BGD Lecture - Sexual Differentiation
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
| This lecture covers embryonic sexual differentiation covering gonad, internal and external genital development. Differences in development are dependent on a protein product of the Y chromosome SRY gene. The paired mesonephric ducts (Wolffian ducts) and paramesonephric ducts (Müllerian ducts) contribute the majority of male and female internal genital tract respectively. I will also introduce some abnormalities of development, that will be covered in the associated practical class. This is one system that continues to develop and change postnatally with puberty and menopause. |
| Lecture Archive |
|---|
| Textbooks | ||||
|---|---|---|---|---|

|
Hill, M.A. (2020). UNSW Embryology (20th ed.) Retrieved April 26, 2024, from https://embryology.med.unsw.edu.au | |||

|
Moore, K.L., Persaud, T.V.N. & Torchia, M.G. (2015). The developing human: clinically oriented embryology (10th ed.). Philadelphia: Saunders. (links only function with UNSW connection)
Chapter 12 Urogenital System
| |||

|
Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R., Francis-West, P.H. & Philippa H. (2015). Larsen's human embryology (5th ed.). New York; Edinburgh: Churchill Livingstone.(links only function with UNSW connection)
Chapter 16 Development of the Reproductive System
| |||

|
Nussey S. and Whitehead S. Endocrinology: An Integrated Approach (2001) Oxford: BIOS Scientific Publishers; ISBN-10: 1-85996-252-1.
Chapter 6 The gonad | |||
| A Recent Review
Bashamboo A, Eozenou C, Rojo S & McElreavey K. (2017). Anomalies in human sex determination provide unique insights into the complex genetic interactions of early gonad development. Clin. Genet. , 91, 143-156. PMID: 27893151 DOI.
| ||||
Objectives
- Understand the development of the gonads in males and females.
- Understand the chromosomal basis of sex determination.
- Understand the differences in male/female internal duct develpoment.
- Understand the origins of the external genitalia.
- Understand the developmental abnormalities in male and female development.
Stages of Sexual Differentiation
- Development of the indifferent gonad - (genital ridge) early embryo
- Differentiation of gonad - (testis or ovary) late embryo, defining event in sexual differentiation
- Differentiation of internal genital organs and ducts - late embryo to fetal
- Differentiation of external genitalia - fetal
- Development of secondary sexual characteristics and reproductive function - puberty
Human Timeline
|
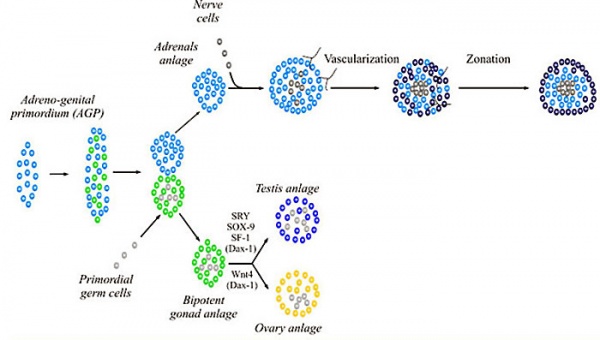
1. Development of the indifferent gonad
