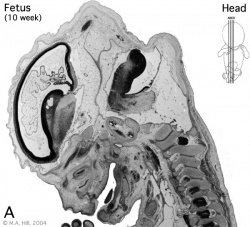
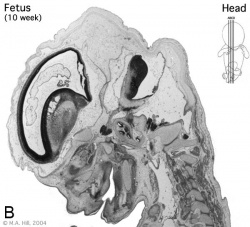
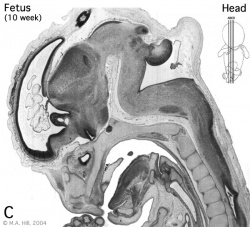
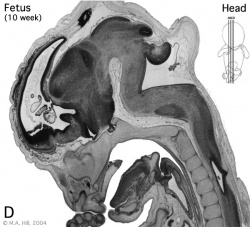
Introduction
The term "Fetal Growth Restriction" (FGR) or Intrauterine Growth Restriction (IUGR) are used to describe when the fetus does not reach full growth potential. This is usually determined by clinical sonography calculations of fetal weight, fetal size, or symmetry.
The fetal period (weeks 9 to 37) is about four times the length of the embryonic period and the clinical term may not relate directly to just the "fetal period", that is a time of extensive growth in size and mass as well as ongoing differentiation of organ systems established in the embryonic period. Clinically this period is generally described as the Second Trimester and Third Trimester. Many of the critical measurements of growth are now carried out by ultrasound and measured at birth including the Birth-Weight.
- Fetal Graphs: Crown-Rump Length (CRL) | Third trimester CRL | Head Circumference | Head Circumference 2nd Trimester | Liver Weight | Pancreas Weight | Thymus Weight | Small Intestine Length | Large Intestine Length | Length and Weight Changes | Fetal Development
Some Recent Findings
- Outcomes in patients with early-onset fetal growth restriction without fetal or genetic anomalies[1] "Early-onset fetal growth restriction is associated with poor pregnancy outcomes, but frequently is due to fetal structural or chromosomal abnormalities. The objective of this study was to determine outcomes in patients with early-onset fetal growth restriction without diagnosed fetal or genetic anomalies and to identify additional risk factors for poor outcomes in these patients. This was retrospective cohort study of singleton pregnancies in women with early-onset growth restriction defined as a sonographic estimated fetal weight <10% diagnosed between 16-28 weeks' gestation. We excluded all women with a fetal structural or chromosomal abnormality diagnosed prenatally. Data on pregnancy characteristics and outcomes were collected and analyzed for estimated fetal weight <10% and ≤5%. A nested case-control study within the cohort of patients with ongoing pregnancies was then performed to identify risk factors associated with poor pregnancy outcome using chi-squared test. One hundred forty-two patients were identified who met inclusion and exclusion criteria and 20 patients were found to have fetal structural or chromosomal abnormalities. In the remaining 122 patients, the incidence of intrauterine fetal demise was 5.7% and there were high rates of preterm birth <37 weeks (20%), birth weight <10% (59.3%), and gestational hypertension (14.1%). Later gestational age at diagnosis and the presence of echogenic bowel and abnormal initial umbilical artery Dopplers were associated with poor pregnancy outcome (22.56 versus 20.86 weeks, p = .046), (17.4 versus 2.2%, OR 9.68, 95%CI 1.65-56.73), and (35.3 versus 0%, OR 4.46, 95%CI 2.65-7.50) respectively. Patients with early-onset fetal growth restriction with no fetal structural or genetic abnormality have a high risk of poor pregnancy outcomes. Gestational age at diagnosis and certain ultrasound findings are associated with poor pregnancy outcome."
