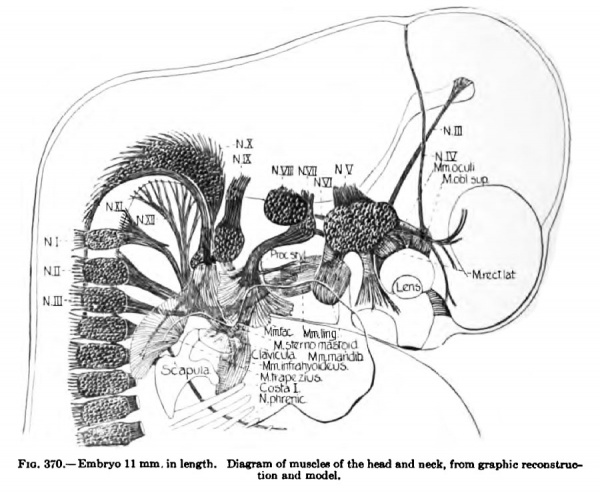
In a 9 mm embryo the rudiments of the neck muscles are beginning to differentiate. At this stage the infrahyoid premuscle mass is especially well seen and apparently develops more rapidly than the other groups (Fig. 367). In early stages the premuscle tissue is continuous above with the tongue premuscle masses and T)elow with the diaphragm masses. It consists of a distinct band of premuscle tissue extending on either side, from the base of the tongue, caudolaterally towards the tip of the first rib. It forms the most ventral group of the neck muscles and already is supplied by the ramus descendens nervus XII. Above the heart towards the base of the tongue region the two lateral masses approach each other and nearly meet in the midventral line; as they descend, however, they become widely separated by the heart and occupy a lateral position between the dorsolateral angle of the heart and the vena cava superior. In later stages as the heart descends into the thorax the muscle masses of the two sides approach each other more and more, the approximation extending from above downwards. In a 14-mm. embryo this approximation is nearly complete. At this stage the muscle mass already shows cleavage into the Mm. stemohyoideus, stemothyroideus, and omohyoideus ; the latter can now be traced to the scapula. This cleavage process begins in embryos of about 11 mm. in length (Figs. 370, 379). In a 20-mm. embryo the muscles have almost the adult form. The muscles of the two sides lie parallel to each other near the midventral line extending from the hyoid and thyroid cartilages to the rudimentary halves of the sternum.
The diaphragm arises from two premuscle masses, one on either side, which are, in a 9-mm. embryo, closely connected with the caudal end of the infrahyoid masses and at this stage lie in the lower cervical region, and, like the infrahyoid masses, there is no direct proof of their origin from the third to the fifth myotomes (Fig. 369). The diaphragm premuscle mass already extends a short distance into pleuroperitoneal septum, although still some distance above the first rib. The plirenic nerve enters the mass. In an 11-mm. embryo the diaphragm muscle mass has descended some distance into the thoracic cavity, carrying with it the nerve, and all connection with the infrahyoid muscle mass has of course disappeared (Fig. 379). In a 14-mm. embryo the diaphragm and its muscle have descended still farther into the thoracic cavity, to occupy more nearly the adult position, and the muscle masses of the two sides have joined. The muscle is best developed in the dorsal portion of the diaphragm in the region of the vena cava inferior and the oesophagus. The crural attachments to the vertebral column do not develop until later.
The scaleni muscles arise from premuscle tissue, ventral to the lower cervical myotomes. From the material at my disposal I have been unable, however, to trace satisfactorily the early development. Already in an embryo 11 mm. in length three scaleni muscles are fairly well differentiated, with the adult attachments. They apparently develop in situ except for the caudal extension of the M. scalenus posterior to the second rib.
The Mm. levator scapulae and serratus anterior, which arise from the premuscle tissue of the lower cervical region and secondarily migrate to the thoracic region, are described with the muscles of the shoulder-girdle.
The Musculature of the Extremities
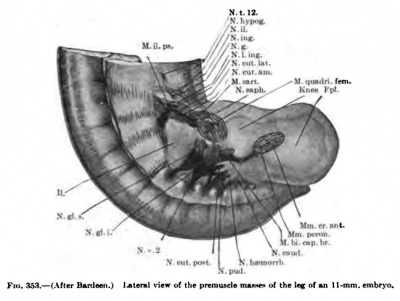
The limb buds first appear, in embryos of about 4 mm in length, as oval projections from the lateral surface of the Wolffian ridge. The arm bud lies opposite the ventral ends of the fifth to eighth cervical and first thoracic segments, and the leg bud opposite the first to fifth lumbar and first sacral segments. At first the limb buds are filled with a closely packed mesenchyme derived from tlie parietal layer of the mesoderm. As the limb buds increase in size, the myotomes extend ventrally in such a manner that the dorsal attachment of the limb bud lies over the ventral portion of the myotomes.
One of the important and much-disputed questions concerning the origin of the limb muscles is the part which the myotomes play or do not play in their origin. It has often been assumed that the myotomes send off large buds into the limbs which combine in various ways to form the muscles, and that the brachial and lumbosacral nerve plexuses were indicators of the complexity of these combinations. Mall (1898) expressed this idea in describing the relations between nerve and muscle in the following sentence: **As the segmental nerves appear, each is immediately connected with its corresponding myotome, and all the muscles arising from a myotome are always innervated by the branches of the nerve which originally belonged to it.* While this remains true for the muscles which actually do come from the myotomes directly, it does not apply to the muscles of the limbs. There are no observations of distinct myotome buds extending into the limbs, such as have been observed in the lower vertebrates or such as can readily be seen extending into the thoracic wall. A number of observers, however, have found in embryos 4.5 to 5 mm. in length a diffuse migration of cells into the limb buds from the ventral and ventrolateral portions of the myotomes that lie opposite the limb buds: Fischel (1895), Ingalls (1907), Kastner (1890), and Kollmann (1891). Ingalls (1907) not only observed this diffuse migration of cells from the ventral portion of the myotomes into the limb buds in an embryo 4.9 mm. in length, but also saw distinct processes from the lateral surface of the myotome near its ventral edge extending lateralwards towards the limb bud.
All observers seem to agree, however, that these migrating cells soon lose their epithelial character and become indistinguishable from the cells of the closely packed mesenchyme filling the limb bud. Kollmann (1891) has pictured these cells from the myotome as forming a continuous band, gradually extending beneath the ectoderm of the limb bud, first on the dorsal then on the ventral side of the limb bud, and continuing on into the ventrolateral body walL This myotome sheath he pictures as lying between the ectoderm and the axial core of somewhat less closely packed mesenchyme. At the distal end of the limb bud this axial core, according to Kollmann, pushes through the muscle band, thus dividing it into a dorsal or extensor layer and a ventral or flexor layer. KoUman's scheme, however, has not been verified by more recent observers.
Bardeen (1907) and Lewis (1902) were unable to find either myotome buds or distinct migrating cells from the myotomes into the limb buds, and it is possible that the myotomes play no part whatever in the origin of the musculature of the limbs. The character of the mesenchyme found in the early limb buds is very similar to that in the branchial arches from which the head muscles arise. The idea that myotomes play a role in the origin of the muscles of the head must be abandoned, even by the most ardent supporter of the segmentation theory. The limbs, phylogenetically and ontogenetically, are of later origin than the branchial arches, and thus there is even less reason for assuming that the limb muscles are derived from the myotomes than that the branchial muscles have a similar origin. As the limbs are of later origin than the trunk musculature, it is but natural to expect the limb musculature to arise independently.
It has been impossible with our present methods for even the strongest adherents of the myotomic origin of the limb muscles to follow the complete history, inasmuch as the cells, if any such are given off from the myotomes, mix with the mesenchyme cells of the limb bud and soon become indistinguishable from them. The description of the differentiation of the limb musculature must begin then, whichever view we may take as to its source, with the first appearance of the premuscle masses in embryos of about 9 mm. in length. Considerable time has thus elapsed between the period when migrating cells have been observed entering the limb buds and the first stages of muscle differentiation.
In both arm and leg the proximal muscles are the first ones to be recognized. There is a progressive proximodistal differentiation, the hand and foot muscles being the last to differentiate. The muscles of the arm differentiate slightly earlier than those of the leg.
The Muscles of the Shoulder-Girdle and the Arm
The Shoulder-girdle
The muscles attached to the shouldergirdle and connecting it with the tnmk may be divided into several groups: (a) the trapezius and stemocleidomastoideus which descend from the occipital region; (b) the rhomboideus major and minor, the levator scapulae, and the serratus anterior, which arise in the lower cervical region; (c) the subclavius and omohyoideus; (d) muscles, arising from the arm bud, that spread out from the arm and shoulder-girdle to the trunk, the latissimus dorsi, and pectoralis major and minor.
