Paper - The Human Pineal Gland and Pineal Cysts
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Cooper ERA. The human pineal gland and pineal cysts. (1932)
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Human Pineal Gland and Pineal Cysts
By Eugenia R. A. Cooper, M.D., M.Sc.
Lecturer in Histology in the University of Manchester
Introduction
This investigation of the human pineal gland was stimulated by the fortunate collection of four pineal cysts. A complete study of the gland was made, from the second foetal month, through childhood to old age, in an endeavour to elucidate the origin of these cysts. From these observations an attempt will be made to show that cystic enlargement of the pineal may be an exaggeration of a normal occurrence rather than a pathological entity.
It is proposed to give a brief survey of the histology of the pineal, dealing first with the appearances during foetal life, then childhood, adolescence and each decade of adult life.
Fifty post-natal and seventeen foetal glands were examined.
Development
The development of the human pineal has been thoroughly and admirably described by Krabbe(6), but a short résumé of the findings in the present research will be given in order to assist subsequent observations.
In the present series, the development of the human pineal gland was investigated in embryos ranging from 25 mm. crown-rump length to full term. Sections were all cut in the sagittal plane.
In the younger embryos the comparatively large anlage of the posterior commissure formed the chief ' factor in the orientation of the gland. This commissure was easily recognisable by its histological appearances.
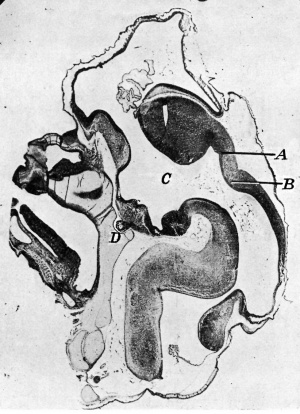
|
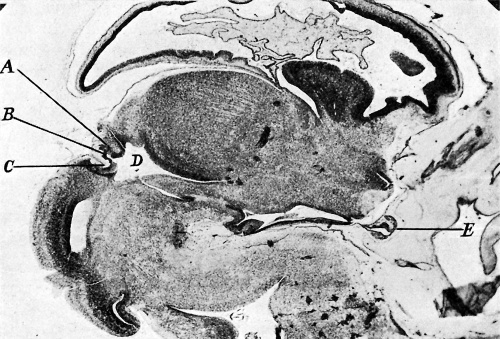
|
| Fig. 1. Sagittal midline section through head of 25 mm. embryo. x 8. A, anlage of pineal gland in the form of a backward hollowextension. B, anlage of posterior commissure. C, third ventricle. D, pituitary body. | Fig. 2. Sagittal section through head of 35 mm. embryo, not quite median. x 10. A, anterior anlage of pineal. B, posterior anlage with divertioulum pineale. C, posterior commisaure. D, third ventricle. E, pituitary body. |
In an embryo of 25 mm. length only a suggestion of a fold anterior to the small invagination of the posterior commissure represented the pineal gland (fig. 1). A little later, in an embryo of 30 mm. length, the anlage of the gland consisted of a backward hollow extension, the pineal diverticulum, of the roof of the hinder part of the diencephalon just in front of the anteriorly projected posterior commissure. In this embryo the anlage of the habenular commissure was discernible, the pineal fold lying between it and the posterior commissure. At this stage the pineal anlage consisted of undifferentiated epithelial cells continuous with those forming the roof-plate of the diencephalon.
The features of the pineal in an embryo of 35 mm. were similar to those described and illustrated by Krabbe(6): a backward hollow extension from the roof of the third ventricle just anterior to the clearly demarcated posterior commissure, represented the posterior anlage, whilst adjacent and united to it was a mass of cells representing the anterior anlage (fig. 2).
Between the third and fourth months of intra-uterine life the anterior pineal anlage had -increased in size and embraced the habenula. The pineal diverticulum, continuous with the third ventricle, was invested by the posterior anlage which joined the posterior commissure as this projected forwards into the ventricle. No line of demarcation between the cells of the two anlagen was discernible as described by Krabbe(6). Even at this stage the constriction of the diverticulum by the approximation of the two anlagen was noticeable (fig. 3).
|
|
| Fig. 3. Sagittal section through pineal region in embryo between the third and fourth months.
|
Fig. 4. Sagittal section through pineal region in a foetus of 4 months.
|
In another example at this age, the gland appeared as a round mass of cells containing a cavity surrounded by tall columnar cells similar to those lining the diverticulum pineale. This cavity, or cavum pineale, was separated from the third ventricle by pineal cells. It was surmised that in this specimen the division of the diverticulum into cavum and recessus pineale had been effected (fig. 4).