- Human papillomavirus infection and intrauterine growth restriction: a data-linkage study[2] "Using unbiased population data, to examine whether having a positive Pap smear, and thus a high probability of Human Papilloma Virus (HPV) infection, is a significant risk factor for intrauterine growth restriction (IUGR) in a subsequent pregnancy. STUDY DESIGN AND METHODS: Two independent population-based databases, namely the South Australian Perinatal Statistics Collection and the South Australian Cervical Screening Database, were deidentified and linked by the SANT Datalinkage Service. Analyses were performed on cases where Pap smear screening data was available for up to 2 years prior to a singleton live birth. RESULTS: A total of 31,827 women met the criteria. Of these, 1311 women (4.1%) had a positive Pap smear within 2 years of the current pregnancy. Those having a positive Pap smear were more likely to have a baby with IUGR than those with negative smear results. For SGA, 5.8% babies were from mothers with positive Pap smears compared to 4.0% with negative smears indicating a 40% higher risk of having an SGA baby (95%CI 20-70%) among women with positive Pap smears. For VLBW, 7.6% mothers had positive Pap smears compared with 4.0% with negative smears (p < .001), which reflects a 90% increased risk (95%CI 40-150%). These associations reduced to 20% (95%CI 1-40%) and 50% (95%CI 10-100%) for SGA and VLBW, respectively, after adjusting for all other significant covariates including maternal age, ethnicity, marital status, occupation, smoking, pregnancy history, and maternal health during pregnancy. CONCLUSIONS: Mothers with a positive Pap smear have an increased risk of IUGR, especially for VLBW, which is independent of other risk factors. The results confirm previous findings in a small study and emphasise the need to consider the risks of both cancer and IUGR in all HPV vaccination programs."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Fetal Growth Restriction | Intrauterine Growth Restriction
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Longitudinal changes in gestational weight gain and the association with intrauterine fetal growth[3] "Ultrasounds were targeted at17, 25, 33, and 37 weeks of gestation. Analyses involved a multi-step process. First, trajectories were estimated across gestation for maternal weight gain and fetal biometrics [abdominal circumference (AC, mm), biparietal diameter (BPD, mm), femur length (FL, mm), and estimated fetal weight (EFW, g)] using linear mixed models. Second, the association between maternal weight changes (per 5kg) and corresponding fetal growth from 0 to 17, 17 to 28, and 28 to 37 weeks was estimated for each fetal parameter adjusting for prepregnancy body mass index, height, parity, chronic diseases, age, smoking, fetal sex, and weight gain up to the respective period as applicable. Third, the probability of fetal SGA, EFW <10th percentile, at the 3rd ultrasound was estimated across the spectrum of maternal weight gain rate by SGA status at the 2nd ultrasound. RESULTS: From 0 to 17 weeks, changes in maternal weight were most strongly associated with changes in BPD [β=0.51 per 5kg (95%CI 0.26, 0.76)] and FL [β=0.46 per 5kg (95%CI 0.26, 0.65)]. From 17 to 28 weeks, AC [β=2.92 per 5kg (95%CI 1.62, 4.22)] and EFW [β=58.7 per 5kg (95%CI 29.5, 88.0)] were more strongly associated with changes in maternal weight. Increased maternal weight gain was significantly associated with a reduced probability of intrauterine SGA; for a normal weight woman with SGA at the 2nd ultrasound, the probability of fetal SGA with a weight gain rate of 0.29kg/w (10th percentile) was 59%, compared to 38% with a rate of 0.67kg/w (90th percentile). CONCLUSION: Among women at high-risk for SGA, maternal weight gain was associated with fetal growth throughout pregnancy, but had a differential relationship with specific biometrics across gestation. For women with fetal SGA identified mid-pregnancy, increased antenatal weight gain was associated with a decreased probability of fetal SGA approximately 7 weeks later."
- Inflammation in rat pregnancy inhibits spiral artery remodeling leading to fetal growth restriction and features of preeclampsia[4] "Fetal growth restriction (FGR) and preeclampsia (PE) are often associated with abnormal maternal inflammation, deficient spiral artery (SA) remodeling, and altered uteroplacental perfusion. Here, we provide evidence of a novel mechanistic link between abnormal maternal inflammation and the development of FGR with features of PE. Using a model in which pregnant rats are administered low-dose lipopolysaccharide (LPS) on gestational days 13.5-16.5, we show that abnormal inflammation resulted in FGR mediated by tumor necrosis factor-α (TNF). Inflammation was also associated with deficient trophoblast invasion and SA remodeling, as well as with altered uteroplacental hemodynamics and placental nitrosative stress. Moreover, inflammation increased maternal mean arterial pressure (MAP) and was associated with renal structural alterations and proteinuria characteristic of PE. Finally, transdermal administration of the nitric oxide (NO) mimetic glyceryl trinitrate prevented altered uteroplacental perfusion, LPS-induced inflammation, placental nitrosative stress, renal structural and functional alterations, increase in MAP, and FGR. These findings demonstrate that maternal inflammation can lead to severe pregnancy complications via a mechanism that involves increased maternal levels of TNF. Our study provides a rationale for the use of antiinflammatory agents or NO-mimetics in the treatment and/or prevention of inflammation-associated pregnancy complications."
