Book - The Early Embryology of the Chick 12
| Embryology - 18 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Patten BM. The Early Embryology of the Chick. (1920) Philadelphia: P. Blakiston's Son and Co.
| Online Editor |
|---|
| This historic 1920 paper by Bradley Patten described the understanding of chicken development. If like me you are interested in development, then these historic embryology textbooks are fascinating in the detail and interpretation of embryology at that given point in time. As with all historic texts, terminology and developmental descriptions may differ from our current understanding. There may also be errors in transcription or interpretation from the original text. Currently only the text has been made available online, figures will be added at a later date. My thanks to the Internet Archive for making the original scanned book available.
By the same author: Patten BM. Developmental defects at the foramen ovale. (1938) Am J Pathol. 14(2):135-162. PMID 19970381 Those interested in historic chicken development should also see the earlier text The Elements of Embryology (1883). Foster M. Balfour FM. Sedgwick A. and Heape W. The Elements of Embryology (1883) Vol. 1. (2nd ed.). London: Macmillan and Co.
Modern Notes |
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Structure of Chicks from Fifty to Fifty-five Hours of Incubation
I. External Features
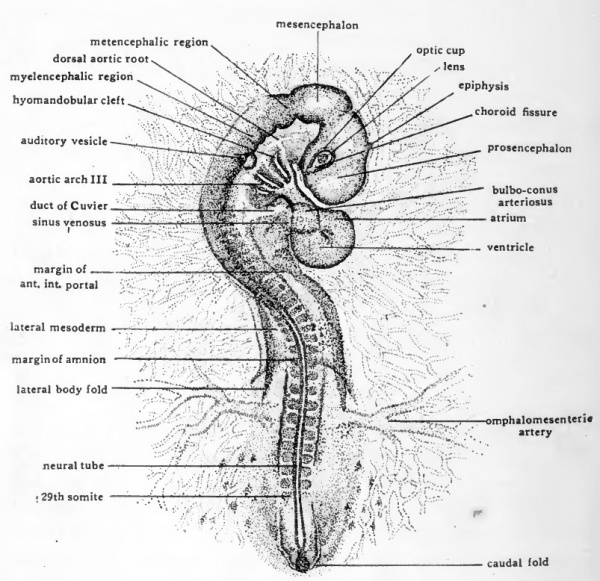
In chicks which have been incubated from 50 to 55 hours (Fig. 34) the entire head region has been freed from the yolk by the progress caudad of the sub-cephalic fold. Torsion has involved the whole anterior half of the embryo and is completed in the cephalic region, so that the head now lies left side down on the yolk. The posterior half of the embryo is still in its original position, ventral surface prone on the yolk. At the extreme posterior end, the beginning of the caudal fold marks off the tail region of the embryo from the extra-embryonic membranes. The head fold of the amnion has progressed caudad, together with the lateral amniotic folds impocketing the embryo nearly to the level of the omphalomesenteric arteries.
The cranial flexure, which was seen beginning in chicks of about 38 hours, has increased rapidly until at this stage the brain is bent nearly double on itself. The axis of the bending being in the mid-brain region, the mesencephalon comes to be the most anteriorly located part of the head and the prosencephalon and myelencephalon lie opposite each other, ventral surface to ventral surface (Fig. 34). The original anterior end ot the prosencephalon is thus brought in close proximity to the heart, and the optic vesicles and the auditory vesicles are brought opposite each other at nearly the same antero-posterior level.
Fig. 34. Dextro-dorsal view ( X 14) of entire embryo of 29 somites (about 55 hours incubation).
At this stage flexion has involved the body farther caudally as well as in the brain region. It is especially marked at about the level of the heart in the region of transition from myelencephalon to spinal cord. Since this is the future neck region of the embryo the flexure at this level is known as the cervical flexure (Fig. 34).
II. The Nervous System
Growth of the Telencephalic Region
The completion of torsion in the head region causes rapid changes in the configuration of the brain as seen in entire embryos from 40 to 50 hours of incubation. The same fundamental regions can, however, be identified throughout this range of development. The anterior part of the brain has undergone rapid enlargement. A slight constriction in the dorsal wall (Fig. 35) indicates the impending division of the prosencephalon into telencephalon and diencephalon. Except for its considerable increase in size no important changes have taken place in the telencephalic region.
