2010 Lecture 16
Genital Development
Introduction
Objectives
- Understand the development of the gonads in males and females
- Understand the chromosomal basis of sex determination
- Understand the differences in male/female internal duct develpoment.
- Understand the origins of the external genitalia
- Understand the developmental abnormalities in male and female development.
Textbooks
- Human Embryology (2nd ed.) Larson Ch10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Lecture Audio Lecture Date: 22-09-2009 Lecture Time: 12:00 Venue: BioMed E Speaker: Mark Hill Genital
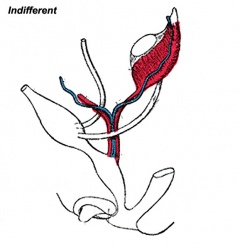
The Four Stages of Sexual Differentiation
There are four stages of sexual differentiation, some of which overlap in time. However, the order in which they occur is as follows:
- Development of the indifferent gonad - also known as the genital ridge
- Differentiation of gonad into testis or ovary
- Differentiation of internal genital organs and ducts
- Differentiation of external genitalia and secondary sexual characteristics
- The first event in sexual differentiation is the development of the indifferent gonad - sometimes called the bipotential gonad or just "genital ridge"
- The first observable sign of sex-specific differentiation in the male is the appearance of testis cords, which are the precursors of the adult seminiferous tubules.
- The first observable sign of sex-specific differentiation in the female is the entry of the germ cells into meiosis.
- Males and females develop both sets of ducts - those that are used in the male and those that are used in the female - and therefore have the potential to develop either way.
- Sex-specific differentiation of the internal ducts and the external genitalia is entirely controlled by hormones. Therefore, the important defining event in sexual differentiation is whether the gential ridge develops as an ovary or a testis. All other events follow on from that decision.
Reveiw of mammalian sex determination
Human Timeline
- 24 days - intermediate mesoderm, pronephros primordium
- 28 days - mesonephros and mesonephric duct
- 35 days - ureteric bud, metanephros, genital ridge
- 42 days - cloacal divison, gonadal primordium - indifferent to first appearance of testis cords
- 49 days - paramesonephric duct, clear gonadal differentiation
- 56 days - paramesonephric duct fusion (female)
- 100 days - primary follicles (ovary)
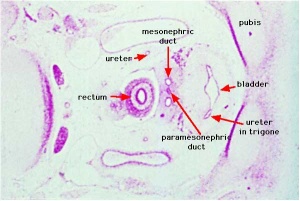
Kidneys and the urogenital system
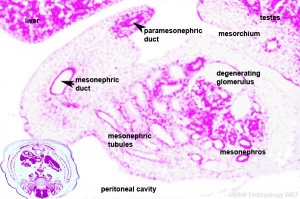
- kidneys and genital ridge develop from intermediate mesoderm, which lies between the lateral plate mesoderm and the somites.
- kidney develops in multiple stages, which occur in a rostrocaudal sequence; pronephros>mesonephros>metanephros (true adult kidney)
- earliest structure to form is the pronephros, in week 4, featuring a pronephric duct with associated nephrogenic mesenchyme.
- pronephros degenerates early on, leaving only the duct system running down to the cloaca – this becomes known as the mesonephric duct (Wolffian duct), in the embryo.
- next stage is the formation of the mesonephros, a series of mesonephric tubules in the mesenchyme that are induced by the mesonephric duct.
- mesonephros is a transient structure in mammals. In fish and amphibians it is the functioning adult kidney but in mammals it serves mainly as the site for gonadal development.
Chromosomal Sex Determination
History
- 1916 Bridges describes the sex chromosomes of Drosophila melanogaster and shows that sex is determined by the X:autosome ratio
- 1923 Painter describes the Sex chromosomes in mammals - it is assumed that sex determining mechanism will be the same as Drosophila
- 1959 Ford et al. find a human XO and female - Turner's syndrome
- 1959 Jacobs and Strong find an XXY male - Kleinfelter's syndrome
- Together these prove that the Y chromosome determines sex and not the X:autosome ratio
- 1989 XX males found that carry a very small portion of Y chromosomal DNA (60kb)
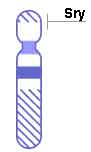
- Using DNA from these males a search for the testis determining gene, TDF or TDY led to the isolation of the gene now calles SRY because it comes from the Sex-determining Region of the Y chromosome. It is the molecular "switch" that determines sex in humans.
