Book - Text-Book of Embryology 6
| Embryology - 27 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Early Development of the Chick
Probably every student has seen a hen's egg, or the egg of some other bird, and knows its size, shape and appearance. If the calcareous shell is broken, the whitish shell-membrane is found closely applied to the inner surface. Enclosed by the membrane is the glairy, transparent '" white" or albumen. Through this can be seen the yellow spherical yolk mass. It requires close observation to discern the delicate transparent vitelline membrane around the yolk. If the egg has been in one position for a few minutes the tiny white germ disk, less than a quarter of an inch in diameter, will appear on the upper side of the yolk. The yolk mass and germ disk together constitute the ovum which was discharged from the Graafian follicle of the ovary. The vitelline membrane is a true cell-membrane, a product of the egg cytoplasm. All the structures on the outside of this are secondary egg membranes deposited by the epithelium of the oviduct as the ovum passed along. If the egg has been fertilized before it is laid the germ disk represents a considerably advanced stage of development, for fertilization occurs in the extreme upper end of the oviduct and during the time the egg is traversing the tube, a period of about 24 hours, the early formative processes have gone on. In order to observe the earliest stages it is necessary therefore to obtain and study the egg before it is laid.
In the chapter on the germ cells it was pointed out that the bird's egg represents the polylecithal type in which the quantity of yolk or deutoplasm reaches the maximum (p. 6) . The cytoplasm, with the nucleus, comprises a small disk, about 3 mm. in diameter and 0.5 mm. thick, which rests upon the yolk. The bulk of the yolk contains no cytoplasm at all, and the transition from pure yolk to pure cytoplasm is rather abrupt. The vast accumulation of yolk in the bird's egg is correlated with the long period that the growing embryo remains within the shell when it must depend upon an internal food supply. The reptilian egg represents the same type. The course of development is greatly modified by the yolk content, and since the mammals are probably descended from forms (reptiles) whose eggs contained much yolk, a study of the developmental processes of a polylecithal egg throws much light upon the development of mammals whose eggs contain but little yolk although resembling in their mode of development their ancestral type.
When the ovum escapes from the ovary it immediately enters the oviduct. The spermatozoa having traversed the oviduct, at once from four to twentyfive of them enter the cytoplasm. Polyspermy is apparently a normal incident, although only one sperm nucleus unites with the egg nucleus. The entrance of the sperms seems to stimulate the formation of the polar bodies (maturation), and as soon as this is accomplished one of the sperm nuclei unites with the mature egg nucleus to form the nucleus of the fertilized ovum.
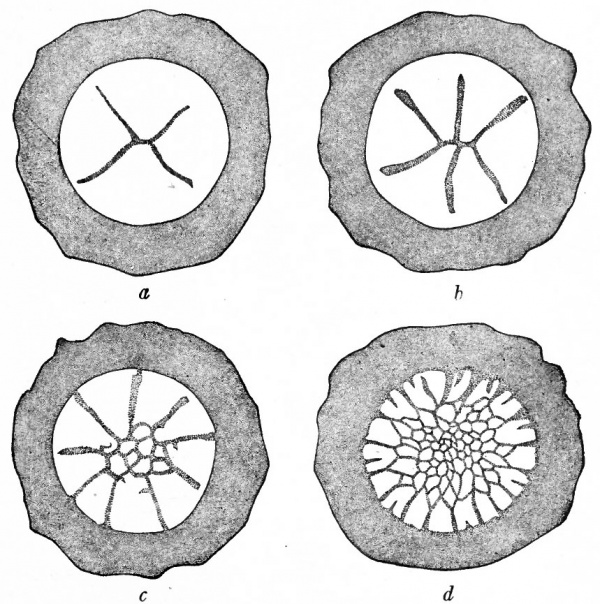
Fig. 38. Cleavage in hen's egg. Coste. Germinal disk and part of yolk, seen from above.
