Book - Text-Book of Embryology 13
| Embryology - 20 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The development of the respiratory system
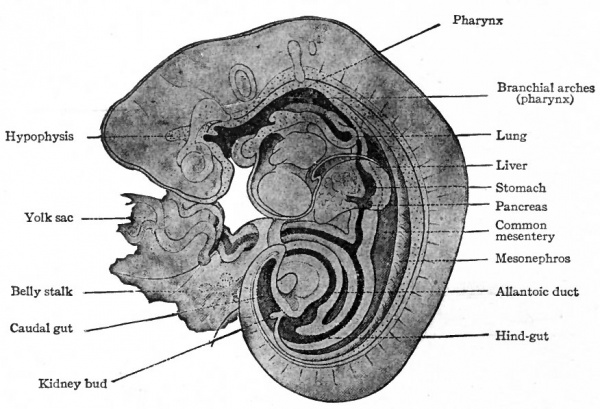
The anlage of the respiratory system appears in human embryos of about 3.2 mm. A hollow, linear evagination the lung groove develops on the ventral side of the oesophageal portion of the primitive gut, extending caudally a short distance from the region of the fourth inner branchial groove. It was once thought that the evagination developed along practically the entire length of the oesophagus anlage, but more recent researches seem to prove that it is confined to the cephalic end. The lung groove soon becomes separated from the gut by a constriction which appears at the caudal end and gradually progresses forward. Thus there is formed a tube which lies ventral to the gut and which opens upon the floor of the latter at the boundary line between the oesophagus and pharynx (Figs. 282 and 246).
Fig. 282. Sagittal section of reconstruction of a human embryo of 5 mm. His, Kollmann.
From this simple tube the entire respiratory system develops. The cephalic end gives rise to the larynx, the opening into the gut being the aditus laryngis. The middle portion gives rise to the trachea. Two outgrowths from the caudal end of the tube, which appear about the time of separation from the oesophagus, develop into the bronchi and their continuations the lungs. The epithelial lining of the system is of course derived from the entoderm. The various kinds of connective tissue are derived from the mesoderm, since the anlage grows into the mesodermal tissue of the ventral mesentery.
The Larynx
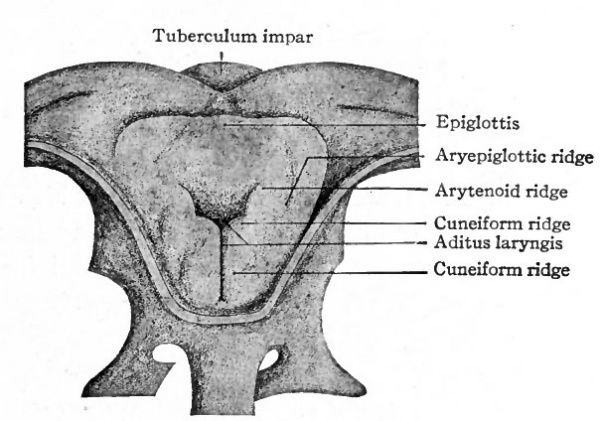
The opening from the gut into the respiratory tube becomes surrounded by a U-shaped elevation the furcula which lies in the floor of the pharynx with its open end directed caudally. Toward the end of the first month each side of the opening (aditus laryngis) becomes elevated, forming the arytenoid ridge. From each of these a secondary elevation arises, forming the cuneiform ridge. The arytenoid ridges come so close together that they practically close the opening except at its cephalic side (Fig. 283). Along with the development of these ridges the apical portion of the furcula becomes a distinct transverse fold at the cephalic rim of the opening. This fold is the anlage of the epiglottis. Laterally the epiglottic fold becomes continuous with the arytenoid ridges, forming the ary epiglottic ridges (Fig. 283).
Fig. 283. From a reconstruction of the larynx of a human embryo of 28 days. Seen from above. Kattius.
During the fourth month a groove-like depression appears on the medial side of each arytenoid ridge, gradually becomes deeper, and leaves on each side of it a fold or lip which bounds the opening. The external lips those nearer ; the pharynx form the superior or false vocal cords; the internal lips form the true vocal cords. At the same time the opening into the larynx, which was closed by the arytenoid ridges, is reestablished. The depression between the vocal cords on each side becomes still deeper to form the ventricle, and a further outgrowth from the ventricle produces the appendage of like ventricle (the laryngeal pouch).
