2009 Lab 10
Therapeutic Use of Stem Cells - Practical Hurdles & Ethical Issues
Introduction
This laboratory will be presented by a guest researcher Dr Antonio Lee and will look at the topic of stem cells, their therapeutic use, practical hurdles and ethical issues.
This topic is also covered in ANAT3231 Cell Biology - Stem Cells in relation to stem cell research and applications.
The laboratory will also allow time for work on the group online project.
Objectives
- Develop a general understanding of current landscape for the field of stem cell technology and therapeutics.
- Able to identify the background, key advancement and future perspectives of a recent scientific break-through in stem cell biology.
- Engage in peer-discussions and share own views on potential ethical and moral issues around a particular scientific advancement.
Lab Tutorial
PART 1.
Brief introduction and re-cap on general concepts in stem cell biology with a focus on therapeutic use of stem cells. As an example, a pre-clinical project using stem cell engineering and chemotherapy to enhance stem cell engraftment for the treatment of muscular dystorphies will be discussed.
Slides: Stem Cells: Introduction & Therapeutic Use
Slides (4 per page): Stem Cells: Introduction & Therapeutic Use (for printing)
With any new concepts or terms, please check Terms list below or the Glossary.
PART 2.
In groups of 4, read the following news article links assigned to your group. Using web search engines and the relevant journal article associated with the news article, each member of the group should address at least one of the following questions from their assigned news article.
- Question 1. What is the background to the existing problem / disease condition?
- Question 2. What approach / method did the research team take to tackle / improve the problem?
- Question 3. What was the breakthrough / major advancement OR failure / drawback? and why might this be of significance?
- Question 4. What are the next steps in moving forward? What are the next or new hurdles to overcome?
- Group 1 : Developmental biology: Asexual healing in Nature News and Views - Nature 461, 354-355 (17 September 2009) Manuscript (PDF)
- Group 2 : Scientists discover clues to what makes human muscle age in EurekAlert! Public release 30 September 2009 Manuscript (PDF)
- Group 3 : Study Shows Mozobil Induces Mobilization Of Stem Cells But Not Myeloma Tumor Cells in The Myeloma Beacon Published online 21 September 2009 10:28 pm Manuscript (PDF)
- Group 4 : Induced Pluripotent Stem Cells Created from Fat Cells in Scientific American Published online 8 September 2009 Manuscript (PDF)
- Group 5 : Human Induced Pluripotent Stem Cells Retain Some Gene Expression Of Donor Cells in Science Daily Published online 19 September 2009 Manuscript (PDF)
Lab 10 Assessment Questions
Answer the questions shown below on your own student page.
- Question 1. Identify and name 3 tissue types which contain adult (somatic) stem cells that were used/studied from the above 5 articles.
- Question 2. Name 2 reprogramming strategies/methods used in generating human induced Pluripotent Stem Cells (iPSCs) from the above 5 articles.
- Question 3. Is the following statement TRUE or FALSE?
- "Unlike the nuclear genome, the mitochondrial DNA in the embryo is derived almost exclusively from the egg; that is, it is of maternal origin."
Questions are also shown listed on the Students Page Lab 10 Assessment
Terms
Tibialis Anterior (TA) - skeletal muscle situated on the lateral side of the tibia and is a direct flexor of the foot at the ankle-joint.
Extensor Digitorum Longus (EDL) - is a pennate muscle, situated at the lateral part of the tibia deep to tibialis anterior and is a direct extensor of the digits of the foot.
Amyotrophic lateral sclerosis (ALS) - A form of motor neuron disease caused by the degeneration of motor neurons, the nerve cells in the central nervous system that control voluntary muscle movement. The condition is often called Lou Gehrig's Disease in North America.
Stem Cell Niche - is a phrase loosely used in the scientific community to describe the microenvironment in which stem cells are found, which interacts with stem cells to regulate stem cell fate. The word 'niche' can be in reference to the in vivo or in vitro stem cell microenvironment.
Satellite Cells - are small mononuclear progenitor cells with virtually no cytoplasm found in mature muscle. They are found sandwiched between the basement membrane and sarcolemma (cell membrane) of individual muscle fibres, and can be difficult to distinguish from the sub-sarcolemmal nuclei of the fibres.
BCNU (bis-chloronitrosourea) - Carmustine or BCNU is a mustard gas-related α-chloro-nitrosourea compound used as an alkylating agent in chemotherapy. It is used in the treatment of several types of brain cancer (including glioma, glioblastoma multiforme, medulloblastoma and astrocytoma), multiple myeloma and lymphoma (Hodgkin's and non-Hodgkin).
MGMT (O-6-methylguanine-DNA methyltransferase) - is a human gene and Methylation of the gene's promoter may play a significant role in carcinogenesis. A 2005 study showed that -- in patients with glioblastoma multiforme, a severe type of brain tumor -- the methylation state of the MGMT gene determined whether tumor cells would be responsive to temozolomide; if the promotor was methylated, temozolomide was effective. [1]
Temozolomide - is an oral alkylating agent which can be used for the treatment of Grade IV astrocytoma -- an aggressive brain tumor, also known as glioblastoma multiforme.
