Ovary Development: Difference between revisions
No edit summary |
|||
| Line 13: | Line 13: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* <ref><pubmed></pubmed></ref> | * '''Mammalian ovary differentiation - A focus on female meiosis'''<ref><pubmed>21964319 </pubmed></ref> "Over the past 50 years, the ovary development has been subject of fewer studies as compare to the male pathway. Nevertheless due to the advancement of genetics, mouse ES cells and the development of genetic models, studies of ovarian differentiation was boosted. This review emphasizes some of new progresses in the research field of the mammalian ovary differentiation that have occurred in recent years with focuses of the period around prophase I of meiosis and of recent roles of small non-RNAs in the ovarian gene expression." | ||
|} | |} | ||
Revision as of 00:17, 10 November 2011
Introduction
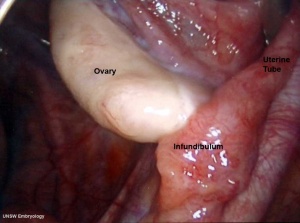
The female gonad is the ovary and is closely associated with female internal genital (reproductive) tract development. In humans, these laterally paired organs lie within the peritoneal cavity. Genes such as Wnt-4 and DAX-1 necessary for initiation of female pathway ovary development, female gonad is not considered a default process.
Initial gonad development in females and males is virtually identical with germ cells migrating into an indifferent gonad. In females with XX, the ovary then begins to develop and the subsequent structure and timecourse of germ cell then differs between males and females. In the ovary oocytes proliferate prior to birth and arrest in meiosis 1.
| Menstrual Cycle | X Chromosome
Some Recent Findings
|
Human Timeline
Approximate Timeline of human development listed below.
24 days - intermediate mesoderm, pronephros primordium
28 days - mesonephros and mesonephric duct
35 days - uteric bud, metanephros, urogenital ridge
42 days - cloacal divison, gonadal primordium (indifferent)
49 days - paramesonephric duct, gonadal differentiation
56 days - paramesonephric duct fusion (female)
100 days - primary follicles (ovary)
Oogenesis
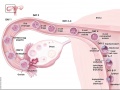
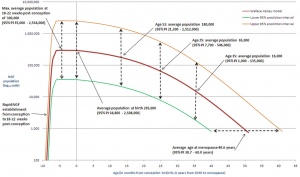
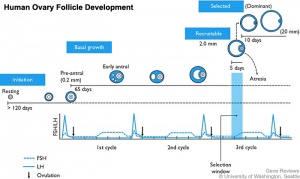
The 2 human ovaries gradually lose follicles both before and after puberty (the beginning of ovulation); beginning with about several million before birth, maximum number at birth, 300-400,000 by puberty and finally by late 40’s have only a few follicles left. Recent studies suggest that the original calculations of ovary follicle numbers at birth were over-estimates and the actual figure should be about 2.5 million.[2] The number of antral follicles detected within the ovary also decreases with increasing materal age.
In humans, a primodial follicle take about 150 days to develop into a preantral follicle (primary) and another 120 days to form an antral follicle (secondary). A number of antral follicles will then "compete" for 14-15 days to become the dominant follicle, which will undergo ovulation.
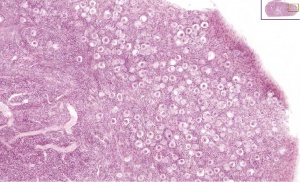
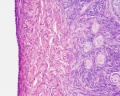
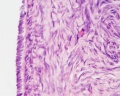
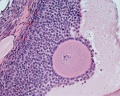
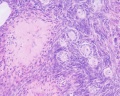
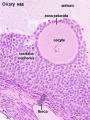
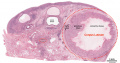
Infant Ovary
This image shows a region (see inset) of the infant ovary cortex.
There are a large number of developing oocytes which will eventually form a dense primordial germ layer at the ovary periphery.
Later stages of follicle development are completely absent and will begin to only appear just prior to puberty.
