2009 Lecture 18
Integumentary System
Introduction
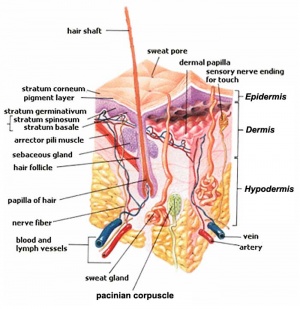
The skin provides a barrier between ourselves and our environment, it also contains specializations in different regions including hair, nails, glands and sensory receptors.
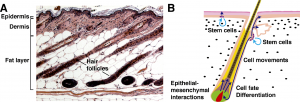
The two major tissue organizations of epithelial (ectoderm, epidermis) and mesenchyme (mesoderm connective tissue, dermis and hypodermis) are shown within skin. In addition, we have aslo extensive populating by melanocytes (neural crest) and sensory nerve endings.
It remains today as possibly the first epithelial specialization from which arose other epithelial specializations now located inside the body. The external skin specializations have many different roles and functions. This system is also an excellent model for distribution or "pattern" that is of interest.
Textbooks
- Human Embryology Larson Chapter 14 p443-455
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 20: P513-529
- Lecture Audio Lecture Date: 29-09-2009 Lecture Time: 12:00 Venue: BioMed E Speaker: Mark Hill Integumentary
Objectives
- Understand the embryonic origin and differentiation of the epidermis and dermis.
- Understand the formation of hair and nails.
- Understand the formation of sweat glands, mammary glands.
- Understand the formation of teeth.
- Brief understanding of associated abnormalities.
Skin Origins
Skin is our largest organ, providing a protective layer between us and our environment
- Ectoderm forms the surface epidermis and the associated glands.
- Mesoderm forms the underlying connective tissue of dermis and hypodermis.
- Neural crest cells also migrate into the forming epidermis and the skin is also populated by specialized sensory endings.
- epithelia/mesenchyme (ectoderm/mesoderm) interaction an inductive manner (last weeks lecture on kidney)
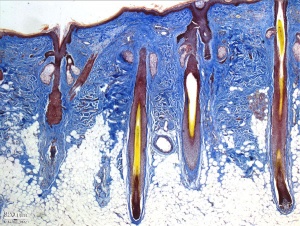
- 2 main types of histological skin - thin (most of body) thick (soles of feet and hands) based on ectoderm, not the thickest skin including dermis (top of back)
Regional Specializations
- skin has different structures associated with different regions of the body
- nails, hail, glands, teeth, eyelashes, eyebrow
Development Overview
4 weeks
- simple ectoderm epithelium over mesenchyme.
1-3 months
- ectoderm - germinative (basal) cell repeated division of generates stratified epithelium.
- mesoderm - somite dermatome spreads out under the epithelium, differentiates into connective tissue and blood vessels.
4 months
- basal cell- proliferation generates folds in basement membrane.
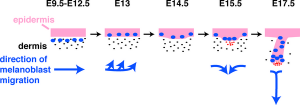
- neural crest cells- (melanocytes) migrate into epithelium. These are the pigment cell of the skin.
- embryonic connective tissue- differentiates into dermis, a loose ct layer over a dense ct layer. Beneath the dense ct layer is another loose ct layer that will form the subcutaneous layer.
- Ectoderm contributes to nails, hair follictles and glands.
- Nails form as thickening of ectoderm epidermis at the tips of fingers and toes. These form germinative cells of nail field.
- Cords of these cells extend into mesoderm forming epithelial columns. These form hair follocles, sebaceous and sweat glands.
5 months
- Hair growth initiated at base of cord, lateral outgrowths form associated sebaceous glands.
- Other cords elongate and coil to form sweat glands.
- Cords in mammary region branch as they elongate to form mammary glands. These glands will complete development in females at puberty. Functional maturity only occurs in late pregnancy.