Cleavage
As soon as the two pronuclei unite in the cytoplasmic disk a spindle appears in preparation for the first division. The spindle is parallel to the surface and the first cleavage is therefore vertical. The cleavage plane is indicated on the surface by a slight furrow near the center of the disk, the margin of the disk being undivided. The first two blastomeres are therefore not completely separated from each other. The new spindle in each primary blastomere forms parallel to the long axis of the cell, and the second division occurs at right angles to the first and is also vertical. The plane of the second cleavage is again indicated by a slight furrow near the center of the disk. The first four blastomeres are separated only near their apices, the peripheral region remaining unaffected (Fig. 38, a). The third cleavage has not been observed in the hen's egg. As a rule the fourth cleavage tends to cut off the apices of the preceding blastomeres so that a central group of small cells is surrounded by a peripheral group of large cells which are not completely divided (Fig. 38, b and c). About this time some of central cells also divide in the horizontal plane. In subsequent divisions the central continue to divide more rapidly than the marginal cells, although among the latter the intercellar boundaries are extending still farther toward the dge of the disk (Fig. 38, d). As divisions succeed one another some of the nuclei of the marginal cells migrate out into the yolk surrounding the disk, the cytoplasm also encroaching upon the yolk and mingling with it. In this manner the disk gradually becomes more extensive in all directions.
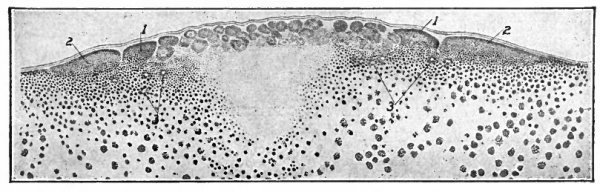
Fig. 39. From a vertical section through the germ disk of a fresh-laid hen's egg. Duval, Hertwig. g.d., Upper layer of germ disk; s.c., segmentation cavity; w.y., white yolk (see Fig. 3).
During this time a narrow space appears between the center of the disk and the underlying yolk. This space is the beginning of the blastoccel or segmentation cavity. Its roof is composed of the smaller central cells; its floor is the yolk, and around its margin it is walled in by the larger partially segmented cells and yolk (Fig. 39). If the living blastoderm is observed from above the area over the blastoccel appears clear and is called the area pellucida; the area peripheral to the blastoccel appears opaque and is known as the area opaca.
It is not difficult at this time to make a comparison between the developing hen's egg and the eggs of the frog and Amphioxus. Obviously the stage described above corresponds to the blastula. The small cells forming the roof of the blastoccel are homologous with the micromeres in the frog's blastula; the yolk mass, which in the bird's egg remains wholly unsegmented except around the margin of the blastoderm, is comparable with the macromeres; the partially segmented cells of the margin of the disk probably corresponds to the transition zone which was designated as the germ ring. In the Amphioxus' ovum there is only a small quantity of yolk, but enough to result in the formation of a few larger cells in the blastula which are homologous with the macromeres in the frog and the yolk mass in the bird. The blastoccel in the bird is reduced to a minimum owing to the fact that the cytoplasm comprises only a small disk which rests upon the relatively great mass of yolk. (Compare Figs. 20, 29 and 39.)
Before going on to the process by which ectoderm and entoderm arise, it is necessary to consider briefly the transition between the margin of the germ disk and the yolk. The incompletely divided marginal cells, which are larger than the central cells, border upon the unsegmented yolk around the disk. This yolk at first contains no nuclei and is called the periblast ring. Then as the marginal cells continue to divide, the peripheral daughter nuclei migrate into the periblast which thus becomes a nucleated yolk ring but is yet wholly unsegmented and merged with the yolk. This nucleated yolk ring receives the name germ wall. The nuclei here continue to divide and then cell boundaries appear. Some of the boundaries become complete, and discrete cells are thus formed which apparently join the group already forming the, cellular disk or blastoderm. Other daughter nuclei migrate farther and other cells are formed and added to the margin of the blastoderm (Fig. 40). In this manner the blastoderm increases in size, extending in all directions farther over the yolk. It is probable that the cytoplasm of these cells is capable of using the yolk to build up into more cytoplasm, a process comparable With digestion and assimilation.
Fig. 40. Cross section through the center of the blastoderm of a pigeon, 14.5 hours after fertilization. Blount, from Lillie. i, Marginal cells; 2, periblast; 3, nuclei in the subgerminal region.
It is possible at this time, or even before, to determine the position of the future embryo relative to the disk. If the disk is viewed from above in its natural position in the shell, with the larger end of the egg toward the left, the edge of the disk toward the observer will indicate the caudal end of the embryo, and the long axis of the embryo will lie at right angles to the long axis of the egg.
