Paper - A Human Embryo with Seven Pairs of Somites Measuring about 2 mm in Length
| Embryology - 28 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Dandy WE. A human embryo with seven pairs of somites measuring about 2 mm in length. (1910) Amer. J Anat. 10: 85-109.
| Online Editor |
|---|
| Carnegie stage 10 | Carnegie No. 391 | Week 4 | 8 somites | Carnegie Collection Contributions to Embryology
Described here by first by Dandy WE. (1910). The apparent lack of fusion of the neural folds described by Dandy is an artifact produced by a crack. There are no camera drawings nor photographs of the intact specimen, and therefore the model reconstructions may not be entirely satisfactory. Described again by Bartelmez and Evans (1926) with additional illustrations (Bartelmez, G. W., and Evans, H. M. 1926. Development of the human embryo during the period of somite formation, including embryos with 2 to 16 pairs of somites. Carnegie Instn. Wash. Publ. 362, Contrib. Embryol, 17, 1-67). Plaster models now at the Carnegie laboratory were made by O. Heard under the supervision of Bartelmez for the 1926 paper. For a detailed description of the stage 10 human embryos also see the historic papers:
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
A Human Embryo with Seven Pairs of Somites Measuring about 2 mm in Length
By
Walter E. Dandy.
From The Anatomical Laboratory, Johns Hopkins University.
With 6 Plates.
Among the youngest human embryos and one of the youngest in Professor Mall’s collection, Embryo No. 391, which he has very kindly permitted me to reconstruct and describe. In general development this embryo is almost identical with, probably a trifle older than, the Kroenier-Pfannestiol embryo, Klb, which measures 1.8 mm. and has six ‘pairs of somites. It is older than E’rernod’s embryo measuring 1.3 mm., Grraf Spee’ s Gle 1.54 mm., and Embryo Frassi 1.17 mm., in the order named. It is younger than the embryos of Unger and Bulle, of 9 and 14 somites respectively, Eternod’s 2.1 mm. embryo, and embryos XLIV (Eff), LXVIII (Lg. 2.15 mm.), VI (B. R. 2.2 mm.) and VII (E 2.1 mm.) of His.
This very rare specimen came into Professor Mall’s possession through the kindness of Dr. R. W. Pearce, of Albany, New York, with the following history from the physician who handed the specimen to him. “The woman had passed her period about two weeks when she performed an abortion with a stick about 8 inches long, which she whittled out for the purpose. This she passed into the uterus and 24 hours later this specimen was aborted. Her purpose in calling me was to see if her object had been attained. I have kept the specimen two years in a bottle of weak formaldehyde.”
Upon receipt the following measurements were made by Professor Mall; ovum 162: 14x12 mm., embryo about 2 mm. The specimen was placed in fresh formalin, stained with alum-cochineal-eosin, imbedded in paraffin and cut into serial sections 10 microns in thickness.
From these sections (163 in number, excluding the bauehstiel), the embryo was reconstructed upon a scale of 200 magnification, giving the model a total length of 32.6 cm. A shrinkage space between the layers of tissue aided materially in the reconstruction without sacrificing tissue. The model is so constructed that every detail may be exposed by cuts and windows.
Upon presentation of an embryo the question naturally arises, is it normal? The history of a mechanical abortion points very strongly toward a normal embryo. It compares very closely with the few other known embryos of this period. The tissues, including the chorion, are excellently preserved and show no degenerative changes.
Age
From the history that the menstrual period passed about two weeks, the age of 13-14 days would fit very nicely into the conventional Reichert—His theory of fertilization and age of embryos. Recent observations however by Mall in 1000 cases with menstrual history have shown that we underestimate the age of young embryos, on an average about 10 days, that fertilization is not restricted to the period immediately before the menstrual period, but may occur at any time in the intermenstrual period. Bryce and Teacher also came to the same conclusions and illustrate this in a well constructed diagram of the menstrual cycle. This obviates the remarkable distortion which has been necessary to harmonize age with size and progress of development, and makes the human embryo much less precocious in its development. In conformity to the above observations the true age of this embryo is probably about 24 days, which would make the time of fertilization occur about ten days before the time for onset of lapsed menstrual period or about eighteen days after beginning of the last menstrual period.
The Adnexa
Chorion. The chorionic membrane is about 0.1 mm. thick, and is covered with many branching villi varying in size up to 1.25 mm. in length and 0.1 mm, in thickness. These villi are more numerous at the point of attachment of the bauchstiel and gradually fade away on all sides until finally a clear zone results from their absence on the opposite pole.
The chorionie membrane and villi have the characteristic inner loose mesenchymatous layer of beautiful branching spindle and stellate cells with anastomosing processes and a somewhat jelly-like inter-eellular substance; an external double epithelial layer, consisting of the inner Langhans layer of small cells with lightly staining nuclei and cytoplasm, and the outer syneytial layer which stains more deeply with eosin and has larger and more densely staining nuclei, but no definite cell boundaries. From the epithelial layer of the chorionic membrane and villi, numerous buds develop, some from the syneytial layer alone, others from both layers of epithelium. These represent proliferation and new formation of villi. The mesenchymatous layer of the ehorionic membrane contains many newly forming capillaries, some of which extend into the villi. The details of these will be considered later in the description of the vascular system.
The Bauchstiel. The bauchstiel does not diiier from that of other young embryos. It consists of loose mesodermal tissue, lined externally by a single layer of flattened mesodermal cells. It is continuous distally with the chorionic mesoderm and proximally with the mesoderm of the umbilical vesicle, amnion and body of the embryo. It contains the allantois, umbilical arteries and veins and their resulting sinuses and branches.
Umbilical vesicle. Tl1e yolk sac has no stalk but is attached to the entire length and breadth of the embryo, with the exception of small portions of the anterior and posterior ends, which are covered by the short head and tail folds of amnion. At its attachment to the embryo, the walls are very thin, consisting of two layers of flattened cells—mesoderm and entoderm. These walls gradually grow thicker distally, due to the degree of development of‘ the blood islands, which also cause a great distortion and knotty appearance of the mesodermal surface. This vascular development extends throughout the Whole length of the umbilical vesicle, but is practically limited to the ventral or distal half, only a few islands being seen in the dorsal half. The greatest development seems to be near the center of the vesicle and is apparently developing to connect with the future vitelline veins. This is in eontradistinction to the findings in Eternod’s 1.3 mm. embryo, in which the posterior region of the umbilical vesicle is drained by the sinus ensiforme. The entodermal lining of the inner surface of the umbilical vesicle forms a distinct layer in places, whereas in other locations, especially in the ventral half, no definite layer of cells can be made out, so intimately is it fused with the knotty and thickened mesoderm.