Real-Time Polymerase Chain Reaction (QPCR) - Also called quantitative real time polymerase chain reaction (Q-PCR/qPCR) or kinetic polymerase chain reaction, is a laboratory technique based on the polymerase chain reaction, which is used to amplify and simultaneously quantify a targeted DNA molecule. It enables both detection and quantification (as absolute number of copies or relative amount when normalized to DNA input or additional normalizing genes) of a specific sequence in a DNA sample.
FISH (fluorescence in situ hybridization) - is a cytogenetic technique used to detect and localize the presence or absence of specific DNA sequences on chromosomes. FISH uses fluorescent probes that bind to only those parts of the chromosome with which they show a high degree of sequence similarity.
Duchenne muscular dystrophy (DMD) - is a severe recessive X-linked form of muscular dystrophy characterized by rapid progression of muscle degeneration, eventually leading to loss of ambulation and death. This affliction affects one in 3500 males, making it the most prevalent of muscular dystrophies.
mdx mice - is a strain of mice that has a hereditory disease of the muscles caused by a mutation on the X-chromosome. It is used as a disease model for human muscular dystrophy.
Dystrophin - is a rod-shaped cytoplasmic protein, and a vital part of a protein complex that connects the cytoskeleton of a muscle fiber to the surrounding extracellular matrix through the cell membrane.
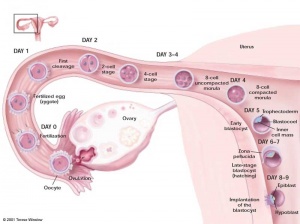
Timeline of Human Embryonic Stem Cell Research
- 1878 First reported attempts to fertilize mammalian eggs outside the body
- 1959 First report of animals (rabbits) produced through IVF in the United States
- 1960's Studies of teratocarcinomas in the testes of several inbred strains of mice indicates they originated from embryonic germ cells. The work establishes embryonal carcinoma (EC) cells as a kind of stem cell
- 1968 Edwards and Bavister fertilize the first human egg in vitro
- 1970's EC cells injected into mouse blastocysts produce chimeric mice. Cultured SC cells are explored as models of embryonic development, although their complement of chromosomes is abnormal
- 1978 Louise Brown, the first IVF baby, is born in England
- 1980 Australia's first IVF baby, Candace Reed, is born in Melbourne
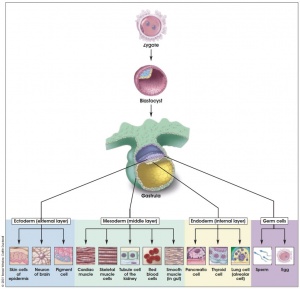
- 1981 Evans and Kaufman, and Martin derive mouse embryonic stem (ES) cells from the inner cell mass of blastocysts. They establish culture conditions for growing pluripotent mouse ES cells in vitro. The ES cells yield cell lines with normal, diploid karyotyes and generate derivatives of all three primary germ layers as well as primordial germ cells. Injecting the ES cells into mice induces the formation of teratomas. The first IVF baby, Elizabeth Carr, is born in the United States.
- 1984–88 Andrews et al., develop pluripotent, genetically identical (clonal) cells called embryonal carcinoma (EC) cells from Tera-2, a cell line of human testicular teratocarcinoma. Cloned human teratoma cells exposed to retinoic acid differentiate into neuron-like cells and other cell types
- 1989 Pera et al., derive a clonal line of human embryonal carcinoma cells, which yields tissues from all three primary germ layers. The cells are aneuploid (fewer or greater than the normal number of chromosomes in the cell) and their potential to differentiate spontaneously in vitro is typically limited. The behavior of human EC cell clones differs from that of mouse ES or EC cells
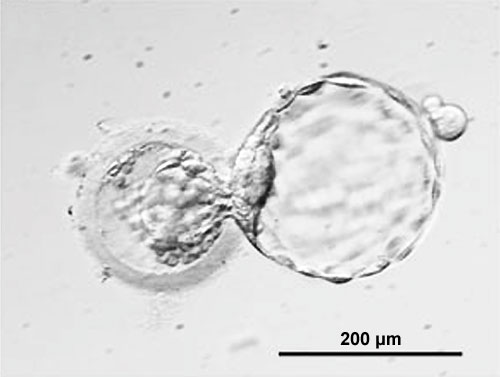
- 1994 Human blastocysts created for reproductive purposes using IVF and donated by patients for research, are generated from the 2-pronuclear stage. The inner cell mass of the blastocyst is maintained in culture and generates aggregates with trophoblast-like cells at the periphery and ES-like cells in the center. The cells retain a complete set of chromosomes (normal karyotype); most cultures retain a stem cell-like morphology, although some inner cell mass clumps differentiate into fibroblasts. The cultures are maintained for two passages
- 1995–96 Non-human primate ES cells are derived and maintained in vitro, first from the inner cell mass of rhesus monkeys, and then from marmosets. The primate ES cells are diploid and have normal karyotypes. They are pluripotent and differentiate into cells types derived from all three primary germ layers. The primate ES cells resemble human EC cells and indicate that it should be possible to derive and maintain human ES cells in vitro.