The Epiphysis
In the mid-dorsal wall of the diencephalic region a small evagination has appeared. This evagination is the epiphysis (Fig. 34 and 35). It is destined to become differentiated into the pineal gland of the adult.
The Infundibulum and Rathke's Pocket
In the flo'br of the diencephalon the infundibular depression has become deepened and lies close to a newly formed ectodermal invagination known as Rathke's pocket (Fig. 35). The epithelium of Rathke's pocket is destined to be separated from the superficial ectoderm and to become permanently associated with the infundibular portion of the diencephalon to form the hypophysis or pituitary body.
The Optic Vesicles
The optic vesicles have undergone changes which completely alter their appearance. In 33-hour chicks they are spheroidal vesicles connected by broad stalks with the Wteral walls of the diencephalon (Fig. 21). At this stage the lumen of each optic vesicle (opticoele) is widely con: tinuous with the lumen of the prosencephalon (prosocoele) (Fig. 28, A). The constriction of the optic stalk which begins to be apparent in 38-hour embryos (Fig. 22) is much more marked in 55-hour chicks.
The most striking and important advance in their development is the invagination of the distal ends of the single-walled optic vesicles to form double walled optic cups (Fig. 36, B). The concavities of the cups are directed laterally. Mesially the cups are continuous with the ventro-lateral walls of the diencephalic region of the original prosencephalon over the narrowed optic stalks. The invaginated layer of the optic cup is termed the sensory layer because it is destined to give rise to the sensory layer of the retina. The layer against which the sensory layer comes to lie after its invagination is termed the pigment layer because it gives rise to the pigmented layer of the retina. The double-walled cups formed by invagination, are also termed secondary optic vesicles in distinction to primary optic vesicles, as they are called before the invagination. The formerly capacious lumen of the primary optic vesicle is practically obliterated in the formation of the optic cup. What remains of the primary opticoele is now but a narrow space between the sensory and the pigment layers of the retina (Fig. 36, B). Later when these two layers fuse this space is entirely obliterated.
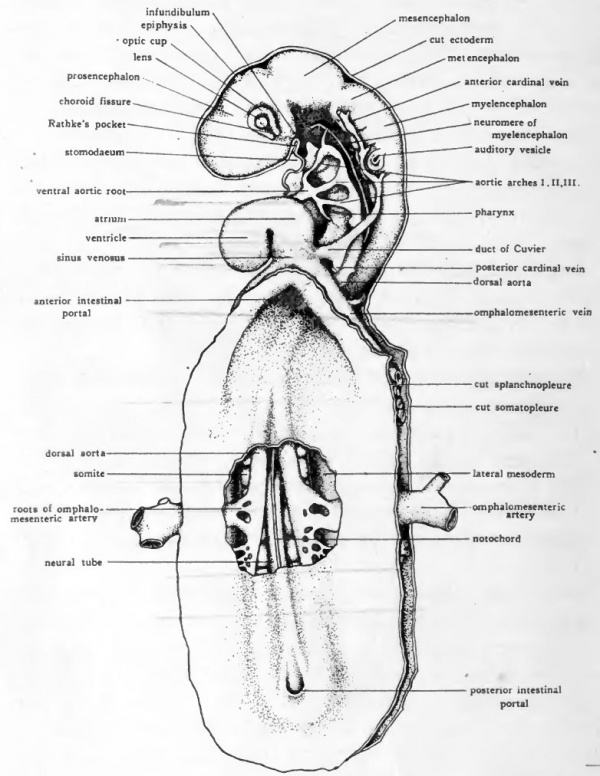
Fig. 35. Diagram of dissection of chick of about 50 hours. (Modified from Prentiss.)
- The spianchnopleure of the yolk-sac cephalic to the anterior intestinal portal, the ectoderm of the left side of the head, and the mesoderm in the pericardial region have been dissected away. A window has been cut in the spianchnopleure of the dorsal wall of the mid-gut to show the origin of the omphalomesenteric arteries.
