Paper - The developing third nerve nucleus in human embryos
| Embryology - 9 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Mann IC. The developing third nerve nucleus in human embryos (1927) J Anat. 61(4): 424-438. PubMed 17104156
| Online Editor | ||
|---|---|---|
| This historic 1927 paper by Ida Mann describes the development of the third cranial nerve nucleus, the oculomotor nucleus (CNIII).
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Developing Third Nerve Nucleus in Human Embryos
Ida C. Mann, M.B., F.R.C.S. Eng.
From the Department of Anatomy, St Mary’s Hospital Medical School (1927)
Work done under a grant from the Medical Research Council.
Introduction
Although a great deal of work has been published on the phylogenetic evolution of the various cell groups commonly considered to form part of the third nerve nucleus in man, there does not appear to have been any exhaustive investigation of their ontogenetic development. The present paper is an attempt to trace the changes of form which the nucleus undergoes, in human embryos, from its earliest appearance until birth. The enquiry has served both to bring out two points of interest with regard to the ontogeny and to add a possible further item of evidence anent the vexed question of the localisation of function in the various cell groups which make up the nucleus. In the first place it becomes obvious that differentiation of shape of the nucleus occurs very much earlier than was previously thought, while one also sees that the ontogenetic changes of form do not correspond in sequence, as is usually stated, with the generally assumed phylogeny. The evidence re localisation of function is concerned with the question of the association of the Edinger-Westphal nucleus with the movements of the pupil and of the nucleus of Perlia with convergence. Before proceeding further in a discussion of the formation of the nucleus
it is necessary to explain the nomenclature to be followed. There are many classifications of the cell groups, some based on anatomical, others on physio- logical observations. The latter are the more elaborate and imply the existence of anatomically separate cell groups corresponding to each of the extrinsic and intrinsic ocular muscles. It must be said at the outset that such cell groups have never been demonstrated conclusively microscopically and that in all probability they do not occur, the localisation of function being of course quite possible without them.
The three best known systems of nomenclature are those of Kappers (comparative), Perlia (anatomical) and Kahler and Pick (physiological). The relation between them can be seen from the following table:
(table to be formatted)
| Kappers | Perlia | Kahler and Pick |
|---|---|---|
| 1. Dorso-lateral
2. Ventro-median 3. Median 4. Accessory |
Levator palp. sup. 1. Dorso-lateral {%gst:erg’;_(§°J::£] {itnefctus superior . _ enor oblique . Anterior ventral Rectus intemus 2' Ve"tr°'med'w’n . Posterior ventral Reotus inferior 3. Median Perlia’s nucleus Convergence Edinger-Westphal Sphincter iridis 4' Accessory lAntero-median lCiliary muscle
The nomenclature of Kappers has the great advantage that it is based on phylogeny and is applicable to a large number of animals and indeed, with modifications, to all the Vertebrates (above the Cyclostomes) so far investigated. Perlia’s system is essentially designed from the point of view of human structure only, while the physiological scheme has little or no anatomical corroboration. The classification of Kappers will therefore be adopted in the following description as having the widest application.
