Testis Development
Introduction
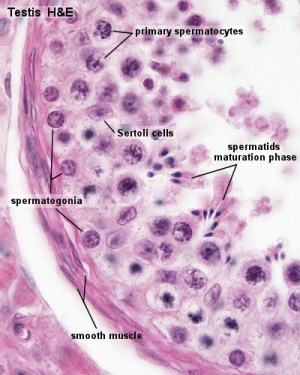
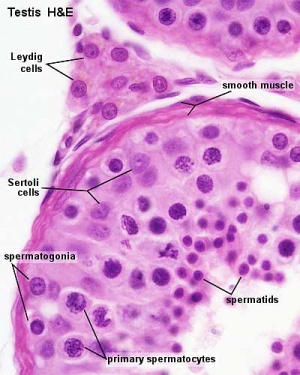
The male gonad is the testis. The initial difference in male and female gonad development are dependent on testis-determining factor (TDF) the protein product of the Y chromosome SRY gene. Recent studies have indicated that additional factors may also be required for full differentiation. Within the developing testis the three main differentiating cell types are: gamete forming cells (spermatogonia), support cells (Sertoli cells) and hormone secreting cells (Leydig or interstitial cells).
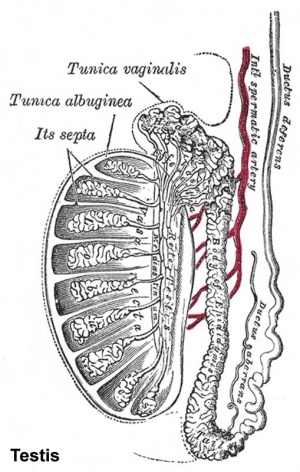
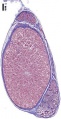
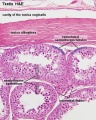
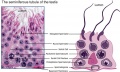
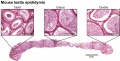
The seminiferous tubules are considered the parenchyma of the testis.
| Y Chromosome | Puberty | original page
Some Recent Findings
|
Development Overview
Sex Determination
- Humans (week 5-6)
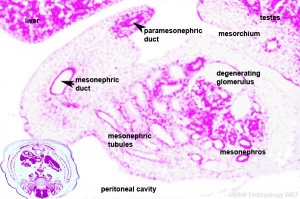
- Germ cells migrate into gonadal ridge
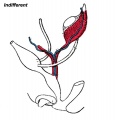
- Gonads (male/female) identical at this stage, Indifferent
Gonad Development
- dependent on sex chromosome
- Y testes
- No Y ovary
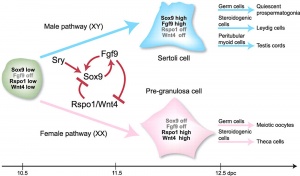
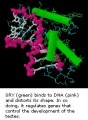
SRY
SRY protein (Testes determining factor, TDF) binds DNA Transcription factor, Bends DNA 70-80 degrees
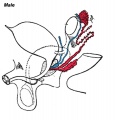
Internal Genital Organs
- All embryos form paired
- Mesonephric duct, see kidney development
- Paramesonephric duct, Humans 7th week Invagination of coelomic epithelium Cord grows and terminates on urogenital sinus
- Male Gonad (testes) secretes Mullerian duct inhibitory factor (MDIF) which causes regression of paramesonephric duct
- Male Gonad (testes) secretes Testosterone which retains mesonephric duct
External Genital Organs
- All embryos initially same (indifferent)
- Testosterone differentiates male
Week 8
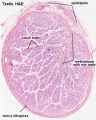
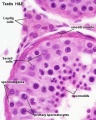
Sertoli Cells
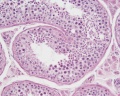
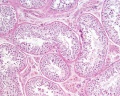
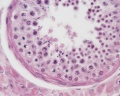
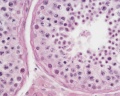
These are the support cells located within the seminiferous tubule. Their differentiation is regulated by the presence of a Y chromosome and in turn regulates Leydig cell differentiation. Sertoli cells direct testis morphogenesis, organizing testis cord formation, establishing testis vasculature and inducing differentiation of peritubular myoid cells and fetal Leydig cells. At puberty the immature Sertoli cells cease to proliferate and differentiate.
