Implantation microRNA
| Embryology - 20 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
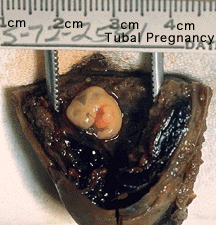
Ectopic pregnancy is a high-risk maternal medical condition with an approximate incidence of 1.5 to 2 % in reported pregnancies. In the USA, data from commercial health plans identified 11,989 ectopic pregnancies during the period from 2002 to 2007.[1] In the Australian state of New South Wales, in 2008 the annual hospitalisation rate for women with an ectopic pregnancy was 12.6 per 1000 births.[2]
Our research group has established a uterine tube Biobank and cell culture techniques in order to investigate factors and markers associated with tubal ectopic pregnancies. This page introduces the project and provides some introductory information for students who may wish to carry out an Honours, Independent Research Project or higher degree in the research laboratory. This is new collaborative research project between clinical researchers from the Women and Childrens Hospital (RHW), basic researchers from the School of Medical Sciences (SoMS) and research support from the HSA Biobank and Bioanalytical Mass Spectrometry Facility ( BMSF).
- Research Links: 2014 Seminar | Dr Mark Hill | Professor William Ledger | Royal Hospital for Women | SOMS | HSA Biobank | BMSF
- Ectopic Page Links: Ectopic Implantation | Implantation | Week 2 | Placenta - Abnormalities | Trophoblast | Trophoblast - Protein Expression | Implantation microRNA
Some Recent Findings
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Ectopic Pregnancy <pubmed limit=5>Ectopic Pregnancy</pubmed> Search term: Implantation microRNA <pubmed limit=5>Implantation microRNA</pubmed> |
MicroRNA Profiles in Spontaneous Decidualized Menstrual Endometrium and Early Pregnancy Decidua with Successfully Implanted Embryos
MicroRNA Profiles in Spontaneous Decidualized Menstrual Endometrium[3]
- To comparatively analyze the human microRNA (miRNA) profiles between spontaneous decidualized menstrual endometrium and early pregnancy decidua by an in-depth sequencing of miRNAs. The specific miRNAs expressed at conception might be involved in pregnancy establishment and expression of let-7f-5p and let-7g-5p was experimentally up-regulated or inhibited to assess the effect on the expression of IGF2BP-1 and IGF2R in vitro, respectively. Samples of endometria and deciduas were obtained from 25 women who suffered from tubal or male factor subfertility and from 35 early pregnant women who underwent pregnancy termination at 6-8 weeks gestation were irrespectively collected and comparatively analyzed by miRNA sequencing and differential expression of known and novel miRNAs was analyzed using bioinformatics. The 2042 miRNA expression was analyzed in the study and the differential expression of six miRNAs was validated by qRT-PCR.
- Down-regulated - miR-34c, miR-92a, miR-181a-5p, and miR-191
- Up-regulated - miR-10a-5p and let-7f-5p
- Expression of IGF2BP-1 and IGF2R declined and increased with overexpression and inhibition of let-7f-5p and let-7g-5p, respectively.
Inflammatory gene networks in term human decidual cells define a potential signature for cytokine-mediated parturition
Inflammatory gene networks in term human decidual cells [4]
- Inflammation is a proximate mediator of preterm birth and fetal injury. During inflammation several microRNAs (22 nucleotide noncoding ribonucleic acid (RNA) molecules) are up-regulated in response to cytokines such as interleukin-1β. MicroRNAs, in most cases, fine-tune gene expression, including both up-regulation and down-regulation of their target genes. However, the role of pro- and antiinflammatory microRNAs in this process is poorly understood.
- The principal goal of the work was to examine the inflammatory genomic profile of human decidual cells challenged with a proinflammatory cytokine known to be present in the setting of preterm parturition. We determined the coding (messenger RNA) and noncoding (microRNA) sequences to construct a network of interacting genes during inflammation using an in vitro model of decidual stromal cells.
- The effects of interleukin-1β exposure on mature microRNA expression were tested in human decidual cell cultures using the multiplexed NanoString platform, whereas the global inflammatory transcriptional response was measured using oligonucleotide microarrays. Differential expression of select transcripts was confirmed by quantitative real time-polymerase chain reaction. Bioinformatics tools were used to infer transcription factor activation and regulatory interactions.
- Interleukin-1β elicited up- and down-regulation of 350 and 78 nonredundant transcripts (false discovery rate < 0.1), respectively, including induction of numerous cytokines, chemokines, and other inflammatory mediators. Whereas this transcriptional response included marked changes in several microRNA gene loci, the pool of fully processed, mature microRNA was comparatively stable following a cytokine challenge. Of a total of 6 mature microRNAs identified as being differentially expressed by NanoString profiling, 2 (miR-146a and miR-155) were validated by quantitative real time-polymerase chain reaction. Using complementary bioinformatics approaches, activation of several inflammatory transcription factors could be inferred downstream of interleukin-1β based on the overall transcriptional response. Further analysis revealed that miR-146a and miR-155 both target genes involved in inflammatory signaling, including Toll-like receptor and mitogen-activated protein kinase pathways.