Amnion. - The amnion is a completely closed cavity, with very thin delicate Walls of flattened cells, the individuality of ec-toderm and mesoderm being everywhere beautifully preserved. The cavity of the amnion is very large, probably due, as suggested by Professor Mall, to the osmosis of large quantities of dilute formalin in which it was preserved for over two years. Anteriorly a rather deep pocket of amnion dips ventral to the heart, evidently a sign of posterior extension of the head fold. The ta.il told is very small, covering the embryo for a distance of only 5 or 6 sections.
General Appearance on the Embryo
The embryo presents an anterior and posterior elevation with a marked dorsal link. (Plate IV.) The anterior elevation is gradual. the posterior very sharp, rising at an angle of about 80”. This kink seems to be partly natural and partly ar1’exaggerated postmortem condition. We should naturally expect a dorsal concavity due to the greater development of the structures in both the anterior and posterior regions of the embryo. The marked aceentuation however may be due, as suggested by Professor Mall, to the large amnion filled with fluid, the weight of which would naturally act upon the point of least resistance. Shrinkage incident to manipulation and imbedding also plays an important role in its aggravation, especially after location of the point of least resistance. This is clearly shown by a comparison of the model with a sketch of the embryo before imbedding, there being an accentuation of the kink by almost 15°.
Ectoderm
Nervous system. The extent of development of the nervous system is a medullary groove, open throughout its entire length. The brain region is divided into three primary Vesicles (Plate VI), the anterior being equal in size to the other two combined. The first or anterior vesicle is long, wide and deep with edges everted and projecting outward over the anterior and lateral Walls of the anterior body elevation. The walls of the second and third vesicles have a tendency toward inversion and the enclosed vesicles are more Sharply defined laterally than the first. The brain passes insensibly into the spinal cord, which is much smaller in all diameters, but is nowhere closed. Posteriorly the medullary groove forms a shallow dilatation gradually fading into the flattened surface of the primitive streak. (Plate IV.) M
No neurcnteric canal is visible, though present in the younger embryos of Frassi, Graf Spec and Eternod and possibly the Kroemer-Pfannenstiel Klb. No traces of spinal or cerebral ganglia or nerves are visible. N o suggestion of anlage of the lens, optic or otio vesicles couldloe detected.
Primitive streak. Posterior-ly and dorsally in the region of the termination of the notochord and neural groove, ectoderm and mesoderm gradually losing their individual morphological characteristics, fuse to form a mass of a single variety of simple undifferentiated cells, which extends to the posterior termination of the embryo. This is the primitive streak (Fig. 7 The entoderm also seems to be a part of this mass since its cells are directly continuous with it, although the charac-tcristic lining of the hinclgut is still maintained throughout. There is no groove except a very shallow flattened
dorsal depression which is gradually lost posteriorly and which is continuous anteriorly with the neural groove. This primitive streak region suggests a storehouse of simple undiiferentiated cells supplying mesoderm, ectoderm and entoderm in the earlier stages, later becoming difierentiated into the characteristic morphology and arrangement of the different layers.
Entoderm
Entoderm lines the ventral surface of embryo, ‘fore and hind gut, allantois and the inner surface of the umbilical vesicle. In the ventral entoderm of the embryo is a median longitudinal groove corresponding to the location of the notochord and caused by its adherence to the ectoderm of the medullary groove. On either side of this groove and parallel to it is a ridge caused by ventral projection of the dorsal aortas. These are no doubt accentuated by postmortem shrinkage. The opening of the foregut is straddled by two prominent ridges which unite above into a large ventral bulging, represented by the overlying coelom, and pericardial cavity with the enclosed heart, respectively.
The foregut is present in 32 sections, representing a length of 320 microns. lt ends blindly anteriorly and is separated by mesoderm from the buccal cavity which is forming by an invagination of ectoderm. There is however no bucco—pharyngeal membrane. One small pharyngeal pouch is present on each side below the first and only complete aortic arch. These pouches are continuous ventrally and unite to form a ventral pouch. There is no contact with the outer ectoderm, although the mesoderm separating them is considerably thinner than elsewhere.
The hindgut is a blind somewhat oval, dilated pouch, 120 microns
in length by sections, but on account of the dorsal kink of the embryo
and the consequent partially longitudinal plane of the sections, the
actual length is somewhat greater. The allantois arises from the
ventral surface of the hindgut. It passes from the embryo into the
bauchstiel in company with and between the umbilical arteries.
After the union of these arteries, it takes its position between the
arterial and venous sinuses (Plate VI) and bends at almost a right
angle to conform to the direction of these sinuses and the bauchstiel.
At this latter point it divides into a short stub measuring about 40
microns and a much longer branch. The terminus of each branch
may be seen in Figs. 8 and 9. The allantois is lined by a single
layer of cubical epithelium; it maintains a lumen to the point of
division, from which it consists of a solid core of cells.
No liver or thyroid anlage has made its appearance. No cloacal or bucco-pharyngeal membranes can be distinguished.
Notochord. With the exception of the Very posterior tip which is entirely free for a distance of three sections, the notochord is everywhere fused with and apparently an integral part of the entoderm. Posteriorly it is Very conspicuous as a relatively large and very compact knob of deeply staining cells on the dorsal wall of the hindgut. This sharply differentiated mass invaginates into the ectoderm, with which is it not here united (Fig. 6). It gradually becomes smaller anteriorly, being represented by a very small thickening of entoderm (Fig. and finally is indistiguishable. Except
in the region of hindgut mentioned above, it, together with the entoderm, is everywhere in contact with the neural groove ectoderm, usually a line of contact being visible; in some places however a line of demarcation is exceedingly difficult to make out. No trace of a notochordal canal could be detected.
Mesoderm
Mesoderm.—Posterior to the somites is a large mass of very simple undifferentiated mesoderm which dorsally passes insensibly into the primitive streak. It is largely responsible for the size and shape of the posterior region of the embryo. Tts only differentiation is laterally, where it becomes somewhat more compactly arranged, lining the spaces which are forming the peritoneal coelom. Anterior to the somites the paraxial mesoderm is also very simple and serves as a wedge of tissue between entoderm and ectoderm. The highest mesodermal differentiation is seen in the formation of the somites, coelom and heart which will now be considered.