|

|
Gonad and adrenal early development (not required to know molecular information)
2. Differentiation of gonad into testis or ovary
Chromosomal Sex Determination
| Males | Females |
|---|---|
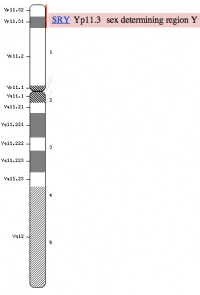
Y Chromosome
|
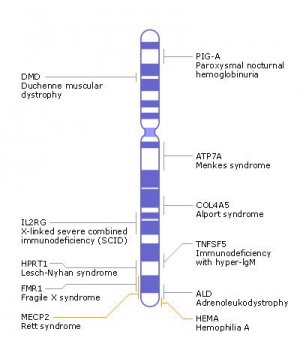
X Chromosome
|
|
|
X Inactivation
|
- Links: MBoC - Figure 20-18. Influence of Sry on gonad development | image (image provides a good overview of the anatomy of sex determination, I will refer to this in the lecture and practical class)
Supporting Cells
- So called because they "support" the germ cells
| Males | Females |
|---|---|
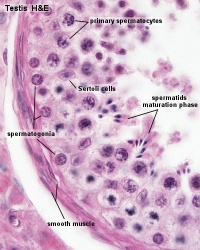
sertoli cells
|
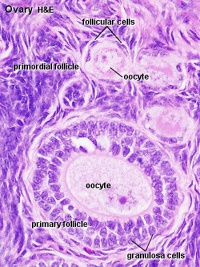
Granulosa cells
|
|
|
|
|
Steroid secreting cell lineage
| Males | Females |
|---|---|
Leydig cells
|
Theca cells
|
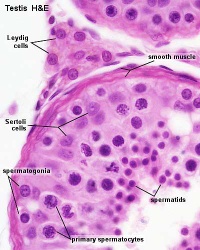
|
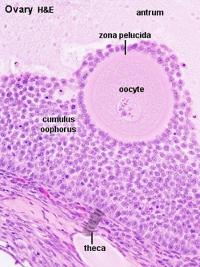
|
Primordial Germ Cells
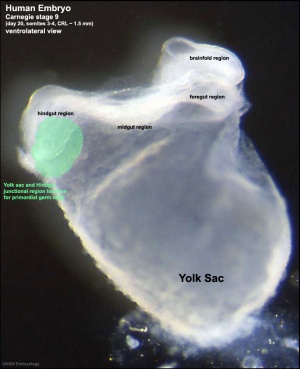
- Primordial germ cells (PGCs) are thought to be the first population of cells to migrate through the primitive streak in early gastrulation (Week 3)
- cells then lie at the hindgut yolk sac junctional region
- later migrate into the genital ridge (germinal ridge) in early embryonic development.
| Mouse Primordial Germ Cell Migration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||
- Not the primordial germ cells which respond to SRY presence or absence, but the supporting cells within the developing gonad.
- Germ cells occasionally migrate by mistake into the developing adrenal gland and in the absence of sertoli cells telling them what to do, abnormally begin to develop as oocytes, even in males
- Links: Germ cell migration pathway
Gametogenesis
- forming PGCs as a small population of migratory cells
- enter the gonad where they undergo several rounds of mitotic cell division
- female - the germ cells enter meiosis and become arrested at the dictyate (diplotene) stage of meiotic prophase 1. All oocytes are at this stage at birth
- male - the germ cells are enclosed by the developing Sertoli cells and are induced to arrest differentiation and cell division as T1 prospermatogonia until after birth.
- Links: Image - Spermatogenesis | Image -Oogenesis
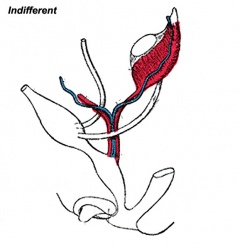
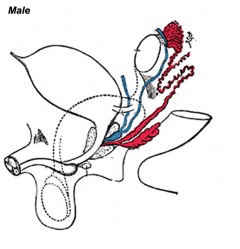
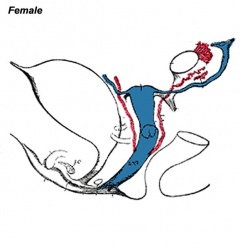
3. Differentiation of internal genital organs and ducts
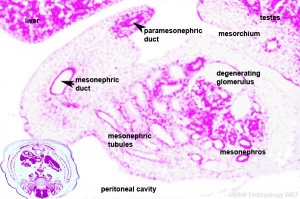
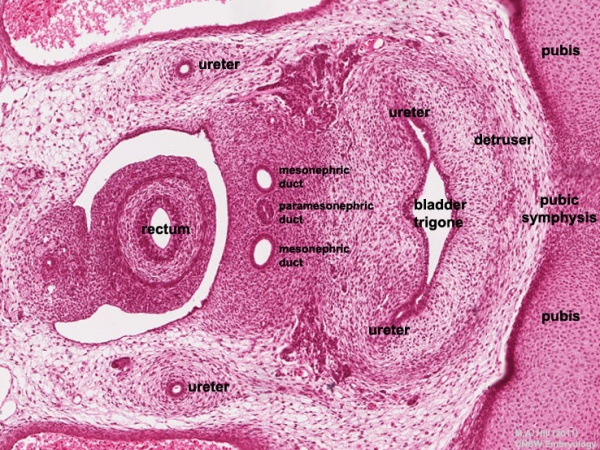
Human embryo (Carnegie stage 22, week 8) pelvic level cross-section.