According to Kappers the phylogenesis of the various cell groups is as follows:
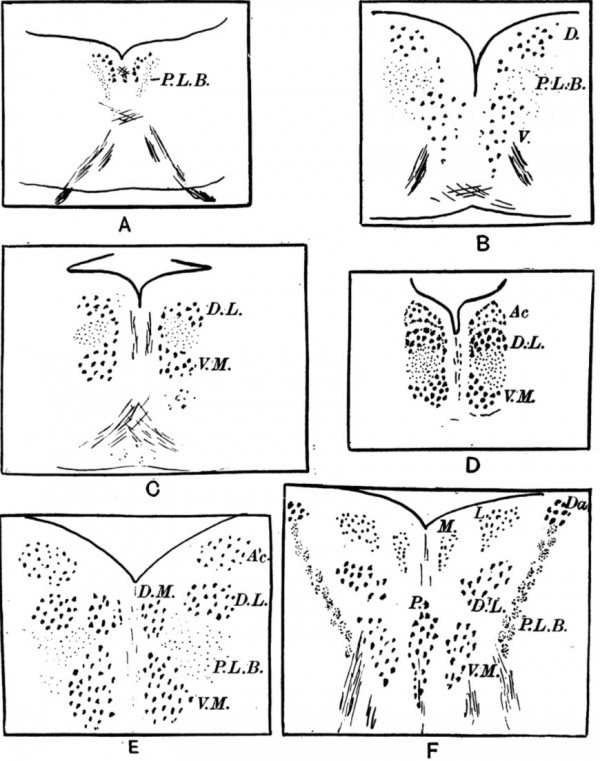
In the Selachian brain the nucleus is represented by a somewhat ill-defined group of cells lying close to the mid-line on each side between the two posterior longitudinal bundles. It is not divided into separate portions, although it is slightly curved in shape, so that the ventral end lies_ nearer the mid-line than the dorsal, with the result that a medial and a dorso-lateral region can be recognised. This is the simplest form of oculo-motor nucleus known (see fig. 1 A).
In Ganoids and Teleosteans is found the first indication of a true separation into two parts. The ventral end of the nucleus has extended medially and downwards so that its lower end is only separated from the base of the brain by the commissura ansulata. It is only joined to the dorsal end of the nucleus by a narrow line of cells, so that in these animals the whole nucleus can be described as consisting of two groups of cells, one, the dorsal group, lying above and slightly outside the other, the ventral group, from which it is separated by some of the fibres of the posterior longitudinal bundle (see fig. 1 B). The ventral group extends further caudally than the dorsal.
Among the Reptiles are found two types of third nerve nucleus, the first resembling in arrangement that of the Ganoids, though much more clearly differentiated, the second showing a third cell group.
The alligator (fig. 1 C) can be taken as an example of the first type. The two cell groups, separated by the posterior longitudinal bundle, are sharply defined and are referred to as dorso-lateral and ventro-median respectively. They end at the same level behind but the dorso-lateral extends further forwards.
The lizard Varanus is of the second type (fig. 1 D). The dorso-lateral and ventro-median groups are clearly marked and, in addition, a third group of smaller cells is present, lying above and extending slightly to the inner side of the dorso-lateral portion. This third group is known as the accessory nucleus. It is not found below the lizards and at its first appearance is characterised by the smallness of its cells.
In Birds (fig. 1 E) the elaboration of the nucleus has progressed much further. The ventro-median group of cells is still present, lying near the middle line, below and internal to the posterior longitudinal bundle. The dorsal group has however divided into two, a dorso-median and a dorso-lateral, lying side by side above the ventro-median group. In addition the accessory nucleus is larger. It lies above the dorso-lateral nucleus.
Fig. 1 A. The third nerve nucleus in a Selachian. P.L.B = posberior longitudinal bundle.
B. The nucleus in Ganoids and Teleosteans. D = dorsa.l nucleus; V = ventrs.l nucleus.
C. The nucleus in the alligator. D.L = dorso-lateral and V.M = ventro-median group of cells.
D. The nucleus in the lizard Varaxnus. Ac =accessory nucleus.
E. The nucleus in the hen. Ac = accessory nucleus; D.M = dorso-median nucleus; D.L = dorso-lateral nucleus; P.L.B : posterior longitudinal bundle; V. M = ventro-median nucleus.
F. The nucleus in man. Daznucleus of Darkschewitsch; M = medial part and L = lateral part of accessory nucleus; D.L = dorso-lateral main nucleus; V.M = ventro-median main nucleus; P = Perlia’s or median nucleus.
In Man the nucleus has reached its greatest known complexity (fig. 1 F). The ventro-median nuclei are present but are separated from each other in the anterior third of the nucleus by a mass of cells lying in the middle line (the median nucleus of Perlia). The dorso-lateral nuclei lie above the ventro- median and above them again lies the small-celled accessory nucleus, now divided into a medial and a lateral portion. The lateral portion is the nucleus of Edinger-\\'estpha.l, the medial portion is sometimes known as the antero-median nucleus. As they are both composed of the same type of cell it is better to consider them both as parts of the accessory nucleus. Yet another cell group, the nucleus of Darksehewitsch, lying above the posterior longi- tudinal bundle outside the accessory nucleus, is sometimes considered, but not by Kappers, as part of the third nucleus in man.