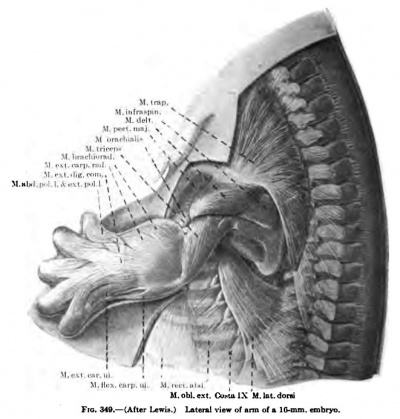
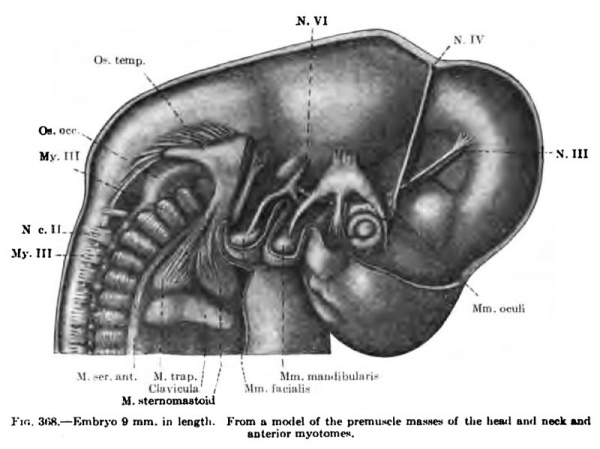
(a) The common rudiment of the Mm. trapezius and stemocleidomastoideus first appears in embryos of about 7 mm. in length, and lies ventral to the two caudal occipital and two anterior cervical myotomes (Fig. 367). It stands, however, more in relation to the branchial arch series than to the myotomic muscles. There are no direct observations on the origin of the cells composing this rudiment, but from its position and nerve supply it may be best considered as the caudal member of the branchial arch series. At this early stage it consists of closely packed cells, indistinguishable from the surrounding mesenchyme cells except for the greater condensation of the mass and the presence of the nervus accessorius which runs for some distance within the mass. The anterior end of the muscle mass lies close to the place where the nervus accessorius leaves the vagus. From this position there is a gradual extension caudalwards towards the arm bud of the premuscle tissue, which in a 9-mm. embryo reaches to about the level of the fourth cervical myotome (Fig. 368). At this stage its caudal end already shows signs of splitting into two divisions for the Mm. sternocleidomastoideus and trapezius. In an 11 mm embryo the two divisions are quite widely separated caudally, the trapezial portion extending to about the sixth cervical nerve and the sternomastoid portion to near the rudimentary clavicle which at this stage lies far anterior to the first rib (Figs. 337, 347, 367). The trapezius has not attached itself to the shoulder-girdle. The trapezius is a thick columnar mass extending from the occipital region caudalwards parallel and close to the vagus nerve. It has extended only slightly towards the spinous processes and is connected with them by a layer of fascia. The thick deep cervical fascia separates it from the more dorsal lying myotomic muscle masses. In a 16-mm. embryo the entire arm and shoulder-girdle have migrated caudally. The Mm. trapezius and sternomastoideus are now separate and distinct throughout their entire lengths. The trapezius has gained its attachment to the spine of the scapula and adjoining portion of the clavicle (Fig. 349). It has also spread as far caudally as the sixth rib and dorsally towards the spinous processes and ligamentum nuchap. The anterior end of the muscle does not extend as yet to the occipital cartilage. Not until after the embryo is over 20 mm. in length does the trapezius acquire its adult extent (Fig. 338). The splitting of the trapezius into the divisions sometimes found in the adult is a secondary process and not to be observed in the early embryo.
The M. stemocleidomastoideus also shows marked advance in a 14-mm, embryo, extending as it does from the mastoid process and occipital region to the clavicle. It has already begun to split into two divisions corresponding to the stemomastoid and cleidomastoid. The accessory motor-nerve supply from the cervical region to the trapezius and stemocleidomastoideus is secondary and does not indicate a myotomic origin for any portion of the muscles.
(b) The exact origin of the tissue which gives rise to the Mm. levator scapulas, serratus anterior, and rhomboidei is uncertain. It has not been possible to trace them back to the myotomes. In a 9 mm embryo the levator scapulae and serratus anterior are beginning to differentiate from the mesenchyme in the region of the ventral ends of the lower cervical myotomes (Fig. 368). The premuscle mass forms at this stage a continuous column, with no distinct attachments to the vertebrae or ribs over which it extends for a short distance. In an 11 mm embryo the muscle mass for the levator scapulae and serratus anterior is well defined, but still forms a column, oval in section, from the cervical region to the thorax (Fig. 337). The thoracic portion, the future serratus, becomes more and more slender towards the caudal end and has distinct digitations to the upper nine ribs, that to the ninth rib containing but a few fibres. Neither muscle has attached itself to the scapula. The attachments to the cervical vertebrae are present, however. The serratus now begins to change into a broad, flat muscle and the lower digitations enlarge, and in a 14 mm embryo it begins to resemble the adult form (Fig. 349). At this stage the scapular attachment is present and evidently plays a part in the change of form and extension of the muscle (Figs. 338, 346).
The development of the Mm. rhomboideus major and minor has not been followed. One can distinguish in a 14-mm. embryo the common muscle mass between the dorsal border of the scapula and the spinous processes. Owing to the relatively anterior position of the shoulder-girdle at this stage, the rhomboid mass only covers the uppermost portion of the M. serratus posterior superior. In later stages, as the arm and shoulder-girdle migrate caudally over the thorax, the rhomboidei come to overlap more and more the serratus posterior until finally the adult condition is attained, in which the caudal border of the M. rhomboideus major lies caudal to that of the serratus post. sup.
(c) The M. subclavius apparently develops in situ from the mesenchyme of the cervical region before the shoulder-girdle migrates caudally. It is sharply defined in a 14-mm. embryo. At this stage it has an anteroposterior direction, as the clavicle is still some distance anterior to the first rib. As the rib grows towards the midventral line the costal attacbmeDt is carried medially, and as this process is taking place the clavicle comes to he closer to the first rib, and thus the subclavius muscle assumes more and more a horizontal position.
The M. omobyoideus splits off from the infrahyoid muscle mass and has already secured its attachment to the scapula in a 14-mm. embryo.
(d) The Mm. latissimus dorsi and teres major are closely associated in their origin from the premuscle sheath of the arm, the teres developing in situ while the latissimus gradually spreads caudally over the side of the thorax. In an 11-mm. embryo it extends to the fourth rib (Fib. 337), at 14 mm. to the eighth or ninth, and at 16 mm. the attachment to the last three ribs is attained, while dorsally the muscle is continuous with the lumbodorsal fascia. Not until the embryo is over 20 mm. in length do the muscle fibres extend to the crest of the ilium (Fig. 338), The teres major is well developed in an embryo 14 mm. long.
The pectoral premusele mass from which both the Mm. pectoralis major and minor arise is clearly indicated in a 9-mm. embryo. It lies in the lower cervical region on the medial side of the arm bud. This premuscle mass is widely continuous with the arm premascle sheath and lies almost entirely anterior to the first rib. In an 11-mm. embryo it reaches about the level of the third rib, but the two muscles still form a single columnar mass attached to the humerus, to the coraccid process, and to the clavicular rudiment (Fig. 347). As the mass differentiates it flattens out and extends eaudoventrally to the region of the distal ends of the upper ribs. In a 14 mm embryo the caudal end of the muscle has extended near to the tip of the fifth rib and the "muscle has begun to assume more the adult form, with fibres arising from the upper five ribs and sternal anlage as well as from the clavicle. At this stage the proximal portion of the muscle has split into the major and minor portions, the one attached by tendon to the humerus and the other to the coraccid process. Both muscles fuse together near the costal attachments. In a 16-mm. embryo the two muscles are quite distinct, the pectoralis major now extending to the sixth rib and showing a distinct cleavage between the costal and clavicular portions (Figs. 339 and 349). The pectoralis minor has now its distinct attachment to the second, third, and fourth ribs.
The pectoralis major early splits into a series of overlapping bundles, and during the migration of the muscle the superficial fibres of each bundle move farther caudally than the deeper ones, giving the overlapping condition found in the adult. The tendon of insertion at first consists of a single sheet, but later from its distal end the second deeper sheet gradually spreads proximally and in an embryo of 40 mm exceeds the superficial or ventral one in breadth. The M. pectoralis major is carried towards the midventral line with the growth of the ribs and sternal rudiments.
The Muscles of the Arm
The remaining or intrinsic muscles of the arm develop in situ and, as they differentiate from the arm blastema, have approximately the same position that they later occupy in the adult. In an embryo of 9 mm. in length the skeletal core has already begun to differentiate as a thick rod in the middle of the arm bud ending distally in the hand plate (Fig. 336). On all sides, however, and at the distal end this skeletal core gradually merges into the surrounding blastemal sheath in which the muscles later appear, although the positions of only the pectoral and latissimus premuscle masses are recognizable at this stage. As the muscles differentiate their tendons likewise form in situ, and the muscles are thus from the first in connection with the skeletal structures by a condensed mesenchymal blastema out of which the tendons later develop.