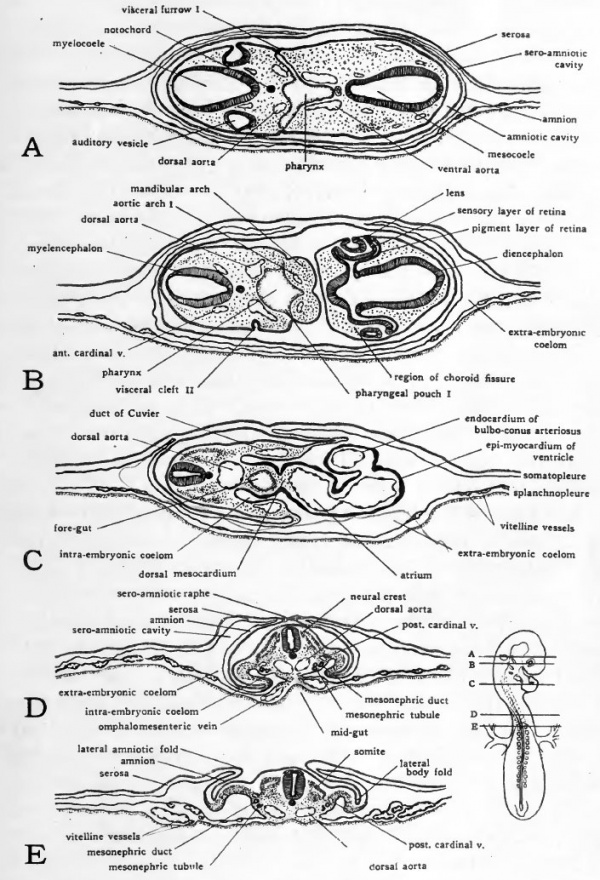
Fig. 36. Diagrams of transverse sections of 55-hour (30-somiite) chick. The location of the sections is indicated on an outline sketch of the entire embryo.
While the secondary optic vesicles are usually spoken of as the optic cups, they are not complete cups. The invagination which gives rise to the secondary optic vesicles, instead of beginning at the most lateral point in the primary optic vesicles, begins at a point somewhat toward their ventral surface and is directed mesiodorsad. As a result the optic cups are formed without any lip on their ventral aspect. They may be likened to cups with a segment broken out of one side. This gap in the optic cup is the choroid fissure (Fig. 35). In Figure 36, B, a section is shown which passes through the head of the embryo on a slight slant so that the right optic cup, being cut to one side of the choroid fissure appears complete while the left optic cup being cut in the region of the fissure shows no ventral lip.
The infolding process by which the optic cups are formed from the primary optic vesicles is continued to the region of the optic stalks. As a result the optic stalks are infolded so that their ventral surfaces become grooved. Later in development the optic nerves and blood vessels come to lie in the grooves thus formed in the optic stalks.
The Lens
The lens of the eye arises independently of the optic vesicles, from the superficial ectoderm of the head. The first indications of lens formation appear in chicks of about 40 hours as local thickenings of the ectoderm immediately overlying the optic vesicles. These placodes of thickened ectoderm sink below the general level of the surface of the head to form small vesicles which extend into the secondary optic vesicles. Their opening to the surface is rapidly constricted and eventually they are disconnected altogether from the superficial ectoderm. At this stage the opening to the outside still persists although it is very small (Fig. 36, B, right eye). In sections which do not pass directly through the opening, the lens vesicle appears completely separated from the overlying ectoderm (Fig. 36, B, left eye).
The derivation of the lens from a placode of thickened epithelium which sinks below the general surface, and eventually loses its connection with the superficial ectoderm, is strikingly similar to the early steps in the derivation of the auditory vesicle. But these primordia once separated from the ectoderm follow divergent lines of differentiation leading to adult conditions which are structurally and functionally totally unlike. The origin of these two structures from cell groups similarly folded off from the same germ layer, but which once established undergo each their own characteristic differentiation, exemplifies a sequence of events so characteristic of developmental processes in general as to call for at least a comment in passing.
The Posterior Part of the Brain and the Cord Region of the Neural Tube
Caudal to the diencephalon the brain shows no great change as compared with the last stages considered. The mesencephalon is somewhat enlarged and the constrictions separating it from the diencephalon cephalically and the metencephalon caudally are more sharply marked. The metencephalon is more clearly marked off from the myelencephalon and its roof is beginning to show thickening. In the myelencephalon the neuromeric constrictions are still evident in the ventral and lateral walls (Figs. 34 and 35). The dorsal wall has become much thinner than the ventral and lateral walls (Fig. 36, A and B) and shows no trace of division between the neuromeres.