Such is the usually accepted phylogenetic history of the third nerve nucleus. Of its ontogeny little is known. Kappers states that the last group to appear in phylogeny, namely the accessory or Edinger-\Vestphal nucleus, is also the last to differentiate in the human foetus. According to both Kappcrs and Tsuchida a four to five months human foetus shows well-developed dorso- lateral and Ventro-median cell groups but no median or accessory nuclei. At seven months, accor(ling to Kappers, the median nucleus is present and the accessory nucleus (lateral part) just beginning. Tsuchida states that the median and accessory nuclei do not appear until after the seventh month.
Tsuchida’s work is open to one criticism. He confined his observations to material in the latter half of foetal life only, when the cells are always difficult to see, those of the accessory nucleus being particularly so, since they usually fail to take the stain. It is probable that had he examined earlier material he would have found that differentiation actually occurs and is easily visible at a much earlier date than he assigned to it.
The present series of specimens begins with a human embryo of 25 mm. I am aware that the position of the nucleus can be recognised by downgrowth of neuroblasts in the corresponding neuromere at a much earlier age, but it is only at about 25 mm that these cells form a well-defined group. In the 25 mm. embryo examined, on either side of the median line of the floor of the aqueduct of Sylvius, can be seen a group of closely aggregated cells, still directly continuous with the ependymal lining of the canal and not differentiated. The area is indelinite in extent, fading into the surrounding brain substance, but in its most marked portion the grouping is quite definite and has the appearance seen in fig. 2.
At no part of the nucleus is there any trace of a connection of these cell groups across the mid-line.
This stage, in which the nucleus consists of two single groups of cells lying beneath the floor of the aqueduct of Sylvius on either side of the middle line and having no median connection, corresponds with the condition of the adult nucleus in Selachians described by Kappers.
Fig. 2. Horizontal section through third nerve nucleus in 25 mm human embryo. The orientation of the brain is similar to that in fig. 3. a = aqueduct of Sylvius; b = nucleus .
Fig. 3. Reconstruction of median sagittal section of brain of 35 mm human embryo (after Frazer). a=nucleus. The line ad indicates the plane of section.
1 This and the succeeding figures are reproduced by kind permission of the Ophthalmological Society of the United Kingdom.
The next stage to be described in the development of the nucleus is that found in a 35 mm. embryo, the appearance of whose brain is shown in fig. 3.
Fig. 4: is a drawing of a horizontal section through the nucleus which can be seen in fig. 3. It will be seen by comparison with fig. 2 that a marked change has occurred in the nucleus. The cells composing it now show the characters of differentiating neuroblasts. They are larger, have more clear protoplasm surrounding the nucleus, and the chromatin net shows a more open mesh than in the previous stage. This change renders the nucleus much more sharply
Fig. 4. Horizontal section of third nerve nucleus of 35 mm. human embryo , along the line cd in fig. 3. a. = median nucleus of Perlia.
defined from the surrounding brain tissue and allows one to make out its limits with much more certainty. It will be seen that a new element has been introduced, namely, an exactly median group of cells, which connects the two lateral portions seen in fig. 2. This is the first appearance of Perlia’s median group of cells. It seems to have been formed by an extension and coalescence across the middle line of the two lateral groups seen in the 25 mm. stage, since, at its earliest appearance, it is continuous with them, and in its later stages appears successively more distinct and separate. On either side of the main lateral cell groups and external to them can be seen two other small cell groups. These are more marked in sections higher than the one shown, and can be recognised extending upwards (dorsalwards) above the upper surface of the larger cell groups. They form a narrow column of cells apparently extending iii an upward and outward (dorso-lateral) direction from the main lateral masses, and, like the central mass, are connected with and apparently arise from them. Their upper limits a.re very ill defined. Their extent and relations are shown in figs. 5 and 6.