Postnatal Oogenesis
There is a dogma in mammalian development that new oocyte and follicle production does not occur during postnatal life. There is substantial data that shows human ovarian changes postnatally are loss by apoptosis of prenatal oocytes. A research group (Tilly JL, Johnson J. 2004, 2007) has recently published experiments using mice, showing potentially other sources/sites (bone marrow) of oocyte (putative germ cell) generation. They recently stated that the argument should be based upon "experimental approaches than simply an absence of evidence, especially from gene expression analyses". Several other research groups (Eggan K etal. 2004 and Veitia etal. 2007) have argued against these findings.
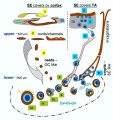
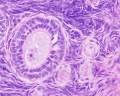
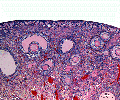
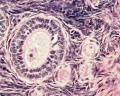
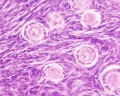
Adult Follicle Structure
A follicle usually contains a single oocyte (egg, ovum, female gamete) and a series of supporting cells and a single fluid-filled space in layers surrounding this cell. The 3 layers below are arranged in layers outward from the oocyte.
Granulosa Cells
- A specific cell type that proliferates in association with the oocyte within the developing follicles of the ovary. These cells form the follicle stratum granulosa and are also given specific names based upon their position within the follicle.
- With development of the antral follicle, there are two populations of granulosa cells with distinct characteristics and functions: mural granulosa cells and cumulus cells.[3]
- mural granulosa cells - an endocrine role by producing steroid hormones and various other ligands
- cumulus cells - play a support role for oocyte development
Alternate Histological Terms
- The membrana granulosa cells sit on the follicular basal lamina and line the antrum as a stratified epitelium. Following ovulation, these granulosa cells contribute to corpus luteum.
- The cumulus oophorus is a column of granulosa cells that attaches the oocyte to the follicle wall. At ovulation, this column of cells is broken or separates to release the oocyte from its follicle attachment.
- The corona radiata are the granulosa cells that directly surround the oocyte, and are released along with it at ovulation. Following ovulation, the corona radiata provide physical protection to the oocyte and are the initial structural barrier that spermatazoa must penetrate during fertilization.
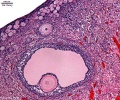
Follicular Fluid
- The antrum is a fluid-filled space in the secondary (antral) follicle
- At ovulation, fluid is released along with the oocyte
- Thought to "carry" the oocyte out of the follicle (like a boat on a wave)
- Aids entry into the uterine tube
Theca Interna
(Greek, thek = box) The ovarian follicle endocrine cells forming the inner layer of the theca folliculi surrounding the developing follicle within the ovary. This vascularized layer of cells respond to leutenizing hormone (LH) synthesizing and secreting androgens (androstendione) transported to glomerulosa cells which process initially into testosterone and then by aromatase into estrogen (estradiol). Theca cells do not begin hormonal functions until puberty.
Theca Externa
(Greek, thek = box) The ovarian follicle stromal cells forming the outer layer of the theca folliculi surrounding the developing follicle within the ovary. Consisting of connective tissue cells, smooth muscle and collagen fibers.
Follicle Classification
There are several different nomenclatures for the stages of follicle maturation. It probably does not matter which naming system you use, as long as you are consistent and use the same set of terminology for all stages. Early stages of follicle development appear to be gonadotropin (Gn) independent and with development become gonadotropin sensitive and then dependent . (UK spelling is gonadotrophin).
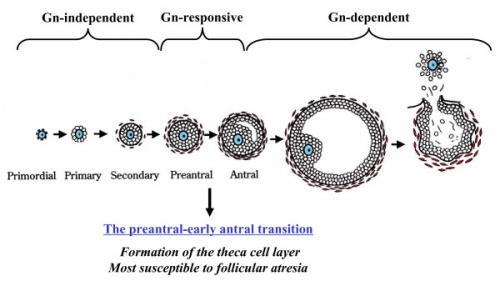
Primordial Follicle
Alternate nomenclature: small follicle or type 1, 2, 3 (25cells)
Primary Follicle
Alternate nomenclature: preantral follicle or type 4 (26-100 cells), type 5 (101-300 cells)
Secondary Follicle
Alternate nomenclature: small and large antral follicle or type 6 (3001-500 cells), type 7 (501-1000 cells)
Preovulatory Follicle
Alternate nomenclature: Graafian follicle or type 8 (>1000 cells)
Atresia
At any one time the majority of follicles are destined not to complete maturation and at any stage (from type 4-7) degeneration of the follicle can occur.