- A randomised controlled trial comparing standard or intensive management of reduced fetal movements after 36 weeks gestation-a feasibility study[5] "Women presenting with reduced fetal movements (RFM) in the third trimester are at increased risk of stillbirth or fetal growth restriction. These outcomes after RFM are related to smaller fetal size on ultrasound scan, oligohydramnios and lower human placental lactogen (hPL) in maternal serum. We performed this study to address whether a randomised controlled trial (RCT) of the management of RFM was feasible with regard to: i) maternal recruitment and retention ii) patient acceptability, iii) adherence to protocol. Additionally, we aimed to confirm the prevalence of poor perinatal outcomes defined as: stillbirth, birthweight <10th centile, umbilical arterial pH <7.1 or unexpected admission to the neonatal intensive care unit. ...An RCT of management of RFM is feasible with a low rate of attrition. Investigations decrease maternal anxiety. Participants in the intensive group were more likely to have IOL for RFM. Further work is required to determine the likely level of intervention in the standard care arm in multiple centres, to develop additional placental biomarkers and to confirm that the composite outcome is valid.Trial registration ISRCTN07944306."
|
Reading
- Human Embryology (3rd ed.) Larson Chapter 15: Fetal development and the Fetus as Patient p481-499
- The Developing Human: Clinically Oriented Embryology (8th ed.) Moore and Persaud Chapter 6: The Fetal Period: Ninth Week to Birth
- Color Atlas of Clinical Embryology (2nd ed.) Moore, Persaud and Shiota Chapter 3: 9th to 38th weeks of human development p50-68
|
|
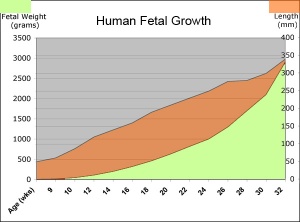
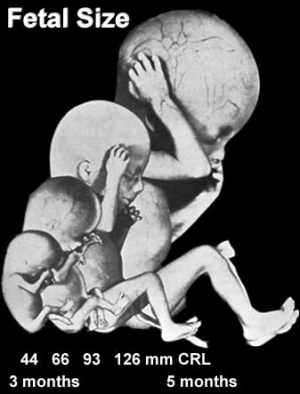
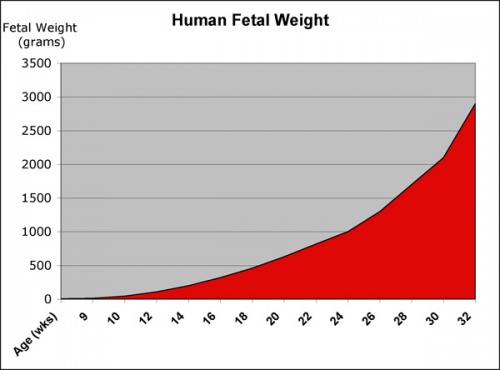
Fetal length and weight change
Second Trimester
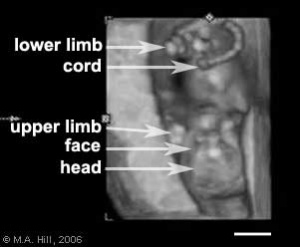
Fetus - second trimester
(ultrasound)
- Second Trimester
- Week 12 - CRL 85 mm, femur length 15 mm, biparietal diameter 25 mm.
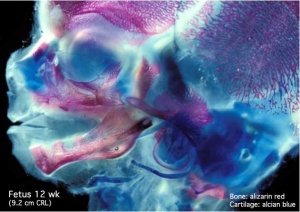
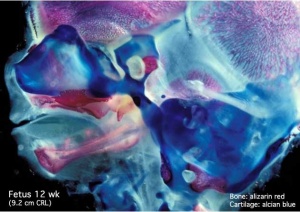
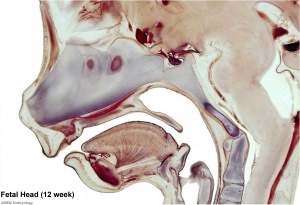
Begin by working through the features present in the early 10 week female fetus. Then look in detail at the head development in a 12 week fetus.
Then look in detail at the head development in a 12 week fetus showing both forms of ossification in the skull.