Sertoli cell functions include:
- regulation of spermatogenesis through endocrine FSH and testosterone
- regulation of the intratubular and intercellular environment adluminal to the tight junctional complexes
- meiotic and post-meiotic germ cells are sequestered by Sertoli-Sertoli junctional complexes
- generate adluminal compartment isolated from both serum and lymph
- attachment of germ cells through unique intermediate filament (desmosome-like junctions) and microfilament (actin- ectoplasmic specializations, ESs) junctions[4]
- to prevent premature sloughing of immature germ cells from the seminiferous epithelium
- desmosome-like junctions are initially present (up to step 8 spermatids)
- ectoplasmic specializations then replace this junction (in step 8 spermatids)
(see also review[5])
Molecular factors:
- Follicle Stimulating Hormone (FSH) -> Krüppel-like factor 4 (KLF4)
- Krüppel-like factor 4 (KLF4) - zinc finger transcription factor, terminal differentiation of epithelial cells.
- Epidermal Growth Factor (EGF)
- Transforming Growth Factor-beta (TGFbeta)
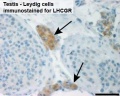
Leydig Cells
Interstitial or Leydig cells, named after german zoologist Franz von Leydig (1821 - 1908).
These cells produce the male testicular androgens and have a role during life prenatally (fetal) and postnatally during puberty onward.
Fetal Leydig Cells
Have a hormonal role in male genitalia differentiation and are lost postnatally. These cells arise approximately at 6 weeks (human) and 12.5 dpc (mouse) and there appears to be differences in hormonal sensitivity between the species. Their initial differentiation requires both luteinizing hormone (LH) and adrenocorticotrophic hormone (ACTH) and therefore normal pituitary development.
Activin A - acts upon Sertoli cells to promote their embryonic proliferation[6]
(More? Endocrine - Pituitary Development)
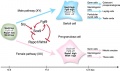
Adult Leydig Cells
Have a hormonal role in puberty, secondary sex characteristics and sexual maturation. Their initial differentiation from peritubular mesenchymal cells does not require gonadotropin, but development and function are dependent upon luteinizing hormone (LH).
The cells differentiate with three discrete stages (newly formed, immature, mature) leading to a decrease in proliferation and increasing testosterone biosynthetic capacity. Insulin-like growth factor I (IGF-I) stimulates proliferation of immature cells and promotes their maturation. Testosterone and estrogen inhibit the process of precursor cell differentiation and may be responsible for the cessation of proliferation in the adult Leydig cells.
Peritubular Myoid Cells
These cells surround the seminiferous tubules and express androgen receptors from fetal life through to adulthood.
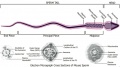
Testis Descent
Testis descent is thought to have 2 phases:
- transabdominal descent - dependent on insulin-like hormone 3 (INSL3).
- inguinoscrotal descent - dependent on androgens.
The regulation of testis descent is still being investigated and several different factors have been identified that may have roles in descent. The first stage of testicular descent occurs 10–15 weeks of gestation with the testes moving to the inguinal region.
The gubernaculum (gubernaculum Hunteri) is the caudal inguinoscrotal ligament that connects the testis to the lower abdomen. The cranial suspensory ligament (mesonephric ligament) is the cranial ligament that connects the tesitis to the posterior abdominal wall.
- Insulin-like factor 3 (INSL3, relaxin-like factor) from fetal leydig cells acting through its receptor (Rxfp2) and BMP and WNT signaling pathways to promote testis descent.
- Calcitonin gene-related peptide (CGRP) from genitofemoral nerve suggested to mediate the inguinoscrotal testicular descent.
- Epidermal growth factor (EGF) may promote by activating the androgen responsive systems.
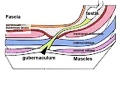
|
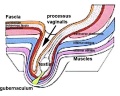
|
|
| Before Descent | End of Descent | Testis Descent Movie |
- Links: OMIM - INSL3
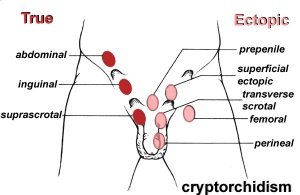
Cryptorchidism
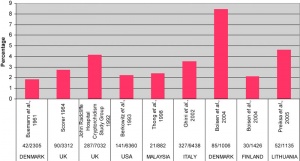
- abnormality of either unilateral or bilateral testicular descent, occurring in up to 30% premature and 3-4% term males.
- Descent may complete postnatally in the first year, failure to descend can result in sterility.
Testis descent is thought to have 2 phases:
- transabdominal descent - dependent on insulin-like hormone 3 (INSL3).
- inguinoscrotal descent - dependent on androgens.
Management of cryptorchidism in children: guidelines.[8] "Cryptorchidism is best diagnosed clinically, and treated by surgical orchiopexy at age 6-12 months, without a routine biopsy. If no testis is palpable, or if other signs of hypovirilisation such as hypospadias are present, the chromosomal sex and hormonal status must be assessed. Laparoscopy is the best way of diagnosing and managing intra-abdominal testes."