The Muscles of the Shoulder and Arm
The Mm. deltoideus, teres minor, supraand infraspinatus arise from a common premuscle mass continuous with the pectoral mass and the common arm sheath (Fig. 336). In an 11-mm. embryo the M. deltoideus has partially split-off from the mass towards its origin from the acromion and clavicula (Fig. 337). In embryos of 14 to 16 mm. in length it has much the adult form (Fig. 349), with usually a distinct slip arising from the fascia over the M. infraspinatus. In a 20-mm. embryo it has practically the adult form and attachments (Figs. 338, 339, 346, 348). The development of the acromion from the cephalic border of the scapula separates in part in an 11-mm. embryo the M. supraspinatus from the M. infraspinatus. The M. supraspinatus lies at first on the medial surface of the scapula (Fig. 347). Later it comes to lie along the cephalic border, as in 16-mm. and 20-mm. embryos (Fig. 350, 351, 346), and only in later stages, with the growth of the cephalic border, does the muscle acquire its position on the lateral surface of the scapula. The Mm. infraspinatus and teres minor are from the first very closely associated and cover in an 11-mm. embryo only a portion of the lateral surface of the scapula (Fig. 337). In a 14-mm. embryo the muscle is quite distinct from the M. deltoideus, but does not cover the whole of the fossa infraspinata even in a 16-mm. or 20-mm. embryo (Figs. 349, 338, 346).
The M. suhscapularis is more or less isolated from the other muscles from its first appearance, and occupies in an 11-mra. embryo only a small portion of the median surface of the scapula, and not until the embryo is more than 20 mm. in length does it occupy the entire medial surface of the scapula (Figs. 350, 351).
The M. triceps brarhii arises along the posterior and lateral surfaces of the humerus extending from the scapula to the ulna, and even in an 11-mm. embryo indications of the three heads are present, while in a 16-ram. embryo thev are verv distinct (Figs. 337, 338, 346, 349, 350, 351).
Fig. 349. (After Lewis.) Lateral view of arm of a 10 mm embryo.
The Mm. biceps brachii, coraccbrachialis, and brachialis are intimately fused together in very early stages and probably arise from a common premuscle mass. The places of origin of the two heads of the biceps at this early stage are close together, and only by the later growth of the scapula do they become separated. The three muscles are to be recognized in embryos of from 14 to 16 mm. in length, and the long tendon of the caput longum is to be recognized in an embryo of 14 mm. The distal end of the common muscle mass differentiates later than the proximal ( Figs, 338, 339, 346, 347, 348, 349. 350, 351).
The Extensor Muscles of the Forearm
The extensors differentiate somewhat earlier than the flexors (Figs. 337, 338, 339, 346, 349). The common extensor premuscle mass on the laterocephalic side of the forearm first splits, in an embryo of about 11 mm. in length, into three groups of muscles, of which the largest and most superficial extends from tbe lateral condyle to the four ulnar digits. It is a thin layer spreading over the ulnar two-thirds of the foreann and distally joining the undifferentiated blastema of the digits. On its radial side proximally it is intimately fused with the second or radial group. From the superficial extensor mass later differentiate the Mm. extensor digitorum communis, extensor carpi ulnaris, and the extensor digiti quinti proprius. (according to Grafenberg (1905), the ext. dig. V. prop, arises in common with the deep extensor mass.) The separation of the muscles of the superficial extensor mass begins in the carpal region and extends proximally (Grafenberg) in the later stages as the tendons differentiate from the blastema of the digits. The radial group extends from the epicondylus lateralis and adjoining portion of the humerus distally along the radial and adjoining extensor or dorsal surface of the forearm. It appears to arise in situ along the radial surface, and not to have wandered there from the extensor surface as claimed by Grafenberg (1905). The mass early divides into two parts at the distal end, one, the brachioradialis, fusing with the distal end of the radius, and the other, the extensor carpi radialis longus and brevis, passing beneath the deep extensor mass to fuse with the blastema at the proximal ends of the second and third digits. The deep extensor mass lies beneath the radial portion of the superficial, becoming itself superficial over the distal radial surface of the radius and carpus and fusing with the blastema of the first and second digits. At a later stage this mass divides into two groups, a radial one for the Mm. abductor pollicis longus and extensor pollicis brevis, and probably the supinator, and an ulnar division for the extensor pollicis longus and extensor indicis proprius.
Not until the embryo is about 20 mm in length does the complete isolation process of the various extensor muscles reach an end. The extensor digiti V. prop, is not split oflf until later.
The Flexor Muscles of the Forearm
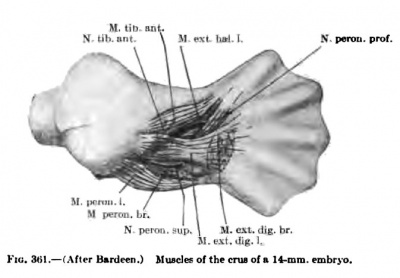
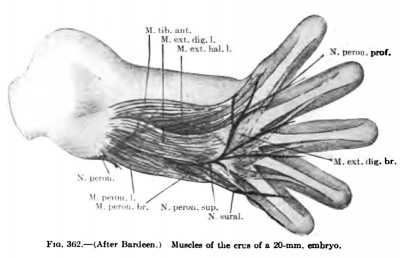
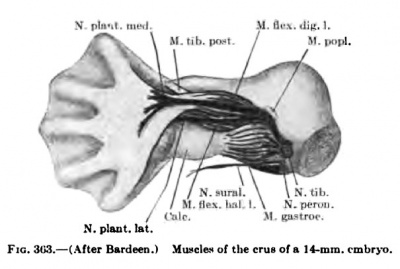
The development of the flexors is more diflBcult to follow than the extensors, and, owing to the concave volar surface of the forearm and carpus, the flexor muscle masses extend much farther distally than do the extensors (Figs. 347, 348, 350, 351). In an 11-mm. embryo, however, one can distinguish a small superficial layer and a voluminous deep layer. The superficial layer lies more on the radial side of the volar surface and already shows indications of a radial mass extending from the epicondylus internus to the blastema at the distal end of the radius; later this mass splits into the Mm. flexor carpi radialis and the pronator teres. The latter extends farther distally on the radius in early stages, but, as the distal part of the radius grows faster than the proximal, the distal attachment of the pronator comes to lie farther and farther from tlie distal end of the radius. The remaining portion of the superficial layer develops into the M. palmaris longus. It is intimately fused with the proximal part of the radial mass, but distally it extends on to the volar surface of the carpus, but with the elongation of the skeleton of the forearm the muscular belly comes to lie more and more over the proximal portion of the forearm.
The deep flexor muscle mass is much more extensive and thicker than the superficial, extending from the epicondylus internus over the entire volar surface of the forearm and carpus into the blastema of the digits. Even in an 11-mm. embryo the ulnar side already shows the beginning of the splitting oflf of the M. flexor carpi ulnaris which reaches to the blastema of the os pisiforme (Fig. 347). As the M. flexor carpi ulnaris increases in size it spreads over the ulnar side of the deep flexor mass and in an embryo 16 mm. in length is quite a distinct muscle.
The remainder of the deep flexor mass, even in an 11-mm. embryo, shows indication of cleavage into a superficial flexor digitorum sublimis and a deeper flexor digitorum profundus. The mass extends from the epicondylus intemus and volar surface of the forearm distally over the carpus without indications at this stage of a longitudinal splitting over the carpal region. The thick portion of the muscle in the carpal region has been designated as a separate M. flexor digitorum brevis by Grafenberg, but it appears to represent merely the distal part of the mass which later recedes to the volar surface of the forearm as the latter increases in length. The tendons of the two layers of the deep flexor mass gradually differentiate from the blastema of the digits and develop in situ as the digits increase in length. In an embryo 20 mm. in length the characteristic adult arrangement of the tendons is attained, although the recession of the muscle bellies on to the forearm is not complete nor the longitudinal splitting which progresses in a distal proximal direction. At this stage the M. flexor pollicis longus is easily recognized, although it is not completely split off from the M. flexor digitorum profundus. The Mm. lumbricales appear to differentiate in situ from the distal portion of the deep flexor mass, and I have first been able to recognize them in an embryo 16 mm. in length, but they probably appear slightly before this stage.
The origin of the M. pronator quadratus is uncertain, and I am not able to determine whether it splits off from the deep flexor mass or arises independently. It is easily recognized in an embryo of 16 mm. according to Grafenberg, it has a greater proximal extent in early stages than in the adult.
The Intrinsic Muscles of the Hand. — The Mm. interossei appear to arise from a common volar muscle mass. In a 16-mm. embryo the first signs of differentiation appear ; later the common muscle mass splits into the various muscles which migrate from the volar surface towards the dorsal surface of the hand between the metacarpals.
The Mm. abductor dig. V. and flexor digiti V. arise from a common muscle blastema, lying more on the ulnar side, and later wander volarwards (Grafenberg). The M. opponens dig. V. differentiates from the common interossei muscle mass (Grafenberg).
The thumb muscles develop from a common muscle mass, except the M. adductor pollicis, which is always separated from the others by the tendon of the M. flexor pollicis longus.