Y chromosome
- Y chromosome - 59 million base pairs, hypervariable in length, mostly non-functional repeats
- Current known protein-coding genes = 48 including SRY
- SRY encodes a 204 amino acid protein that is a member of the HMG (High mobility group) box class of DNA-binding proteins. Transcription factors bind to specific sites of DNA and regulates the transcription (expression) of other genes.
X chromosome
- 155 million base pairs
- In contrast to the Y chromosome, the X chromosome contains about 5% of the haploid genome and encodes house-keeping and specialized functions.
- Genes such as Wnt-4 and DAX-1 necessary for initiation of female pathway ovary development
- An early discovery (1961) was that in order to have correct levels of X chromosome gene/protein expression (gene dosage), females must "inactivate" a single copy of the X chromosome in each and every cell. The initiator of the X inactivation process was discovered (1991) to be regulated by a region on the inactivating X chromosome encoding an X inactive specific transcript (XIST), that acts as RNA and does not encode a protein.
- The genetic content of the X chromosome has been strongly conserved between species because these genes have become adapted to working as a single dose - Ohno's law
- X inactivation occurs randomly throughout the embryo, generating a mosaic of maternal and paternally derived X chromosome activity in all tissues and organs. This can be seen in the fur colour of tortoiseshell cats.
Cellular mechanism of sex determination
MBoC - Figure 20-18. Influence of Sry on gonad development
The supporting cells
- So called because they "support" the germ cells
Males
- In the male they develop as Sertoli cells
- SRY is expressed in the supporting cells and the immediate result is to transform them into Sertoli cells that surround the germ cells and form testis cords
- SRY is not expressed in the other cell types of the gonad - therefore the Sertoli cells instruct the germ cells and the steroid secreting cells to take the male path of development
- Embryonic Sertoli cells secrete anti-Mullerian hormone (AMH)
- Adult Sertoli cells line the inside of the seminiferous tubules and support spermatogenesis.
Females
- In the female they develop as follicle cells (granulosa cells)
- Follicle cells surround and nurture the developing oocytes
- In response to FSH, follicle cells proliferate
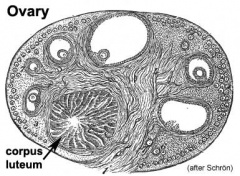
- After ovulation, these cells become luteal cells of the corpus luteum secreting progesterone and oestrogens
The steroid secreting cell lineage
Male
- Develop into Leydig cells which sit outside the seminiferous tubules
- Secrete testosterone in response to luteinizing hormone from the pituitary
Female
- Develop into theca cells that secrete androstenedione which can be converted by the follicle cells into estrogens
Primordial Germ Cells
Primordial Germ Cell Migration
- thought to be the first population of cells to migrate through the primitive streak in early gastrulation.
- This population of cells then lie at the hindgut yolk sac junctional region and later migrate into the genital ridge (germinal ridge) in early embryonic development.
- It is not the primordial germ cells which respond to SRY presence or absence, but the supporting cells within the developing gonad.
- Germ cells sometimes migrate by mistake into the developing adreanl gland and in the absence of sertoli cells telling them what to do, they begin to develop as oocytes, even in males
Gametogenesis
- forming PGCs as a small population of migratory cells
- enter the gonad where they undergo several rounds of cell division
- in the female the germ cells enter meiosis and become arrested at the dictyate (diplotene) stage of meiotic prophase. All oocytes are at this stage at birth
- in the male, the germ cells are enclosed by the developing Sertoli cells and are induced to arrest differentiaiton and cell division as T1 prospermatogonia until after birth.
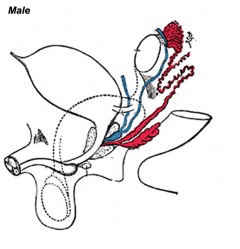
Differentiation of internal genital organs and ducts 1. Male
Media:Male_gonad_2.mov This looped animation shows the development of the male gonad showing medullary sex cords.