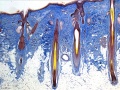
Epidermis
- week 4-5 early skin is a single ectodermal layer, stratum germinativum basal layer
- week 11 forms intermediate layer
- periderm then lost replaced by stratum corneum, keratinization and desquamification
- week 10 epidermial ridges are formed by proliferation
Neural crest cells
- Neural crest cells migrate into skin (late embryonic) form melanoblasts
- day 40-50 differentiate into then melanocytes - form pigment granules
- different content of melanin (Greek, melas = "black") accounts for differet skin colour
Dermis
- lateral plate mesodermal in origin
- forms connective tissue
- afferent nerves influence dermal ridge formation
Blood Vessels
- lateral plate mesodermal in origin
- week 5 blood vessels form in mesenchyme
- form capillary beds, extensive remodelling with development
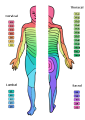
Skin Dermatomes
- pattern of skin innervation
- area supplied by single spinal nerve
- motor and sensory DRG
- cutaneous nerve area
Keratin
- large family of intermediate filament protein, 17+ isoforms
- skin disease associated with mutations in keratin genes
Keratins are the major structural proteins of the vertebrate epidermis and its appendages, constituting up to 85% of a fully differentiated keratinocyte. Together with actin microfilaments and microtubules, keratin filaments make up the cytoskeletons of vertebrate epithelial cells. Traced as far back in the evolutionary kingdom as mollusks, keratins belong to the superfamily of intermediate filament (IF) proteins that form alpha-helical coiled-coil dimers which associate laterally and end-to-end to form 10-nm diameter filaments. The evolutionary transition between organisms bearing an exoskeleton and those with an endoskeleton seemed to cause considerable change in keratin. Keratins expanded from a single gene to a multigene family. Of the approximately 60 IF genes in the human genome, half encode keratins, and at least 18 of these are expressed in skin. Vertebrate keratins are subdivided into two sequence types (I and II) that are typically coexpressed as specific pairs with complex expression patterns. The filament-forming capacity of a pair is dependent upon its intrinsic ability to self-assemble into coiled-coil heterodimers, a feature not required of the invertebrate keratins (Weber et al 1988). Approximately 20,000 heterodimers of type I and type II keratins assemble into an IF. Mutations that perturb keratin filament assembly in vitro can cause blistering human skin disorders in vivo. (from Review Article)
Development of Glands
- 2 main types - sebaceous and sweat
- both ectodermal in origin
- form as ingrowth of ectoderm into the mesoderm
Sebaceous
- associated with hair development
- except plans penis and labia minora
- these glands secrete vernix
Sweat Glands
- mostly eccrine some apocrine
- apocrine in axilla, pubic and nipple regions
- see also mammary gland development
Vernix Caseosa
- (Latin, vernix caseosa = varnish)
- covers fetal skin- secretion from sebaceous glands
- protects skin from extraembryonic fluids amnion, urine
- slippery and helps with parturition
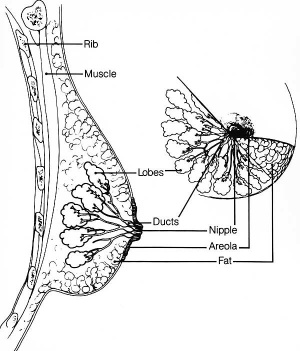
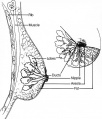
Mammary Glands
UNSW Embryology - Mammary Glands
- week 6 epidermis downgrowth into dermis, modified sweat glands
- epithelia/mesenchyme inductive interaction, mesenchyme forms connective tissue and fat
- mammary ridges - mammary bud formation, pair of ventral regions axilla to inguinal
- pectoral regions generate breasts
- buds branch to form lactiferous ducts, only main duct formed at birth
- mammary pit - forms fetal period
- areola - depressed region at gland, proliferation of connective tissue postnatally
- prior to puberty male and female glands the same
Puberty
- sex hormone estrogen stimulate growth, full development approx 20 years
- growth also influenced by other hormones - progereterone, prolactin, corticoids, growth hormone
- mainly fat and connective tissue deposition
Pregnancy
- raised estrogens and progesterone stimulate gland development
- hemispherical shape due to fat deposition
- lactation supports development
Breast cancer
- In 1994, two breast cancer susceptibility genes were identified: BRCA1 on chromosome 17 BRCA2 on chromosome 13
- When an individual carries a mutation in either BRCA1 or BRCA2, they are at an increased risk of being diagnosed with breast or ovarian cancer at some point in their lives. Normal function of these genes was to participate in repairing radiation-induced breaks in double-stranded DNA. It is though that mutations in BRCA1 or BRCA2 might disable this mechanism, leading to more errors in DNA replication and ultimately to cancerous growth.