There are seven pairs of somites completely segmented. Each of the first six pairs contains a cavity, unconnected laterally with the coelom as described by Keibel in the reconstruction of the Kroemel-Pfannenstiel embryo, Klb. The first pair is apparently the most
highly differentiated, being the largest, having the largest cavity and
most compact peripheral arrangement of cells and is connected laterally by thc most clearly defined and differentiatedi intermediate cell
mass} These characteristics are less pronounced in each successive
posterior somite until the last is the smallest, contains no cavity,
has no cellular arrangement, and shows no other signs of differentiation. This would seem to indicate that the cavity is of secondary
origin; Graaf Spee however finds a small mesoderrnal slit in embryo
Gle which he thinks is a myocoele, although no somites are present.
In addition to the completely formed somites, another pair is just
beginning to show signs of constriction from the posterior paraxial
mesoderm. Anteriorly there is also another pair of somites about
half segmented with a very uniform compact mass of cells but containing no cavity. This rudimentary pair probahly represents the first pair of somites, which according to experiments in the chick
by Marion Hubbard and Patterson, is established first in time and
position, but develops slowly and maintains its connection with the
anterior paraxial mesoderm, thus causing the formation of somites
to take place only posteriorly.
Coelom. In his article on the coelom for the new Keibel-Mall
Embryology (now in the press), Professor Mall used this embryo
as the second stage of human coelomic development. Graaf Spee’s
Grle 1.54 mm. is the first human embryo to show any sign of ccelom
formation, this being a very small bilateral slit in the mesoderm of
the anterior portion of the embryo—the beginning of the pericardial
coelom. This slit is said to communicate externally by a very small
channel. The next stage, represented by this embryo, shows a very
large and well developed single, united pericardial cavity. (Figs. 1 and 11.)
The posterior continuation of the pericardial cavity on each side (Figs. 11 and 15) probably represents the pleural coelorn, which
passes insensibly into the peritoneal coelom, now forming from
multiple foci. A glance at Fig. 11 will show the extreme irregularity
and entire absence of any metameric arrangement in the formation
of the peritoneal coelom. Numerous irregular pockets of varying
size dip into the mesoderm from the extracoelom. This communication with the exterior may be either primary or secondary. The
diagram of the reconstructed coelom, however, would seem to indicate the former might be the case, because posteriorly a rather long
continuous slit is present in the mesoderm, the pockets of which are
shorter, much wider at the external opening, and diminish in size
internally. There are, however, several small independent cavities,
having no visible connection with each other or with the extracoelom,
which strongly suggest an independent formation by internal mesodermal cleavage, beginning in multiple small foci, which may later
connect with the other cavities and thus indirectly through them to the
extracoelom. Judging from the structure of this specimen alone it
seems probable that the result may be a combination of both processes
which are in reality ultimately one and the same. process differing
only in posi_tion. This irregular formation has been described by Bonnet in young sheep embryos, both in pericardial and peritoneal
coelom, the latter only communieating with the extracoelom.
The pericardial cavity communicates freely with the pleural, which in turn passes into the peritoneal eoelom and through it communication is made on each side with the cxoeoelom at the level of somite IV (Fig. 11). ‘There is therefore a complete circuitous canal, by which it is possible to travel from the extracoelom on one side through the embryonic coelom to the extraeoelom on the other side.
Heart. The mesodermal walls of the heart are the result of confluence, fusion and absorption of the walls of the pericardial
eoelom. The heart is attaclied dorsally throughout its whole length
by a mesocardium; ventrally and laterally it is free in the pericardial cavity. The heart roughly fills about one—third of the
perieardial cavity, is bent upon itself almost at a right angle, with
convexity to right and concavity to the left side of the embryo. The
endothelial heart is a simple tube of flattened cells lying within the
mesodermal walls but greatly shrunken and collapsed from fixation.
Nephric system. There is no evidence of the VVolfi‘ian bodies or
duets. Three independent small cavities are seen in the lateral
masses of somites I and II on the right side and somite II on the
left side, none of, which connect with coelom. These cavities show
a partially differentiated wall of more or less distinct cubical epithelium 3) and are probably the earliest stages of the pronephric tubules. Below somite II the lateral masses become very vague and
indistinct. Fig. 11 shows a number of cavities, some independent,
others connecting with the coelom, but agreeing in position with the
future lateral masses. ‘They are merely small undifferentiated slits
in the mesoderm, have no definite lining and may or may not be
homologous with those mentioned above as pronephrie tubules and in
earlier stages of development. They may, however, represent foei
or coelomic development, as is suggested by several small spaces
similar in position and connecting with the coelom,
Vascular system. - The description of the vascular system of this
embryo might appear very unorthodox indeed, were it not for the
previous description by Eternod of a 1.3 mm. human embryo, which
for a decade has remained alone and unconfirmed, which shows, however, all the signs of a normal embryo. The question of a primitive
yolk circulation has always been regarded as an established and indisputable fact, based almost entirely upon comparative embryology.
Recent observations in human embryology, however, have shown
the human ovum to differ markedly from other comparative forms
in the very early stages of development. This embryo, together with
the early embryos which have been described, particularly that of
Eternod, seems to indicate that the primitive human vascular system diiiers from the conventional comparative system in that the
umbilical circulation is the predecessor of the time—honored vitelline circulation.
In studying the vascular system, every capillary was traced as far
as high magnification would permit. The recent Work of H. M. Evans,
With injections of fresh specimens of young chick and pig embryos,
has however demonstrated the existence of large hitherto unrecognizable capillary beds and has shown the comparative inefiiciency of
studying fixed uninjected tissues for blood—vessels, even under the
most favorable conditions. We must therefore always bear in mind
the possibility of collapsed vessels, and that other things being equal,
too much emphasis cannot be placed upon negative evidence. Even
if there should be some small capillary beds, which are unrecognizable by the microscope in the absence of injections, it would merely
be evidence in favor of vascular connection between the umbilical
vesicle and the embryonic circulation, and would not in any Way
affect the question of priority, because of the relatively greater development of the umbilical vessels. A, glance at Fig. 15 and Fig. 12 will conclusively show that the functioning system is umbilical or
chorionic and that if a connection could be traced through the large
gap between the umbilical vesicle and the small sprout of the possible
vitelline vein, it would be comparatively insignificant. Since the
embryonic vessels are full of blood and there is no apparent connection with the blood-forming area in the umbilical vesicle, the question naturally arises, how are we to account for the presence of the
blood corpuscles? This is explained by finding many beautiful examples of endothelial proliferation of blood corpuscles from the capillaries in the chorionic membrane (Fig. 10)., probably supplying the
embryo with blood until the time of connection with the yolk sac.