- The paramesonephric duct (red, left) degenerates under the influence of anti-Mullerian hormone (AMH) also known as Mullerian inhibiting substance (MIS)or Mullerian duct inhibitory factor (MDIF) secreted by sertoli cells.
- The mesonephric duct (purple) is maintained and differentiates under the influence of Testosterone secreted by Leydig cells. Within the testes these mesonephric tubules grow towards the testis cords and will form the rete testis. The mesonephric duct extending out of the gonad forms the ductus deferens.
- The testis cords (orange) containing the Sertoli cells and the germ cells (which are arrested as T1 prospermatogonia until after birth) later differentiate into seminiferous tubules which become hollow and actively produce spermatazoa during puberty.
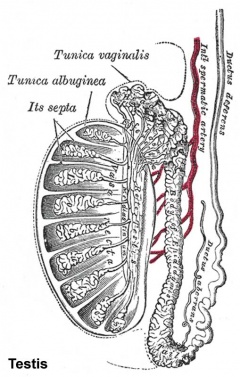
The tunica albuginea (white) covers the testis and bands extend inward to form connective tissue septa.
Anti-Mullerian Hormone
Anti-Mullerian hormone (AMH) or Mullerian Inhibiting Substance (MIS) hormone with at least two gonadal related functions:
- In males, it is produced by embryonic Sertoli cells and causes the loss of the paramesonephric (Mullerian) duct system that forms the internal female genital tract.
- In females, it is produced after puberty by follicle cells and suppresses the development of other primary follicles, thus restricting the number of follicles stimulated by FSH.
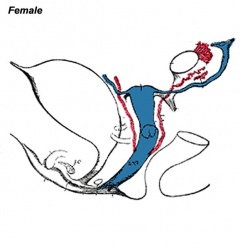
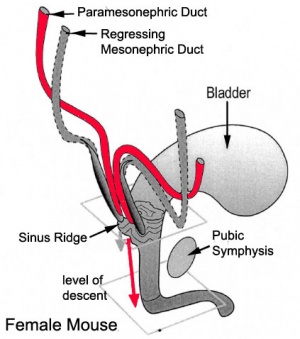
Differentiation of internal genital organs and ducts 2. Female
Female movie This looped animation shows the development of the female gonad showing cortical sex cords.
- The mesonephric duct (purple) degenerates, small remnants may remain as epoophoron and paroophoron (in the mesentry of the ovary) and Gartner's cycts (near vagina).
- The paramesonephric duct (red, left) grows forming the oviducts (fallopian tubes) and the end opens into the peritoneal cavity and terminates in fimbria (finger-like extensions). Away from the ovary, the two paramesonephric ducts fuse in the midline to form the uterus.
- After entry of the germ cell into meiosis they are called oocytes and they are surrounded by the derivatives of the supporting cell lineage - the follicle cells or granulosa cells.
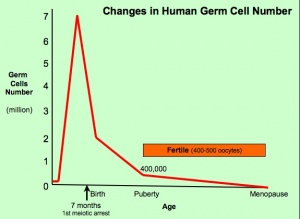
- About 95% of the germ cells that entered meiosis in the female will be lost by a process called follicular atresia (see graph. Only about 400,000 remain at the time of puberty.
- Week 7 – duct regression or preservation begins
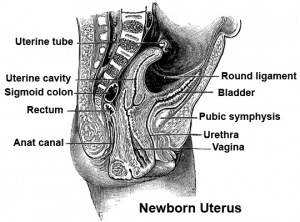
Vagina Development Uterus and vagina
- The embryonic origin of the vagina has been a historically hotly debated issue with several different contributions and origins described.
- One description shows the vagina arising by downward growth of Wolffian and Mullerian ducts. The sinovaginal bulbs are the caudal ends of the Wolffian ducts. Vaginal development is also under negative control of androgens.
External Genitalia
External Genital | Endocrinology - Diagram of the development of the external genitalia
- The external genitalia are initially identical and undergo male and female differentiation under the influence or absence of steroidal sex hormones.