In the cord region of the neural tube the lateral walls have become thickened at the expense of the lumen so that the neural canal appears slit-like in sections of embryos of this age (Fig. 36, E) rather than eliptical as it is immediately after the closure of the neural folds. At this stage the closure of the neural tube is completed throughout its entire length. The last regions to close were at the cephalic and caudal ends of the neural groove. In younger stages where they remained open these regions were known as the anterior neuropore and the sinus rhomboidalis, respectively.
The Neural Crest
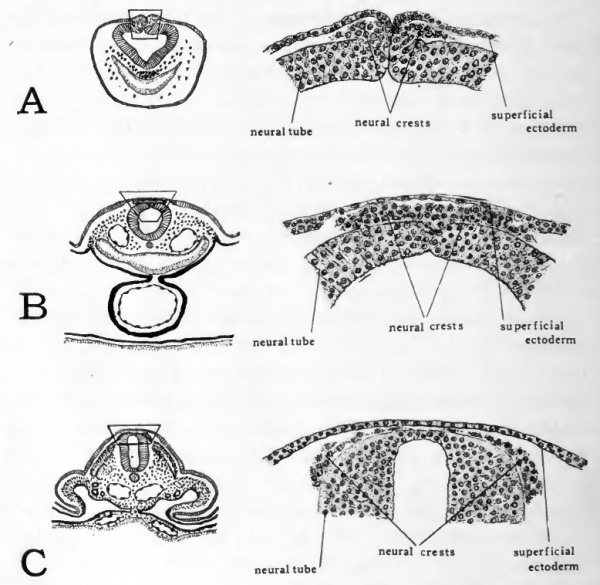
In the closure of the nairal tube the superficial ectoderm which at first lay on either side of the neural groove, continuous with the neural plate ectoderm, becomes fused in the mid-line and separated from the neural plate to constitute an unbroken ectodermal covering (Cf. Figs. 17, B, and 28, B). At the same time the lateral margins of the neural plate become fused to complete the neural tube. There are cells lying originally at Ihe edges of the neural folds which are not involved in the fusion of either the superficial ectoderm or the neural plate. These cells form a pair of longitudinal aggregations extending one on either side of the mid-dorsal line in the angles between the superficial ectoderm and the neural tube (Fig. 37, A). With the fusion of the edges of the neural folds to complete the neural tube, and the fusion of the superficial ectoderm dorsal to the neural tube, these two longitudinal cell masses become for a time confluent in the mid-line (Fig. 37, jB). But because this aggregation of cells arises from paired components and soon again separates into right and left parts it is to be considered as potentially paired. On account of its position dorsal to the neural tube it is known as the neural crest.
Fig. 37. Drawings from transverse sections to show origin of neural crest cells. The location of the area drawn is indicated on the small sketch to the left of each drawing.
- A, anterior rhombencephalic region of 30-hour chick; B, posterior rhombencephalic region of 36-hour chick; C, mid-dorsal region of cord in 55-hour chick.
The neural crest should not be confused with the margin of the neural fold with which it is associated before the closure of the neural tube. The margin of the neural fold involves cells which go into the superficial ectoderm and into the neural tube, as well as those which are concerned in the formation of the neural crest.
When first established the neural crest is continuous anteroposteriorly. As development proceeds, the cells of the neural crest migrate ventro-laterally on either side of the spinal cord (Fig. 37, C), and at the same time become segmen tally clustered. The segmen tally arranged cell groups thus derived from the neural crest give rise to the dorsal root ganglia of the spinal nerves, and in the head region to the ganglia of the sensory cranial nerves. (For a later stage of the dorsal root ganglia see Figure 44.)
III. The Digestive Tract
The Fore-gut
The manner in which the three primary regions of the gut-tract are established has already been considered in a general way (see Chapter XI and Fig. 31). In 50 to 55-hour chicks the fore-gut has acquired considerable length. It extends from the anterior intestinal portal cephalad almost to the infundibulum (Fig. 35).
As the first region of the tract to be established, the fore-gut is naturally the most advanced in differentiation. We can already recognize a pharyngeal and an oesophageal portion. The pharyngeal region lies ventral to the myelencephalon and is encircled by the aortic arches (Fig. 35). The pharynx is somewhat flattened dorso-ventrally and has a considerably larger lumen than the oesophageal part of the fore-gut (Cf . Fig. 36, B and C).