Male
This looped animation shows the development of the male gonad showing medullary sex cords.
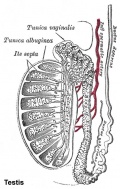
The tunica albuginea (white) covers the testis and bands extend inward to form connective tissue septa. |
|
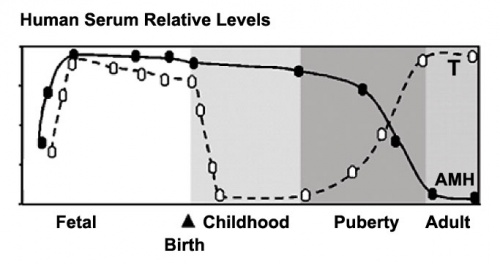
Anti-Mullerian Hormone
Anti-Mullerian hormone (AMH) or Mullerian Inhibiting Substance (MIS) hormone with at least two gonadal related functions:
- In males, it is produced by embryonic Sertoli cells and causes the loss of the paramesonephric (Mullerian) duct system that forms the internal female genital tract.
- In females, it is produced after puberty by follicle cells and suppresses the development of other primary follicles, thus restricting the number of follicles stimulated by FSH.
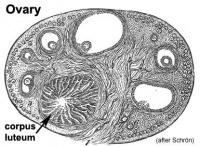
Female
This looped animation shows the development of the female gonad showing cortical sex cords.
|
|
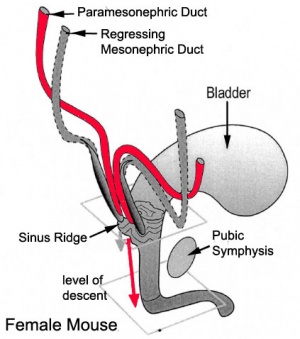
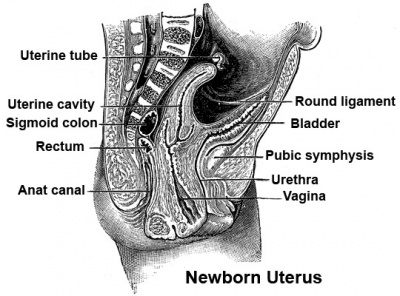
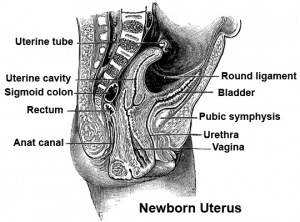
Uterus Development
- Week 7 – duct preservation or regression begins
Paramesonephric duct development
Vagina Development
- The embryonic origin of the vagina has been a historically hotly debated issue with several different contributions and origins described.
- Current molecular studies show the whole vagina is derived from the intermediate mesoderm-derived Paramesonephric (Müllerian) duct[1] (see also earlier review [2])
- bone morphogenic protein 4 (BMP4) reshapes the duct into the vaginal primordium.
- exhibits different features from the uterus
- stratified squamous epithelium
- insensitivity to anti-Müllerian hormone
- Links: Vagina Development
4. Differentiation of External Genitalia
Endocrinology - Diagram of the development of the external genitalia | image |

Genital external development |
Dihydrotestosterone (DHT)
Male presence of DHT
Female absence of DHT
|
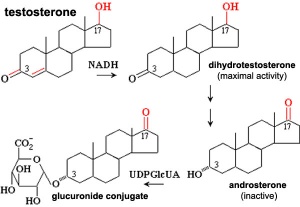
Testosterone metabolism |
Female
This looped animation shows the development of external female genitalia from the indifferent external structure, covering the approximate period of week 9 to 12.
|
|
Note the original cloacal membrane becomes separated into the urogenital membrane and anal membrane. The urogenital folds beneath the genital tubercle remain separate (unfused), forming the inner labia minora and second outer skin folds form the larger labia majora either side of the developing vestibule of the vagina. Note at the top of the animation, the changing relative size of the genital tubercle as it forms the glans of the clitoris. |
Male Genitalia Development
Endocrinology - Box 6.6 The roles of testosterone (T) and 5α-dihydrotestosterone (DHT)
This looped animation shows the development of external male genitalia from the indifferent external structure, covering the approximate period of week 9 to 12.
| Male External |
| Page | Play |
Note the original cloacal membrane becomes separated into the urogenital membrane and anal membrane (identical to female). The urogenital folds beneath the genital tubercle begin to fuse in the midline. The skin folds either side for the scrotum, which too has a midline fusion, the raphe. The scrotal sac is initially empty and is an attachment site for the gubernaculum, descent of the testes begins generally during week 26 and may take several days.