- Indifferent stage ‐ cloaca divided by proliferating mesenchyme forming the urorectal septum which separates the ventral urogenital sinus from the dorsal rectum.
- Difference stage ‐ locally in this region the presence or absence of dihydrotestosterone (DHT), generated from testosterone, determines male/female development.
- Presence‐ DHT locally in this region leads to genital tubercle growth, maintenance and fusion of external male genitalia.
- Absence‐ of DHT, genital tubercle remains small, bends caudally to form the clitoris. Urethral folds persist, do not fuse, and form labia minora. The open urogenital sinus forms a cleft into which urethra and vagina open. The labioscrotal swellings become the labia majora.
Female Genitalia Development
This looped animation shows the development of external female genitalia from the indifferent external structure, covering the approximate period of week 9 to 12.
![]()
![]() Urorectal septum and division of the cloacal membrane
Urorectal septum and division of the cloacal membrane
Note the original cloacal membrane becomes separated into the urogenital membrane and anal membrane. The urogenital folds beneath the genital tubercle remain separate (unfused), forming the inner labia minora and second outer skin folds form the larger labia majora either side of the developing vestibule of the vagina. Note at the top of the animation, the changing relative size of the genital tubercle as it forms the glans of the clitoris.
Male Genitalia Development
Endocrinology - Box 6.6 The roles of testosterone (T) and 5α-dihydrotestosterone (DHT)
This looped animation shows the development of external male genitalia from the indifferent external structure, covering the approximate period of week 9 to 12.
Note the original cloacal membrane becomes separated into the urogenital membrane and anal membrane (identical to female). The urogenital folds beneath the genital tubercle begin to fuse in the midline. The skin folds either side for the scrotum, which too has a midline fusion, the raphe. The scrotal sac is initially empty and is an attachment site for the gubernaculum, descent of the testes begins generally during week 26 and may take several days.
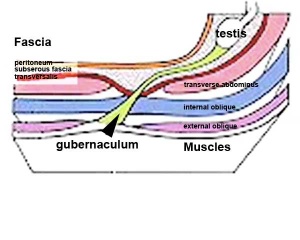
Gonad Descent
- Both kidney and gonads develop retroperitoneally, with the gonads moving into the abdomen or eventually into the scrotal sacs.
- During fetal development the gubernaculum and fetal growth in both male and female, changes the gonads’ relative positions finally reaching their adult locations.
Both female and male gonads undergo anatomical descent.
- Ovaries ‐ undergo caudal and lateral shifts to be suspended in the broad ligament of the uterus, gubernaculum does not shorten, it attaches to paramesonephric ducts, causing medial movement into the pelvis.
- Testes ‐ two anatomical phases in descent, transabdominal and transinguinal, under the influence of the shortening gubernaculum.
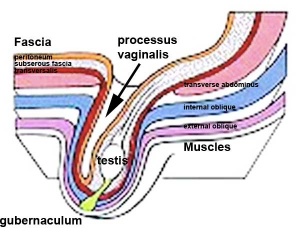
The testis (white) lies in the subserous fascia (spotted) a cavity processus vaginalis evaginates into the scrotum, and the gubernaculum (green) attached to the testis shortens drawing it into the scotal sac. As it descends it passes through the inguinal canal which extends from the deep ring (transversalis fascia) to the superficial ring (external oblique muscle). Descent of the testes into the scrotal sac begins generally during week 26 and may take several days. The animation shows the path of a single testis.
Incomplete or failed descent can occur unilaterally or bilaterally, is more common in premature births, and can be completed postnatally.
Data from a recent study of male human fetal (between 10 and 35 weeks) gonad position.
- 10 to 23 weeks - (9.45%) had migrated from the abdomen and were situated in the inguinal canal
- 24 to 26 weeks - (57.9%) had migrated from the abdomen
- 27 to 29 weeks - (16.7%) had not descended to the scrotum
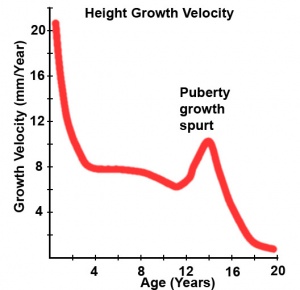
Postnatal - Puberty
Puberty can occur over a broad range of time and differently for each sex:
- girls (age 7 to 13)
- boys (age 9 to 15)
The physical characteristics that can be generally measured are: genital stage, pubic hair, axillary hair, menarche, breast, voice change and facial hair.