The Muscles of the Leg
The muscles of the leg, like those of the arm, develop from the premuscle sheath of condensed mesenchyme which surrounds the axial skeletal rudiment. In embryos of about 11 mm. in length this common muscle blastema begins in the proximal part of the leg to differentiate into the muscles. according to Graf enberg (1904), the nerves enter a common muscle mass for each group and later this mass splits into the various muscles of the group. Graf enberg 's description thus corresponds with that given for the arm. Bardeon (1907), however, finds that the individual muscles differentiate separately from the general muscle blastema, first as small centres about their nerves, and from these centres the muscle gradually extends towards origin and insertion. Whether this extension is brought about by differentiation of the mesenchyme into muscle or by active proliferation of cells of the original muscle nucleus is not clear from Bardeen's account. If we assume, however, that in these early stages the mesenchyme continues to be transformed into muscle about the original centre, Bardeen's observations come more into accord with the observations of Graf enberg and Lewis (1902). Bardeen's (1907) accoimt is here adopted as a basis for the following section on the development of the muscles of the leg.
The Femoral Group
The femoral group, comprising the Mm. iliopsoas, iliacus, pectineus, quadriceps femoris, and sartorius, arises from the muscle blastema on the ventral side of the thigh and into it pass the N. femoralis and its branches.
The M. iliopsoas arises from that portion of the femoral blastema which embraces the N. femoralis as it passes into the limb bud. In subsequent development the M. iliacus spreads out over the ilium and the M. psoas major extends upwards along the roots of the femoral nerve to form its attachment to the vertebral column (Fig. 355), and in close union the two muscles extend distallv to be attached to the trochanter minor. The M. psoas minor seems to split off from the M. psoas, but this is uncertain.
The origin of the rudiment of the M. pectineus is uncertain, but it probably arises either from the iliopsoas mass or the M. adductor longus. In a 14-mm. embryo it is quite distinct, extending from the pubis to the femur (Fig. 354). A portion of the muscle probably arises in connection with the M. obturator extemus.
The rudiment of the M. quadriceps femoris first appears in an embryo of about 11 mm. in length as a single mass lying over the anterolateral aspect of the middle of the shaft of the femur (Figs. 337, 347, 352, 353). In a slightly older embryo the mass begins to show definite differentiation into the four portions, the Mm, rectus femoris, vastus lateralis, vastus intermedins, and vastus medialis (Fig. 354). In a 20-mm. embryo the various muscles of this group are all clearly demarcated and attached to the skeletal apparatus by distinct tendons (Figs. 338, 346, 355).
The M. sartorius appears to arise from a distinct rudiment proximal to that of the quadriceps mass (Fig. 353), and differentiation gradually extends towards the ilium and tibia. The M. sartorius is at first relatively larger than the quadriceps group, but by the fourth month the quadriceps have overtaken it and at birth it is only a little larger relatively than in the adult (Figs. 354, 355, 358, 357, 339, 348).
Fig. 353. — (After B«nloeii.)
Fig. 354. — (After Bsrdeen.) Lateral view of the miudeg of the-thigh of a 11 nim. embryo.
The Obturator Group
The obturator group probably arises from a common muscle mass Ij-ing along the anteromedial portion of tlie femur (Figs. 347, 352), Cleavage is first apparent, in an 11-mra, embrj^o, in the proximal section of the region, into masses, one for the obturator portion of the M, adductor magnus and possibly also the M. obturator esternus, the other for the Mm. adductor longus and brevis and tlie gracilis. In an embryo of 14 mm, the individual muscles may be clearly recognized, although the tendons are not as yet well differentiated (Figs. 356, 359). But by the time an embryo is 20 mm. in length the tendons of origin and insertion are well differentiated to the skeletal attachments {Figs. 348, 357, 358, 359, 360) and the obturator and sciatic portions of the M. adductor magnus have fused.
The Gluteal Muscles. — according to Grafenberg, all the hip musculature, including the Mm. glutieus maximus, medius, and minimus, tensor fascia; latie,piriformis,obturator intemus, gemellus superior and inferior, and quadratus femoris, arise from a coneshaped mass found at the distal end of the pelvis during the fifth week. Bardeen deri\es these muscles from four mdiments which first appear about the ends of their nerves.
The superior gluteal group consists of the Mm. glutteua medius and minimus, piriformis, and tensor fasciae lata;, which are intimately united in embryos 11 mm. in length (Figs. 337, 353). In a 14-mm. embryo the M. tensor fascia latffi has split off from the lateral edge of the two gluteals. according to Grafenberg, the M. tensor fascis latae is at first inserted into the rudiment of the trochanter major, but after splitting off from the gluteal it migrates laterally and loses its attachments to the trochanter (Figs. 338, 346). At this stage the M. piriformis is still closely fused with the gluteals, which lie over the acetabulum, extending from the femoral margin of the ilium to the rudiment of the trochanter major. The Mm. gluta^us medius and minimus gradually extend over the surface of the ala oss, ilium. Grafenberg finds that the M. piriformis is from the first attached to the sacrum, but, according to Bardeen, it is at first separate and only later extends to its sacral attachment.
Fig. 355. — (Aft«r Bardeen.) Latenl view of the muscles ot the thigh of a 20-mm. embryo.
The rudiment of the M. glutteus maximus is separate from that of the other gluteal muscles (Bardeeu) and slightly fused with the rudiment of the short head of the biceps. At first it only slightly overlaps the M. gluteus medius in the trochanteric region, but it gradually extends over this muscle to the ilium, sacrum, and coccyx, and becomes separated into two portions, one being inserted into the femur and the other into the fascia latte. In the embryo each portion has a separate nerve.
The M. quadratus femoris (Figs. 347, 352, 359, 360) seems to arise early from a distinct rudiment tying between the anlage of the tuber ischiadicum and the trochanter major, but close to the rndiment for the Mm. obturator internus and gemelli on the ischial Hide of the hip-joint (Bardeen). The Mm. obturator internus and gemelli are at first closely united, but, as the obturator extends over the pelvic surface of the foramen obturatum from its original attachment to the ischium, the two gemelli retain the original attaciiment and are thus split off from the obturator. The nerve of the M. obturator int. is carried into the pelvis with the extension of the muscle.
The Posterior Thigh Muscles
The hamstring muscles, including the Mm. semiteudinosus, semimembranosus, biceps femoris, and the sciatic portion of the M. adductor magnus, differentiate from the muscle blastema on the dorsal (plantar) side of the thigh (Figs. 347, 352). They are all distinctly differentiated in a 14-mm. embryo (Fig. 356). The M. semimembranosus arises from a special rudiment in close association with the sciatic portion of the M. adductor magnus (Bardeen). In a 20-mm. embryo the tendons of origin and insertion are already developed (Figs. 348, 357). The M. semiteudinosus, according to Bardeen, arises from two separate rudiments corresponding to its two nerves. G-riifenberg, however, finds but a single rudiment. The muscle is well differentiated in a 20-mm. embryo, but its tendon of insertion is attached relatively more distally on the tibia than in the adult.
The caput longum, M. biceps femoris, arises from a special rudiment near the tuber isehiadicum closely fused with that of the M. semi tend! Qosus. The caput breve arises as a separate rudiment continuous proximally with the M. glutffius maximus and extends distally along tlie fibular margin of the femur. In a 14-mm. embiyo it begins to fuse with the caput longum {Figs. 356, 360).
The Extensors of the Foot
During the sixth week the rudiment of the peroneal muscles becomes separated from the long extensors of the toes and the M. tibialis anterior, and at the same time the rudiment of each peroneal muscle begins to become distinct (Figs. 361, ^62). They differentiate in situ, gradually extending from the dorsolateral surface of the proximal end of the fibula. The tendon of insertion of the M. peronseus longus gradually differentiates: in a 14-mm. embryo it can be traced to the base of the fifth metatarsal, in a 20-mm. embryo part way across the sole of the foot, and in a 30-mm. embryo to the first metatarsal, but not until later does the tendon become free in its sheath. The tendon is at first lateral to the rudiment of the malleolus lateralis ; later (20 mm.) it passes behind it. The muscle also extends proximally to the tibia attachment. The M. peronaBus brevis arises relatively more proximal than in the adult position and its tendon splits off from that of the peronaeus longus. Schomburg describes an intimate union between it and the M. extensor digitorum brevis persisting until the third month, but Bardeen was unable to find it.
Fig. 359. (After Bmrdecn.) Deep mua
Fig. 360. (After Banlwo.) D«p musdo of the thigli vl a SO mm. embryo.
In the early stages (Figs. 337, 353) the common extensor mass of the foot is connected with the peroneal mass, but in a 14-mm. embryo the two masses have become distinct and the extensor mass has already differentiated into the Mm. tibialis anterior, extensor digitorum longus, and extensor hallucis longus (Figs. 338, 339, 346, 361, 362). The Mm. tibialis anterior at this stage can be traced into a broad tendon as far as the region of the OS cuneiforme primum, and in a 20-mm. embryo it has the adult attachments (Figs. 338, 339, 346, 361, 362). The M. extensor digitorum longus is differentiated from the central portion of the muscle mass and is relatively more on the fibular side than in the adult. At first it ends distally in a broad flat plate which later, in a 20-mm. embryo, gives off the broad tendons to the digits. according to Schomberg (1900), the M. peronaeus tertius is early distinct from the M. extensor digitorum longus, but Bardeen maintains that it later splits off from the M. extensor digitorum longus with which it is often fused in later life. The M* extensor hallucis longus early splits off from the deep portion of the extension mass, although its tendon remains longer fused with that of the M. extensor digitorum longus. The M. ext. dig. brevis differentiates in Mtu beneath the extensor plate somewhat later than the other extensor muscles of the fo6t (Figs. 346, 361).