Gastrulation
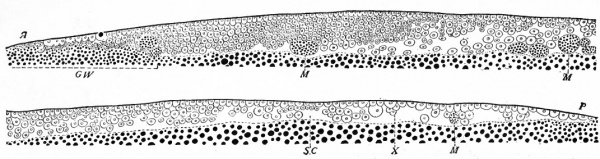
Fig. 41. Median longitudinal section through the blastoderm of a pigeon, 31 hours after fertilization. Patterson, from Lillie. A, anterior end of the blastoderm; G. W., germ wall; M, yolk masses of the blastocoel; P, posterior margin of blastoderm; S.C. blastocoel; X, cells which later may become members of the single layer. Anterior half of section is shown above, the posterior half below.
In the bird as in the lower forms gastrulation is the process by which the single layered blastula is converted into the double layered gastrula. In the bird the blastula consists of a disk of cells, the blastoderm, resting upon the yolk, the blastoccel being a shallow cavity beneath the center of the disk. Between the twentieth and tenth hours before the egg is laid the cells of the blastoderm are rearranged so that a sector of the posterior or caudal third becomes thinner and composed ^)f only a single layer of cells (Fig. 41). In front of the sector there is a gradual increase in thickness and at the anterior or cephalic border the blastoderm may be seven cells thick. Around the caudal edge of the sector the germ wall is interrupted so that the margin of the disk rests dir ec tly upon the yolk . The blastocoel during this time becomes larger.
Fig. 42. Median longitudinal section through the blastoderm of a pigeon, 36 hours after fertilization; process of gastrulation. Patterson, from Lillie. A, anterior end of the blastoderm;B, blastopore; E, entoderm; EC, ectoderm; F, floor of the blastocoel; G. W., germ wall; M, yolk masses in blastocoel; N, nuclei of germ wall; O, extension of blastoderm beyond germ wall; P, posterior margin of blastoderm; R, region of invagination and inflection of cells around dorsal lip of blastopore; V, vitelline membrane; Z, anterior limit of invaginated and migrating entodermal cells. S.C. Anterior half of section is shown above, the posterior half below.
Gastrulation is initiated by the tucking or rolling under of the caudal margin of the sector. The cells thus rolled in or involuted continue to proliferate and at the same time seem to migrate forward toward the cephalic border and outward toward the lateral borders of the blastoderm (Fig. 42). They do not at first form a complete layer but are more or less scattered. This new layer of cells is the entoderm while the original layer which now lies over it comprises the ectoderm. The two layers are continuous at the margin of the sector where involution began. This margin is the anterior lip of the bias to pore, a minute cleft between the margin and the yolk behind it, which leads from the exterior into the space, now the archenter on, beneath the double layered blastoderm. Concomitant with involution there is a considerable thickening of the lip of the blastopore where the ectoderm and entoderm are continuous.
It has been noted that the germ wall is interrupted along the posterior margin of the sector after the disk has here been reduced to one layer of cells. The margin of the sector is obviously a crescent, so that the blastopore also is originally crescent-shaped (Fig. 43, A). Then as gastrulation proceeds the horns of the crescent are withdrawn toward the median line, and concomitantly the two free ends of the germ wall approach each other (Fig. 43, B). Eventually the ends of the germ wall meet and the blastopore is closed ; and since the germ wall lies behind the closed blastopore, the latter is no longer situated on the edge of the disk but is included within it (Fig. 43, C).
Fig. 43. Diagrammatic reconstructions showing surface views of blastoderms of the pigeon. Patterson, from Lillie. A , from same blastoderm as shown in Fig. 41 , the line CD indicating the plane of section of Fig. 41; the numbers 1-7 indicate the thickness of the blastoderm in numbers of cells; the broken line around i includes the sector which is one cell thick, at the posterior margin of which invagination begins; GW, germ wall. B, from same blastoderm as shown in Fig. 42; the arrows at the posterior margin indicate the advance and approach of the two halves of the margin; E, indicates extent of entoderm; O, extension of disk margin beyond germ wall; PA, outer margin of area pellucida; R, margin where invagination is progressing (lip of blastopore) ; Y and Z together indicate region of germ wall. C, from a blastoderm of pigeon 38 hours after fertilization; E indicates extent of entoderm; R, mass of cells where blastopore closed; SG, portion of blastoccel not yet crossed by migrating entodermal cells; other abbreviations as in B.