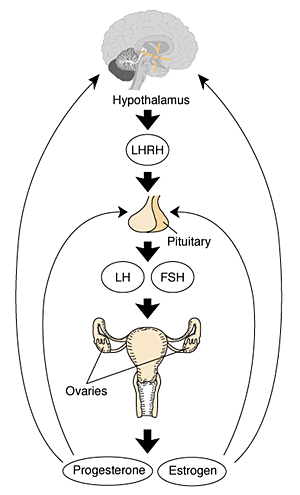
Female
In females, menarche (the first menstruation or a period) usually occurs after the other secondary sex characteristics, and will continue until menopause (permanent cessation of reproductive fertility).
The diagram shows the hormonal regulation pathway from the brain to the ovary and subsequent impact on uterine changes during the menstral cycle.
LHRH = Luteinizing Hormone-Releasing Hormone, also called gonadotropin-releasing hormone (GnRH). This peptide hormone is a decapeptide (10 amino acids) with a short half life (<15 minutes).
LH = Luteinizing Hormone
FSH = Follicle Stimulating Hormone
A similar endocrine axis is also found for regulation of the male gonad.
Puberty Abnormalities
- Precocious Puberty - Premature development of the signs of puberty which can occur in both girls (before age 7 or 8) and in boys (before age 9).
- Delayed Puberty - Determined in boys by a lack of increase in testicular volume by the age of 14 years. In girls, no breast development by the age of 13.5 years and a lack of menstruation by the age of 16 years. There can also be a "pubertal arrest" where there is no progress in puberty over 2 year period.
Sex Differences in Adult and Developing Brains
- not known significance of brain sex differences
- transient sex differences in gene expression in developing brains may cause permanent differences in brain structure
- may prevent as well, by compensating for potentially differentiating effects of sex differences in gonadal hormone levels and sex chromosomal gene expression
- Brains of males and females differ
- in regions specialized for reproduction
- in other regions (controlling cognition, etc) where sex differences are not necessarily expected
- Differentially susceptible to neurological and psychiatric disease
2 sources of sexually dimorphic information
- complement of sex chromosome genes
- mix of gonadal hormones
Abnormalities
Sex Reversal
- Where chromosomal sex does not match phenotypic sex i.e. XX males or XY females
XX males - usually caused by a transfer of some Y chromosome DNA onto the X chromosome
- Gonads develop as testes, everything looks normal internally and externally but infertile due to a failure of spermatogenesis
- Similar to Kleinfelters syndrome (XXY)
XY females - usually steroidal origin
- Main cause is Androgen Insensitivity Syndrome (AIS) Complete (CAIS) Partial (PAIS) and Mild (MAIS) usually caused by mutations of the gene encoding the androgen receptor AR gene located on the X chromosome
- 5-alpha-reductase deficiency - again leads to a lack of complete steroidal induction of external genitalia
- Rare mutations in key sex determining genes including deletion or mutations of SRY
Human genital abnormalities are currently described as "Disorders of Sex Development" (DSD) and include: chromosomal, gonadal dysfunction, tract abnormalities, external genitalia and gonadal descent. Genital Abnormalities
Congenital adrenal hyperplasia
- impairment of cortisol production by the adrenal cortex, is one of the most common causes of intersex genitalia at birth
- genetically male (XY) infants born with undervirilized genitalia are often assigned and reared as girls.
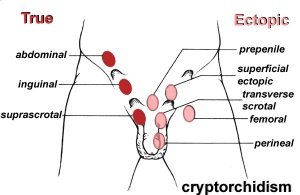
Cryptorchidism
- abnormality of either unilateral or bilateral testicular descent, occurring in up to 30% premature and 3-4% term males.
- Descent may complete postnatally in the first year, failure to descend can result in sterility.
Testis descent is thought to have 2 phases:
- transabdominal descent - dependent on insulin-like hormone 3 (INSL3).