- Stimulation of decidual cells with interleukin-1β alters the expression of microRNAs that function to temper proinflammatory signaling. In this setting, some microRNAs may be involved in tissue-level inflammation during the bulk of gestation and assist in pregnancy maintenance.
- Up-regulated - MiRNA 147b, MiRNA 3142 MiRNA 146a MiRNA 155, MIR155 host gene (nonprotein coding) Long intergenic nonprotein coding RNA 944 Long intergenic nonprotein coding RNA 473 Long intergenic nonprotein coding RNA 1137 MiRNA 4632 MiRNA 154 MiRNA 3182 MiRNA 4683 MiRNA 1185-1 MiRNA 645 Long intergenic nonprotein coding RNA 312 MiRNA 889 MiRNA 3614 MiRNA 299 MiRNA 4751 MiRNA 1246
- Down-regulated - MiRNA 4680 MiRNA 452, miRNA 224 MiRNA 622 MiRNA 196b Long intergenic nonprotein coding RNA 339 Long intergenic nonprotein coding RNA 478 MiRNA 143, miRNA 145, MIR143 host gene (nonprotein coding) MiRNA 424 MiRNA 503 Long intergenic nonprotein coding RNA 1085
- Nanostring Analysis - Based on the absolute value of the expression data, miR-21, miR-143, miR-145, and miR-4454 as well as several members of the let-7, miR-10, miR-15, miR-29, and miR-125 families were among the top 50 most highly expressed mature miRNAs in untreated term decidual cells
whereas members of the miR-181, miR-183, and miR-200 families, which undergo down-regulation following decidualization of endometrial stromal cells,41 were among the least abundant miRNAs (data not shown). Differentially expressed mature miRNAs in IL-1β-stimulated term decidual cells by NanoString profiling (Linear fold change was used)
- Up-regulated - miR-146a-5p, miR-525-5p
- Down regulated - miR-143-3p, miR-145-5p, miR-924, miR-4454
MicroRNA-181a is involved in the regulation of human endometrial stromal cell decidualization by inhibiting Krüppel-like factor 12
MicroRNA-181a endometrial stromal cell decidualization[5]
- The transformation of endometrium into decidua is essential for normal implantation of the blastocyst. However, the post-transcriptional regulation and the miRNAs involved in decidualization remain poorly understood. Here, we examined microRNA-181a (miR-181a) expression in decidualized human endometrial stromal cell (hESC). In addition, we investigated the functional effect of miR-181a on hESC decidualization in vitro.
- Quantitative real-time PCR (qRT-PCR) was used to detect the profile of miR-181a in decidualized hESC. qRT-PCR, enzyme-linked fluorescent assay, and immunofluorescence assay were performed to investigate decidualization marker genes' expression after enhancing or inhibition of miR-181a expression in hESC. Luciferase reporter assay, western blotting, qRT-PCR, and immunofluorescence assay were carried out to identify the relationship between miR-181a and Krüppel-like factor 12 (KLF12).
- miR-181a expression levels increased dramatically in hESC treated with 8-Br-cAMP and MPA. Increased miR-181a expression promoted hESC decidualization-related gene expression and morphological transformation; conversely, inhibition of miR-181a expression compromised hESC decidualization in vitro. Further analysis confirmed that miR-181a interacted with the 3' untranslated region of the transcription factor KLF12 and down-regulated KLF12 at the transcriptional and translational levels. KLF12 overexpression abolished miR-181a-induced decidualization.
- Our findings suggest that miR-181a plays a functionally important role in human endometrial stromal cell decidualization in vitro by inhibiting KLF12.
miRNA signature and Dicer requirement during human endometrial stromal decidualization in vitro
Dicer during human endometrial stromal decidualization[6]PLoS One. 2012;7(7):e41080. doi: 10.1371/journal.pone.0041080. Epub 2012 Jul 20.