Fig. 5 shows a reconstruction of the nucleus seen from above. It will be noted that three definite cell groups can now be recognised, though they are at this stage all connected. A median group is connected on either side with a large group of cells from which a narrow column is growing upwards and outwards in a dorso-lateral direction. It will be seen that the median nucleus extends beyond the lateral groups posteriorly, but does not come up as far as their anterior ends. The dorso-lateral groups are anterior to the centre of the whole nucleus. We can now, therefore, recognise the three main groups described by Kappers—a ventro-median lying alongside the middle line, a dorso-lateral extending upwards outside this, and a median uniting the two ventro-median groups. We have seen that in phylogeny the first definite indication of a division into a dorso-lateral and a ventro-median group is found in the Ganoids and Teleosteans. In them the ventro-median group is the larger, and lies in relation with the lower part of the side wall of the cleft at the bottom of the aqueduct. It is separated from the dorso-lateral part by the fibres of the posterior longitudinal bundle. The dorso-lateral part does not extend so far postcriorly as the Ventro-median. This arrangement is similar to that found in the 35 mm. embryo. except that in the human subject a median nucleus is present connecting the two ventro-median portions. The median nucleus of the 35 mm human embryo is a simple connection and shows no forward prolongation as it does later. There is so far no trace of any cell group lying alongside the mid-line in front of the central connection and internal to the dorso-lateral and ventro-median groups, i.e. in the situation of the accessory nucleus.
Fig. 5. Reconstruction of third nerve nucleus of 35 mm. human embryo seen from above.
Fig. 6. Reconstruction of third nerve nucleus of 35 mm. human embryo seen from the left side. Black portion = median nucleus; lined portion = ventro-median cell group.
Fig. 7. Reconstruction of third nerve nucleus of 48 mm. human embryo seen from above. Black portion = median nucleus; lined portion = ventro-median cell group; dotted portion = accessory nucleus. Dorso-lateral nucleus not shaded.
The next stage for consideration is the 4-8 mm. In the specimen I examined the various cell groups showed a uniform staining and the cells were all similar in appearance. The cell groups were easy to recognise as the specimen was darkly stained. and at this early stage the histological structure of the central nervous system is always more clearly seen than in older specimens. This probably explains the failure of Tsuchida to find the Edinger-\Vestphal nucleus in a live mouths’ foetus, since the nucleus at its first appearance resembles the rest of the nucleus of the third, while, when it differentiates it becomes fainter and fainter and the cells further apart, so that it is less easy to see in the adult than in the 48 mm. embryo. The following changes (see figs. 7, 8 and 9) have occurred: the dorso-lateral group has extended antero- posteriorly and now equals the ventro-median in length. It still retains its continuity with the ventro-median group at its anterior and posterior ex- tremities, but in its middle part it is separate. The anterior part of the ventra- medizm group is on a slightly higher level than the posterior (i.e. nearer the level of the floor of the aqueduct).
Fig. 8. Reconstruction of third nerve nucleus of 48 mm. human embryo seen from the left side. For explanation of parts see previous figure.
Fig. 9. Transverse section through third nerve nucleus of 48 mm. human embryo. a = dorso-lateral cell group; b =accessory cell group; c = ventro-median cell group; d = median cell group.
The median nucleus has lost its continuity with the ventro-median group. It is actually larger, but not relatively so, since the ventro-median group has extended more rapidly, and now reaches further back than the median nucleus—the reverse of the 35 mm stage. The most marked change, however, is in the form of the median group. This shows two largeioutgrowths, one anterior and one posterior. They will be referred to as the anterior and posterior portions of the accessory nucleus. The anterior has a blunt rounded end which extends forwards and lies beside the mid-line, internal to and above the anterior division of the ventro-median group. The posterior is shorter and passes a little backwards from the median nucleus and then extends in two thin processes upwards and downwards, the upper one lying internal to the posterior part of the dorso-lateral group.
Fig. 7 shows the appearance from above, fig. 8 from the side. Fig. 9 shows a transverse section through the middle of the third nerve nucleus.