The Flexors of the Foot
In an 11 mm embryo the common flexor mass begins* to show signs of differentiation into the muscle rudiments (Figs. 347, 352). In a 14 mm embryo the two muscle groups are fairly distinct, a superficial, proximolateral group for the Mm. gastrocnemius, soleus, and plantaris, and a deep, more medial group for the Mm. flex. hal. long., flex. dig. long., popliteus, and tibialis posterior (Figs. 363, 364). The gastrocnemius group is connected with the blastema of the calcaneus and the two long flexor muscles with the flat aponeurotic ** foot-plate, " from which tendons extend to the blastema of the digits. The gastrocnemiussoleus group gradually spreads from its original lateral position towards the medial side of the leg to attain the tibial attachment, and the two heads of the M. gastrocnemius develop during the second half of the second month, the medial head attaining its attachment later than the lateral (Figs. 348, 365, 366). The M. plantaris seems to split off at a comparatively late stage from the lateral head of the M. gastrocnemius.
The M. popliteus probably develops in situ from the proximal end of the deep flexor mass. It is well developed in a 20-mm. embryo (Figs. 363, 366). The rudiment of the M. flexor hallucis longus is quite distinct in a 14-mm. embryo {Fig. 364). It differentiates from the lateral side of the deep flexor mass, ending distally at this stage in the common tendon plate from which its tendon differentiates at a considerably later stage. The M. flexor digitorum longus is differentiated from the medial portion of the deep flexor mass in a 14-mm. embrj'o, and ends distally in the common tendon plate from which the tendons are beginning to radiate to the digits (Figs. 348, 36.3). The tibial attachment does not begin to take place until the embryo is about 20 mm. in length. The M. tibialis posterior differentiates from the deeper tibial part of the deep flexor mass over the lower half of the tibia and extends later Id a proximolateral direction. Its tendon is early differentiated.
ii.— (After Bnrdee
The Plantar Muscles
The rudiment of the M. quadratus plantie has dififerentiated in a 14-mm, embryo. Sehomburg (1900) finds it fused with the M. flexor hallucis longus, but Bardeen did not find this attachment. The M. abductor digiti quinti differentiates at the same stage immediately distal to the tuber calcanei and later moves to a more lateral position (Figs. 364, 366). The Mm. flexor brevis and opponens digiti quinti arise from a common rudiment somewhat later than the above muscles, and even in a 20-mm. embryo thev are not clearly differentiated (Fig. 366).
Fig. 363. (After Bardeen) I Muscle of the eruB ot a l*-iDra. embryo.
Fig. 364.
Fig. 365. ((After Bardeen) Huwie" oE tlie cruB of a H-miii. embryo.
The Mm. interossei develop on the plantar surface and the dorsal interossei wander between the metatarsals to the dorsal surfare: Ruge (1878), Sehomburg (1900). They first appear in embryos over 20 mm. in length (Fig. 366).
The M. adductor hallucis arises at the base of the second metatarsal and wanders to its adult position (Ruge, 1878), and, according to Ruge, the transverse and oblique heads arise from a common rudiment, but, according to Sehomburg (1900), from separate mdiments.
The Mm. lumbricates do not ditferentiate until late, in embryos over 20 mm. in length, and, according to Sehomburg, develop as separate rudiments near the distal extremities of the metatarsals and wander towards their attachments to the tendons of the M. flexor digitomm longus. They arise from the medial plantar layer.
The M, flexor digitomm brevis develops relatively late. In a 20-mm. embryo the rudiment is just beginning to differentiate on the surface of the tendon plate of the long flexor muscles over the middle cuneiform cartilage (Fig. 365). Its tendons develop later, as does its proximal extension to the tuber caleanei.
The M. abductor hallucis also develops relatively late. In a 20-mm. embryo it can just be distinguished on the medial edge of the plantar surface of the foot in close association with the M. flexor hallucis brevis (Figs. 365, 366). With the torsion of the foot the abductor extends proximally to be attached to the tuber calcanei.
The M. flexor hallucis brevis, like the other muscles in this group, only begins to appear in a 20-mm. embryo (Fig. 366); later it splits into two parts, the lateral head coming into relation with the M. adductor and the medial with the M. abductor hallucis.
The Muscles of the Head
Muscles of the Orbit
It has commonly been assumed that the muscles of the orbit are derived from anterior head somites ; the first somite giving rise to the Mm. levator palpebrsB superioris, rectus superior, rectus internus, rectus inferior, and obliquus inferior, supplied by the N. oculomotorius ; the second, to M. obliquus superior, supplied by the N. trochlearis; and the third, to the M. rectus lateralis, supplied by the N. abducens. Anterior head somites have, however, never been observed in man or mammals. Zimmermann (1898) observed in a 3.5-mm. embryo several small head cavities in the region where later the eye muscles develop. It is uncertain even that they represent rudimentary head somites, and no connection has been established between them and the eye muscles. On the contrary, the observations of Renter (1897) on the pig and my own observations on human embryos, which follow here, show that all the eye muscles arise from a common premuscle mass, which occupies, both in the pig and in man, the same general position in the early embryo.
This common premuscle mass is first to be recognized in human embryos of about 7 mm. in length (Fig. 367). It consists of a lens-shaped mass of condensed mesenchyme, outlined from the surrounding mesenchyme by a capillary network over its surface. This premuscle mass lies dorsal to the optic stalk, between it and the ganglion Gasseri, and medial to the optic cup. The N. ophthalmicus passes in front of and lateral to the mass, and the N. maxillaris behind and lateral to it. At this stage only the N. oculomotorius enters the anterior end of the muscle mass. The Nn. trochlearis and abduceus do not appear until later.
In a 9 mm embryo the eye premuscle mass occupies much the same position as in the earlier stage, lying on the dorsal side of the optic stalk and medial to the N. ophthalmicus and maxillaris (Figs. 368, 369). It has enlarged somewhat and extends along the caudal side of the optic stalk. It also begins to show cleavage into separate muscle masses, each supplied by its respective nerve.
The N. trochlearis now enters the anterior portion of the mass which later forms the rudiment of the M. ohliquns superior. The N. abducens enters the caudal end of the mass, which has begun to extend out along the path of the nerve and also shows indica tions of separation from the rest of the mass which lies closer to the optic stalk and into which the N. oeiilomotorius runs. The muscle mass at this stage has no very distinct attachment either to the precartilage or to the sclera, but there exists a direct continuity with the mesenchyme from which tliese structures differentiate.
In an 11 mm embryo the pre-muscle mass has still farther enlarged and partially split into the rudiments supplied by the three nerves. The anlagen lie on the dorsal and caudal sides of the optic nerve and for the most part medial to the eyeball (Figs. 370, 379). They are continuous laterally with the primitive sclera, which is now beginning to form as a definite condensation of the mesenchyme about the eyeball, and medially with the precartilaginous tissue about the optic nerve. The M. rectus lateralis has extended farther out along the path of the N. abducens and has as yet no skeletal attachment. The N. ophthalmicus passes above these muscle masses and nearly at right angles to them.
Fig. 367. Embryo 7 mm in Length Diagram with premuscle masses of the head anterior myotomes, from model.
Fig. 368. Embryo 9 mm in Length From a model of the premuscle masses of the head and neck and anterior myotomes.
As differentiation progresses tlie muscle mass of the N. ocuJomotorius gradually extends around the optic nerve and splits into the various muscles supplied by this nerve. In a 14-mm. embryo all the orbital muscles are to be distinguished and have nearly the adult relations to the bulbus oculi. The M. obliquus inferior, however, does not completely separate off from the M. rectus inferior until a later stage.
The Muscles of the Mandibular Arch
The mesoderm of the mandibular arch gives rise to the muscles of mastication, including the Mm. temporalis, masseter, pterygoideus externus and interus, and probably to the M. mylohyoideus. The Mm. tensor tympani and tensor veli palatini are also derived from this arch. In a 7-mm. embryo the mandibular arch is filled with a uniformly closely packed mesenchyme, with only the slightest trace of condensation about the peripheral end of the N. mandibularis (Fig. 367). In a 9-mm. embryo, however, this condensation is clearly to be recognized (Fig. 368). This preranscle mass, in which the N. mandibularis ends, lies at about the middle of the arch. It is a simple mass, without indications of splitting and not sharply outlined from the mesenchyme which fills the arch. In an 11-mra. embn-o this egg-shaped i)remuscle mass has increased in size, but still shows no indications of splitting into the various muscles (Fig. 370). The differentiation probably takes place in a manner very similar to that described by Eeuter in the pig. The premuscle mass is from the very beginning closely associated with the condensed mesenchyme for the mandible, and with the differentiation of the proximal end of the mandible the premuscle mass is partially split into a Y-shaped mass, the handle representing the M. temporalis, the outer limb corresponding to the M. masseter, and the inner deeper limb, separated from the outer by the proximal end of the mandible, the mass for the Mm. pterygoideus externus and internus. In a 14-mm. embryo this process of differentiation has progressed still farther, and the Mm. pterygoideus externus and internus are partially separated by Meckel's cartilage. The processus coronoideus only partially separates the masseter from the pterygoid mass at this stage. With the continued differentiation of the membranous mandible and adjoining portions of the skull to which the muscles of mastication are attached comes the gradual differentiation of these muscles. The muscle:: are at no time attached to Meckel's cartilage, but are always in relation with the membranous mandible. In a 20-mm. embryo the various muscles of mastication are easily to be recognized, but have only a slight resemblance to the adult form. The M. temporalis is very small in proportion to the size of the head and gradually extends over a much greater proportional area during later stages. The M. masseter is at first attached only to the medial and lower surfaces of the arcus zygomaticus.