A survey of the literature gives very limited information on the
primitive vascular development. ‘The first suggestion of a vascular
anlage is a very indistinct knotty appearance in Peters’ embryo;
Keibel, however, who made a reconstruction of this ovum, says this
appearance is merely suggestive and that no positive conclusions
can be drawn. None of the several other young ova show any vascular formation. Embryo Frassi 1.17 mm. shows the first blood
vessels, which are distributed in the chorion, bauchstiel and ventral
pole of the yolk sac. No vessels were observed in the body of the
embryo. Graf Spee’s Gle 1.54 mm. is probably next in point of age,
followed very closely by Eternod’s 1.3 mm. embryo. Graf Spee
describes the first anlage of the heart, otherwise there are no vessels
in the body of the embryo; blood islands are present in the ventral
pole of the yolk sac, but no mention is made of vessels in the bauchstiel or chorion. Eternod’s 1.3 mm. embryo is the first specimen to
present a complete circulation. It has a very well developed umbilical circulation and the villi are partly vascularized, but no vitelline vessels could be detected. The Kroemer-Pfannenstiel embryo
Klb. of approximately the same age as the embryo under consideration is said to have a very large umbilical artery, unaccompanied
however by a corresponding vein, also omphalomesenteric veins are
present but no corresponding arteries. The vessels in the yolk sac are
full of corpuscles, but no mention is made of any chorionic vessels.
This summary of the earliest vascular development is by no means
evidence in favor of a primitive vitelline circulation. Although the
facts are very meager on account of the scarcity of material and
nothing positive can be deduced, nevertheless there seems to be some
evidence in support of a primitive umbilical circulation. The first
embryo having a complete circulation is Eternod’s (1.3 mm.), with
a well developed umbilical circulation. From the other remaining
specimen (Kroemer-Pfannestiel Klb.) between Eternod’s and our
embryo it would be hard to draw definite conclusions on account of
the presence of the artery of one system and the vein of another,
forming no complete circulation.
The presence of capillaries in the chorion, bauchstiel and yolk sac
in Embryo Frassi would seem to indicate that the mesoderm which
forms all of the above structures, is endowed with a high power of vascnlarity in its early history, irrespective of position, and that from
this vascular formation, whether from single or multiple points of
origin, two main primitive vascular areas originate practically synchronously and develop independently toward the embryo. Eternod’s
embryo together with the one under consideration, which are the
two earliest embryos with a complete circulation, would seem to indicate thatthe umbilical circulation was the first to attain this function.
As mentioned before, this embryo agrees in general very closely
with Eternod’ s 1.3 mm. embryo, differing only in a few details. The
yolk vascular system is developing rapidly and consists of numerous
large blood islands, forming irregular, disconnected channels and
masses of blood—foI'Ining tissue, situated almost entirely on the ventral
half of the umbilical vesicle (Fig. 12), extending from the anterior
to the posterior poles and more marked in the region of the future
vitelline veins. A few very small blood islands extend along the
dorsal half of the yolk sac, but never is there any visible vascular
connection within the embryo and always a considerable distance
exists between the blood islands and the vessels of the body of the
embryo.
The umbilical circulation consists of a simple vascular cycle
comprising the umbilical veins, heart, dorsal aortas and umbilical
arteries, with capillary connection in the chorion and cliorionic villi.
A number of villi show the early stages of vaseularization, which are
not limited to the region around the attachment of the bauehstiel, but
extend anteriorly beyond a point corresponding with the anterior limit
of the embryo. There appear to be several seemingly independent
small systems of capillaries in the chorionie membran-e, i-nto which
drain the capillaries of the villi. Although these systems seem to have
no connection with each other, the difiiculty of following capillaries
in uninjected specimens would make a positive statement impossible. As mentioned above, many of these chorionic vessels show
beautiful examples of what. appears to be blood formation from
endothelial proliferations (Fig. 10) and is probably the source of
the blood for the present circulation.
Two large independent bl.ood_ reservoirs (Figs. 8, 9 and 15) in the bauchstiel connect distally with the ehorionic vessels; proximally the smaller gives off the paired umbilical veins; the larger is formed by the confluence of the umbilical arteries.
The umbilical veins run in the dorsolateral portion of the body
of the embryo, at the point of origin of the amnion. The left vein
gives one, the right vein two, very short branches to the amnion at
the level of the posterior region of the embryo. Nothing comparable
to the sinus ensiforme of Eternod’s 1.3 mm. embryo was observed.
While still in the dorsal position and just before its course ventralward, each umbilical vein gives ofi a short branch which runs ven-
trally through the body of the embryo toward the umbilical vesicle.
On one side it terminates before reaching the yolk sac, on the other
side it runs a short distance in the wall of the yolk sac and seems
to end blindly. On each side a couple of spaces, presumably capillaries, although they do not contain any blood, are in the immediate
vicinity of these short branches, but no connection can be detected.
These short branches and possible capillaries may be the beginnings
of the vitelline veins, although the dorsal point of origin from the
umbilical veins is rather against this view, unless there is a subsequent ventral wandering, as has been observed in many other vessels.
This is the only visible evidence of the possibility of the vitelline
veins. (Plate IV.)
The umbilical veins now run ventrally around the ecelom and
unite to form the heart just anterior to the origin of the foregut.
It is a simple tube of endothelium throughout. From its anterior
extremity one complete aortic arch is given ofi on each side (Fig.
1) and in addition, four short stubs on the right and two on the
left sides, vvhich represent rudiments of future aortic arches. No
corresponding buds are given ofi by the cephalic portion of the
dorsal aortas to connect with these. The heart is a step in advance
of that of the Kroemer—Pfannenstiel embryo, which has one aortic
arch on each side, but a paired heart is still present. Eternod’s
1.3 mm. has a slightly higher development, in that three and possibly four complete arches are present on each side.