The four cell groups recognised by Kappers are now present though they are as yet interconnected. The dorso-lateral group is connected in parts with the ventro-niedian, while the accessory nucleus is present as an outgrowth from the median nucleus and shows no distinctive characters, being only recognis- able from its position.
A further differentiation has occurred in the 75 mm. foetus next examined. In the first place the dorso-lateral cell group has completely separated from the ventro-median. It lies above it and extends slightly in front of it (figs. 10, 11 and 12).
The median nucleus has also grown forwards in the mid-line and ends in a sharp point. This has been described by Perlia and has been seen in anthro- poid apes, but is not present in the lower forms.
The accessory nucleus extends further forwards, curving towards the middle line, so that, just in front of the median nucleus, and also just short of its anterior end, there is a small area where it meets its fellow of the opposite side. This joining of the anterior portions of the accessory nuclei across the middle line is of interest, as, though it only occurs as a secondary and transient stage in man, it has been decribed as occurring in the adult chimpanzee (Brouwer), while in certain animals which possess an accessory nucleus, but no median nucleus, the accessory nuclei are fused (c.g. Rodents, Edentates, Marsupials).
Fig. 10. Reconstruction of right half of third nerve nucleus of 75 mm. human foetus, seen from above. Shading as before. The more finely dotted portion represents the differentiated portion of the accessory nucleus.
The posterior portion of the accessory nucleus still blends with the median nucleus and extends backwards from this, dividing into an upper and a lower tongue. These are not so long nor so marked as before (at 48 mm.). The most important change, however, at this stage is that the cells lying on the upper aspect of the middle portion of the accessory nucleus (and presumably part of this) are now beginning to take on the adult characteristics of the cells of the Edinger-Westphal and antero-median nuclei, namely, they are smaller and the nucleus appears less deeply stained. Therefore at this stage a definite Edinger-Westphal nucleus of the adult type can be seen, though the mass of undifferentiated neuroblasts which form it had already budded out from the median nucleus by the 48 mm. stage (see figs. 10, 11, 12).
Fig. 11. Third nerve nucleus of 75 mm. human foetus seen from left side.
Fig. 12. Transverse section through third nerve nucleus of 75 mm. human foetus passing through anterior end of nucleus. a:nuelcus of Darkschewitsch; b - nucleus of Edinger—Westphal; c = nucleus ruber.
In this embryo three cell groups can be recognised lying dorso—latera_l again to the dorso-lateral nucleus, in the positions indicated in figs. 10 and 11. The anterior one can be recognised as the nucleus of Darkschewitsch (a in fig. 12), the middle one is probably the interstitial nucleus of Cajal, which in the adult is almost continuous with Darkschewitsch, lying among the fibres of the posterior longitudinal bundle immediately posterior to it. The posterior one cannot with cer- tainty be named at present. These three cell groups are all interconnected, and appear rather in the nature of localised thickenings in a continuous cell column than as three perfectly separated nuclei.
In a 100 mm. foetus the main differences from the 75 mm. stage are in the direction of greater separation of the cell groups from each other. The dorso-lateral and ventro-median groups are distinct as in the 75 mm. specimen, and occupy the same positions.
The median nucleus with its anterior prolongation has almost completely sepa- rated from the accessory nucleus.
The accessory nucleus has now divided into two, an antero-median group corresponding to the portion which was fused across the mid-line in the 75 mm., and a posterior or nucleus of Edinger-Westphal. The antero-median group no longer meets its fellow in the mid-line. It does not yet show the differentiation into small cells.
Fig. 13. Reconstruction of third nerve nucleus (rightside) of 100 mm human foetus seen from above.
The posterior part or Edinger-Westphal nucleus is only connected by a few scattered cells with the median nucleus. The cells on its upper and inner side are small and faintly stained. The upper prolongation or tongue can just be distinguished in a lateral view (fig. 14). The main trend of the nucleus, however, is in a curve concave towards the median nucleus, and with a sort of tail passing downwards and backwards in front of and internal to the ventro-median nuclei and behind and external to the median. This corresponds to the situation of the Edinger-Westphal nucleus in Perlia’s well-known diagram.