Gonad Descent
- Both kidney and gonads develop retroperitoneally, with the gonads moving into the abdomen or eventually into the scrotal sacs.
- During fetal development the gubernaculum and fetal growth in both male and female, changes the gonads’ relative positions finally reaching their adult locations.
Both female and male gonads undergo anatomical descent.
- Ovaries ‐ undergo caudal and lateral shifts to be suspended in the broad ligament of the uterus, gubernaculum does not shorten, it attaches to paramesonephric ducts, causing medial movement into the pelvis.
- Testes ‐ two anatomical phases in descent, transabdominal and transinguinal, under the influence of the shortening gubernaculum.
|
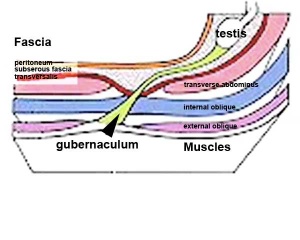 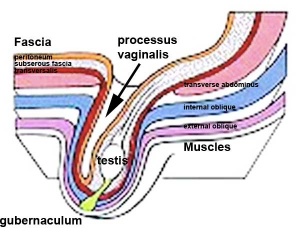
|
The testis (white) lies in the subserous fascia (spotted) a cavity processus vaginalis evaginates into the scrotum, and the gubernaculum (green) attached to the testis shortens drawing it into the scrotal sac. As it descends it passes through the inguinal canal which extends from the deep ring (transversalis fascia) to the superficial ring (external oblique muscle). Descent of the testes into the scrotal sac begins generally during week 26 and may take several days. The animation shows the path of a single testis.
Data from a recent study of male human fetal (between 10 and 35 weeks) gonad position.
- 10 to 23 weeks - (9.45%) had migrated from the abdomen and were situated in the inguinal canal
- 24 to 26 weeks - (57.9%) had migrated from the abdomen
- 27 to 29 weeks - (16.7%) had not descended to the scrotum
Incomplete or failed descent can occur unilaterally or bilaterally, is more common in premature births, and can be completed postnatally.
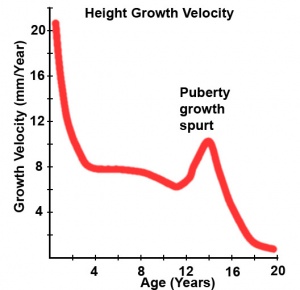
5. Postnatal - Puberty
Puberty can occur over a broad range of time and differently for each sex:
- girls (age 7 to 13)
- boys (age 9 to 15)
The physical characteristics that can be generally measured are: genital stage, pubic hair, axillary hair, menarche, breast, voice change and facial hair.
Male
- testosterone - adult testes produce about 6-10 mg /day in males (~0.5 mg / day in females) carried in circulation by a specific carrier globulin.
- masculinizing androgen - also at puberty, spermatogenesis in males
- development of secondary sexual characteristics - body and facial hair growth (male pattern baldness)
- anabolic effect - metabolism towards conservation of amino acids, promoting protein synthesis, muscle development
- neural - libido in both sexes, male pattern behaviour
- {{Sertoli cell))s (Sustentacular) - produce anti-mullerian hormone (AMH) to puberty.
- AMH - anti-Müllerian hormone (Müllerian inhibiting factor (MIF), Müllerian-inhibiting hormone (MIH), and Müllerian-inhibiting substance (MIS)).