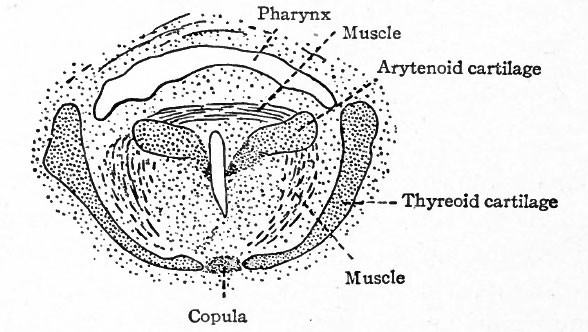
The mesodermal tissue external to the epithelium (entoderm) of the larynx gives rise to the various kinds of connective tissue including the laryngeal cartilages. By the end of the fourth week condensations appear in the mesenchymal tissue, which are the forerunners of the cartilages, but true cartilage does not appear until the seventh week. The anlagen of the thyreoid cartilage are two mesenchymal plates, one on each side, which are bilaterally symmetrical and correspond to the lateral parts of the adult cartilage (Fig. 284, A). These plates gradually grow ventrally and unite and fuse in the midventral line (Fig. 285) . Two centers of chondrification appear in each plate (Fig. 284, A,) and enlarge until the entire plate is converted into cartilage, the middle becoming elastic in character, the rest hyalin.
Fig. 284. From reconstructions of the mesenchymal condensations which represent the hyoid and thyreoid cartilages in an embryo of 40 days. A, Ventral view; B, lateral view from right. Kallius.
- Inf.hy., Inferior (greater) horn of hyoid; Sup.hy., superior (lesser) horn of hyoid; Thyr., thyreoid. The portions indicated by black lines represent chondrification centers.
Fig. 285. From a transverse section through the pharynx and larynx of a human embryo of 48 mm. Nicolas.
Originally the cephalic edge of each thyreoid plate is connected with the inferior horn of the hyoid cartilage (Fig. 284, B). This connection is subsequently lost, but a remnant of the connecting cartilage persists as the triticeous cartilage in the lateral hyothyreoid ligament. The anlagen of the arytenoid cartilages develop in the arytenoid ridges as condensations of the mesenchyme, which later are converted into true cartilage (Fig. 285). The apex and vocal process of each arytenoid become elastic, the main body becomes hyalin. The corniculate cartilages (cartilages of Santorini) are split off from the cephalic ends of the arytenoids and are of the elastic variety. The cricoid cartilage, like the others, is preceded by a condensation of mesenchyme. Chondrification begins on each side and then progresses around dorsally and ventrally until a complete hyalin ring is formed. From its developmental resemblance to the tracheal rings, the cricoid is sometimes regarded as the most cephalic of that series. The epiglottic cartilage develops in the epiglottic ridge as two separate pieces which subsequently fuse. It is of the elastic variety. The cuneiform cartilages (cartilages of Wrisberg) are split off from the two pieces of the epiglottic, and are of the elastic type.
Attempts have been made to determine which branchial arches are represented by the laryngeal cartilages. It seems quite definitely settled that the thyreoid is derived in part, at least, from the fourth arch. There is much doubt as regards the others, for there is great difficulty in determining their derivation in the human embryo, since the arches disappear at such an early stage. Furthermore, some of these cartilages may represent arches which are present in lower forms but do not appear in the higher Mammals.
The larynx is situated much farther cranially in the foetus and in the newborn child than in the adult. In a five months fcetus it extends into the nasopharyngeal cavity, whence it migrates caudally to its adult position. The laryngeal skeleton becomes ossified during postnatal life. Ossification begins in the thyreoid and cricoid cartilages at the age of eighteen to twenty years, and in the arytenoids a few years later. Three centers appear in the thyreoid one on each side near the inferior cornu and one in the medial line between the two wings. In the cricoid, ossification begins near the upper border on each side, in the arytenoids at the lower borders. Ossification usually begins earlier and proceeds more rapidly in the male than in the female.