Histology Images
Ovary histology: Tunica Albuginea x20 | Tunica albuginea, Germinal epithelium x40 | Primary follicle, primordial follicle, oocyte, x40 | Secondary follicle, cumulus oophorus, zona pelucida, granulosa cells, oocyte x20 | Corpus luteum, theca lutein cells, granulosa lutein cells, Loupe | Corpus luteum, theca lutein cells, granulosa lutein cells, x10 | Corpus luteum, theca lutein cells, granulosa lutein cells, x40 | Corpus albicans, primary follicle, primordial follicle, granulosa cells, oocyte x20 | Menstrual Cycle | Ovary Development
Follicle Factors
There are both external endocrine factors and internal follicle factors that can influence the development and atresia of ovarian follicles.
External Factors
Leutenizing Hormone (LH)
- from the anterior pituitary
- stimulate the theca interna to synthesize and secrete androgens (androstendione) transported to granulosa cells
- granulosa cells process initially into testosterone and then by aromatase into estrogen (estradiol)
Follicle-stimulating hormone (FSH)
- from the anterior pituitary
- initiates follicle growth through the granulosa cells
- involved in selecting the most advanced (sensitive) follicle to proceed to ovulation
Internal Factors
Oocyte Factors
- Growth Differentiation Factor-9 (GDF-9) - involved in the differentiation of theca cells during this early stage of follicular development OMIM 601918
- Bone morphogenetic protein 15 (BMP15)
- Fibroblast growth factor 8B (FGF8B)
Granulosal Factor(s)
- stimulates the recruitment of theca cells from cortical stromal cells
Thecal Factor(s)
- appear to be several inhibitors of apoptotic cell death
- Epidermal growth factor (EGF)
- Transforming growth factor alpha (TGF-α)
- keratinocyte growth factor (KGF)
- hepatocyte growth factor (HGF)
- Bone morphogenetic protein 7 (BMP-7) also known as osteogenic protein-1 or OP-1
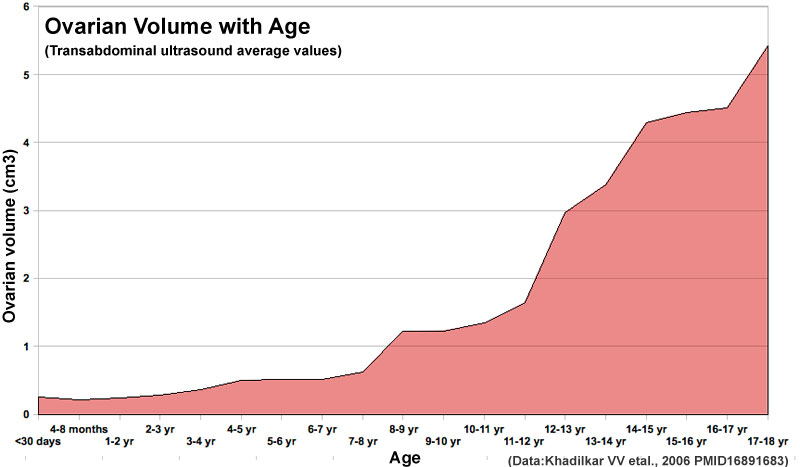
Postnatal Growth
Human ovary postnatal volume growth[6]
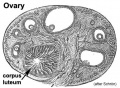
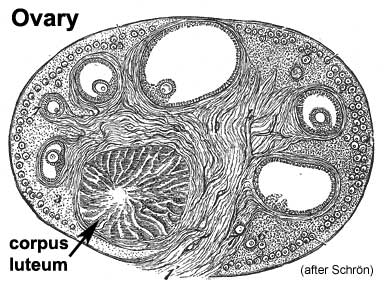
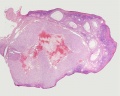
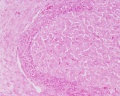
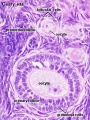
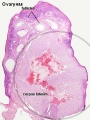
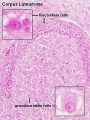
Corpus Luteum
(Latin, corpus = body, luteum = yellow) The remains of ovarian follicle formed after ovulation that acts as an endocrine organ (produce progesterone and oestrogens) supporting pregnancy and preventing menstruation (loss of the endometrial lining). Formed during the luteal phase (secretory phase) of the menstrual cycle by proliferation of both follicular granulosa cells (granulosa lutein cells) and thecal cells (theca lutein cells), which produce progesterone and oestrogens.