Fig. 370. Embryo 11 mm in length. Diagram of muscles of the head and neck, from graphic reconstruction and model.
The M. mylohyoideus apparently differentiates more rapidly than the other muscles of mastication, and is to be recognized by its nerve supply in an 11 mm embryo, and in a 14 mm embryo it has much the adult relations. I have been unable to determine its origin from the common muscle mass.
In a 14-mm. embryo the Mm. tensor tympani and tensor veli palatini are to be recognized and are connected with the pterygoid mass from which they probably arise. The M. tensor tympani has already gained its insertion to the malleus. The later development of these muscles is bound up with the development of the base of the skull, the tuba auditiva, and the soft palate.
The Facial Muscles
(The Muscles of the Hyoid Arch.) The facial muscle group includes all the muscles supplied by the N. facialis. The subcutaneous muscles of expression and certain muscles about the facial openings concerned with the vegetative functions belong to this group.
Futamura (1906) has given the most elaborate recent account of their origin and development, and the following is based upon his work.
This entire facial musculature arises from the closely packed mesenchyme which fills the second branchial or hyoid arch. In a 9 mm embryo the main stem of the N. facialis, which is very simple at this stage, passes into the hyoid arch to end brush-like in a single mass of premusele blastema that occupies the ventrolateral portion of the arch (Figs. 367, 368, 371). The condition is very similar to that described by Futamura as existing in a 10 mm embryo. It is from this premusele mass that the entire musculature supplied by the N. facialis arises (Futamura). This premusele mass increases in size (Fig. 370), and in an embryo 13.7 mm in length has begun to spread out not only ventralwards but also dorsalwards and aborally (Fig. 372). It also extends towards the shoulder to form the platysma. The medial side of the mass becomes thickened to form the rudiment of the Mm. stylohyoideus, digastricus, and stajwdius. It extends from the root of the tongue along the posterolateral side of Beichert's cartilage towards the ear capsule.
As the muscle rudiment spreads out the N. facialis becomes divided into several branches which follow the wanderings of the muscle tissue. Thus, in a 13.8 mm embryo the nerve stem distal to the chorda t>'mpaiii divides into three branches, a thin one directed medially into the rudiment of the Mm. digastrieus and stylohyoideus, a second large branch, the N. auricularis posterior, and a third large branch which soon divides into the R. temporalis, maxillaris, and cervicofacialie.
Fig. 371. Human embryo 27-30 days. (After Futamura, Anat. Hefte, Bd. 30, Fig. 27, on p.440 X50 dia. Sagittal projection from frontal sections.) a. eye; Cv, guiglJDD triganiDi: (i, <ii, Uu. runiu primus, MGimdiu. and tertiiu N. trifemtni; ^, juncdoD of Xhx r. u N. triganioi with N. petroflus supcjf. maj.; f, N. linsuaJii; Cj., chorda tympani ; JV.p.t, nuv'.. N. petroaue (uperfidalU major; G.vii, g
In a 15.5 mm embryo the platysma rudiment has extended orally over the hyoid arch and caudally to the region of the sternum and shoulder-girdle. It has also pushed medially and begins to unite with its fellow of the opposite side (Fig. 373, 374). The spreading of the muscle headwards takes place along two paths, separated by the aniage of the outer ear. The occipital portion gives rise to the Mm. auricularis posterior, transversus nuchfe, transversus and obliquus auriculie, but at this stage still forming a continuous membrane.
Fig. 372. (After Futamura, Anat. Hefte, Bd. 30, Fig. 2, on p. 441.) Human Embryo of 31-34 days. Sagittal projection from aacittal series. XSSdis. EipluationssiD Gsurs 371. fi.p.i.miit., N.petrtma
The facial portion of the platysma splits at the upper part of the neck into two layers, a superficial, lightly staining one and a deeper, more intensely staining one. A capillary network often separates the two layers. Futamura designates the superficial layer as the platysma faciei and the deep layer as the sphincter colli.
The platysma faciei gradually extends over the lower jaw and cheek to the forehead, eye, and temporal region, while the most anterior part goes to the lower lip. At the angle of the mouth the two layers are very difficult to separate from one another. In a 7-wk. embryo (Figs. 375, 376) the platysma faciei over the side of the head and ahove the ear unites with the platysma occipitale, which has also in the meantime extended cranialwards and broadened out to touch its fellow on the opposite side. The piatj-sma occipitale has, however, now lost its connection with the platysma colli.
Fig. 373. (After Futamura, Anat. Hefte, Bd. 30. Fig.4, A,onp.4. , SacitUtI proieclion from frontal wria. X20 d>a. Deep layer, ojk.. ortncul«rii oculi: g nfen; Qj-t.p., theanbi^ of the M. quadrmtuB ^bij fluperiohe proprjUB; a, the partji or Che flphinet^ cQlti soiog to the lower letenJ ode of the eye; 0.ar., orbiculima owie; a.a., orbicularU auHcuIif: N js.p,, oervuB auHcularis poeterior; b' utd b". bnncha uF the faciei goini Is the posterior end anterior nirfaca of the -eAr; RA.f., runiu t^npomfaeiaiiB; Rxj,, ramus oervioofacialia; tj.. Bphiocter colli.
The sphincter colli, the deep layer, which imderlies the platysma faciei, forms at first by the sixth week a sheet over the face, with sphincter-like differentiations about the mouth, nose, eye, and ear, to form the primitive Mm. orbicularis oris, orbicularis narium, orbicularis oculi, and orbicularis auriculae (Fig. 373). The differentiation of the sphincter colli begins earlier than the superficial layer and is far advanced in embryos 8-9 weeks old(Fig. 378). The lateral portion of the sphincter colli over the cheek degenerates, as do also the primitive Mm. orbicularis oculi and narium. That part of the sphincter colli between the M. orbicularis oculi and angle of the mouth becomes attached to the maxilla to form the rudiment of the M. quadratus labii superioris. Lateral to this the M. zygomati<ius appears to develop from the sphincter colli. The M. orbicularis oris, which comes from this layer, continues to increase in size and, connected with the mouth but having attachment to the skeleton, are differentiated from the sphincter colli
Fig. 374. (After Futamura, Anat. Hefte, Bd. 30. Fig. 4. B. on p. 44fi.) SuperSdkl layer of the culsture of the nine embryo as in Fit. 373. pji., platysou occj|»(«le: «, onl foaea: p./., i: P.C.. pUtyBom colli.
the Mm. eaninns, incisivus labii superioris and inferioris, and nasalis. The latter replaces partly tlie M. sphincter narium. The M. triangularis is associated with the M. caninus. The M. risorius is derived later {thirteenth to seventeenth week) from the M. triangularis. The M. buCoinator is derived from that portion of the sphincter colli l>ing between the upper and lower lips, and as the mouth decreases in size the muscle comes to lie deeper.
The superficial layer, the platysma faciei, spreads out over the face and head in a continuous sheet, and in a Z-weeks embryo has united over the ear with the platysma occipitale to form the M. fronto-auriculo-occi pi talis (Fig. 375). As the sphincters of the deep layer degenerate, there are gradually formed out of the superficial layer a new orbicalaris oculi and orbicularis anricolse (Figs. 375, 377). The medial and lateral part of the M. quadratus labii superioris is derived from this superficial layer and also the Mm. mentalis and quadratus labii inferioris (Fig. 377). From the M. fronto-auriculo-occipitalis are derived the Mm. auricularis saperior and anterior, and, through degeneration of the medial portion of the M. fronto-auriculo-occipitalis, the Mm. frontalis and occipitalis become widely separated but joined by the galea aponeurotica which probably represents the degenerated portion of the muscle.
From the platysma occipitalis not only arises the M. occipitalis but also the Mm. auricularis posterior and transversus nuchie through degeneration of the intermediate parts.
Fig. 375. (After Futamura, Anat. Hefte, Bd. 30, Fig.6.A.onp.44B.) Superficial layer of th< embryo u ia Fig 376. Fji.o^ M. FronU>4uriculo-occipitALis; o.oej>„ U. orbicul&ria oeuLi (Inun : AJ.. R. Umporalia: I.U., H. levalor Isbii auperiorifl proprius (alKque mui); Ojir., orb ona; A, max., R. mAxillariB; R. mar., R. morgiDalifi; R. fot^, R. colli; R.c.f., R. MrrviiyifAciKJiv; b" the branchefl of the facial nerve coinK to the antvriar and pofftehor Burfaocfl of tJie ear; o aurioul)E; H.a,p^ R, suriculAris poctcrior.