As an example of the explanation which Embryology offers of certain peculiarities of structure in the adult, the case of the recurrent laryngeal nerve may be cited. The heart and aortic arches are primarily situated in the cervical region. At that time a branch of the vagus on each side, passes behind the fourth aortic arch to reach the larynx. As the heart and arches recede into the thorax, the nerve is pulled caudally between its origin and termination, so that in the adult the left nerve bends around the arch of the aorta and the right around the subclavian artery.
The Trachea
The portion of the original tube between the larynx and the two caudal outgrowths which form the bronchi and lungs, develops into the trachea. It lies ventral to the oesophagus and is surrounded by mesodermal tissue which is destined to give rise to the connective tis. 'ie, including the cartilage, of the adult trachea (Figs. 246 and 282). The development of the tracheal rings is very similar to that of the laryngeal cartilages. During the eighth or ninth week condensations appear in the mesenchyme, which are later transformed into hyalin cartilage. The rings are not complete but remain open on the dorsal side. At birth the trachea is collapsed, the ventral side being concave and the dorsal ends of each ring being in contact After respiration begins it is dilated and becomes more or less rigid. Ossification of the tracheal rings begins in the male at the age of about forty years, in the female at about sixty. The glands of the trachea represent evaginations from the epithelial linings.
The Lungs
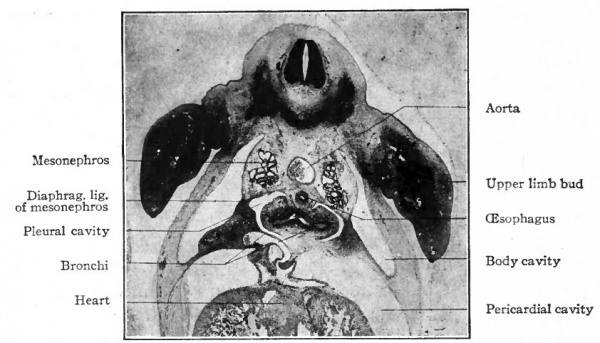
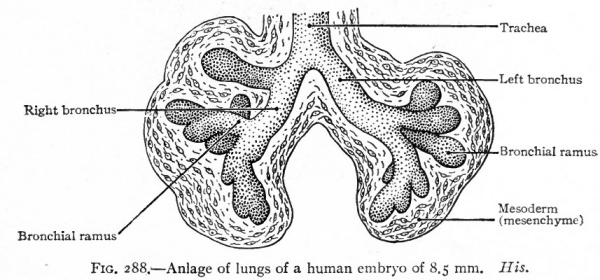
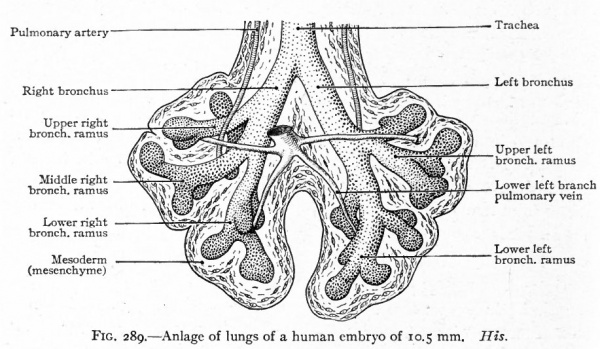
As has been stated (p. 330), the caudal end of the original tube evaginates to form two hollow buds which are the beginnings. of the two lungs (Fig. 286). The evagination takes place soon after or even along with the separation of the lung groove from the gut. The right bud soon gives rise to three secondary buds, the forerunners of the three lobes of the right lung. The left bud gives rise to two secondary buds, the forerunners of the two lobes of the left lung (Fig. 287). The primary buds may be said to represent the two bronchi arising from the trachea, the five secondary buds to represent the bronchial rami which extend into the five lobes of the lungs. Successive evaginations from each of the five buds take place and form an extensive arborization for each lobe (Figs. 288 and 289).
Fig. 286. Transverse section of a 14 mm. pig embryo, at the level of the upper limb buds, showing especially the two bronchi.
