Book - The Early Embryology of the Chick 11
| Embryology - 14 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Patten BM. The Early Embryology of the Chick. (1920) Philadelphia: P. Blakiston's Son and Co.
| Online Editor |
|---|
| This historic 1920 paper by Bradley Patten described the understanding of chicken development. If like me you are interested in development, then these historic embryology textbooks are fascinating in the detail and interpretation of embryology at that given point in time. As with all historic texts, terminology and developmental descriptions may differ from our current understanding. There may also be errors in transcription or interpretation from the original text. Currently only the text has been made available online, figures will be added at a later date. My thanks to the Internet Archive for making the original scanned book available.
By the same author: Patten BM. Developmental defects at the foramen ovale. (1938) Am J Pathol. 14(2):135-162. PMID 19970381 Those interested in historic chicken development should also see the earlier text The Elements of Embryology (1883). Foster M. Balfour FM. Sedgwick A. and Heape W. The Elements of Embryology (1883) Vol. 1. (2nd ed.). London: Macmillan and Co.
Modern Notes |
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Extra-embryonic Membranes
The Folding off of the Body of the Embryo
In bird embryos the somatopleure and splanchnopleure extend over the yolk peripherally, beyond the region where the body of the embryo is being formed. Distal to the body of the embryo the layers are termed extra-embryonic. At first the body of the chick has no definite boundaries and consequently embryonic and extraembryonic layers are directly continuous without there being any definite boundary at which we may say one ends and the other begins. As the body of the embryo takes form, a series of folds develop about it, undercut it, and finally nearly separate it from the yolk. The folds which thus definitely estabhsh the boundaries between intra-embryonic and extra-embryonic regions are known as the limiting body folds or simply the body folds.
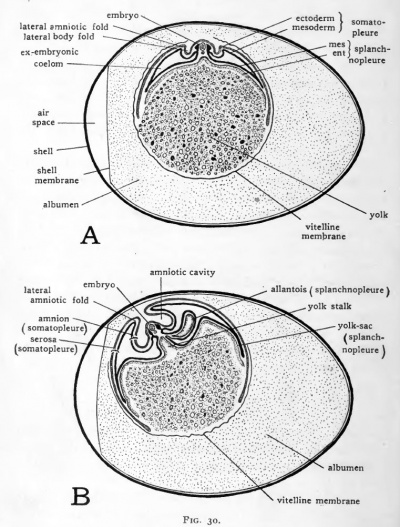
The first of the body folds to appear is the fold which marks the boundary of the head. By the end of the first day of incubation the head has grown anteriorly and the fold originally bounding it appears to have undercut and separated it anteriorly from the blastoderm (Figs. 15 and 17, ). The cephalic limiting fold at this stage is crescentic, concave caudally. As this fold continues to progress caudad, its posterior extremities become continuous with folds which develop along either side of the embryo. Because of the fact that these folds bound the body of the embryo laterally, they are known as the lateral body folds (lateral limiting sulci). The lateral body folds, at first shallow (Fig. 28, Z)) become deeper, undercutting the body of the embryo from either side and further separating it from the yolk (Fig. 36, and Fig. 30).
During the third day a fold appears bounding the posterior region of the embryo (Fig. 31, C). This caudal fold undercuts the tail of the embryo forming a subcaudal pocket just as the sub-cephalic fold undercuts the head. The combined effect of the development of the sub-cephalic, lateral body, and the subcaudal folds is to constrict off the embryo more and more from the yolk (Figs. 30 and 32). These folds which establish the, contour of the embryo indicate at the same time the boundary between the tissues which are built into the body of the embryo, and the so-called extra-embryonic tissues which serve temporary purposes during development but are not incorporated in the structure of the adult body.
The Establishment of the Yolk-sac and the Delimitation of the Embryonic Gut
The extra-embryonic membranes of the chick are four in number, the yolk-sac, the amnion, the serosa and the allantois. The yolk-sac is the first of these to make its appearance. The splanchnopleure of the chick instead of forming a closed gut, as happens in forms with little yolk, grows over the yolk surface. The primitive gut has a cellular wall dorsally only, while the yolk acts as a temporary floor (Fig. 31, .4). The extra-embryonic extension of the splanchnopleure eventually forms a sac-like investment for the yolk (Figs. 30 and 32).