The processes of development thus far described go on while the egg is traversing the oviduct. Development ceases when the egg is laid and cools; it begins again only if the temperature is raised. If the temperature remains below about 25 (Centigrade) there is no appreciable development, but if brought up to about 38, which is the optimum, development progresses normally. And from now on, the ages of embryos are reckoned from the beginning of incubation; not from the time the egg is laid nor from the time cleavage begins.
Fig. 44. Surface views of blastoderms of Haliplana, showing formation of primitive streak. Schauinsland.
Gastrulation in the bird seems to be a simple process as compared with that in the frog; in some respects it is even simpler than in Amphioxus. Rapid cell proliferation is of course a common incident in all three cases, particularly along the lip of the blastopore. In Amphioxus invagination plays the important part; involution and epiboly are less prominent. In the frog invagination is greatly reduced, while involution and epiboly are the most conspicuous features. In the bird invagination and epiboly can scarcely be said to occur at all; involution appears to be the essential process, and with it a specially marked migration of entodermal cells beneath the ectoderm. If an immediate cause for the differences in the three forms is sought, the yolk content of the egg offers itself as a mechanical influence which must be accepted as a most important factor.
Fig. 45. Surface view of embryonic disk of chick. Bonnet.
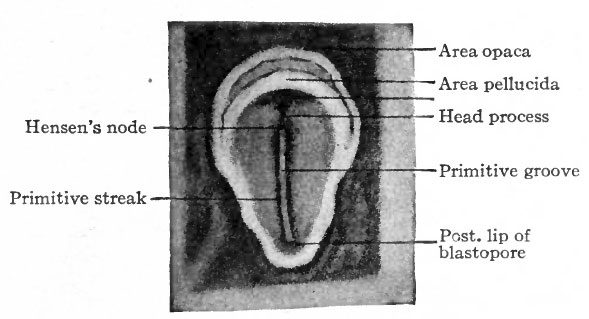
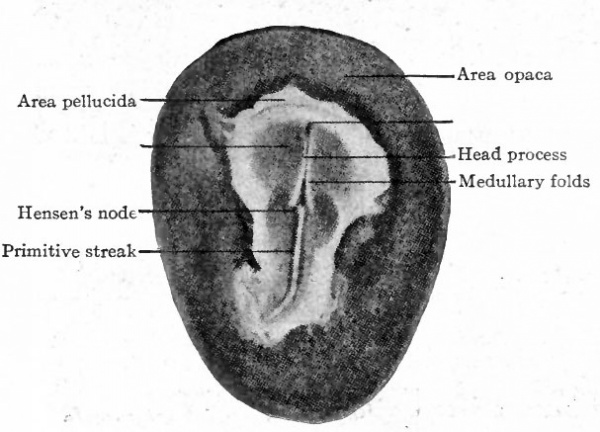
When incubation commences certain changes in the appearance of the Wastoderm can be seen on the surface.'Lpurmg the first day a narrow band, which is slightly more opaque than the Surrounding area, appears in front of the closed blastopore and extends forward more than half way across the area pellucida. It seems to grow from the blastopore; as a matter of fact, however, the blastopore recedes and leaves the band in its trail. This is the primitive streak (Fig. 44 and Fig. 45). While the streak grows the area pellucida elongates in the same direction and becomes oval, the broader end being anterior. Then a transparent line appears along the center of the streak and terminates in front in a slight enlargement. In front of this enlargement the streak is a little more opaque than elsewhere. The transparent line indicates the primitive groove, which is flanked by the primitive folds, and its broadened terminus is the primitive pit; the denser portion of the streak in front of the pit is the primitive knot (Hensen's knot). Following the development of the primitive streak there appears in front of it a narrow band, less conspicuous than the streak but continuous with and extending forward from the primitive knot. This is known as the primitive axis or head process (Fig. 46). During these changes in appearance the blastoderm also increases in total area.
Fig. 46. Surface view of chick blastoderm. Bonnet.