The manner in which the bronchial rami branch is not definitely known. Some maintain that the branching is dichotomous, that is, each bud gives rise to two equal buds and each of these to two others, and so on. In order to assume the adult form, however, one of the buds places itself in line with the preceding bud or bronchus while the other places itself as a lateral outgrowth. Others hold that the growth is monopodial, that is, that the original bud grows in a more or less direct line and the others develop as lateral outgrowths. When the evaginations that produce the bronchial rami are completed, each terminal (respiratory) bronchus subdivides into three to six narrow tubules, the alveolar ducts. The latter again branch into several wider compartments, the atria, from which several .air sacs are given off. The walls of the air sacs are evaginated to form many closely set air cells which represent the ultimate branches of the air passages of the lungs.
Fig. 287. Anlage of lungs of a human embryo of 4.3 mm. His.
Fig. 288. Anlage of lungs of a human embryo of 8.5 mm. His.
While there is a general tendency toward bilateral symmetry in the various sets of bronchial rami, the lobes of the lungs are asymmetrical. This asymmetry is indicated in the five secondary buds that arise from the two primary, since three arise on the right side and only two on the left. The three on the right represent the upper, middle and lower lobes of the right lung (Fig. 287). The upper is known as the eparterial from the fact that its bronchus lies dorsad to the pulmonary artery. No lobe develops on the left side corresponding to the upper (eparterial) on the right. There is a possibility that it is absent in order to allow the arch of the aorta to migrate caudally as it normally does (see p. 254). One of the larger ventral bronchial rami of the left lung is absent, owing to the inclination of the heart toward the left side; but as a compensation the corresponding ramus of the right lung develops more extensively and projects into the space between the pericardium and diaphragm as the infracardiac ramus.
Fig. 289. Anlage of lungs of a human embryo of 10.5 mm. His.
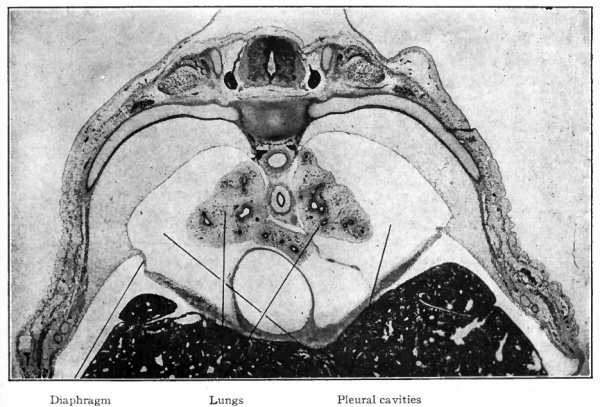
From the fact that the anlage of the respiratory system is enclosed within the mesentery between the gut and the pericardial cavity, and that its caudal end becomes enclosed within the dorsal edge of the septum transversum, it is obvious that the lungs will push their way into the dorsal parietal recesses or pleural cavities (Figs. 290 and 295). The way in which the lungs and pleural cavities enlarge and separate the pericardium from the body wall on each side and from the diaphragm is described on page 346 (see Figs. 296 and 297). The mesodermal tissue that surrounds the primary lung buds is in part pushed before the numerous outgrowths and in part remains among them (Figs. 287, 288, 289). The part around the lungs, with its covering of mesothelium, comes to form the visceral layer of the pleura which closely invests the entire surface of the lungs and dips down between the lobes. At the roots of the lungs it is continuous with the parietal layer of the pleura lining the inner surface of the pleural cavities. The mesodermal tissue among the bronchi and their terminations gives rise to the connective tissue that separates the lobes and lobules and invests all the structures in the interior of the lungs. This connective tissue at first constitutes a large part of the lungs, but as development proceeds, the more rapid growth of the respiratory parts results in the relatively small amount of connective tissue characteristic of the adult lung.
Changes in the Lungs at Birth. At birth the lungs undergo rapid and remarkable changes in consequence of their assuming the respiratory function. These changes affect their size, form, position, texture, weight, etc., and furnish probably the only certain means of distinguishing between a still-born child and one that has breathed. In the foetus at term the lungs are small, possess rather sharp margins and lie in the dorsal part of the pleural cavities.