Puberty
In humans at puberty, hormonal and morphological changes occur within the gonad and other systems (secondary sex characteristics). Within the testis the immature Sertoli cells cease to proliferate and differentiate. Spermatogonium proliferate and spermatogenesis begins, and it takes about 70 days for cells to mature from the diploid spermatogonium to a primary spermatocyte. This maturation occurs in waves along the seminiferous tubules.
Links: Puberty Development
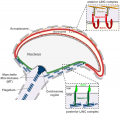
Blood-Testis Barrier
Within the testis seminiferous tubules the Sertoli cells located near the basement membrane act as an initial cellular barrier with many functions, but often described as forming a "blood-testis barrier". (see review[9]
Functions:
- prevent substances reaching the developing spermatozoa (through drug transporters)
- establish a basal and adluminal (apical) compartment (specialized microenvironment)
- provide an immunological privilege status of the testis (anti-sperm antibodies are not developed)
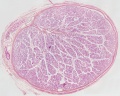
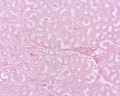
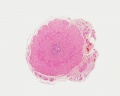
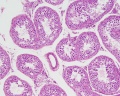
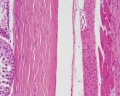
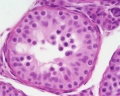
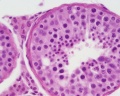
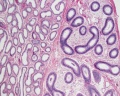
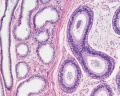
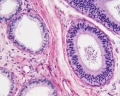
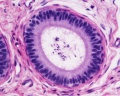
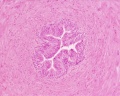
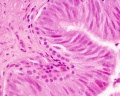
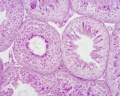
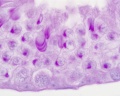
Histology
Molecular
Sry
- Y chromosome gene for a transcription factor
- member of the high mobility group (HMG)-box family of DNA binding proteins
- human - 204 amino acid protein [10]
- Links: Sry
Sox9
- autosomal transcription factor
- Development of XY females - presence of only a single functional copy of the transcription factor encoding genes SOX9, SF1, or WT1 (Note- not all XY humans are sex-reversed if only a single copy of a normal SF1 or WT1 allele is present)
- A nuclear export signal within the high mobility group domain regulates the nucleocytoplasmic translocation of SOX9 during sexual determination[11]
Other roles
- Cartilage - essential for chondrocyte differentiation
- Hearing - otic placode formation, maintenance of progenitors in the otic epithelium
- Links: Sox9 | Cartilage Development | Inner Ear Development
Fog2
- transcription factor, named Friend of Gata2
- human - (8q23) 1,151 amino acid nuclear protein that contains 8 zinc finger motifs[12]
- dosage critical for fetal testis development in mice[13]
- Links: Fog2
Gata4
- transcription factor
- dosage critical for fetal testis development in mice[13]
References
- ↑ <pubmed>20807406</pubmed>| PMC2940779 | BMC Biol.
- ↑ <pubmed>20413907</pubmed>| PDF
- ↑ <pubmed>20040496</pubmed>
- ↑ <pubmed>20403872</pubmed>
- ↑ <pubmed>7711190</pubmed>| Biol Reprod.
- ↑ <pubmed>20498064</pubmed>|http://www.pnas.org/content/107/23/10526.long PNAS]
- ↑ <pubmed>18032558</pubmed>| Hum Reprod Update.
- ↑ <pubmed>18726735</pubmed>
- ↑ <pubmed>21134990</pubmed>| J Endocrinol.
- ↑ <pubmed>8434602</pubmed>
- ↑ <pubmed>12169669</pubmed>| PMC123233 | PNAS
- ↑ <pubmed>10438528</pubmed>
- ↑ 13.0 13.1 <pubmed>17848526</pubmed>PMC1986601 | PNAS
Reviews
Articles
21147849 <pubmed>20664245</pubmed> <pubmed>20610195</pubmed> <pubmed>15379965</pubmed>
Search PubMed
Search Pubmed: Testis Development | Sry | Sox9
Additional Images
External Links
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Genital Links: genital | Lecture - Medicine | Lecture - Science | Lecture Movie | Medicine - Practical | primordial germ cell | meiosis | endocrine gonad | Genital Movies | genital abnormalities | Assisted Reproductive Technology | puberty | Category:Genital
| ||||
|
| Y Chromosome | Week 1 - Spermatogenesis | Ovary | Puberty | original page
Cite this page: Hill, M.A. (2024, June 18) Embryology Testis Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Testis_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G