Fetal Head Growth
Second and third trimesters
Third Trimester
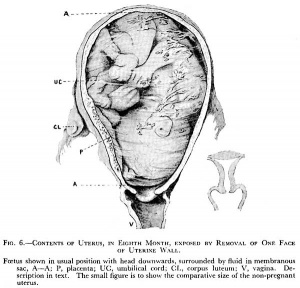
Fetus - third trimester
(historic image)
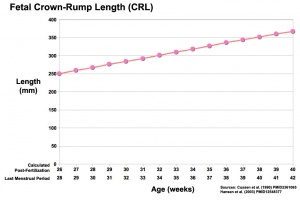
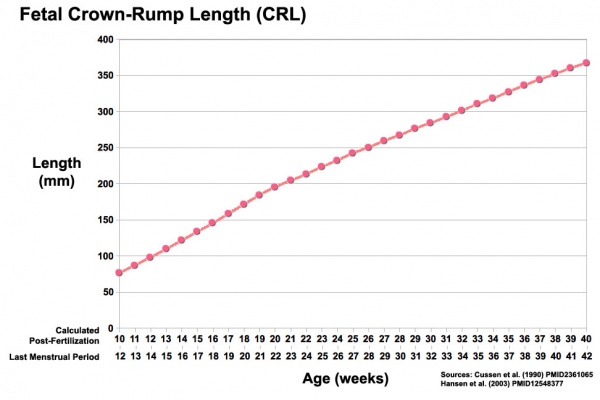
Third trimester Crown-Rump Length
- Vibration acoustically of maternal abdominal wall induces startle respone in fetus.
- Month 7 - respiratory bronchioles proliferate and end in alveolar ducts and sacs.
- Week 37 to 38 Birth.
- Links: Third Trimester
References
- ↑ Gupta S, Naert M, Lam-Rachlin J, Monteagudo A, Rebarber A, Saltzman D & Fox NS. (2019). Outcomes in patients with early-onset fetal growth restriction without fetal or genetic anomalies. J. Matern. Fetal. Neonatal. Med. , 32, 2662-2666. PMID: 29478342 DOI.
- ↑ Ford JH, Li M, Scheil W & Roder D. (2019). Human papillomavirus infection and intrauterine growth restriction: a data-linkage study. J. Matern. Fetal. Neonatal. Med. , 32, 279-285. PMID: 28889772 DOI.
- ↑ Hinkle SN, Johns AM, Albert PS, Kim S & Grantz KL. (2015). Longitudinal changes in gestational weight gain and the association with intrauterine fetal growth. Eur. J. Obstet. Gynecol. Reprod. Biol. , 190, 41-7. PMID: 25978857 DOI.
- ↑ Cotechini T, Komisarenko M, Sperou A, Macdonald-Goodfellow S, Adams MA & Graham CH. (2014). Inflammation in rat pregnancy inhibits spiral artery remodeling leading to fetal growth restriction and features of preeclampsia. J. Exp. Med. , 211, 165-79. PMID: 24395887 DOI.
- ↑ Heazell AE, Bernatavicius G, Roberts SA, Garrod A, Whitworth MK, Johnstone ED, Gillham JC & Lavender T. (2013). A randomised controlled trial comparing standard or intensive management of reduced fetal movements after 36 weeks gestation--a feasibility study. BMC Pregnancy Childbirth , 13, 95. PMID: 23590451 DOI.
Journals
Reviews
Wang C, Gao R, Huang L, Hu P, Zhu L & Chen WQ. (2020). Effect of prenatal nutritional intervention on foetal growth restriction: a real-world study in Shenzhen, China. J. Matern. Fetal. Neonatal. Med. , , 1-10. PMID: 32627652 DOI.
Thompson JL, Kuller JA & Rhee EH. (2012). Antenatal surveillance of fetal growth restriction. Obstet Gynecol Surv , 67, 554-65. PMID: 22990459 DOI.
Grivell RM, Wong L & Bhatia V. (2012). Regimens of fetal surveillance for impaired fetal growth. Cochrane Database Syst Rev , , CD007113. PMID: 22696366 DOI.
Nardozza LM, Araujo Júnior E, Barbosa MM, Caetano AC, Lee DJ & Moron AF. (2012). Fetal growth restriction: current knowledge to the general Obs/Gyn. Arch. Gynecol. Obstet. , 286, 1-13. PMID: 22526452 DOI.
Articles
Badr DA, Carlin A, Kang X, Cos Sanchez T, Olivier C, Jani JC & Bevilacqua E. (2020). Evaluation of the new expert consensus-based definition of selective fetal growth restriction in monochorionic pregnancies. J. Matern. Fetal. Neonatal. Med. , , 1-7. PMID: 32627604 DOI.
Radulescu L, Ferechide D & Popa F. (2013). The importance of fetal gender in intrauterine growth restriction. J Med Life , 6, 38-9. PMID: 23599816
Search PubMed
Search Pubmed: Fetal Growth Restriction
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 4) Embryology Abnormal Development - Fetal Growth Restriction. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Fetal_Growth_Restriction
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G