Fig. 290. Transverse section of a pig embryo of 35 mm., showing the developing lungs (bronchial rami surrounded by mesoderm). The oesophagus is seen between the two lungs; above the oesophagus is the' aorta. The dark mass in the lower part of the figure is the liver. Photograph.
After respiration they enlarge, fill practically the entire pleural cavities and naturally become more rounded at their margins. The introduction of air into the air passages converts the compact, gland-like, foetal lung into a loose, spongy tissue. The specific gravity is changed from 1.056 to 0.342. While there is a gradual increase in the weight of the lungs during development, there is a very sudden increase at birth when the blood is freely admitted to them through the pulmonary arteries. The weight of the lungs relative to that of the body changes from about i to 70 before birth, to about i to 35 or 40 after birth.
Anomalies
The Larynx
The larynx may be excessively large or unusually small. Occasionally the epiglottic cartilage consists of two pieces, indicating a failure of the two anlagen to fuse (p. 332). Similar defects may occur in the other cartilages that are derived from more than one anlage. The ventricle on either side may be abnormally large with an exaggerated appendage (laryngeal pouch) . This condition resembles that in the anthropoid apes.
The Trachea
The trachea is sometimes absent, in which case the bronchi arise immediately below the larynx, indicating a failure on the part of the original tube to elongate. The trachea may be abnormally short. Rarely there is a direct communication between the trachea and oesophagus, probably due to an incomplete separation of the lung groove from the gut (p. 330) . The cartilaginous rings may vary in number as a result of abnormal splittings and fusions.
The Lungs
Rarely the eparterial bronchial ramus on the right side arises as a branch of the trachea and not as a branch of the bronchus (p. 335). This condition is normal in certain Mammals (ox, sheep) . Rarely an eparterial bronchial ramus is present on the left side, thus producing a third lobe for the left lung. In some animals an eparterial ramus is normally present on each side, the larger bronchial rami thus being bilaterally symmetrical. Variation in size and number of lobes is not infrequent. Supernumerary or accessory lobes, formed either by evaginations from the original anlage or by independent evaginations from the gut, are met with in rare cases.
Occasionally some portion of either lung is defective. The bronchial bud that would normally give rise to the lung tissue in that region fails to develop properly, and the result is a number of rami, without the ultimate terminations, surrounded by vascular tissue. The rami may remain normal or. may become dilated and form krge bronchial cysts.
References for Further Study
BONNET, R.: Lehrbuch der Entwickelungsgeschichte. Berlin, 1907.
Flint JM. The development of the lungs. (1906) Amer. J Anat. 6: 1-137.
GOPPERT, E.: Die Entwickelung des Mundes und der Mundhohle mit Drusen uud Zunge; die Entwickelung der Schwimmblase, der Lunge und des KehlkopfesderWirbeltiere. In Hertwig's Handbuch der vergleich. u. experiment. Entwickelungslehre der Wirbeltiere, Bd. II, Teil I, 1902.
HERTWIG, O.: Lehrbuch der Entwickelungsgeschichte des Menschen und der Wirbeltiere. Jena, 1906.
His, W.: Zur Bildungsgeschichte der Lungen beim menschlichen Embryo. Arch. /. Anat. u. Physiol., Anat. Abth., 1887.
KALLIUS, E.: Beitrage zur Entwickelungsgeschichte des Kehlkopfes. Anat. Hejte, Bd. IX, 1897.
KOLLMANN, J.: Lehrbuch der Entwickelungsgeschichte des Menschen. Jena, 1898.
KOLLMANN, J.: Handatlas der Entwickelungsgeschichte des Menschen. Jena, 1907.
HUNTINGTON, CEO. S.: A Critique of the Theories of Pulmonary Evolution in the Mammalia. Am. Jour, of Anat., Vol. XXVII, No. 2, 1920.
McMurrich JP. The Development Of The Human Body. (1914) P. Blakiston's Son & Co., Philadelphia, Pennsylvania.
PIERSOL, G. A.: Teratology. In Wood's Reference Handbook of the Medical Sciences, Vol. VII, 1904.
SYMINGTON, J.: On the Relations of Larynx and Trachea to the Vertebral Column in the Foetus and Child. Journ. of Anat. and Physiol., Vol. IX.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 20) Embryology Book - Text-Book of Embryology 13. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_13
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G