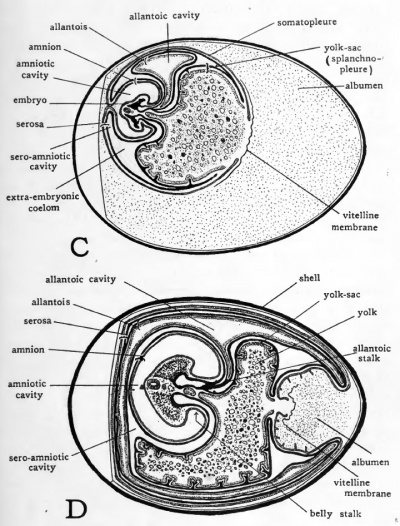
Concomitant with the spreading of the extra-embryonic splanchnopleure about the yolk, the intra-embryonic splanchnopleure is undergoing a series of changes which result in the establishment of a completely walled gut in the body of the embryo. The interrelations of the various steps in the formation of the gut and of the yolk-sac make it necessary to repeat some points and anticipate other points concerning the formation of the gut, in order that their relation to yolk-sac formation may not be overlooked.
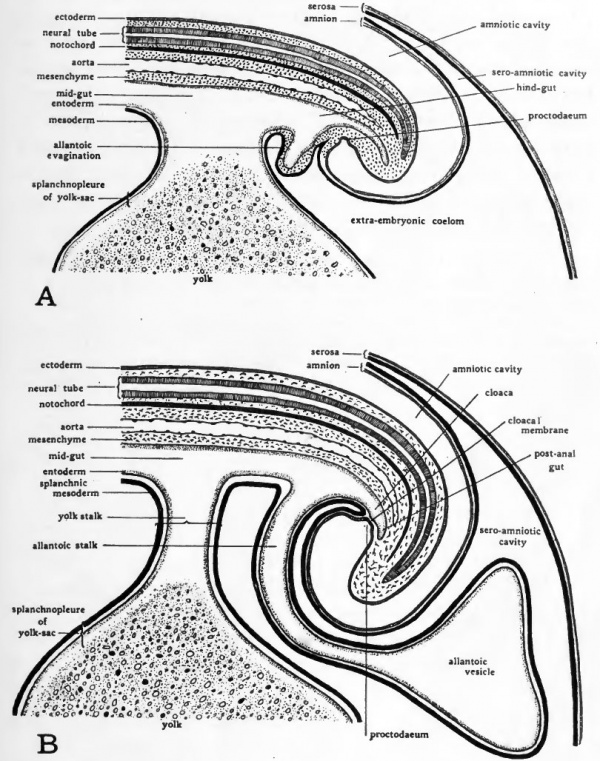
It will be recalled that the first part of the primitive gut to acquire a cellular floor is its cephalic region. The same folding process by which the head is separated from the blastoderm involves the entoderm of the gut. The part of the primitive gut which acquires a floor as the sub-cephalic fold progresses caudad is termed the fore-gut (Fig. 31, B). During the third day of incubation the caudal fold undercuts the posterior end of the embryo. The splanchnopleure of the gut is involved in the progress of the sub-caudal fold so that a hind-gut is established in a manner analogous to the formation of the foregut (Fig. 31, C). The part of the gut which still remains open to the yolk is known as the mid-gut. As the embryo is constricted off from the yolk by the progress of the sub-cephalic and sub-caudal folds, the fore-gut and hind-gut are increased in extent at the expense of the mid-gut. The mid-gut is finally diminished until it opens ventrally by a small aperture which 6-^ares out, like an inverted funnel, into the yolk-sac (Fig. 31, D). This opening is the yolk duct and its wall constitutes the yolk stalk.