The Stomodaemn
There is at this stage no mouth opening into the pharynx. However, the location where the opening will be formed is indicated by the approximation of a ventral outpocketing near the anterior end of the pharynx, to a depression formed in the adjacent ectoderm of the ventral surface of the head (Fig. 35). The ectodermal depression, known as the stomodaeum, deepens until its floor lies in contact with the entoderm of the pharyngeal out-pocketing (Fig. 35). The thin layer of tissue formed by the apposition of the stomodaeal ectoderm to the pharyngeal entoderm is known as the oral plate.
Later in development the oral plate, breaks through bringing the stomodaeum and the pharynx into open communication. Growth of surrounding structures deepens the original stomodaeal depression, and it becomes the oral cavity. The region of the oral plate in the embryo becomes, in the adult, the region of transition from oral cavity to pharynx.
The Pre-oral Gut
It will be noted by reference to Figure 35 that the oral opening is not established at the extreme cephalic end of the pharynx. The part of the pharynx which extends cephalic to the mouth opening is known as the pre-oral gut. After the rupture of the oral plate, the pre-oral gut eventually disappears, but an indication of it persists for a time as a small diverticulum termed Seessell's pocket(Cf.Figs. 35 and 43).
The Mid-gut
Although the mid-gut is still the most extensive of the three primary divisions of the digestive tract, it presents little of interest. It is nothing more than a region where the gut still lies open to the yolk. It does not have even a fixed identity. As fast as any part of the mid-gut acquires a ventral wall by the closing-in process involved in the progress of the subcephalic and subcaudal folds it ceases to be mid-gut and becomes fore-gut or hind-gut. Differentiation and local specilizations appear in the digestive tract only in regions which have ceased to be mid-gut.
The Hind-gut
The hind-gut first appears in embryos of about 55 hours (Fig. 35). The method of its formation is similar to that by which the fore-gut was established. The sub-caudal fold undercuts the tail region and walls off a gut pocket. The hind-gut is lengthened at the expense of the mid-gut as the sub-caudal fold progresses cephalad and is also lengthened by its own growth caudad. It shows no local specializations until later in development.
IV. The Visceral Clefts and Visceral Arches
At this stage the chick embryo has unmistakable visceral arches and visceral clefts. Although only transitory, they are morphologically of great importance not only from the comparative view point, and because of their significance as structures exemplifying recapitulation, but also because of their participation in the formation of the embryonic arterial system, of some of the ductless glands, of the eustachian tube, and of the face and jaws.
The visceral clefts are formed by the meeting of ectodermal depressions, the visceral furrows, with divericula from the lateral walls of the pharynx, the pharyngeal pouches. During most of the time the visceral furrows are conspicuous features in entire embryos, they may be seen in sections to be closed by a thin double layer of tissue composed of the ectoderm of the floor of the visceral furrow and the entoderm at the distal extremity of' the pharyngeal pouch (Fig. 36, A). The breaking through of this thin double layer of tissue brings the pharyngeal pouches into communication with the visceral furrows thereby estabhshing open visceral clefts. In birds an open condition of the clefts is transitory. In the chick the most posterior of the series of clefts never becomes open. Although some of the clefts never become open and others open for but a short time the term cleft is usually used to designate these structures which are potentially clefts, whether open or not.
The position of the visceral clefts is best seen in entire embryos. They are commonly designated by number beginning with the first cleft posterior to the mouth and proceeding caudal. The first post-oral cleft appears earliest in development and is discernible at about 46 hours of incubation. Visceral cleft 11 appears soon after, and by 50 to 55 hours three clefts have been formed (Fig. 34).
Between adjacent visceral clefts, the lateral body walls about the pharynx are thickened. Each of these lateral thickenings in the mid- ventral line meets and merges with the corresponding thickening of the opposite side of the body. Thus the pharynx is encompassed laterally and ventrally by a series of arch-like thickenings, the visceral or gill arches. The visceral arches like the visceral clefts are designated by number, beginning at the anterior end of the series. Visceral arch 1 lies cephalic to the first post-oral cleft, between it and the mouth region. Because of the part it plays in the formation of the mandible it is also designated as the mandibular arch. Visceral arch II is frequently termed the hyoid arch, and visceral cleft 1, because of its position between the mandibular and hyoid arches, is known as the hyomandibular cleft. Posterior to the hyoid arch the visceral arches and clefts are ordinarily designated by their post-oral numbers only.
There are other structures which are just beginning to be differentiated in the pharyngeal region and fore-gut of embryos of this stage, but it seems better to consider them in connection with later stages when their significance will be more readily grasped.