The dorsal aorta, paired throughout, begin anteriorly in a small dilatation and terminate caudally at the posterior border of the fore-gut in a much larger dilatation, from which the umbilical arteries are the direct continuation (Plate VI). Each aorta gives off a series of eleven lateral branches, beginning as very minute and rather indefinite branches posterior to the first somite; these become progressively larger and reach a climax at the posterior dilatation of the aorta. In the somite region they are solid buds (Fig. 5), the last five arising from the dilatation have a lumen, but end blindly in the mesoderm of the splanchnopleure (Fig. 5). On the right side the last three branches seem to end in a common blood island, giving the impression of a small capillary plexus; this was, however, not observed on the other side. These branches probably represent the segmental arteries of Mall destined for the umbilical vesicle. In the somite region there are six of these branches for the seven somites; on one side all six arise between the somites; on the other side three arise intersegrncntally, the other three come ofl nearer the center of the somites. From the dorsal and caudal end of the posterior aortic dilatation there is a very short stub on each side, which may represent the beginning of the caudal aorta. Otherwise no dorsal branches of the aortas were observed.
The umbilical arteries, somewhat larger than the aortas, are direct posterior continuations of the posterior aortic dilatation. In the bauchstiel they unite to form a large sinus (Fig. 9) which lies posterior to the smaller venous sinus. (Fig. 8.) It sends ofi' numerous large branches to the chorion, some of which anastomose and redistribute branches to the chorionic membrane and villi.
It is a pleasure to extend my heartiest thanks to Professor Mall for his kindness in allowing me the privilege of describing this very rare specimen, for his valuable advice and suggestions and for the use of two manuscripts which are not yet in print.
A Human Embryo with Seven Pairs of Somites. 99
Bibliography
BENEKE, R. Ein sehr junges menschliches Ei. Deutsehe med. Wochenschix, Jg. 30, p. 1304.
BROMAN, IVAR. Ueber die Entwicklung, “Wanderung” und Variation der Bauehaortenzweige bei den Wirbeltieren. Ergebn. d. Anat. u. Ent-Wgeseh., Bd. 16, 1906.
Baron, J. H., and TEACHER, J. H. An Early Human Ovum. Glasgow, 1908.
ETERNOD, A. C. F. 11 y a un lecithophore dans l’e1nbryon humain. Biblegraphie anatomique, T. 15, 1906.
. Communication sur un oeuf humain avec embryon excessivement jeune. Comptes Bend. du X1. Congres int. Méd., Rome, 1894.
. Sur un oeuf humain de 163 mm. avec embryon de 2.1 mm. Actes de la Soc. helv. des Sc. nat., 1896.
. Premiers stades de la circulation sanguine dans 1’oeuf et Pembryon humain. Anat. Anz., Bd. XV, 1898.
EVANS, H. M. On the Earliest Blood Vessels in the Anterior Limb Buds of Birds. Amer. Jour. Anat., April, 1909.
. On the Development of the Aorta, Cardinal and Umbilical Veins and other Blood Vessels in the Vertebrate Embryo from Capillaries. Anat. Record, Vol. III, 1909.
FRASSI, L. Ueber ein junges menschliches El in situ. Archiv f. mierosk. Anat., Bd. 70, 1907.
. Weitere Ergebuisse des Studinms eines jungen menschlichen Eies in situ. Archiv f. mikr. Anat, Bd. 71, 1908.
GAGE, SUSANA PHELPS. Three Weeks’ Human Embryo. American Jour. Anat., Vol. IV, 1905.
HIS, WILHELM. Anatomie menschlicher Embryonen. Leipzig, 1880-1885.
INGALLS, N. W. Beschreibung‘ eines menschlichen Embryo von 4.9 cm. Archiv f. mikr. Anatomie, Bd. 70, 1907.
KEIBEL, FRANZ. Ueber den Schwanz des menschlichen Embryo. Archiv f. Anat. u. Physiol., Anat. Abt., Jg. 1891, pp. 356-398.
. Ein se-hr junges menschliches Ei. Archiv f. Anat. u. Physlol., Anat. Abt., .Tg. 1890, pp. 250-267.
Normentafel zur Entwicklungsgesehichte der Wirbeltiere. Jena, 1908.
KOLLMAN, JUILIUs. Handatlas der Entwicklungsgeschichte des Menschen. Jena, 1907.
Knommm. Wachsmodell eines jungen menschl. Embryo. Verhand. d. Ges. f. Gyniikologie, 1903.
Lnrmossrzxs, M. V. Die Entwicklung der Ganglien-Anlagen bei dem menschllchen Embryo. Archiv f. Anat. u. Physiol., Anat. Abt., Vol. —, 1891;
LILLIE, FRANK R. The Development of the Chick. New York, 1908. (Ref. to Marion Hubbard and J. T. Patterson.) (Pp. 111-114.)
MAGCAILLUM, JOHN BRUCE. Note on the Wolflian Body of Higher Mammals. Amer. Journ. Anat., Vol. I, 1902.
MALL, F. P. Age of Embryos and Foetuses. Keibel-Mall Embryology (In press) (manuscript by permision of author).
MALL, F. P. On the Development of the Human Diaphragm. Bull. Johns Hopkins Hosp., Vol. XII, 1901.
. A Human Embryo of the Second Week. Anat. Anz., Jg. 8, 1893, pp. 630-633.
. Human Embryo, twenty-six days old. Journal of Morphology, Vol. V, 1891.
. Development of the Human Coelom. Journal of Morphology, Vol. XII, 1897.
Development of the Human Coelom. Keibel—Mall Embryology (in press), Leipzig and Philadelphia, 1910.
MINOT, C. S. Human Embryology. New York, 1892.
QUAIN’S Anatomy, Vol. I, London, 1908.
SPEE, FERDINAND, GRAF V. Beobachtungen an einer menschlichen Kennscheibe mit’ offener Medullarrinne und Canalis neurentericus. Archiv f. Anat. u. Physiol, Anat. Abt., 1889, pp. 159-176.
Neue Beobachtungen fiber sehr frfihe Entwicklungsstufen des Inenschlichen Eies. Archiv f. Anat. 11. Physio1., Anat. Abt., 1896, pp. 1-27.
TANDLER, JULIUS. Ueber Vornierenrudimente beim menschliohen Embryo. Anat. Hefte, Bd. 28, 1905.