The primitive streak and the structures associated with it can be interpreted properly only in terms of sections. A transverse section through the streak near its center shows both ectoderm and entoderm merged with an intermediate layer which is obviously mesoderm (Fig. 47, A ) . It is the thickness of mass resulting from the fusion of the three layers which gives the opaque appearance of the primitive streak when seen from the surface. The primitive groove is a linear depression in the dorsal side of the streak, and the primitive folds are the elevations flanking the depression. The ectoderm is thickened perceptibly for some distance on both sides of the groove, thus forming the early neural plate. Beyond the neural plate the non-neural ectoderm extends laterally to the edge of the blastoderm, in fact forming its margin. The entoderm is a thin layer which extends laterally until it merges with the yolk to form the germ wall. The cavity beneath is the archenteron, extending from the germ wall on one side to that on the opposite side. The mesoderm at this time is not an extensive layer, for it constitutes only a portion of the mass of the primitive streak and extends laterally only a short distance between the other two layers as scattered irregular cells.
Fig. 47. Transverse sections of blastoderm of chick (21 hours' incubation). Hertwig.
- a, Section through primitive groove, posterior to Hensen's node.
- b, Section through Hensen's node.
A transverse section through the primitive pit shows essentially the same structural arrangement as in the streak farther caudally (Fig. 47, B). In some birds the pit opens into the archenteron, but not in the chick. The region of the primitive knot also shows the same arrangement, the knot itself being an elevation just in front of the pit. Caudally the primitive groove becomes more shallow and finally disappears, the caudal end of the streak broadening out as the primitive plate.
The morphological significance of the primitive streak is a question which has not yet been unequivocally answered. It is generally agreed, but not universally, that the streak is the homologue of the blastopore in the lower animals on the ground that all three germ layers are fused as they are in the lip of the blastopore, that it marks the caudal end of the embryo as does the blastopore, and that in some birds the primitive pit opens into the archenteron in the same manner as the blastopore. It has already been pointed out that the caudal margin of the sector where the blastoderm has been reduced to the thickness of one layer of cells was rolled or tucked under when gastrulation began, and that the germ wall was lacking along this margin. It was also stated that as gastrulation proceeded the two ends of the germ wall approached each other and eventually met behind the margin of the sector, and that the two horns of the crescentic groove were withdrawn toward the median line and finally closed (Fig. 43). Immediately after these phenomena the primitive streak appears, extending forward from the center where the horns of the crescent were drawn in and closed. It would seem therefore that the formation of the primitive streak is a continuation of the gastrulation process.
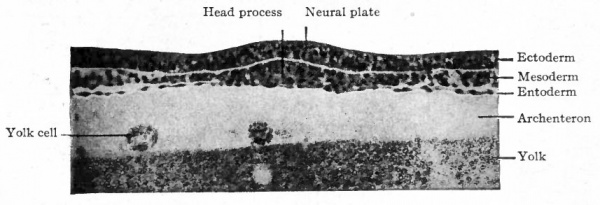
Fig. 48. Transverse section of blastoderm of chick (21 hours' incubation). Hertwig. Section through head process, anterior to Hensen's node.
In Fig. 46 there can be seen the slightly opaque band extending forward from the primitive streak which has been designated the primitive axis or head process. In cross section (Fig. 48) it is obvious that the opacity is due to the fused mass of entoderm and mesoderm, while the ectoderm here is a separate layer. In a longitudinal section which includes both axis and streak (Fig. 49) the ectoderm is observed to fuse with the other two layers at the anterior end of the streak. It is probable that the primitive axis is not the result of a forward growth from the end of the streak, but is the result of the separation of the ectoderm from the other two layers from before backward. That is, if one imagines the primitive streak at its full development before the axis has appeared, and then imagines a wedge started just beneath the ectoderm and driven backward, one can readily see that the ectoderm will be separated from the underlying and still fused mesoderm and entoderm. As this continues the axis thus becomes longer. The streak does not become correspondingly shorter, however, because it increases at the caudal end; in other words, as the primitive axis increases in length the primitive streak recedes or is carried backward by additions to its own organization. This exemplifies again the general principle that growth and differentiation in the early stages proceed from before backward.