V. The Circulatory System
The Heart
In embryos of 30 to 40 hours incubation we traced the expansion of the heart till it was bent to the right of the embryo in the form of a U-shaped tube (Figs. 19, 21, 23). The disappearance of the dorsal mesocardium except at its posterior end, leaves the mid-region of the heart lying unattached and extending to the right, into the pericardial region of the coelom. The heart is fixed with reference to the body of the embryo at its cephalic end where the ventral aortic roots lie embedded beneath the floor of the pharynx, and caudally in the sinus region where it is attached by the omphalomesenteric veins, by the ducts of Cuvier, and by the persistent portion of the dorsal mesocardium.
During the period between 30 and 55 hours of incubation the heart itself is growing more rapidly than is the body of the embryo in the region where the heart lies. Since its cephalic and caudal ends are fixed, the unattached mid-region of the heart becomes at first U-shaped and then twisted on itself to form a loop. The atrial region of the heart is forced somewhat to the left, and the conus region is thrown across the atrial region by being twisted to the right and dorsally. The ventricular region constitutes the loop proper (Cf. Figs. 22, 29 and 34). This twisting process reverses the original cephalocaudal relations of the atrial and ventricular regions)/ Before the twisting, the atrial region of the heart was caudal to the ventricular region as it is in the adult fish heart. In the twisting of the heart the atrial region, by reason of its association with the fixed sinus region of the heart, undergoes relativeh' little change in position. ^The ventricular region is carried over the dextral side of the atrium and comes to lie caudal to it, thus arriving in the relative position it occupies in the adult heart.
The bending and subsequent twisting of the heartlead toward its division into separate chambers. As yet, however, no indication of the actual partitioning off of the heart is apparent. It is still essentially a tubular organ through which the blood passes directly without any division into separate channels or currents.
The Aortic Arches
In 33 to 38 hour chicks the ventral aortas communicate with the dorsal aortae over a single pair of aortic arches which bend around the anterior end of the pharynx (Figs. 23 and 24) . With the formation of the visceral arches new aortic arches appear. The original pair of aortic arches comes to lie in the mandibular arch, and the new aortic arches are formed caudal to the first pair, one pair in each visceral arch. In chicks of 50 to 55 hours, three pairs of aortic arches have been established and a fourth is usually beginning to form (Figs. 34, 35, and 36, A and J5).
The Fusion of the Dorsal Aortae
The dorsal aortae arise as vessels paired throughout their entire length (Fig. 23). As development progresses they fuse in the mid-line to form the unpaired dorsal aorta familiar in adult anatomy. This fusion takes place first at about the level of the sinus venosus and progresses thence cephalad and caudad. Cephalically it never extends to the pharyngeal region. Caudally the whole length of the aorta is eventually involved. At this stage the fusion has progressed caudad to about the level of the 14th somite (Figs. 34, 35, 36).
The Cardinal and Omphalomesenteric Vessels
The relationships of the cardinal veins and the omphalomesenteric vessels are little changed from the conditions in 40 to 50 hour chicks. The posterior cardinals have elongated, keeping pace with the caudal progress of differentiation in the mesoderm. They lie just dorsal to the intermediate mesoderm in the angle formed between it and the somites (Fig. 36, D). The entrance of the omphalomesenteric veins into the sinus venosus, and the origin of the omphalomesenteric arteries from the dorsal aortae show little change from conditions familiar from the study of younger embryos.