THOMPSON, PETER. Description of a Human Embryo of Twenty_three Paired Somites. Journ. of Anal. and Physiol., Vol. XLI, 1907.
WILLIAMS, J. VVI-IITRIDGE. Textbook of Obstetrics, New York, 1907.
Explanation of Plates
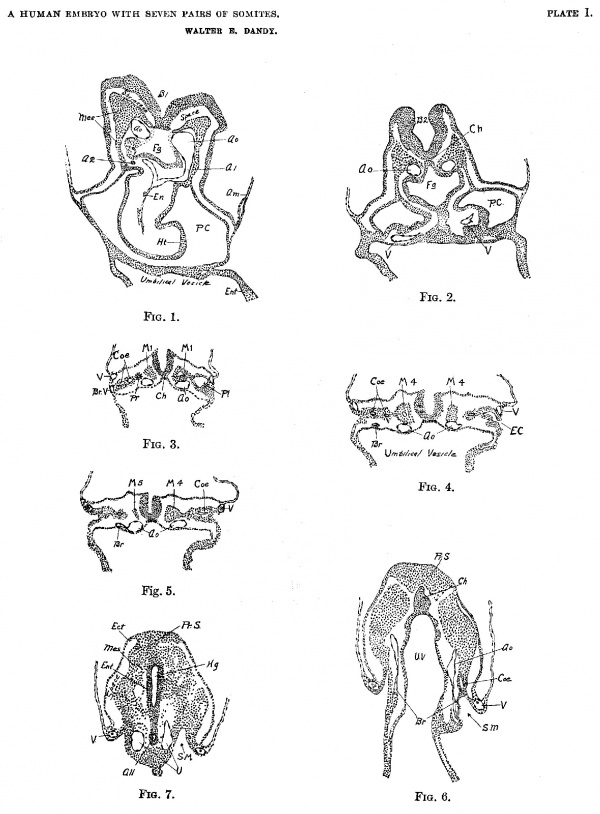
Plate 1
(FIGS. 1—7).
Fig. 1. Section 22, X 50. B1, First primary brain vesicle; Oo, Aorta; A1, Complete View of longitudinal section of tirst aortic arch; A2, Cross section of stub of second aortic arch; En, Endothelial heart; Ht, Mesodermal wall of heart; P.C., Pericardial cavity; Fg, foregut; Am, Amnion; Mes, Mesoderm; Ent, Entoderm.
Fig. 2. Section 53, X 50. Ch, Chorda; Ao, Dorsal aorta; V, Umbilical vein; Fg, Foregut; P.C., Pericardial cavity; B2, Second primary vesicle of the brain.
Fig. 3. Section 76, X 50. Pr, Pronephros; Coe, Coelom; Pl, Pleural Cuelom; V, Umbilical vein; Br V, Branch of umbilical vein, only suggestion of a vitelline vein, it unites with umbilical vein two sections below; Oh, Chorda; M1, First Somite in each side.
Fig. 4. Section 93, X 50. Br, Cross section of lateral branch of dorsal aorta; V, Umbilical vein; M4, Somite IV, with cavity (anterior tip of somite IV on opposite side) ; Coe, Coelom; E.C., Communication of coelom with exterior; Ch, Chorda.
Fig. 5. Section 101, X 50. Br. Lateral branch of Aorta; Ao, Aorta; V, Umbilical vein; M4, Somite IV with cavity; M5, Tip of Somite V; Coe, Coelom.
Fig. 6. Section 137, X 50. A0, Dorsal aorta; Br, Lateral branch of aorta (branch on left side terminates in a mass of mesodermal cells which may be a small blood island); Ch, Chorda at point of greatest development; Coe, Coelom; S.M, Slit in mesoderm. (See also Fig. 11.) Pr. S, Beginning of primitive streak. U. V, Umbilical vesicle.
Fig. 7. Section 152, X 50. Shows primitive streak region (Pr. S.) from fusion of ectoderm, mesoderm and entfoderm. Hg, Hindgut; A11, Allantois, near point of origin from hindgut; U, Umbilical artery; V, Umbilical vein; S. M, Slit in mesoderm. Ect, Ectoderm; Ent, Entoderm; Mes, Meoderm.
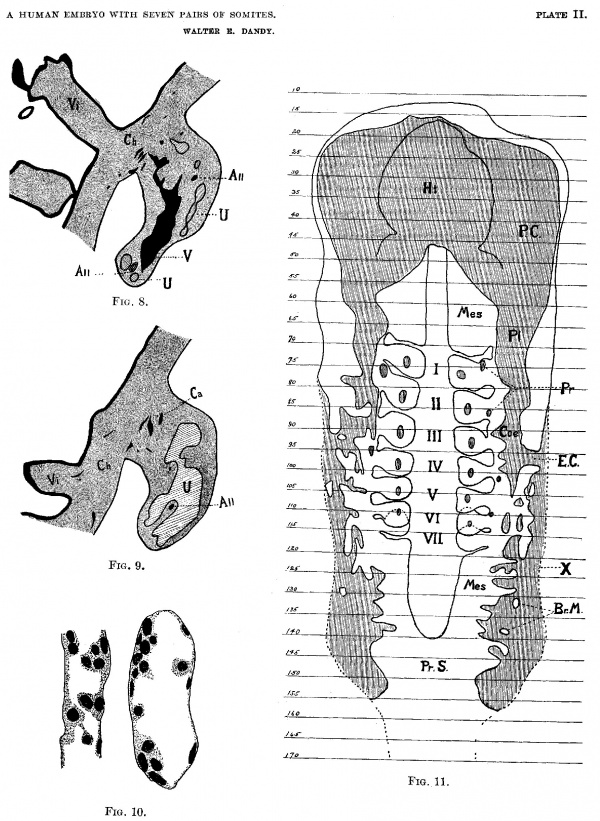
Plate II
(FIGS. 8-11).
Fig. 8. Section 177, X 25. Outline of section to show size of the sinus formed by union of umbilical veins; V, Umbilical venous sinus (from union of the umbilical veins); All, Allantois, shown in two places, the lower is before division, the upper is terminus of long branch; Ch, Chorion; Vi, Chorionic villus, showing syncytial bud; U, Umbilical artery, shown in two places below, before union to form umbilical arterial sinus, and above, tip of sinus and large branches.