Fig. 14. Third nerve nucleus of 100 mm. human foetus seen from left side.
Fig. 15. Transverse section through third nerve nucleus of four months’ human foetus (100 mm.). a = Edinger-Westphal nucleus; b = dorso-lateral part of main nucleus; c = ventro-median part of main nucleus.
As in the 75 mm. embryo, the cell column with three thickenings, of which the anterior one is the nucleus of Darkschewitsch, can be seen dorso-lateral to the main nucleus.
Fig. 16. Transirerse section through third nerve nucleus ofpfull term foetus. a =Ed.inger-Westphal nucleus; b = main nucleus; c =posterior longitudinal bundle; d =nucleus ruber.
Fig. 15 is a section through the Edinger-Westphal portion of the accessory nucleus just in front of the median nucleus, showing the differentiation of the upper part of it.
Fig. 16 is a section through the Edinger-Westphal nucleus in a full term brain showing the differentiation now complete.
From the examination of the foregoing stages it is obvious that the onto- genetic development does not follow the assumed phylogeny. It differs from this in one important particular, namely, the appearance of the median nucleus before the accessory. The most probable explanation for this apparent discrepancy may be found in a consideration of thefunction of the various nuclei. It has been suggested by many observers (Hensen, Volkers, Kahler and Pick, Bruce, Westphal, Brouwer) that the accessory nucleus is concerned with the innervation of the sphincter iridis and the median nucleus with the act of convergence. If this is the case the reversal of the appearances of these two nuclei in human ontogeny may be simply an expression of the correlation of the development of an organ with the development of its nerve supply. Suppose that the median nucleus is concerned with convergence. It appears between 25 and 35 mm. and correlated with this is the fact that the maximum alteration of the optic axes in human embryos (i.e. from 120° to 72°) has also occurred by the 40 mm. stage. The peripheral mechanism of convergence is therefore developing pari passu with the nucleus and both are completed together. The peripheral mechanism for the light reaction does not however begin to develop until after the maximum alteration in the optic axes has already occurred. The forward growth of the margin of the optic cup to form the ectodermal portion of the iris from which the sphincter pupillae differen- tiates does not commence until the 48 mm stage, at which time, as we have seen, an undifferentiated outgrowth from the main mass of the third nerve nucleus can be seen in the situation of the future nucleus accessorius. The cells forming this outgrowth differentiate slowly so that by the fifth month it is showing adult characters over its major portion. At the same time the sphincter pupillae has differentiated from the margin of the optic cup, though it is not yet vascularised.
There is therefore the possibility that this apparent reversal in order of development of the various parts of the nucleus in human embryos may be correlated with the late development of the sphincter pupillae, and may therefore constitute a further point in the evidence in favour of the connection of the accessory nucleus with the intrinsic eye muscles.
References
BROUWER, B. (1918). “K1inisch-anatomjsche Untersuchungen fiber den Oculomotoriuskern.” Zeitach. f. d. gesell. Neurologie 11.. Psychiatric, Bd. XL, S. 152.
BRUCE, A. (1889). Proc. Roy. Soc. Edin. vol. XVII, p. 168.
HENSEN and Vommns (1898). Graefe’s Archiv, Bd. xxrv, S. 1.
KAHLER and PICK (1881). Prag. Zeitschr. fiir Augenheillc. Bd. II, S. 30.
KAPPERS, A. (1920). Vergleichende Amztomie des N erven.system.s. Haarlem.
PATON, L. and MANN, I. C. (1925). Trans. Ophth. Soc. vol. xLv.
TSUCBIDA (1906). “Uber die Ursprungskerne der Augenbewegungsnerven.‘ Hirnanatomischen I nstitut in Ziirich, Heft 2, Wiesbaden.
Cite this page: Hill, M.A. (2026, April 9) Embryology Paper - The developing third nerve nucleus in human embryos. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_developing_third_nerve_nucleus_in_human_embryos
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G