Abnormal Development - Tuberculosis
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
The gram-positive bacterium Mycobacterium tuberculosis causes the disease Tuberculosis (TB) usually initially infecting the lungs. The infection can cross the placenta to infect the fetus infecting many different systems (liver, bones, kidneys, spleen, gastrointestinal tract, skin, lymph nodes).
More than two billion people, one third of the world's total population, are infected with TB bacilli, an airborne infectious disease that is preventable and curable. The Bacillus Calmette-Guérin (BCG) vaccine was first used in 1921 as a vaccine for tuberculosis disease and also used in some countries to prevent childhood tuberculous meningitis and miliary disease.
Congenital tuberculosis cases are rare with a relatively high mortality rate.[1]
Postnatal infant infection can occur as a result of inhalation of bacilli at or soon after birth, ingestion of infected breast milk, or contamination of traumatized skin or mucous membranes.
| Bacterial Links: bacterial infection | syphilis | gonorrhea | tuberculosis | listeria | salmonella | TORCH | Environmental | Category:Bacteria |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Tuberculosis |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page. |
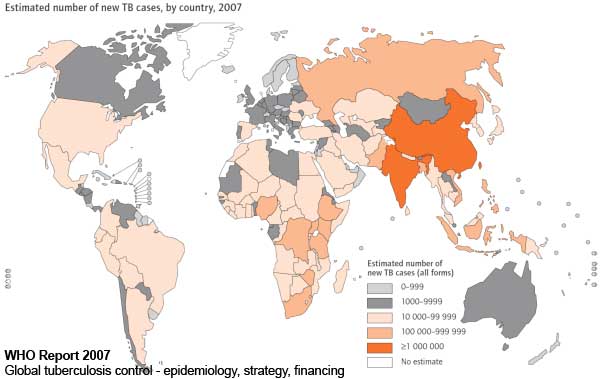
Global Tuberculosis (new cases 2007)
Drug-Resistant Tuberculosis
Extensively drug-resistant tuberculosis (XDR-TB2) is a highly drug-resistant strain subset of multidrug-resistant TB (MDR-TB) that have significantly worse outcomes, has now been reported in more than 50 countries. (WHO data)
Multidrug-resistant tuberculosis (MDR-TB) is defined as resistance to the two most powerful first-line anti-TB drugs (isoniazid and rifampicin).
Extensively drug-resistant tuberculosis (XDR-TB2) is defined as MDR-TB plus resistance to the most powerful second-line anti-TB drugs (any fluoroquinolone and any of the three injectable drugs: amikacin, capreomycin and kanamycin).
Australian NHMRC Guidelines
NHMRC Guidelines "are intended to promote health, prevent harm, encourage best practice and reduce waste" have replaced the earlier historic 1988 NHMRC recommendations.
- Links: Clinical Practice Guidelines - Pregnancy Care (2019) | PDF | Related Documents | Australian Clinical Practice Guidelines | NHMRC guidelines
| Historic - NHMRC (1988) Recommendations | ||
|---|---|---|
Neonates be assessed for follow-up care under the following conditions:
|
2006 Communicable Diseases Intelligence
Volume 30 Number 1, March 2006 - The BCG vaccine: information and recommendations for use in Australia
BCG vaccination is not recommended for general use in the Australian population.
BCG is recommended for:
- Aboriginal neonates in areas of high incidence of TB (e.g. Northern Territory, Far North Queensland, northern areas of Western Australia and South Australia).
- neonates and children 5 years and under who will be travelling or living in countries or areas with a high prevalence of TB for extended periods.
- neonates born to parents with leprosy or a family history of leprosy.
In addition to these recommendations BCG may be considered in the following:
- Children over 5 years who will be travelling or living in countries or areas with a high prevalence of TB for extended periods.
- Health care workers (HCWs) who may be at high risk of exposure to drug resistant cases.
References
- ↑ Peng W, Yang J & Liu E. (2011). Analysis of 170 cases of congenital TB reported in the literature between 1946 and 2009. Pediatr. Pulmonol. , 46, 1215-24. PMID: 21626715 DOI.
- ↑ Kaufmann SH. (2011). Fact and fiction in tuberculosis vaccine research: 10 years later. Lancet Infect Dis , 11, 633-40. PMID: 21798463 DOI.
- ↑ Lin HC, Lin HC & Chen SF. (2010). Increased risk of low birthweight and small for gestational age infants among women with tuberculosis. BJOG , 117, 585-90. PMID: 20156210 DOI.
Reviews
Borrell S & Gagneux S. (2011). Strain diversity, epistasis and the evolution of drug resistance in Mycobacterium tuberculosis. Clin. Microbiol. Infect. , 17, 815-20. PMID: 21682802 DOI.
Articles
Peker E, Bozdoğan E & Doğan M. (2010). A rare tuberculosis form: congenital tuberculosis. Tuberk Toraks , 58, 93-6. PMID: 20517736
Neyaz Z, Gadodia A, Gamanagatti S & Sarthi M. (2008). Imaging findings of congenital tuberculosis in three infants. Singapore Med J , 49, e42-6. PMID: 18301825
Vilarinho LC. (2006). Congenital tuberculosis: a case report. Braz J Infect Dis , 10, 368-70. PMID: 17293929
Chen A & Shih SL. (2004). Congenital tuberculosis in two infants. AJR Am J Roentgenol , 182, 253-6. PMID: 14684547 DOI.
Stähelin-Massik J, Carrel T, Duppenthaler A, Zeilinger G & Gnehm HE. (2002). Congenital tuberculosis in a premature infant. Swiss Med Wkly , 132, 598-602. PMID: 12571760 DOI.
Search Pubmed
Search NCBI Bookshelf: Medical Microbiology - Tuberculosis Search
Search PubMed: Mycobacterium Tuberculosis
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- CDC (USA) CDC (USA) - Tuberculosis | CDC (USA) - Tuberculosis Vaccine
- NHMRC (Australia) NHMRC (Australia) - BCG vaccine
- WHO WHO - tuberculosis | WHO Report 2007 - Global tuberculosis control - epidemiology, strategy, financing)
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 6) Embryology Abnormal Development - Tuberculosis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Tuberculosis
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G