Female
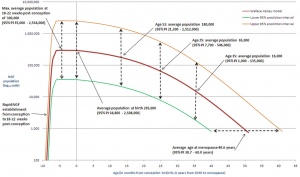
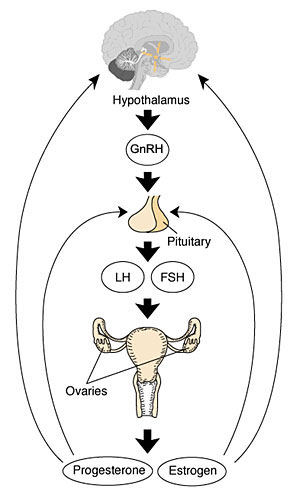
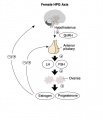
In females, menarche (the first menstruation or a period) usually occurs after the other secondary sex characteristics, and will continue until menopause (permanent cessation of reproductive fertility).
The diagram shows the hormonal regulation pathway from the brain to the ovary and subsequent impact on uterine changes during the menstral cycle.
- GnRH = Gonadotropin-releasing hormone (GnRH). This peptide hormone is a decapeptide (10 amino acids) with a short half life (<15 minutes).
- LH = Luteinizing Hormone
- FSH = Follicle Stimulating Hormone
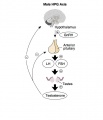
A similar endocrine axis is also found for regulation of the male gonad.
Puberty Abnormalities
- Precocious Puberty - Premature development of the signs of puberty which can occur in both girls (before age 7 or 8) and in boys (before age 9).
- Delayed Puberty - Determined in boys by a lack of increase in testicular volume by the age of 14 years. In girls, no breast development by the age of 13.5 years and a lack of menstruation by the age of 16 years. There can also be a "pubertal arrest" where there is no progress in puberty over 2 year period.
Sex Differences in Adult and Developing Brains
Brains of males and females differ
- in regions specialized for reproduction
- in other regions (controlling cognition, etc) where sex differences are not necessarily expected
- differentially susceptible to neurological and psychiatric disease
2 sources of sexually dimorphic information
- complement of sex chromosome genes
- mix of gonadal hormones
- not known significance of brain sex differences
- transient sex differences in gene expression in developing brains may cause permanent differences in brain structure
Abnormalities
Human genital abnormalities are currently described as "Disorders of Sex Development" (DSD).
| New Terminology | Previous Terminology |
|---|---|
| DSD | Intersex |
| 46,XY DSD | Male pseudohermaphrodite undervirilization of an XY male undermasculinization of an XY male |
| 46,XX DSD | Female pseudohermaphrodite overvirilization of an XX female masculinization of an XX female |
| Ovotesticular DSD | True hermaphrodite |
| 46,XX testicular DSD | XX male or XX sex reversal |
| 46,XY complete gonadal dysgenesis | XY sex reversal |
Include: chromosomal, gonadal dysfunction, tract abnormalities, external genitalia and gonadal descent.
Sex Reversal
- Where chromosomal sex does not match phenotypic sex i.e. XX males or XY females.
XX males - usually caused by a transfer of some Y chromosome DNA onto the X chromosome
- Gonads develop as testes, everything looks normal internally and externally but infertile due to a failure of spermatogenesis
- Similar to Template:Kleinfelter's syndrome (XXY)
XY females - usually steroidal origin
- Main cause is Androgen Insensitivity Syndrome (AIS) Complete (CAIS) Partial (PAIS) and Mild (MAIS) usually caused by mutations of the gene encoding the androgen receptor AR gene located on the X chromosome
- 5-alpha-reductase deficiency - again leads to a lack of complete steroidal induction of external genitalia
- Rare mutations in key sex determining genes including deletion or mutations of SRY
- Links: genital abnormalities
Congenital Adrenal Hyperplasia
- impairment of cortisol production by the adrenal cortex, is one of the most common causes of DSD genitalia at birth.
- depending upon the synthesising enzyme affected a range of genital and hypertensive abnormalities can occur in the 2 sexes.
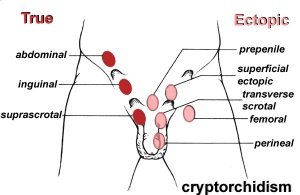
Cryptorchidism
- abnormality of either unilateral or bilateral testicular descent, occurring in up to 30% premature and 3-4% term males.
- descent may complete postnatally in the first year, failure to descend can result in sterility.
Testis descent is thought to have 2 phases:
- transabdominal descent - dependent on insulin-like hormone 3 (INSL3).
- inguinoscrotal descent - dependent on androgens.