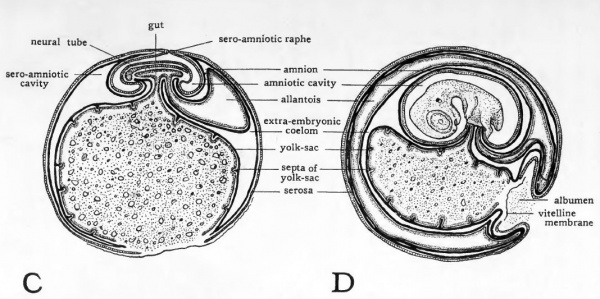
Fig. 30. Schematic diagrams to show the extra-embryonic membranes of the chick. (After Duval.)
- The diagrams represent longitudinal sections through the entire egg. The body of the embryo, being oriented approximately at right angles to the long-axis of the egg, is cut transversely.
|
|
The walls of the yolk-sac are still continuous with the walls, of the gut along the constricted yolk-stalk thus formed, but the boundary between the intra-embryonic splanchnopleure of the gut and the extra-embryonic splanchnopleure of the yolk-sac can now be established definitely at the yolk-stalk.
As the neck of the yolk-sac is constricted the omphalomesenteric arteries and omphalomesenteric veins, caught in the same series of foldings, are brought together and traverse the yolkstalk side by side. The vascular network in the splanchnopleure of the yolk-sac which in young chicks was seen spreading over the yolk eventually nearly encompasses it. The embryo's store of food material thus comes to be suspended from the gut of the mid-body region in a sac provided with a circulatory arc of its own, the vitelline arc. Apparently no yolk passes directly through the yolk-duct into the intestine. Absorption of the yolk is effected by the epithelium of the yolk-sac and the food material is transferred to the embryo by the vitelline circulation. In older embryos (Fig. 30, C and D) the epithelium of the yolk-sac undergoes a series of foldings which greatly increase its surface area and thereby the amount of absorption it can accomplish.
During development the albumen loses water, becomes more viscid , and rapidly decreases in bulk. The growth of the allantois, an extra-embryonic structure which we have yet to consider, forces the albumen toward the distal end of the yolksac (Fig. 30, D). The manner in which the albumen is encompassed between the yolk-sac and folds of the allantois and serosa belong to later stages of development than those with which we are concerned. Suffice it to say that the albumen like the yolk, is surrounded by extra-embryonic membranes by which it is absorbed and transferred over the extra-embryonic circulation to the embryo.
Fig. 31. Schematic longitudinal-section diagrams of the chick showing four stages in the formation of the gut tract. The embryos are represented as unaffected by torsion.
- A, chick toward the end of the first day of incubation; no regional differentiation of primitive gut is as yet apparent.
- B, toward the end of the second day; fore-gut established.
- C, chick of about three days; fore-gut, mid-gut and hindgut established.
- D, chick of about four days; fore-gut and hind-gut increased in length at expense of mid-gut; yolk-stalk formed.
Toward the end of the period of incubation, usually on the 19th day, the remains of the yolk-sac are enclosed within the body walls of the embryo. After its inclusion in the embryo both the wall and the remaining contents of the yolk-sac rapidly disappear, their absorption being practically completed in the first six days after hatching.
The Amnion and the Serosa
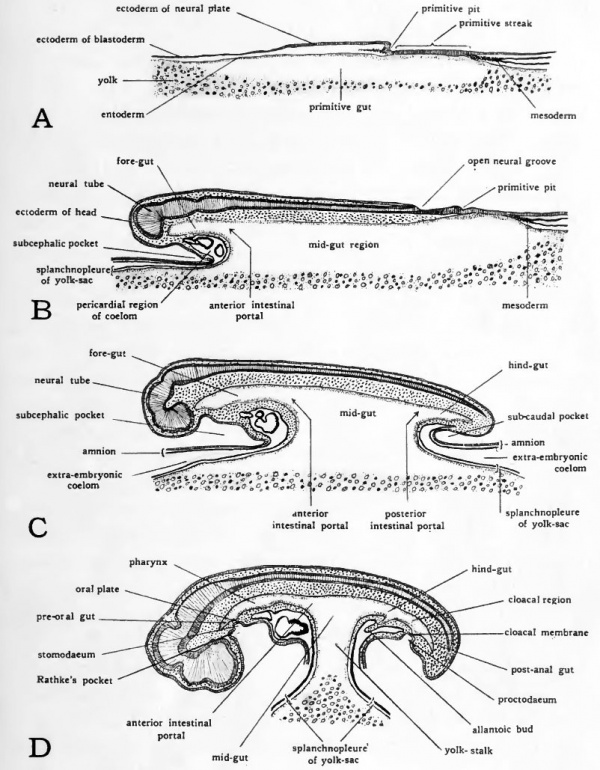
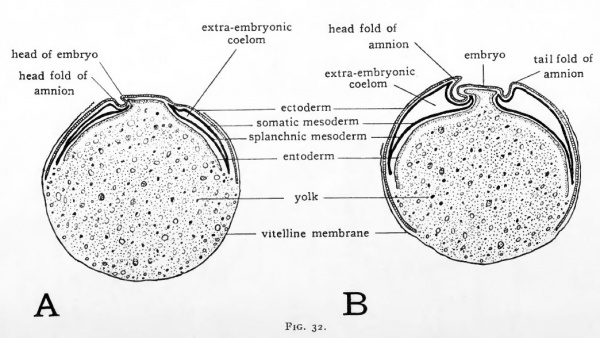