Origin of the Mesoderm
The presence of the mesoderm between the other two layers in and lateral to the primitive streak has already been noted (Fig. 47). The cells composing the mesoderm appear to arise in the streak and migrate laterally as irregular elements which are so scattered that they do not at first form a complete layer. Whether they originate from ectoderm or entoderm is difficult to determine. The interpretation by those who have studied the problem most carefully is that the early mesoderm cells originate and differentiate from the thickened ectoderm along the primitive groove. The migrating cells multiply rapidly and soon a complete layer is formed which extends across the pellucid area until its margin overlaps the opaque area. The growth of the mesoderm is at first most rapid around the caudal end of the primitive streak, then it extends across the clear area laterally, and finally reaches forward on the two sides as horns which meet in front of the developing embryo but leave an area (the proamnion) in the head region unoccupied by mesoderm until much later.
Fig. 49. Median longitudinal section of a blastoderm of the chick just after the appearance of the primitive axis (see Fig. 46). Lillie, Ect., ectoderm; Ent., entoderm; G. W. , germ wall; H.F., head fold; H. Pr., primitive axis; med. pl., neural plate; Mes., mesoderm; pr.f., primitive fold; pr. gr., floor of primitive groove (primitive streak); pr. kn., primitive node; pr.p., primitive pit; pr. pl., primitive plate.
When the mesoderm overlaps the opaque area this area thus becomes three-layered, comprising ectoderm, mesoderm and germ wall. The mesoderm, if it does not actually merge with the germ wall, at least establishes intimate contact with it. While the mesodermal cells that arose in the primitive streak continue to proliferate, thus augmenting the layer generally, there is also evidence that new mesodermal cells are added by differentiation of the entodermal elements of the germ wall. This probable origin of mesodermal cells from the yolk cells of the germ wall is comparable with the formation of mesoderm around the gut in the frog (p. 60). As the germ wall recedes by encroachment of the entoderm upon the yolk, and the pellucid area becomes correspondingly more extensive, the mesoderm likewise increases in extent.
Almost as soon as the margin of the mesoderm overlaps the germ wall and begins to extend over the area opaca, small dense clusters of cells appear in the mesoderm in close relation to the yolk cells. These are the blood islands from which arise the early blood vessels and blood cells, and the area occupied by them is known as the area vasculosa (Fig. 51). They appear, first caudal to the primitive streak and then farther forward on both sides, almost keeping pace with the mesoderm' itself. The islands increase in size and coalesce irregularly to form a network or plexus which is one of the conspicuous features of the blastoderm (Fig. 156). The cells at the periphery become flat and arranged edge to edge to form endothelial tubes or vessels within which the central cells are contained as primitive blood cells (Fig. 158). The plexus then gradually extends across the pellucid area toward the axis of the blastoderm where the embryonic body is developing. It is obvious that blood vessels and blood cells develop relatively earlier in the bird than in either Amphioxus or the frog.
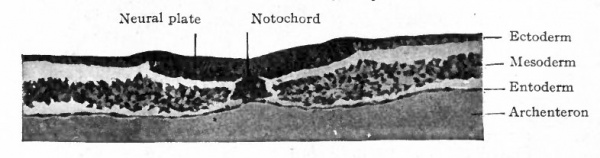
Fig. 50. Transverse section of blastoderm of chick (40 hours' incubation). Hertwig. Section taken short distance anterior to Hensen' node.
The notocord develops out of the fused mass of entoderm and mesoderm of the primitive axis, whether from the one layer or the other being difficult to determine. It thus becomes a rod of cells extending forward from the front end of the primitive streak and separating the mesoderm on one side from that on the other. The formation of the notocord as the axial structure of the future embryo destroys the primitive axis, and the entoderm now becomes a distinct and separate layer (Fig. 50) .
Summing up, it may be said that about the beginning of the second day of incubation the mesoderm comprises a sheet of cells between ectoderm and entoderm. It extends caudally and laterally from the primitive streak where it i? merged with the other two layers. In front of the streak it extends laterally from the notocord (which here separates the sheet into the two lateral portions) and forward like a horn over the area opaca on each side. Over a portion of the area opaca the network of blood islands and vessels forms a prominent differentiated part of the mesoderm.