Breast Cancer Detection - reduce mortality is through early detection (general screening of the population for BRCA1 and BRCA2 is not yet recommended). New strategies to find anti-cancer drugs are constantly being developed. The latest, called 'synthetic lethal screening' looks for new drug targets in organisms such as yeast and fruit flies. In the same way that studies in yeast recently helped to identify the functions of BRCA1 and BRCA2, it is thought that drugs that work in more primative organisms will also be applicable to humans.
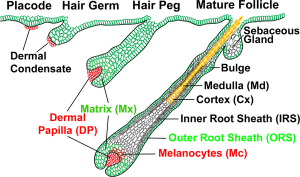
Hair Development
Hair formation, or follicle development, is an example of two distinct developmental processes: epithelio-mesenchymal interactions and pattern formation. The differentiated hair follicle will eventually contain 20 or more different cell types. Melanocytes, which provide the hair colour, have a neural crest origin, and with ageing their numbers decline leading to whitening (grey) of the hair process.
Hair follicle development in humans begins as an epithelial-mesenchymal interaction at week 9 - 12. This initial lanugo hair is replaced in the late fetal or early neonate by vellus and terminal hairs. A second round of development occurs during puberty under the influence of steroidal hormones (More? Puberty).
- Before birth we have embryonic hair that has an imporant role in binding the skins waxy protective coating against our watery environment.
- After birth we have early postnatal hair that is gradually replaced by the mature form.
- At puberty we have a second round of hair formation under endocrine regulation by sex hormones.
- The hair follicle is also a site for stem cells, allowing replacement of the follicle.
Hair Follicle
- follicle forms in stratum germinativum of epidermis
- hair bud then hair bulb forms hair
- mesenchyme forms hair papilla
- germinal matrix cells become keratinized to form hair shaft
- week 12 - lanugo hair (Latin, lana = wool) - first hair formed replaced postnatally, role in binding vernix to skin
- arrector pili muscle - develop in mesenchyme and form the muscles that move hair.
- hair colour - melanocytes (neural crest) produce melanin which influences hair colour.
- Puberty - coarse hair in pubis and axilla in both male and female (in males also on face and other body regions chest, etc)
Fetal Hair
- Month 5 - hair appears on the head and beginning of vernix caseosa deposition.
- Month 6 - body is covered by fine hairs (lanugo) and the deposit of vernix caseosa is considerable. Skin papilla are developed and the free border of the nail projects from the corium of the dermis.
- Month 8 - skin now completely coated with vernix caseosa, and the lanugo begins to disappear. Skin is also pink in colour and subcutaneous fat being deposited (hypodermis layer).
- Month 9 - lanugo has largely disappeared from the trunk.
Lanugo Hair
- From about the third month lanugo hair (Latin, lana = wool) hiar is initially formed and it has a role in binding vernix to skin.
- Hair grows over the entire body at the same rate, so the hairs are the same length, and is shed abut 4 weeks before birth. Premature infants can still be covered with these hairs.