Undescended Ovaries
- reasonably rare gonad abnormality, often detected following clinical assessment of fertility problems and may also be associated with other uterine malformations (unicornuate uterus).
- Due to the relative positions of the male (external) and female (internal) gonads and the pathways for their movement, failure of gonad descent is more apparent and common in male cryptorchidism than female undescended ovaries.
Hydrocele
- Male Hydrocele is a fluid-filled cavity of either testis or spermatic cord, where peritoneal fluid passes into a patent processus vaginalis.
- Female Hydrocele is a similar, but rarer, fluid-filled cavity occuring in the female as a pouch of peritoneum extending into the labium majorum (canal of Nuck).
Tract Abnormalities
Many different forms
- uterus: associated with other anomolies, unicornuate uterus
- vagina: agenesis, atresia
- Ductus Deferens: Unilateral or bilateral absence, failure of mesonephric duct to differentiate
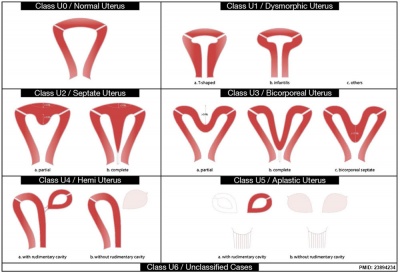
|

|
| Uterine abnormalities | Unicornate uterus |
Uterine Duplication (uterus didelphys, double uterus, uterus didelphis) A rare uterine developmental abnormality where the paramesonephric ducts (Mullerian ducts) completely fail to fuse generating two separate uterus parts each connected to the cervix and having an ovary each.
Septate Uterus
Cervical: cervical agenesis, cervical duplication
Vaginal: Mayer-Rokitansky syndrome (MRK anomaly, Rokitansky-Küster-Hauser syndrome, RKH syndrome, RKH) congenital absence of the vagina, dyspareunia, vaginal agenesis.
External Genitalia - Hypospadias
hypospadias
|
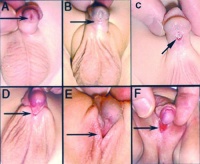
Hypospadia classifications |
Movies
|
|
|
|
|
|
|
|
| Mouse Primordial Germ Cell Migration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||
References
- ↑ Cunha GR, Kurita T, Cao M, Shen J, Robboy S & Baskin L. (2017). Molecular mechanisms of development of the human fetal female reproductive tract. Differentiation , 97, 54-72. PMID: 29053991 DOI.
- ↑ Cai Y. (2009). Revisiting old vaginal topics: conversion of the Müllerian vagina and origin of the "sinus" vagina. Int. J. Dev. Biol. , 53, 925-34. PMID: 19598112 DOI.
Reviews
Roly ZY, Backhouse B, Cutting A, Tan TY, Sinclair AH, Ayers KL, Major AT & Smith CA. (2018). The cell biology and molecular genetics of Müllerian duct development. Wiley Interdiscip Rev Dev Biol , , . PMID: 29350886 DOI.
Kim JH, MacLaughlin DT & Donahoe PK. (2014). Müllerian inhibiting substance/anti-Müllerian hormone: A novel treatment for gynecologic tumors. Obstet Gynecol Sci , 57, 343-57. PMID: 25264524 DOI.
Feingold KR, Anawalt B, Boyce A, Chrousos G, Dungan K, Grossman A, Hershman JM, Kaltsas G, Koch C, Kopp P, Korbonits M, McLachlan R, Morley JE, New M, Perreault L, Purnell J, Rebar R, Singer F, Trence DL, Vinik A, Wilson DP, Rey R, Josso N & Racine C. (2000). Sexual Differentiation. , , . PMID: 25905232
Wilhelm D, Palmer S & Koopman P. (2007). Sex determination and gonadal development in mammals. Physiol. Rev. , 87, 1-28. PMID: 17237341 DOI.
BGDB: Lecture - Gastrointestinal System | Practical - Gastrointestinal System | Lecture - Face and Ear | Practical - Face and Ear | Lecture - Endocrine | Lecture - Sexual Differentiation | Practical - Sexual Differentiation | Tutorial
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology BGD Lecture - Sexual Differentiation. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGD_Lecture_-_Sexual_Differentiation
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G