From this time on, the changes in the mesoderm are rapid and extensive. The cells multiply rapidly, resulting in a thickening of the layer generally. Along each side of the notocord the paraxial? portion becomes distinctly thicker and then shades off into the thinner lateral portion. A short distance ahead of the primitive streak the paraxial band becomes marked off transversely into blocks by a loosening of the cells between the blocks. These are bilaterally symmetrical and are at once recognized as mesodermal somites (Fig. 51). The pair that appears first is the second pair of somites in the series, the first pair of the series being represented by a close aggregation of cells in front of the first cleft. This has been shown experimentally by delicately injuring the first pair with an electric needle and then allowing the blastoderm to continue to develop for several hours; it was found that the other somites of the series appeared behind the injury. A third pair appears behind the second, then a fourth pair, and so on through the series.
Fig. 51. Dorsal view of chick embryo with ten pairs of mesodermal somites. Bonnet.
Developing from before backward, the somites here as in Amphioxus and the frog again illustrate the general principle that growth progresses from before backward. The first somites appear only a short distance in front of the primitive streak, about where the anterior end of the primitive axis was located, and the paraxial band of mesoderm between the somites and the streak is wholly unsegmented. Then, as successive somites appear and the band becomes thus far segmented, new cells are constantly added at the caudal end of the band as the primitive streak recedes, so that eventually the whole series of somites (more than 40 pairs in the chick) is still followed by the primitive streak which now lies at' the caudal end of the embryo.
Fig. 52. Transverse section of chick embryo (2 days incubation). Photograph. The parietal mesoderm (lying above the coelom) is not labeled. The two large vessels under the primitive segments are the primitive aortae. Spaces separating germ layers are due to shrinkage.
Each somite is composed of massive epithelium whose cells converge toward a small central cavity (myoccel). The latter cavity, however, contains, a few loosely arranged cells (Fig. 52). Laterally each somite is continuous with a much thinner mass or plate of cells, known as the intermediate cell mass or nephrotome, which in turn merges with the lateral mesoderm of the area pellucida.
The sheet of lateral mesoderm becomes separated into two plates or layers, an outer which is apposed to ectoderm and an inner apposed to entoderm. In many places small clefts appear among the cells, grow larger and finally coalesce to form a continuous cavity, the ccelom, which in its greatest extent reaches from the outer edge of the nephrotome to the margin of the area vasculosa. The cleft-like ccelom develops on the ectodermal side of the blood islands and vessels, so that these structures are at first confined to the inner layer of mesoderm which lies close to the entoderm. The ccelom is the rudiment of the serous cavities of the adult the pleural, pericardial and peritoneal cavities. The layer of mesoderm apposed to ectoderm is known as the parietal or somatic layer, while that apposed to entodeim is called the visceral or splanchnic layer (Fig. 52). By comparison with Amphioxus and the frog it is seen that ectoderm and somatic mesoderm constitute the body wall and that entoderm and visceral mesoderm comprise the wall of the archenteron or gut cavity, although in the chick the germ layers at this stage are still spread out on the surface of the large yolk mass.
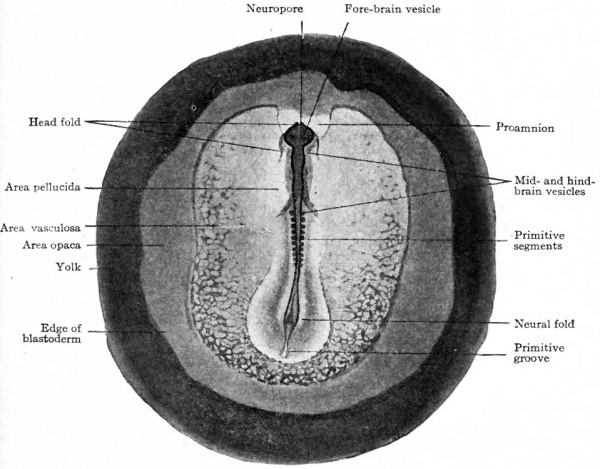
Another structure of early origin, as in the lower forms dealt with in the preceding chapters, is the neural plate. When the primitive axis appears, the ectoderm is moderately thickened into a broad band over the axis itself and extending backward over the primitive streak. This thickened ectoderm represents the rudiment of the nervous system and is known as the neural or medullary plate (Fig. 50). Laterally it gradually becomes thinner and shades off into the non-neural ectoderm. As development proceeds the anterior end of the plate becomes broader, indicating already the heavier brain region. The plate is composed of columnar epithelium possibly pseudo-stratified. When the somites begin to develop, a broad groove the neural groove appears along the center of the plate, thus producing ridges the neural ridges or folds along the sides of the groove. The ridges become higher and bend in toward the median line until they meet over the groove. The meeting occurs first in the region of the brain; then fusion occurs and a tube the neural tube is thus formed (Fig. 52). The meeting and fusion gradually progress backward. It is obvious from these conditions that the neural tube in the main is formed from before backward; in fact even when closure has occurred in the brain region, the caudal region exhibits still the flat neural plate while the intermediate portion shows all stages between the two extremes.