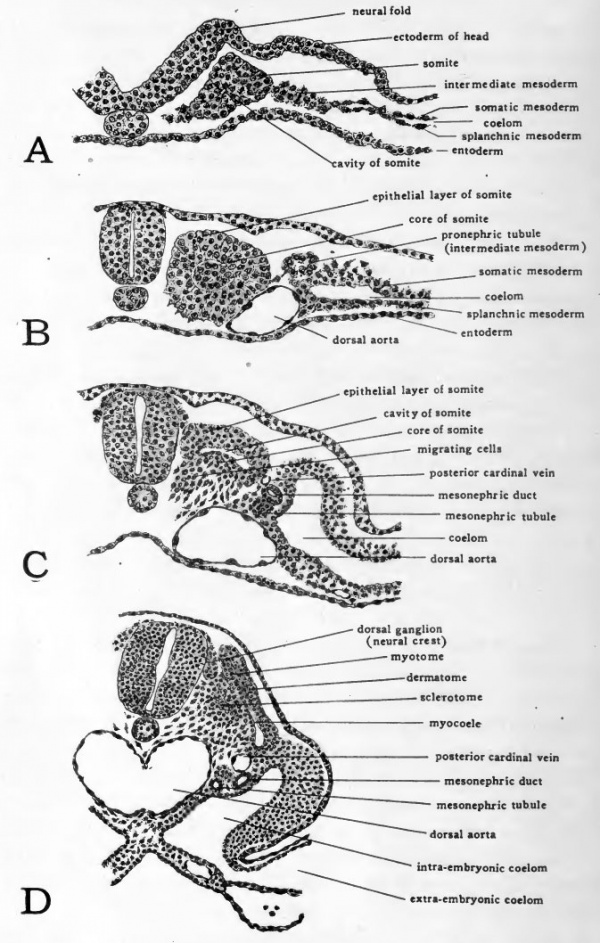
VI. The Differentiation of the Somites
When the somites are first formed they consist of a nearly solid mass of cells derived from the dorsal mesoderm (Fig. 37, A). The cells composing them show a more or less radial arrangement. In the center of the somite a cavity is usually discernible. This cavity is at first extremely minute. In somites which have been recently formed it may be altogether wanting.
Fig. 38. Drawings from transverse sections to show the differentiation of the somites.
- A, second somite of 4-somite chick; B, ninth somite of 12-somite chick; C, twentieth somite of 30-somite chick; D, seventeenth somite of 33-somite chick.
As the somite becomes more sharply marked off the radial arrangement of the outer zone of cells appears more definitely (Fig. 38, B) The boundaries of the central cavity are considerably extended but its lumen is almost completely filled by a core of irregularly arranged cells. In sections which pass through the middle of the somite, this central core of cells is seen to arise from the lateral wall of the somite where it is continuous with the intermediate mesoderm.
A little later in development the outer zone of cells on the ventro-mesial face of the somite loses its originally definite boundaries and becomes merged with the central core of cells. This ill-defined cell aggregation, known as the sclerotome, becomes mesenchymal in characteristics, and extends ventromesiad from the somite of either side toward the notochord (Fig. 38, C and D). The cells of the sclerotomes of either side continue to converge about the notochord and later take part in the formation of the axial skeleton.
During the formation of the sclerotome the dorsal part of the original outer cell-zone of the somite has maintained its definite boundaries and epithelial characteristics. The part of this outer zone which lies parallel to the ectoderm is known as the dermatome (Fig. 38, C and D). It later becomes associated with the ectoderm and forms the deeper layers of the integument, the ectoderm giving rise to the epithelial layer only.
The dorso-mesial portion of the outer zone of the somite becomes the myotome. It is folded somewhat laterad from its original position next to the neural tube (Fig. 38, C) and comes to lie ventro-mesial to the dermatome and parallel to it (Fig. 38, D). (A later stage in the differentiation of the somite is shown in Figure 44). The portion of the original cavity which persists for a time between the dermatome and myotome is termed the myocoele. The myotomes undergo the most extensive growth of any of the parts of the somite, giving rise eventually to the entire skeletal musculature of the body.
Vll. The Urinary System
In the section-diagrams of Figure 36, D and E, certain parts of the urinary system which have been established in chicks of 50 to 55 hours will be found located and labeled. The urinary system is relatively late in becoming differentiated. Only a few of the early steps in its formation can at this time be made out. Many structures which later become of great importance are not represented even by primordial cell aggregations. Except for those well grounded in comparative anatomy, any logical discussion of the structures which have appeared must anticipate much that occurs later in development. Consideration of the mode of origin and significance of the nephric organs appearing at this stage has, therefore, been deferred.
- Next: Day 3 to 4
The Early Embryology of the Chick: Introduction | Gametes and Fertilization | Segmentation | Entoderm | Primitive Streak and Mesoderm | Primitive Streak to Somites | 24 Hours | 24 to 33 Hours | 33 to 39 Hours | 40 to 50 Hours | Extra-embryonic Membranes | 50 to 55 Hours | Day 3 to 4 | References | Figures | Site links: Embryology History | Chicken Development
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 18) Embryology Book - The Early Embryology of the Chick 12. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_The_Early_Embryology_of_the_Chick_12
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G