The amnion and the serosa are so closely associated in their origin that they must be con, sidered together. Both are derived from the extra-embryonic Somatopleure. The amnion encloses the embryo as a saccular investment and the cavity thus formed between the amnion and the embryo becomes filled with a watery fluid. Suspended I in this amniotic fluid, the embryo is free to change its shape and position, and external pressure upon it is equahzed. MusI cle fibers develop in the amnion, which by their contraction gently agitate the amniotic fluid. The movement thus imparted to the embryo apparently aids in keeping it free and 1 preventing adhesions and resultant malformations. The first indication of amnion formation appears in chicks of about 30 hours incubation. The head of the embryo sinks into the yolk somewhat, and at the same time the extra-embryonic somatopleure anterior to the head is thrown into a fold, the head fold of the amnion (Fig. 32, ^). In dorsal aspect the margin of this fold is crescentic in shape with its concavity directed toward the head of the embryo. The head fold of the amnion must not be confused with the sub-cephalic fold which arises earlier in development and undercuts the head.
As the embryo increases in length its head grows anteriorly into the amniotic fold. Growth in the somatopleure itself tends to extend the amniotic fold caudad over the head of the embryo ([[#Fig032|Fig. 32, A). By continuation of these two growth processes the head soon comes to lie in a double walled pocket of extra-embryonic somatopleure which covers the head like a cap (Fig. 29). The free edge of the amniotic pocket retains its original crescentic shape as, in its progress caudad, it covers more and more of the embryo.
The caudally-directed limbs of the head fold of the amnion are continued posteriorly along either side of the embryo as the lateral amniotic folds. The lateral folds of the amnion grow dorso-mesiad, eventually meeting in the mid-line dorsal to the embryo (Fig. 30, A-C).
During the third day, the tail-fold of the amnion develops about the caudal region of the embryo. Its manner of development is similar to that of the head fold of the amnion but its direction of growth is reversed, its concavity being directed anteriorly and its progression being cephalad (Fig. )
Continued growth of the head, lateral, and tail folds of the amnion results in their meeting above the embryo. At the point where the folds meet, they become fused in a scar-like thickening termed the amniotic raphe (sero-amniotic raphe). (Fig. 32, C). The way in which the somatopleure has been folded about the embryo leaves the amniotic cavity completely lined by ectoderm which is continuous with the superficial ectoderm of the embryo at the region where the yolk-stalk enters the body (Fig. 30, D).
All the amniotic folds involve doubling the somatopleure on itself. Only the inner layer of the somatopleuric fold is involved in the formation of the amniotic cavity. The outer layer of somatopleure becomes the serosa (Fig. 30, ). Thai cavity between serosa and amnion (sero-amniotic cavity) is part of the extra-embryonic coelom. The continuity of the extra-embryonic coelom with the intra-embryonic coelom is most apparent in early stages (Fig. 30, A and B). They remain, however, in open communication in the yolk-stalk region until relatively late in development.