Fig. 9. Section 188, X 25. Outline of section to show size of sinus formed from union of umbilical arteries. U, Umbilical arterial sinus; All, The terminus of the short branch of the allantois; Ca, Capillaries and small branches of sinus in chorion; Ch, Chorionic Membrane; Vi, Chorionic villus, showing capillary entering.
Fig. 10. X 525. Capillaries from chorion, showing apparent formation of blood corpuscles from endothelium of capillaries.
Fig. 11. Reconstruction of coelom, showing relation to somites; Arabic numbers on side represent the numbers of sections; Roman numerals in center represent the paired somites; P.C., Pericardial coelom; Pl, Pleural coelom; Coe, Peritoneal coelom; E.C., External Communication of the coelom: Pr, Pronephros; Ht, Projection of the heart in the pericardial cavity; X. Outer limit of body wall; Mes, Paraxial mesoblat; Br.M., Bridge of mesodermal-tissue extending across the mesodermal slit; Pr.S., Primitive streak.
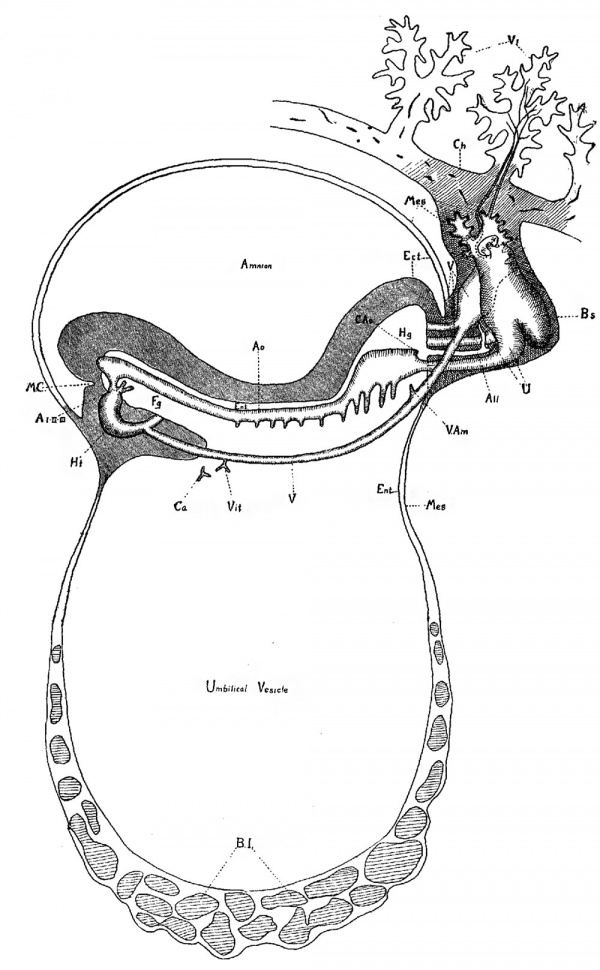
Plate III
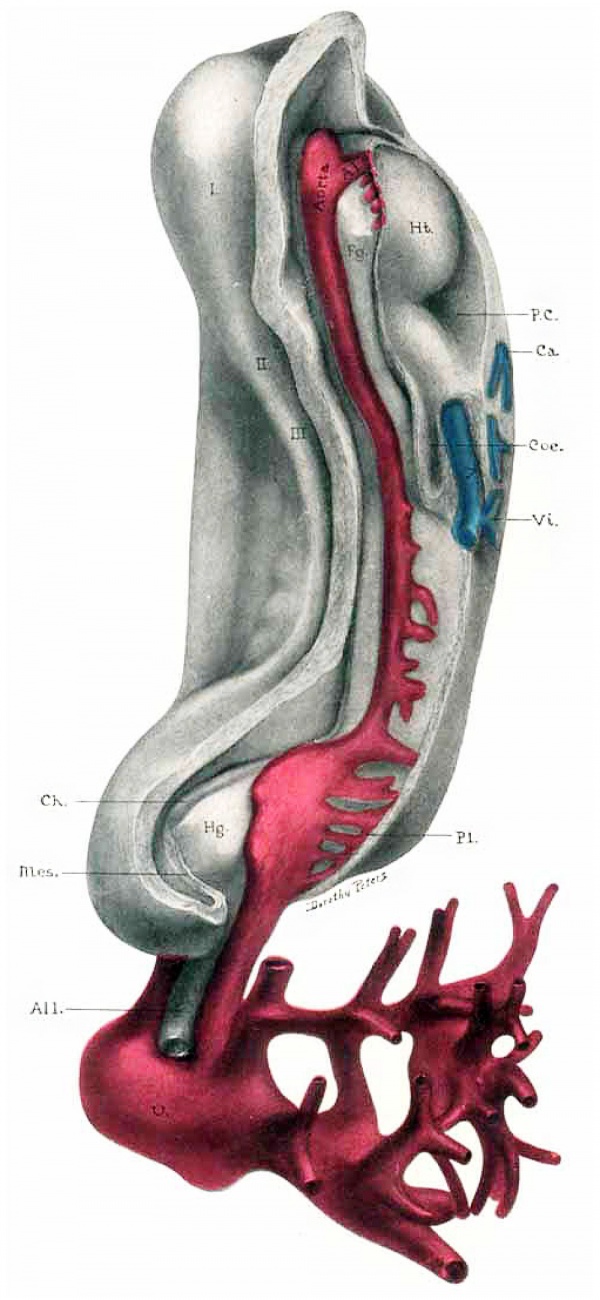
Fig. 12. Diagram of a sagittal View of embryo to show the vascular system.
Ht, Heart; A I-II-III, First, second and third aortic arches; A0, Dorsal aorta; C.Ao, Caudal aorta; U, Umbilical artery (unite in the bauchstiel); V, Umbilical vein (unite in the Bauchstiel) ; Vit, Branch of umbilical vein, is only suggestion of a possible vitelline vein; V. am, Venous branch to amnion; Ca, Capillaries; do not contain blood and do not connect with veins; B.I., Blood Islands; Ect, Ectoderm; Mes, Mesoderm; Ent, Entoderm; Fg, Foregut; Hg, Hindgut; All, Allantois; Oh, Chorionic membrane; V1, Chorionic villi, showing vascularization; B.S, Bauchstiel; M.C., Mouth Cavity.