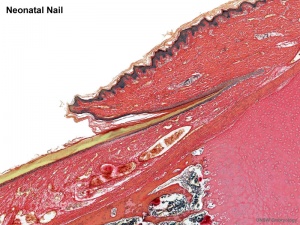
Neonatal Hair
Newborn infants have two types of hair:
- Vellus Hairs - short hairs, only a centimetre or two long, and contain little or no pigment, follicles that produce them do not have sebaceous glands and never produce any other kind of hairs
- Terminal Hairs - long hairs that grow on the head and in many people on the body, arms and legs, produced by follicles with sebaceous glands, the hairs in these follicles gradually become thinner and shorter until they look like vellus hairs
Hair Follicle Phases
There are several phases of hair follicle growth.
- Anagen Phase - active phase
- Catagen Phase - apoptosis-driven involution, end of active growing phase of the life cycle of the hair, between growing phase (anagen) and resting stage (telogen).
- Telogen Phase - hair follicle resting phase of hair growth cycle.
Puberty Hair Development
The appearance of pubic hair occurs along with ther secondary sexual characteristics (also Tanner staged) and is under endocrine control.
- Estrogens- (1 beta-estradiol, E2) involved in skin physiology and are potent hair growth modulators.
- Testosterone- Face, trunk and extremities increases hair follicle anagen phase (active) and increases also hair growth rate, thickness, medullation and pigmentation. Effects due to high hormone levels and target organ conversion to 5 alpha-dihydrotestosterone. Pubic hair develops even in absence of 5 alpha-reductase effect.
| Tanner Stage | Pubic Hair Development |
| 1 | None |
| 2 | Few darker hairs along labia or at base of penis |
| 3 | Curly pigmented hairs across pubes |
| 4 | Small adult configuration |
| 5 | Adult configuration with spread onto inner thighs |
| 6 | Adult configuration with spread to linea alba |
Table based upon the Tanner stages of secondary sexual development. (Tanner JM. Growth at Adolescence. 2nd ed. Oxford: Blackwell Scientific, 1962.)
Nail Development
- Forelimb before hindlimb - week 10 fingernails, week 14 toe nails
- nail field - appears at tip and migrates to dorsal surface
- thickened epidermis - surrounding cells form nail fold
- keratinization of proximal nail fold forms nail plate
Nails reach Digit Tip
- week 32 fingernails
- week 36 toenails
- nail growth indicator of prematurity
Nail Terms
- nail plate - visible part of the nail
- nail bed - skin beneath the nail plate
- cuticle - tissue that overlaps the plate and rims the base of the nail
- nail folds - skin folds that frame and support the nail on three sides
- lunula - half-moon at the base of the nail
- matrix - hidden part of the nail unit under the cuticle
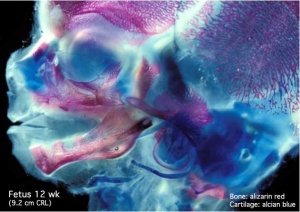
Embryo Images - Human (day 64) primary nail fields
Teeth
- integumentary system specialization by epitheilal/mesenchymal interactions in development and develops with a major contribution from the neural crest.
- ectoderm of the first pharyngeal arch and neural crest, ectomesenchymal cells.
- week 6 - odontogenesis begins, tooth bud
- 4 morphological stages describing the early tooth development: bud, cap, bell, and terminal differentiation
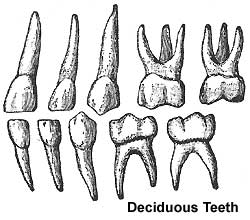
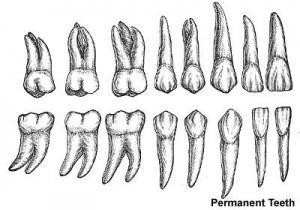
- 2 sets of teeth: 20 deciduous teeth, 32 permanent teeth
- differential rates of growth, shed at different times over 20 year period
- ectoderm, mesoderm and neural crest mesenchyme contribute
- inductive influence of neural crest with overlying ectoderm
- tooth growth occurs in ossifying jaws
odontoblasts
- neural crest-derived mesenchymal cells which differentiate under the influence of the enamel epithelium. Cells secrete predentin, calcifies to form dentin.
ameloblasts
- inner enamel epithelium forms pre-ameloblasts differentiate and produce enamel
periodontal ligament
- tooth is not anchored directly onto its bony socket (alveolar bone) but held in place by the periodontal ligament (PDL), a specialized connective tissue structure that surrounds the tooth root coating of cementum.