The rapid peripheral growth of the somatopleure carries the V serosa about the yolk-sac, which it eventually envelops. The albumen-sac also is surrounded by folds of serosa, and the allantois after its establishment develops within the serosa, between it and the amnion. Thus the serosa eventually encompasses the embryo itself and all the other extra-embryonic membranes. The relationships of the serosa and allantois and the functional significance of the serosa will be taken up after the allantois has been considered.
Fig. 32.
The Allantois
The allantois differs from the amnion and i serosa in that it arises primarily within the body of the embryo, Its proximal portion is intra-embryonic throughout development. Its distal portion, however, is carried outside the confines of the intra-embryonic coelom and becomes associated with the other extra-embryonic membranes. Like the other extra|embryonic membranes the distal portion of the allantois functions only during the incubation period and is not incorporated into the structure of the adult body.
The allantois first appears late in the third day of incubation. It rises as a diverticulum from the ventral wall of the hind-gut and its walls are, therefore, splanchnopleure. Its relationships to structures within the embryo will be better understood when chicks of three and four days incubation have been studied, but its general location can be appreciated from the schematic diagrams of Figures 32 and 33.
During the fourth day of development the allantois pushes out of the body of the embryo into the extra-embryonic coelom. Its proximal portion lies parallel to the yolk-stalk and just caudal to it. When the distal portion of the allantois has grown clear of the embryo it becomes enlarged (Fig. 32, C). Its narrow proximal portion is known as the allantoic stalk the enlarged distal portion as the allantoic vesicle. Fluid accumulating in the allantois distends it so the appearance of its terminal portion in entire embryos is somewhat balloon-like (Fig. 40).
The allantoic vesicle enlarges very rapidly from the fourth ( to the tenth day of incubation. Extending into the sero-amniotic cavity it becomes flattened and finally encompasses f the embryo and the yolk-sac (Fig. 30, C, D). In this process the mesodermic layer of the allantois becomes fused with the adjacent mesodermic layer of the serosa. There is thus formed a double layer of mesoderm, the serosal component of which is somatic mesoderm and the allantoic component of which is splanchnic mesoderm. In this double layer of mesoderm an extremely rich vascular network develops which is connected with the embryonic circulation by the allantoic arteries and veins. It is through this circulation that the allantois carries on its primary function of oxygenating the blood of the embryo and relieving it of carbon dioxide. This is made possible by the position occupied by the allantois, close beneath the porous shell (Fig. 30). In addition to its primary respiratory function the allantois serves as a reservoir for the secretions coming from the developing excretory organs and also takes part in the absorption of the albumen.
Fig. 33. Schematic longitudinal-section diagrams of the caudal half of the embryo to show the formation of the allantois.
- A, chick of about three days incubation; B, chick of about four lays incubation.
The fusion of the allantoic mesoderm and blood vessels with the serosa is of particular interest because of its homology with the establishment of the chorion in the higher mammals.1 The chorion of mammalian embryos arises by the fusion of allantoic vessels and mesoderm with the inner wall of the serosa, and constitutes the embryos's organ of attachment to the uterine wall. In mammalian embryos the allantoic, or umbilical circulation as it is usually called in mammals, serves more than a respiratory function. In the absence of any appreciable amount of yolk, the mammalian embryo derives its nutrition through the allantoic circulation from the uterine blood of the mother. Thus the mammalian allantoic circulation carries out the functions which in the chick are divided between the vitelline and the allantoic circulations.
1By reason of this homology the serosa of the chick is sometimes called chorion. It seems less likely to lead to confusion if the use of the term chorion is restricted to mammalian forms, especially as the serosa alone is the homologue of only part of the mammalian chorion. In some books the term outer or false amnion will be found used to designate the structure called serosa in this book. The term false amnion is not, however, in general use in this country.
- Next: 50 to 55 Hours
The Early Embryology of the Chick: Introduction | Gametes and Fertilization | Segmentation | Entoderm | Primitive Streak and Mesoderm | Primitive Streak to Somites | 24 Hours | 24 to 33 Hours | 33 to 39 Hours | 40 to 50 Hours | Extra-embryonic Membranes | 50 to 55 Hours | Day 3 to 4 | References | Figures | Site links: Embryology History | Chicken Development
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 14) Embryology Book - The Early Embryology of the Chick 11. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_The_Early_Embryology_of_the_Chick_11
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G