- Decidualization is a morphological and biochemical transformation of endometrial stromal fibroblast into differentiated decidual cells, which is critical for embryo implantation and pregnancy establishment. The complex regulatory networks have been elucidated at both the transcriptome and the proteome levels, however very little is known about the post-transcriptional regulation of this process. miRNAs regulate multiple physiological pathways and their de-regulation is associated with human disorders including gynaecological conditions such as endometriosis and preeclampsia. In this study we profile the miRNAs expression throughout human endometrial stromal (hESCs) decidualization and analyze the requirement of the miRNA biogenesis enzyme Dicer during this process. A total of 26 miRNAs were upregulated and 17 miRNAs downregulated in decidualized hESCs compared to non-decidualized hESCs. Three miRNAs families, miR-181, miR-183 and miR-200, are down-regulated during the decidualization process. Using miRNAs target prediction algorithms we have identified the potential targets and pathways regulated by these miRNAs. The knockdown of Dicer has a minor effect on hESCs during in vitro decidualization. We have analyzed a battery of decidualization markers such as cell morphology, Prolactin, IGFBP-1, MPIF-1 and TIMP-3 secretion as well as HOXA10, COX2, SP1, C/EBPß and FOXO1 expression in decidualized hESCs with decreased Dicer function. We found decreased levels of HOXA10 and altered intracellular organization of actin filaments in Dicer knockdown decidualized hESCs compared to control. Our results provide the miRNA signature of hESC during the decidualization process in vitro. We also provide the first functional characterization of Dicer during human endometrial decidualization although surprisingly we found that Dicer plays a minor role regulating this process suggesting that alternative biogenesis miRNAs pathways must be involved in human endometrial decidualization.
- miR-181, miR-183 and miR-200 miRNAs Families Members are Similarly down-regulated during in vitro Decidualization
- For the mir-181 family, which includes six miRNAs precursors (mir-181a-1, mir-181a-2, mir-181b-1, mir-181b-2, mir-181c and mir-181d) , the corresponding mature miRNA expression in decidual cells decreased
- miR-96 and miR-135b Regulates FOXO and HOXA10 Expression and IGFBP-1 Secretion
- two miRNAs that decrease their expression during stromal differentiation
- other miRNA family includes miR-96, miR-182 and miR-183 with potential molecular pathways implicated in actin cytoskeleton reorganization, such as RAC1 and ITGß1, and both genes are implicated in endometrial decidualization
- total 33 differentially expressed miRNAs during our in vitro decidualization model, only two (miR-181b and miR-181d) have been previously identified in the Qian et al study
- miRNA Target Prediction - To identify potential miRNA target genes, we used four publicly available target prediction algorithms: targetscan (http://www.targetscan.org/), miRanda (microRNA.org), microCosm (http://www.ebi.ac.uk/enright-srv/microcosm/htdocs/targets/v5/) and PicTar (http://pictar.mdc-berlin.de/). The Diana miR-Path database is a web-based computational tool developed to identify the molecular pathways potentially altered by the expression of single or multiple microRNAs [21] (http://diana.cslab.ece.ntua.gr/pathways/).
Differential expression of microRNAs with progression of gestation and inflammation in the human chorioamniotic membranes
microRNAs of gestation and inflammation in the human chorioamniotic membranes[7]
- The aim of this study was to identify differential expression of microRNAs (miRNAs) in chorioamniotic membranes with advancing gestation, labor, and inflammation.
- Expression profiles of 157 miRNAs in the chorioamniotic membranes were obtained from patients in the following groups: 1) term not in labor (n = 10); 2) term in labor (n = 10); 3) preterm labor with histologic chorioamionitis (n = 9); and 4) without histologic chorioamnionitis (n = 10).
- More than 95% of the miRNAs screened were expressed. Gestational age-dependent changes in expression were observed for 13 miRNAs. No differences in miRNA expression were observed between women without labor and women in labor. Membranes with chorioamnionitis displayed increased expression of miR-223 and miR-338. Gene Ontology analysis of genes targeted by differentially expressed miRNAs revealed enrichment for specific biological process categories.
- Chorioamniotic membranes with advancing gestational age and chorioamnionitis are associated with the differential expression of a subset of miRNAs.
DIANA-miRPath
A miRNA pathway analysis web-server, providing accurate statistics, while being able to accommodate advanced pipelines. miRPath can utilize predicted miRNA targets (in CDS or 3’-UTR regions) provided by the DIANA-microT-CDS algorithm or even experimentally validated miRNA interactions derived from DIANA-TarBase v6.0. These interactions (predicted and/or validated) can be subsequently combined with sophisticated merging and meta-analysis algorithms. miRPath v2.0 can perform advanced analysis pipelines, such as hierarchical clustering of miRNAs and pathways based on the levels of their interactions. Furthermore, users can easily create heat maps of miRNAs vs pathways interactions. Other supported features include the identification of pathological single nucleotide polymorphisms (SNPs) in miRNA binding sites, as well as the “Reverse Search module”, where the user can identify all the predicted or experimentally validated miRNAs significantly targeting a selected pathway.
- Links: DIANA-miRPath
References
Reviews
<pubmed>26707514</pubmed> <pubmed>26784241</pubmed> <pubmed>26771608</pubmed> <pubmed></pubmed>
Articles
<pubmed></pubmed>
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- miRNA Target Prediction targetscan | [microRNA.org miRanda] | microCosm | PicTar
- DIANA-miRPath
- Royal Hospital for Women (RWH)
- School of Medical Sciences (SoMS)
- HSA Biobank
- Bioanalytical Mass Spectrometry Facility (BMSF)
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 20) Embryology Implantation microRNA. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Implantation_microRNA
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G