- ligament also act as; a shock absorber, transmitter of chewing forces (from tooth to bone), sensory information (heat, cold, pressure and pain).
- collagen fiber bundles within the ligament are called "Sharpey's fibres".
Teeth Postnatal
Deciduous teeth
- 6-24 months erupt from gums by pushing toward surface
- 2 years - all deciduous teeth present
Permanent teeth
- 6 years until early adult
- tooth bud lie in gums beneath deciduous teeth
- osteoclasts resorb deciduous teeth roots
- growth affects face shape
Molecular
- Role of homeobox genes in the patterning, specification, and differentiation of ectodermal appendages in mammals. Duverger O, Morasso MI. J Cell Physiol. 2008 Aug;216(2):337-46. Review. PMCID: 18459147 | Figure 1 Key steps in the development of three major ectodermal appendages
Abnormalities
The list below represents only a selection of associated abnormalities.
Skin
- Ehlers-Danlos Syndrome - (EDS I and EDS II) loose-jointedness and fragile, bruisable skin that heals with peculiar scars. The syndrome is caused by mutation in the collagen gene. Infants are born prematurely due to premature rupture of fetal membranes.
- Epidermolysis Bullosa Simplex - autosomal dominant disease of keratin, generating skin fagility and non-scarring blisters of the skin caused by little or no trauma. Four clinical subtypes: 1. EBS - Weber-Cockayne - mild blistering of the hands and feet 2. EBS - Koebner, 3. EBS - mottled pigmentation, 4. EBS - Dowling-Meara - generalized blistering which can be fatal.
- Autosomal Recessive Congenital Ichthyosis - an excessive keratinization disorder.
- Cutis Aplasia - congenital absence of the skin, particularly on the scalp, larger defects may extend to the dura or meninges. Generally isolated lesions, but can also be associated with a variety of other genetic disorders. Heals as a flat scar or keloid lump.
- Incontinentia Pigmenti - X-linked dominant disorder with most but not all cases affecting females. The skin changes follow characteristic four stages. In the neonatal period the first stage is noted with blisters often preceded or accompanied by erythema. These involve any part of the body but usually not the face. They do not cross the midline. These lesions are best seen in the second photograph in the groin and suprapubic region. The lesions follow a linear distribution in the limbs and circumferentially around the trunk. Crops of lesions may occur over a period of weeks to few months. During that stage, peripheral eosinophilia may be noted. The second stage follows and is characterised by hyperkeratosis or verrucous changes. At times the 2 stages occur simultaneously as noted in the first and third photograph. The third stage is that of hyperpigmentation typically appearing as streaks or whorls. It may be present throughout childhood. The fourth stage seen in teenage or adults is that of pale or atrophic streaks.
- Haemangiomas - relatively common (10% of infants), more common more common in preterm infants and girls. Initially present neonatally as a small "spot" or blanched vascular area which grows over the next 6 months before gradually involuting, usually over the next few years.
Breast
- occurs in 1% of female population
- polymastia - extra breast
- polytheli - extra nipple, supernumerary nipple (relatively common in males)
Hair
- androgenetic alopecia - male- and female-pattern hair loss.
- telogen effluvium - alteration of the normal hair cycle, due to many different stress stimuli (severe stress, chemotherapy, childbirth, major surgery, severe chronic illness, rarely occurance in vaccination)
- alopecia areata - autoimmune disease, form antibodies against some hair follicles, distinct circular pattern of hair loss.
Nail
- Congenital hyponychia or anonychia - (hyponychium is the thickened epidermis beneath the free distal end of the digit) fingernails and toenails are absent without significant bone anomalies.