It has already been noted, that on the medial side of the platysma arise the rudiments of the Mm. digastricus and stylohyoideus. In the sixth week this mass is already well developed, extending from the posterior side of the otic capsule in a concave bow towards the angle of the lower jaw. At this stage it is in intimate relation with Eeichert's cartilage. With the growth and lengthening of Beichert's cartilage comes the complete separation of the digastric rudiment from the platysma. In the early stages this anlage is supplied entirely by the N. facialis, the N. mylohyoideua entering only the M. mylohyoideus. At first the M. digastricus consists of only a single belly. From this rudiment splits off gradually the M. stylohyoideus. Parallel with this process occurs a constriction and degeneration in the middle of the digastricus, dividing it into two bellies. During the eighth to ninth week this process is quite complete. The innervation of the anterior belly of the digastric by the N. mylohyoideus is first to be recognized in a 7-weeks embryo when the tendon begins to form.
Fig. 376. — (After Futamura, Anat. Hefte, Bd. 30, Fig. 6. B, on p. tlSO.) Humaii «mbryo o( 7 weekn. 8B(itt«l pnjsotioa Iram fronlal Kctions. X 12 dia. Deeij layer, o-t., U. arUeularis oouli (From iphinctsr oolli); mJ.. U. mBxillolsbiBles; o.or., M. orbiculariB nris: buc.. M. buCoinslor: oin.. H. eaninuB; tv„ M. lygODUitiouBi «.c., aptuDctar cidli: Sjv., R. lygmutieua; R. buc., R. buceiiuitoriiu; N,a,p., N. niuicularis p<Mt«rior, RJ., R. lempontlis; Aj;./.. R. nrvioolsculis; A.iRai..R.auiillaris.
The M. stapedius appears to come from the proximal end of the rudiment of the M. digastricus. It gradually becomes separated and enclosed within the otie cartilages. In a 14-miii. embryo it is quite separate from the digastric and lies in close relation to the stem of the N. facialis.
The Mm. levator veli palatini and uvulie arise from the facial mass. Their development and complete separation from the rest of the facial muscles are bound up with the development of the palate and the tuba nuditiva. The M. uvulie is at first a paired muscle, but with the formation of the soft palate the two muscles unite in the midline.
Fig. 377. — (After Futamura, Anat. Hefte, Bd. 30. Fii. S. A, on p. 456.) Human fetus of 84 meki. Sagittal projection from nsittal seriea. X 14 dia. Superfieial layer of Ihe fadal miuculalure. t.a.o^ U. frontt>-auncuk>-occipJl«ILi>: ox,. M. orbleularin oruli: U.;, M. levator lalni njperioris stcque nasi; H. indsivuB Ubii inlerioris; r., M. triaD«utarii<; Pj.. platynna «tli: a.p,. M. aurieularis poeterior: oji^ orbiculans auriculs: N.a.p.. auricularis pwterior: Rj., ramus tanparaJis; R.c, ramus colli: S.mar,
The Pharyngeal Muscles
Very little is known concerning the development of the pharyngeal muscles, but the constrictors of the pharynx, as well as the Mm. stylopharyngeus and palatoglossus, probably arise from the third gill arch. The premuscle tissue of this arch is already recognizable in a 9-mm. embryo and into it goes the ninth nerve. The third and fourth gill arches at this stage have already sunken some distance below the surface, and thus the premuscle mass comes to lie in the depths in this region. In an ll-mm. embryo the Mm. stylopharyngeus and constrictors are closely united together at the side of the pharynx, the M. stylopharyngeus having its attachment to the precartilaginous horizontal styloid process near its medial end. With the growth and shifting of the various structures in this region this portion of the styloid gradually assumes a more lateral position, and in a 14-mm. embryo the muscle takes a medial direction toward the thyroid precartilage and the constrictors. One can also distinguish at this stage the attachment of the M. constrictor pharyngis medius to the hyoid and the M. constrictor pharyngis inferior to the thyroid cartilages. These two constrictors are still united into a common mass which is rapidly extending over the dorsum of the pharjoix. Concerning the M. constrictor pharyngis superior nothing is known and I am uncertain as to whether it arises in common with the others. Its relation with the bucoinator suggests that it may arise from the facial mass. The M. constrictor pharyngis inferior is at first continuous with the M. cricothyreoideus. In a 20-mm embryo the pharyngeal muscles are quite distinct and the constrictors have grown round to the mid-dorsal line of the pharynx.
Fig. 378. — (After Futamura, Anat. Hefte, Bd. 30, R*. 8, B, on p. «7.) Deep Uyw of the r&ci*! miuculstureof tb«Mme«nbryouiDFil.377, withhighermaanificAtion. mn.. M.caninus: n., M.DBBBlis; i.lj,, H. iordsivus labii superioria; bae„ M. buCoinator; (^ trianEulu-is (abceschnittea); iJ,i,, incisivua labii inr«riorie; ({i.t., M. qiudntui Isbli inferioiis; m., meoUiU.
The Intrinsic Muscles of the Larynx
The muscles of the larynx probably arise from the ventral ends of the third and fourth gill arches, which early fuse to form a mass of closely packed mesenchyme out of which later diflferentiate the cartilages and muscle*. Soulie and Bardier (1907) found it difficult to recognize laryngeal muscles in a 14-mm. embryo. But in a 19-mm. embryo they were able to recognize clearly the Mm. interaryta^noideus, crico-arytsenoideus posterior, cricothyreoideus, and thyreo-cricoarytaenoideus. They were unable to discover any traces of a sphincter laryngis as described by Strazza (1889), but found that in 32to 40-mm. embryos the M. thyreo-crico-arytsBuoideus is clearly distinguishable from the M. thyreo-arytaenoideus and towards the middle of the fifth month all the muscles are recognizable.
I have noticed that in a 14-mm. embryo one can distinguish the various muscles, the Mm. arytaenoideus, crico-arytsenoideus posterior and lateralis, thyreo-arytaenoideus, and the cricothyreoideus. I do not find, as Strazza, the muscles forming a common constrictor of the larynx. I find, with Strazza, that the constrictor of the pharynx is continuous with the M. cricothyreoideus. With the farther differentiation of the cartilages the muscles become more distinct and in a 20-mm. embryo they have much the adult form.
The Tongue Musculature
It is usually assumaed that the tongue musculature is derived from the occipital myotomes which appear to be serially related to the N. hypoglossus. There is, however, no direct evidence whatever for this statement, and we are inclined to believe from our studies that the tongue musculature is derived from the mesoderm of the floor of the mouth. In a 7-mm. embryo the mesenchyme in the floor of the mouth is similar to that in the mandibular and hyoid arches from which later the musculature of these two arches develops. In a 9-mm. embryo the floor of the mouth has increased in thickness and the groove between the mandibular and hyoid arches has disappeared. In the mesenchyme of this thickened floor are two bilateral masses similar in appearance to the jaw and facial masses found in the mandibular and hyoid arches at this same stage. These bilateral tonsrue premuscle masses extend from the region in which the mandible later develops to the hyoid region, and are here continuous with a medial mass of condensed mesenchyme which extends into the larynx region and also with the infrahyoid premuscle bands (Fig. 369). The caudal end of this latter mass is in turn continuous with the diaphragm premuscle mass. This lingual-infrahyoid-diaphragmatic band is probably a primitive ventral visceral muscle complex and in no way concerned with the myotomic system. The N. hj-poglossus enters the caudal end of the tongue premuscle mass. One is unable at this stage to distinguish any of the individual muscles.
In an 11-mm. embryo (Figs. 370, 379) each homogeneous premuscle mass has split into two masses, a medial ventral mass for the Mm. geniohyoideus and genioglossus and a dorsolateral mass for the Mm. hyoglossus, styloglossus, and chondroglossus. The medial ventral mass extends from the region of the future symphysis menti to the prehyoid mass and dorsally it expands into the tongue region. The main stem of the N. hypoglossus enters its caudal end and passes longitudinally nearly to the anterior end. The dorsolateral mass extends from the prehyoid and medial portion of the styloid process into the dorsolateral region of the tongue for a short distance. A branch of the N. hypoglossus enters its ventral surface. The styloid process at this stage has very nearly a horizontal position and extends nearly to the midline.
With the differentiation and development of these muscle masses the tongue gradually becomes raised more and more above the mandibular arch. In a 14-mm. embryo this process is advanced, keeping pace with the differentiation of the mandibular arch in which are now plainly to be recognized Meckel's cartilage and the partially enclosing membranous mandible. From the membranous mandible arise the radiating M. genioglossus,. extending a considerable distance fan-like into the tongue, and the M. geniohyoideus, extending to the hyoid precartilage. Over the dorsum and dorsolateral region of the tongue extend the M. hyoglossus and M. styloglossus, from the hyoid precartilage and styloid process respectively, to the tip of the tongue. They lie dorsal and lateral to the radiating genioglossus. Their origins, though distinct, are still close together and parallel. The N. hypoglossus gives off branches to the M. geniohyoideus, then to the Mm. hyoglossus and styloglossus, and passes through the M. genioglossus to its tip, giving off numerous lateral branches into this muscle. There is at this stage apparently very little interlacing of the tongue muscle, nor are we able to recognize either the intrinsic muscles — the Mm. longitudinalis superior and inferior, transversus linguae, and verticalis linguae — or the M. glossopalatinus.