- 1998 Thomson et al., derive human ES cells from the inner cell mass of normal human blastocysts donated by couples undergoing treatment for infertility. The cells are cultured through many passages, retain their normal karyotypes, maintain high levels of telomerase activity, and express a panel of markers typical of human EC cells non-human primate ES cells. Several (non-clonal) cell lines are established that form teratomas when injected into immune-deficient mice. The teratomas include cell types derived from all three primary germ layers, demonstrating the pluripotency of human ES cells. Gearhart and colleagues derive human embryonic germ (EG) cells from the gonadal ridge and mesenchyma of 5- to 9-week fetal tissue that resulted from elective abortions. They grow EG cells in vitro for approximately 20 passages, and the cells maintain normal karyotypes. The cells spontaneously form aggregates that differentiate spontaneously, and ultimately contain derivatives of all three primary germ layers. Other indications of their pluripotency include the expression of a panel of markers typical of mouse ES and EG cells. The EG cells do not form teratomas when injected into immune-deficient mice
- 2000 Scientists in Singapore and Australia led by Pera, Trounson, and Bongso derive human ES cells from the inner cell mass of blastocysts donated by couples undergoing treatment for infertility. The ES cells proliferate for extended periods in vitro, maintain normal karyotypes, differentiate spontaneously into somatic cell lineages derived from all three primary germ layers, and form teratomas when injected into immune-deficient mice.
- 2001 As human ES cell lines are shared and new lines are derived, more research groups report methods to direct the differentiation of the cells in vitro. Many of the methods are aimed at generating human tissues for transplantation purposes, including pancreatic islet cells, neurons that release dopamine, and cardiac muscle cells.
References
Lee et al. Methylguanine DNA Methyltransferase-Mediated Drug Resistance-Based Selective Enrichment and Engraftment of Transplanted Stem Cells in Skeletal Muscle. Stem Cells (2009) vol. 27 (5) pp. 1098-1108 Manuscript (PDF)
Textbooks
- Molecular Biology of the Cell Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 Chapter 19 Cellular Mechanisms of Development p1037-1039 | Figure 22-4. The definition of a stem cell | Figure 22-19. Renewal of the gut lining | Figure 22-5. Two ways for a stem cell to produce daughters with different fates
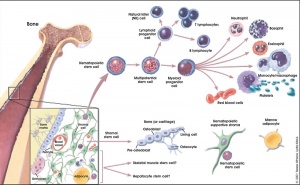
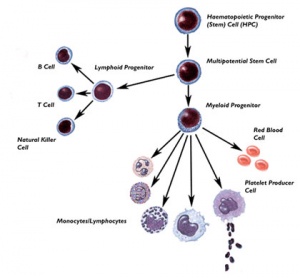
- Molecular Cell Biology Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E. New York: W. H. Freeman & Co.; c1999 Chapter 23. Cell Interactions in Development Figure 24-8. Formation of differentiated blood cells from hematopoietic stem cells in the bone marrow
- The Cell- A Molecular Approach Cooper, Geoffrey M. Sunderland (MA): Sinauer Associates, Inc.; c2000 IV. Cell Regulation Chapter 14. Cell Proliferation in Development and Differentiation Stem Cells
Search
- Bookshelf stem cell |
Links
- UNSW Embryology Stem Cells | Stem Cells - Ethics | Stem Cells - Cord Blood | Stem Cells - Adult
- Australian Stem Cell Centre Australian Stem Cell Centre | Public Education | Fact Sheets | Australasian Society for Stem Cell Research
- NIH Stem Cell Information Home Page | Stem Cell Bank (NSCB) | Stem Cells: Scientific Progress and Future Research Directions 2001 | Regenerative Medicine 2006
- International Consortium of Stem Cell Networks Italic textout-icscn/ International Consortium of Stem Cell Networks | Stem Cell Legislation - World Map | FAQs
- STEM CELLS Journal Stem Cells Portal
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Course Content 2009
Embryology Introduction | Cell Division/Fertilization | Cell Division/Fertilization | Week 1&2 Development | Week 3 Development | Lab 2 | Mesoderm Development | Ectoderm, Early Neural, Neural Crest | Lab 3 | Early Vascular Development | Placenta | Lab 4 | Endoderm, Early Gastrointestinal | Respiratory Development | Lab 5 | Head Development | Neural Crest Development | Lab 6 | Musculoskeletal Development | Limb Development | Lab 7 | Kidney | Genital | Lab 8 | Sensory - Ear | Integumentary | Lab 9 | Sensory - Eye | Endocrine | Lab 10 | Late Vascular Development | Fetal | Lab 11 | Birth, Postnatal | Revision | Lab 12 | Lecture Audio | Course Timetable
Cite this page: Hill, M.A. (2026, Haziran 9) Embryology 2009 Lab 10. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2009_Lab_10
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G