- Total anonychia congenita - all absent, is a rare condition and may have an autosomal dominant inheritance patternis a rare condition, potentially autosomal dominant inheritance.
- Nail-patella syndrome - small, poorly developed nails and kneecaps, autosomal dominant inheritance.
- Ectodermal dysplasias - group of syndromes all deriving from abnormalities of the ectodermal structures.
- Brachydactylies
Teeth
- adontia - total lack of tooth development.
- amelogenesis imperfecta - abnormal tooth enamel formation (AMELX, ENAM, KLK4, MMP20).
- dentinogenesis imperfecta - discoloured teeth with an opalescent sheen, dentin does not support enamel (dentin sialophosphoprotein mutation)
- dens evaginatus - dental anomaly mainly affecting premolars in people of Mongolian origin.
- hypodontia - lack of development of one or more teeth.
- hypohidrotic ectodermal dysplasia - maldevelopment of one or more ectodermal-derived tissues.
- microdontia - small teeth.
Links
- UNSW Embryology - Introduction | Detailed Overview of Skin components | Abnormalities | Stage 13/14 | Stage 22 | Histology | Dermatomes | Glands | Mammary Glands | Teeth | Hair | Nails
- Teeth - University of Helsinki - Gene Expression in Tooth | American Dental Association Overview - Tooth | Columbia University Medical Centre - Illustrations: How a Tooth Decays | Merck - Tooth disorders | Nemours Foundation - Teething Tots
- Neonatal Dermatology - NZ National Women's Health
- Dermatology Image Atlas - DermAtlas
References
- Before We Are Born (5th ed.) Moore and Persaud Chapter 21: P481-496
- Essentials of Human Embryology Larson Chapter 14: P303-315
- Color Atlas of Clinical Embryology Moore Persaud and Shiota Chapter 15: p231-236
- Nature - Milestones in Cutaneous Biology "highlights groundbreaking advances in cutaneous biology over the past 100 years."
- International Journal Developmental Biology 2004 - Skin Development special issue
Online Textbooks
Developmental Biology 6th ed. Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000. Development of the hair follicles in fetal human skin | Image - Coordinated differentiation and morphogenesis in the mammalian tooth | Tooth Development
Eurekah Bioscience Collection Role of GLI proteins in embryonic hair follicle development
Molecular Biology of the Cell FGF5 is a negative regulator of hair formation
Images
Terms
dermal papilla - the extensions of the dermis into the epidermis.
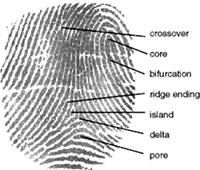
dermatoglyphic patterns - (Greek, derma = "skin", glyph = "carving") fingers, palms, toes, and soles skin patterns.
epidermal growth factor receptor - expressed on cells in the epidermis basal layer, signaling stimulates both epidermal growth and wound healing and also mediates an inhibition of differentiation.
rete ridge- the extensions of the epidermis into the dermis. These epidermal surface thickenings extend downward between underlying connective tissue dermal papillae. This is also the site of initial eccrine gland differentiation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Course Content 2009
Embryology Introduction | Cell Division/Fertilization | Cell Division/Fertilization | Week 1&2 Development | Week 3 Development | Lab 2 | Mesoderm Development | Ectoderm, Early Neural, Neural Crest | Lab 3 | Early Vascular Development | Placenta | Lab 4 | Endoderm, Early Gastrointestinal | Respiratory Development | Lab 5 | Head Development | Neural Crest Development | Lab 6 | Musculoskeletal Development | Limb Development | Lab 7 | Kidney | Genital | Lab 8 | Sensory - Ear | Integumentary | Lab 9 | Sensory - Eye | Endocrine | Lab 10 | Late Vascular Development | Fetal | Lab 11 | Birth, Postnatal | Revision | Lab 12 | Lecture Audio | Course Timetable
Cite this page: Hill, M.A. (2026, April 21) Embryology 2009 Lecture 18. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2009_Lecture_18
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G