Body Form
In Amphioxus and the frog the cylindrical body form of the embryo results from simple elongation of the gastrula. In these forms the yolk is wholly encompassed by the egg cytoplasm, is contained in the entodermal cells of the gastrula, and is wholly enclosed within the embryonic body, being gradually consumed as the organism develops. In the bird only a small portion of the great quantity of yolk is enclosed within the cytoplasm of the mature egg; the cytoplasm is really only a small disk resting upon the yolk. As development proceeds the cytoplasmic disk takes the only active part; in the gastrula the two primary germ layers are simply spread out on the surface of the yolk mass; the appearance of the mesoderm does not alter the general conditions. Finally, however, in the latter part of the incubation period, the germ layers grow over the entire surface of the yolk like membranes; but this does not imply that the yolk is enclosed within the embryonic body.
The body of the embryo arises from a relatively small portion of the blastoderm, and its position is indicated by the primitive streak and head process which lie in its long axis. As they develop, the neural tube and mesodermal somites produce a thickening of the blastoderm which becomes slightly elevated like a rounded ridge above the general level of the surface (Fig. 52). Just in front of the cephalic end of the neural tube a transverse crescentic groove, the head fold, appears and sharply delimits the cephalic end of the body. The rapidly growing brain then projects over the groove, giving the appearance that the germ layers at the bottom of the groove are being tucked beneath the head-end of the embryo. The horns of the crescentic head fold extend caudally along the sides of the thickening produced by the neural tube and somites, thus delimiting the body region laterally. These lateral grooves become deeper from before backward and eventually reach the level of the primitive streak. Finally a transverse fold, the tail fold, appears caudal to the primitive streak and becomes continuous on each side with the lateral fold. The whole embryonic body thus becomes surrounded and delimited by a gutter.
The gutter or groove becomes deeper as the embryo continues to grow; the head-end of the body projects farther over the head fold and the tailend farther over the tail fold. The effect is much as if the body was being constricted or pinched off from the remainder of the blastoderm. The remainder of the blastoderm, composed of the extraembryonic portion of the germ layers, engages in the development of the yolk sac and certain other appendages which are useful during the period of incubation. The yolk sac and not the embryonic body contains the yolk that was present in the ovum. The yolk sac is an appendage of the gut, it is true, but the yolk substance is carried to the embryo by the blood circulating through the vitelline vessels.
- Next: Mammalian
References for Further Study
BLOUNT, MARY: The early Development of the Pigeon's Egg with especial Reference to the Supernumerary Sperm Nuclei, the Periblast and the Germ Wall. Biological Biilletin, Vol. 13, 1917.
DUVAL, M.: Atlas d'Embryologie. Paris, 1889.
HARPER, E. H.: The Fertilization and early Development of the Pigeon's Egg. Am. Journal of Anat., Vol. 3, 1904.
KELLICOTT, W. E.: Chordate Development. Chap. IV, 1913.
LILLIE, FRANK R.: The Development of the Chick. 2nd Ed., 1908.
PATTERSON, J. T.: Studies on the Early Development of the Hen's Egg. I. History of the Early Cleavage and of the Accessory Cleavage. Journal of Morphology, Vol. 21, 1910.
PATTERSON, J. T.: Gastrulation in the Pigeon's Egg. A Morphological and Experimental Study. Journal of Morphology, Vol. 20, 1909.
PEEBLES, FLORENCE: The Location of the Chick Embryo upon the Blastoderm. Journal of Experimental Zoology, Vol. i, 1904.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 27) Embryology Book - Text-Book of Embryology 6. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_6
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G