- inguinoscrotal descent - dependent on androgens.
Undescended Ovaries
- reasonably rare gonad abnormality, often detected following clinical assessment of fertility problems and may also be associated with other uterine malformations (unicornuate uterus).
- Due to the relative positions of the male (external) and female (internal) gonads and the pathways for their movement, failure of gonad descent is more apparent and common in male cryptorchidism than female undescended ovaries.
Hydrocele
- Male Hydrocele is a fluid-filled cavity of either testis or spermatic cord, where peritoneal fluid passes into a patent processus vaginalis.
- Female Hydrocele is a similar, but rarer, fluid-filled cavity occuring in the female as a pouch of peritoneum extending into the labium majorum (canal of Nuck).
Tract Abnormalities
Many different forms
- Uterine: associated with other anomolies, unicornuate uterus
- Vagina: agenesis, atresia
- Ductus Deferens: Unilateral or bilateral absence, failure of mesonephric duct to differentiate
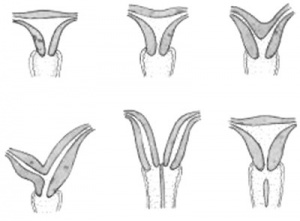
Uterine Duplication (uterus didelphys, double uterus, uterus didelphis) A rare uterine developmental abnormality where the paramesonephric ducts (Mullerian ducts) completely fail to fuse generating two separate uterus parts each connected to the cervix and having an ovary each.
Septate Uterus
Cervical: cervical agenesis, cervical duplication
Vaginal: Mayer-Rokitansky syndrome (MRK anomaly, Rokitansky-Küster-Hauser syndrome, RKH syndrome, RKH) congenital absence of the vagina, dyspareunia, vaginal agenesis.
External Genitalia - Hypospadia
- most common penis abnormality (1 in 300) from a failure of male urogenital folds to fuse in various regions and resulting in a proximally displaced urethral meatus.
- The cause is unknown, but suggested to involve many factors either indivdually or in combination including: familial inheritance, low birth weight, assisted reproductive technology, advanced maternal age, paternal subfertility and endocrine-disrupting chemicals. Infants with hypospadias should not undergo circumcision.
Movies
| Urogenital Sinus | Urogenital Septum | Trigone | Renal Nephron |
| Uterus | Female External | Male External | Testis Descent |
References
Textbooks
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
Online Textbooks
- Developmental Biology (6th ed.) Gilbert Chapter14 Intermediate Mesoderm
Search
- Bookshelf genital development | gonad development | sex determination
Online Links
- UNSW Embryology Abnormalities | Y chromosome | Male | X chromosome | Female | Stage 13/14 Embryo | Stage 22 Embryo | Stage 22 Highpower
- UNSW Embryology Movies: Urogenital Movies
- Embryo Images Unit: Embryo Images Online | Urongenital Development | Internal Genitalia | Definitive Kidney | External Genitalia
- Histology: Male Reproductive System | Female Reproductive System
Introduction
This section of notes covers genital development. Differences in development are dependent on a protein product of the Y chromosome SRY gene. Mesonephric duct (Wolffian Duct) and paramesonephric (Mullerian Duct) contribute the majority of male and female internal genital tract respectively.
Lecture Archive
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Course Content 2010
Embryology Introduction | Cell Division/Fertilization | Lab 1 | Week 1&2 Development | Week 3 Development | Lab 2 | Mesoderm Development | Ectoderm, Early Neural, Neural Crest | Lab 3 | Early Vascular Development | Placenta | Lab 4 | Endoderm, Early Gastrointestinal | Respiratory Development | Lab 5 | Head Development | Neural Crest Development | Lab 6 | Musculoskeletal Development | Limb Development | Lab 7 | Kidney | Genital | Lab 8 | Sensory | Stem Cells | Stem Cells | Endocrine | Lab 10 | Late Vascular Development | Integumentary | Lab 11 | Birth, Postnatal | Revision | Lab 12 | Lecture Audio | Course Timetable
Cite this page: Hill, M.A. (2026, Haziran 9) Embryology 2010 Lecture 16. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2010_Lecture_16
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G