Peak luteal function during the menstrual cycle, determined by maximum luteal area, progesterone concentration and estradiol concentration, is observed about 6 days following ovulation. [7]
If fertilization and pregnancy does not occur, the corpus luteum degenerates to form the corpus albicans.
Historically, Regnier de Graaf (1641 – 1673) was the first observer in the ovary of a cow as a yellow structure, the yellow colour was caused by accumulation of steroidal hormones.
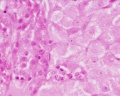
Human corpus luteum (light and electron micrograph)[8]
Corpus Albicans
(Latin, corpus = body, albicans = whitish) The histological structure formed by luteolysis of the corpus luteum in the ovary. If implantation does not occur and the hormone hCG is not released the corpus luteum degenerates and the structure is white, not yellow, because of the absence of steroid hormone synthesis/accumulation.
Histology
- Links: Hematoxylin and Eosin | Histology Stains
References
- ↑ <pubmed>21964319 </pubmed>
- ↑ <pubmed>20111701</pubmed>
- ↑ <pubmed>19755486</pubmed>
- ↑ Oocyte-granulosa-theca cell interactions during preantral follicular development. Orisaka M, Tajima K, Tsang BK, Kotsuji F. J Ovarian Res. 2009 Jul 9;2(1):9. PMID: 19589134 | J Ovarian Res.
- ↑ <pubmed>19538736</pubmed>| PMC2711087 | BMC Dev Biol.
- ↑ <pubmed>16891683</pubmed>
- ↑ <pubmed>15846762</pubmed>
- ↑ <pubmed>16870952</pubmed>| Mol Hum Reprod.
Reviews
- Complementary pathways in mammalian female sex determination. Nef S, Vassalli JD. J Biol. 2009 Sep 2;8(8):74. Review. PMID: 19735582
- On the origin of the maternal age effect in trisomy 21 Down syndrome: the Oocyte Mosaicism Selection model. Hultén MA, Patel S, Jonasson J, Iwarsson E. Reproduction. 2010 Jan;139(1):1-9. Epub . Review. PMID: 19755486
Articles
- The forkhead transcription factor FOXL2 is expressed in somatic cells of the human ovary prior to follicle formation. Duffin K, Bayne RA, Childs AJ, Collins C, Anderson RA. Mol Hum Reprod. 2009 Dec;15(12):771-7. Epub 2009 Aug 25. PMID: 19706741 | Mol Hum Reprod.
Search Pubmed
Search Pubmed: Ovary Development | Follicle Development | Follicle Atresia
Additional Images
- 1471-213X-9-36-3.jpg
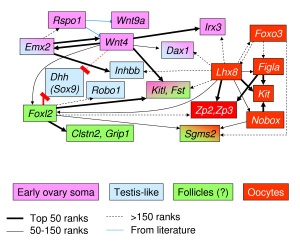
Putative interactions among developmental genes inferred from the comparison of several knockout models of ovarian dysgenesis.
External Links
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Genital Links: genital | Lecture - Medicine | Lecture - Science | Lecture Movie | Medicine - Practical | primordial germ cell | meiosis | endocrine gonad | Genital Movies | genital abnormalities | Assisted Reproductive Technology | puberty | Category:Genital
| ||||
|
| Menstrual Cycle | X Chromosome | Genital System - Female | Oogenesis
Cite this page: Hill, M.A. (2024, June 16) Embryology Ovary Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Ovary_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G

![Human corpus luteum (light and electron micrograph)[8]](/embryology/images/thumb/5/5b/Human_corpus_luteum_-_light-and-electron-micrograph.jpg/120px-Human_corpus_luteum_-_light-and-electron-micrograph.jpg)