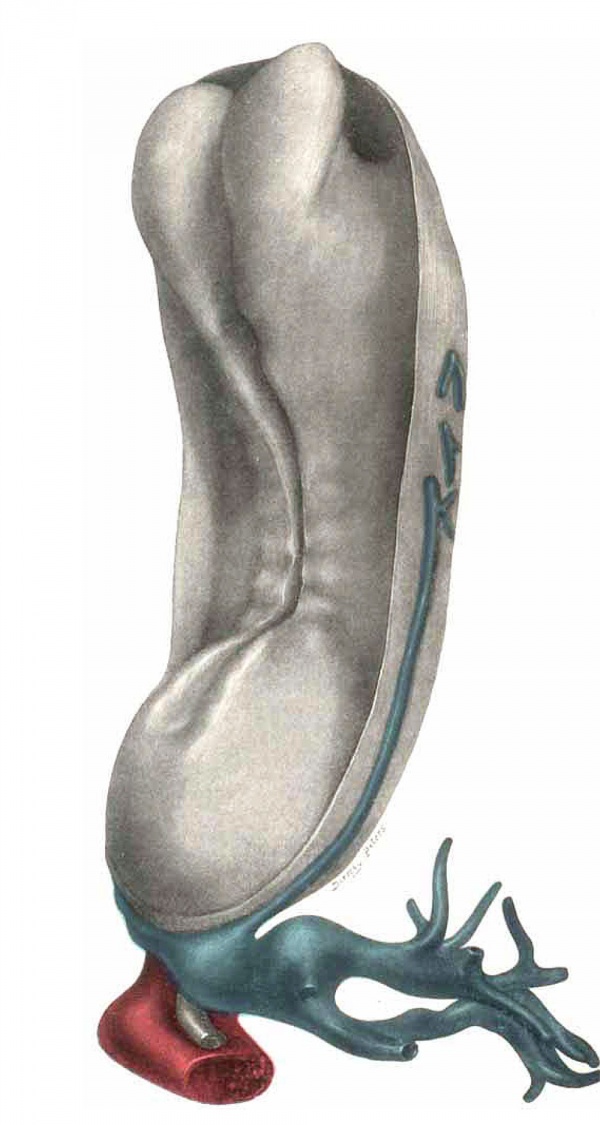
Plate IV
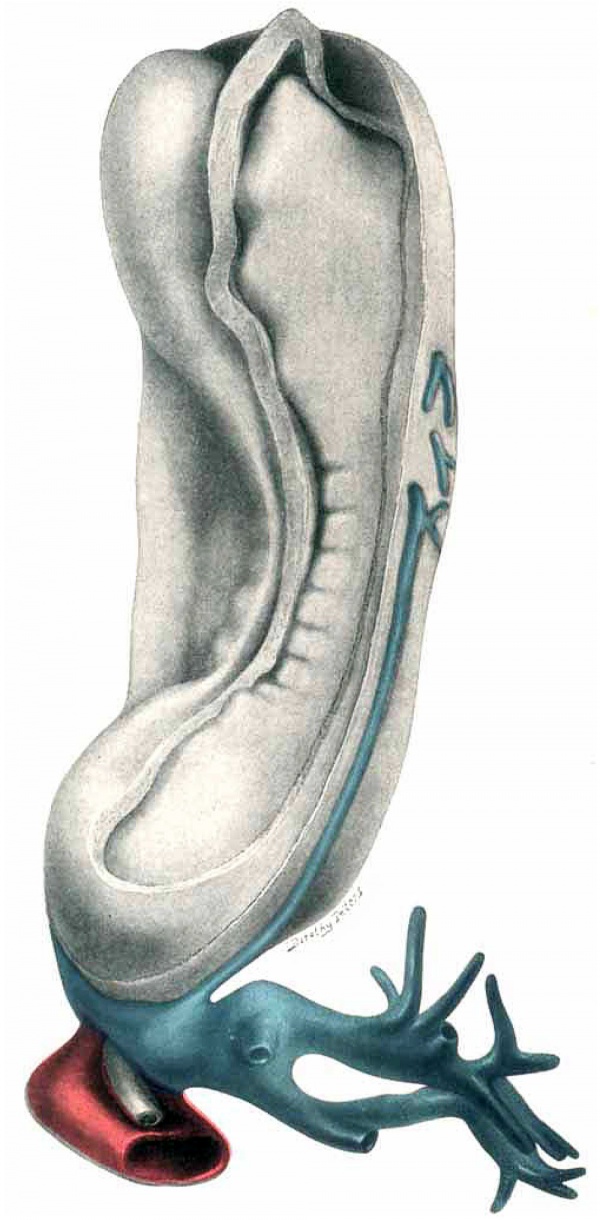
Fig. 13. Dorsol-lateral View of model of the embryo, with the amnion removed. Umbilical cord with possible vitelline branch (?) seen on right side. (The vein is projected on exterior for purpose of clearness.) Large umbilical venons and arterial sinuses posteriorly, with allantois lying between. Arterial sinus is partially cut away, venous is intact. Neural canal is open throughout, and shows the three primary vesicles of the brain.
Plate V
Fig. 14. Same view as Plate IV; window of ectoderm removed, exposing the somites and mesoderm.
Plate VI
Fig. 15. Same View as in Plates IV and V. Mesoderxn is removed. I, II, III-Three primary brain vesicles; 1~First aortic arch; studs of arches 2, 3, 4, 5 are seen just posterior; U—Umbilical arterial sinus; .V-Umbilical veins (umbilical veins and umbilical venous sinus have been cut away to show the arterial system). Vi, Bl'21ll(fll of umbilical vein, only suggestion of possible vitelline vein: Fg—Foregut; Hg-Hindgut; All-Allantois Ch-Chorda; Ht-Heart; Coe-Coelom; l’.C.—Pericardi3.l ccelom; Ca-Capillary, but no connection; Pl—Plexus of lateral aortic branches (shown only in this place). Mes-Mesoderm in primitive streak region separated from the ectoderm by a shrinkage space, which is only present laterally, in this region. (See also Fig. 7.)
Cite this page: Hill, M.A. (2024, April 28) Embryology Paper - A Human Embryo with Seven Pairs of Somites Measuring about 2 mm in Length. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_A_Human_Embryo_with_Seven_Pairs_of_Somites_Measuring_about_2_mm_in_Length
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G