In a 20 mm embryo, however, all the muscles are clearly differentiated and increased in size. The origin of the M. styloglossus has been carried to a more lateral position and enters the tongue at an angle to the M. hyoglossus. The latter has spread out more in its attachment to the hyoid cartilage. The M. glossopalatinus is now recognizable, extending from the laterally placed soft palate to the lateral surface of the tongue. The great development of the intrinsic muscles is most noticeable at this stage, but concerning their origin there are no observations. It is uncertain whether they are derived from the hyoid and mandibular tongue muscles or independently from the mesenchymal matrix. At this stage the interlacing of the tongue musculature is quite advanced.
Literature
Bardeen: The Development of the Musculature of the Body Wall in the Pig, Including its Histogenesis and its Relation to the Myotomes and to the Skebtal and Nervous Apparatus, Johns Hopkins Hosp. Rep., vol. ix, 1900.
Bardeen CR. Development and variation of the nerves and the musculature of the inferior extremity and of the neighboring regions of the trunk in man. (1907) Amer. J Anat. 6: 259-386.
Bardeen CR. and Lewis WH. The development of the limbs, body-wall and back. (1901) Amer. J Anat. 1: 1-36.
Collin: Recherches sur le developpement du muscle sphincter de Tiris chez les oiseaux, Bibliographic Anatomique, t. 12, p. 183, 1903.
Felix: Teilungserscheinungen an quergestreiften Muskelfasem menschlieher Embryonen, Anat. Anz., Bd. 2, 1888. Ueber Wachsthum der quergestreiften Muskulatur naeh Beobachtungen am Menschen, 2^itschrift f . wiss. Zool., Bd. 48, p. 224, 1888.
FiSCHEL, A.: Zur Entwieklung der ventralen Rumpf u. Extremitatenmuskulatur bei Vogeln und Saugetieren, Morph. Jabrbuch, Bd. 23, 1895.
Futamura: Ueber die Entwieklung der Faeialismuskulatur des Menschen, mit 27 Textabbildungen, Anat. Hefte, Bd. 30, 1906.
GU)DLEWSKi : Die Entwickelung des Skeletund Herzmuskelgewebes der Saugetiere, Archiv f. mikr. Anat., Bd. 60, 1902.
Grafekberg, E. : Die Entwieklung der mensehl. Beckenmuskulatur, Anat. Hefte, Bd. 23, 1904. Die Entwieklung der Knochen, Muskeln und Nerven der Hand und der fiir die Bewegungen der Hand bestimmten Muskeln des Unterarms, mit 19 Abbildungen im Text, Anat. Hefte, Bd. 30, 1905.
Harrison, R. G. : An Experimental Study of the Relation of the Nervous System to the Developing Musculature in the Embryo of the Frog, Am. Joum. of Anat., vol. iii, 1904.
Heerpordt, C. F.: Studien ueber den Muse. Dilatator Pupillae, etc., Anat. Hefte, Bd. 14, p. 487 to 558, 1900.
Heidenhain, M. : Ueber das Vorkommen von Intercellularbriicken zwischen glatten Muskelzellen und Epithelzellen, etc., Anat. Anz., Bd. 8, 1893. Herzog, H. : Ueber die Entwieklung der Binnenmuskulatur des Auges, Archiv f. mikr. Anat., Bd. 60, p. 517-586, 1902.
Ingalls NW. Description of a human embryo of 4.9 mm (Beschreibung eines menschlichen Embryos von 4.9 mm). (1907) Arch. f. mik. Anat., 70: 506-576.
Kotzenberg, W. : Zur Entwieklung der Ringmuskelschicht an den Bronchien der Saugetiere, Archiv f. mikr. Anat., Bd. 60, p. 460, 1902. Kazzander: Beitrag zur Lehre ueber die Entwieklung der Kaumuskeln, Anat. Anz., Bd. 6, p. 224, 1891.
Keibel, F. : Studien zur Entwicklungsgeschichte des Schweines II. Morphologische Arbeiten, Bd. 5, 1895. Koelliker: Handbuch der Gewebelehre des Menschen, S. 253, 1889.
Kollmann: Die Rumpfsegmente menschlieher Embryonen von 13-35 Ui-wirbeln, Archiv f. Anat. u. Phys., Anat. Abt., 1891.
Kroesing: Ueber die Riickbildung und Entwieklung der quergestreiften Muskelfasem, Virch. Arch., Bd. 128, 1892. Leonowa, O. von: Zur pathologischen Entwieklung des Centralnervensystems (ein Fall von Anencephalie kombiniert mit totaler Amyelie), Neurol. Centralblatt, Jahrg. 12, 1893.
Lewis WH. The development of the arm in man. (1902) Amer. J Anat. 1(2): 145-184. Observations on the Pectoralis Major Muscle in Man, Johns Hopkins Hosp. Bull., vol. xii, 1901. Wandering Pigmented Cells Arising from the Epithelium of the Optic Cup, with Observations on the Origin of the M. Sphincter Pupillse in the Chick, Am. Journ. of Anat., vol. ii, 1903.
Maccallum, J. B. : On the Histogenesis of the Striated Muscle-fibre and the Growth of the Human Sartorius Muscle. Johns Hopkins Hosp. Bull., 1898. Mall: Development of the Ventral Abdominal Walls in Man, Jour, of Morph., vol. xiv, 1898.
McGiLL, Caroline: The Histogenesis of Smooth Muscle in the Alimentary Canal and Respiratory Tract of the Pig, Intemat. Monatschrift. Anat. u. Phv?., Bd. 24, 1907.
Marchesint and Ferrari: Untersuchungen ueber die glatten und die gestreiften Muskelfasem, Anat. Anz., Bd. 11, 1895. Meek, A.: Preliminary note on the Posl-embryonal History of Striped Muscle fibres in Mammalia, Anat Anz. 14, 1898, and 15, 1899. On the Postembryonal History of Yoluntary Muscles in Mammals, Joum. of Anat. and Physiol., London, vol. xxxiii, p. 546-608, 1899.
MoRPURGO, B. : Ueber die postembryonale Entwicklung der quergestreif ten Muskeln von weissen Ratten, Anat. Anz., 15, p. 200-206, 1898. Ueber die Verhaltnisse der Kemwucherung zmn Langenwachstmn an den quergestreiften Muskelfasem der weissen Ratten, Anat. Anz., Bd. 16, p. 88-91, 1899.
Neumann, E. : Ueber die vermeintliehe Abhangigkeit der Entstehung der Muskeln von den sensiblen Nerven, Arch, f . Entw.-Meeh., Bd. 16, p. 642-650, 1903.
NussBAUM, M. : Entwicklungsgeschichte des menschlichen Auges, Grafe-Samisch Arehiv, Bd. 2, 1900. Die Entwickelung der Binnenmuskeln des Auges der Wirbeltiere, Arch. f. mikr. Anat., Bd. 58, 1902.
Paneth: Die Entwickelung von quergestreiften Muskelfasem aus Sarkoplasten, Sitzungsberichte d. Kais. Akad. d. Wissenschaft, III Abt., Bd. 92, 1885.
POPOWSKY, J.: Zur Entwickelungsgeschichte der Dammuskulatur beim Menseben, 2 Taf., Anat. Hefte, Bd. 12, 1899. Reuter: Ueber die Entwickelung der Kaumuskulatur beim Schwein, Anat. Hefte, Bd. 7, 1896. Ueber die Entwickelung der Augenmuskeln beim Schwein, Anat. Hefte, Bd. 9, 1897. Ruge: Entwicklungsvorgange an der muskulatur d. menschl. Fusses, Morph. Jahrb., Bd. 4, Suppl., 1878.
ScnAFFER! Beitrjige zur Histologic und Histogenesis der quergestreiften Muskelfasem des Menschen und einiger Wirbeltiere, S. Ber. d. Acad. d. Wiss. Wien, 3 Abt., B. 102, p. 7-148, 1893.
ScHOMBURG, H. : Untersuchungen der Entwickelung der Muskeln u. Knochen des menschl. Fusses, Von der med. Facultat d. Universitat Gottingen gekronte Preisschrift, 1900.
Strazza, G. : Zur Lehre ueber die Entwicklung der Kehlkopf muskeln. MittheiL aus dem Embryologischen Institute der K. K. Univ. in Wien, Der Zweiten Folge, 3 Heft, 1899. Stohr, Ph.: Entwickelungsgeschichte der menschlichen Wollhaares, Anat. Hefte, Bd. 23, 1902.
SouLiE, A., et Bardier, E. : Recherches sur le developpement du larynx chez Fhomme, Jour. d'Anatomie et de la Phys., t. 43, p. 137-240, 1907. SziLi, A.: Zur Anatomie und Entwickelungsgeschichte der hinteren Irisschichten mit besonderer Beriicksichtigung des Musculus sphincter iridis des Menschen, Anat. Anz., Bd. 20, p. 161, 1901.
ZiMMERMANN *. Ueber. Kopf hohlenrudimente beim Menschen, Arch, f . mikr. Anat, Bd. 53, 1899.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Book - Manual of Human Embryology 12. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Manual_of_Human_Embryology_12
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G