Towards the middle of intra-uterine life, vascularisation of the pineal had commenced by a projection of vascular connective tissue into the periphery of the organ. Furthermore it was found that two types of cells were present; fine fibres and associated small cells, with pale nuclei containing one or two nucleoli, were believed to be neuroglial elements. The glial fibres ramified amongst the undifferentiated cells which were recognisable by their very deeply stained nuclei and small amount of cytoplasm. The neuroglial network of the posterior commissure was unmistakable and continued into the pineal mass. The connective tissue elements were distinguished by their elongated pale nuclei.
In sections from embryos during the later months of intra-uterine life, the architecture of the pineal was similar to that of post-natal glands. The recessus pineale, lined by ependyma, projected into the pineal anteriorly and was embraced by the neuroglial peduncles. Vascular connective tissue entered the periphery of the organ at many points, and the connective tissue septum originally separating the two pineal anlagen was observed. The neuroglial network of the pineal peduncles invaded the cellular mass and joined a similar network already present in the gland.
During the eighth foetal month the undifferentiated pineal cells had become transformed into the so-called pineal parenchyma. These cells were of much greater size, the pale nucleus, containing one or two nucleolar masses, being surrounded by a clearly defined hyaline or faintly granular mass of cytoplasm (fig. 5).
Infancy
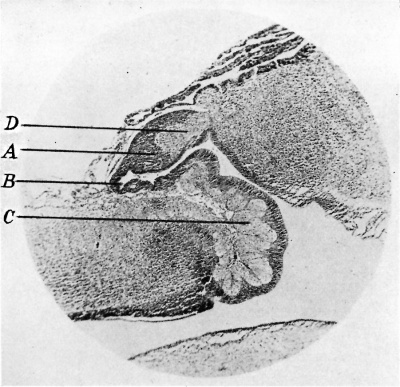
During infancy the pineal was found to have assumed characteristics which persisted more or less throughout life. The parenchymatous mass fitted over the backward extension of the third ventricle, i.e. the pineal recess. The gland was prolonged forwards embracing the pineal recess and forming the pineal peduncles (fig. 6). A capsule derived from the pia mater enveloped the gland, and invaded it peripherally with fine blood vessels.
A neuroglial network was seen to form the peduncles and to insinuate itself as a band between the blind end of the pineal recess and the rest of the organ (fig. 6). From this band the glial network radiated throughout the gland, enmeshing the parenchymal cells (fig. 7). Furthermore a narrow glial band occupied the periphery of the pineal, separating the parenchyma from the pial capsule. The parenchymal cells were similar to those described as differentiating during the eighth month of foetal life (fig. 5). Mitotic changes were seen in the parenchymal cell nuclei in the infantile glands. Four of the infantile specimens examined were “ solid,” in other words the cavum pineale was completely obliterated. In another case, from an infant aged 12 months, two small cavities were present in the gland. The larger one was surrounded by a wall of concentrically arranged glial fibres separating it from the parenchyma. The smaller one lay within the glial band adjacent to the blind end of the pineal recess. No cells were seen lining these cavities which were empty.
Within the “solid” glands mentioned above were one or two masses of glia devoid of parenchymal cells. These masses are known as “glial plaques,” and it is suggested that they are formed during the constriction of the pineal diverticulum and the attempted obliteration of the pineal cave. It seems possible that glia is instrumental in closing the primitive cavities of the pineal. Two specimens from infants aged 22 months presented variable features. One was “solid” and consisted of closely packed typical parenchymal cells enveloped in glial network. In the centre. of the other specimen was a large oval cavity surrounded by a wide band of concentric glial‘ fibres (fig. 8). The cavity was empty, but a few irregular cells were seen lining the mural glia. This appearance again suggests that glia plays an important part in the closure of the early pineal cave, but that the obliteration may be incomplete, leaving a cavity enveloped in glia. The original caval ependyma may disintegrate and disappear so that there is no cell lining to the cavities, but, on the other hand, some of these cells may persist. This important feature will be referred to again later.
Two specimens were obtained from children of 4 and 8 years and both were cavitated. The pineal from the child of 8 years contained four cavities, the largest lying in the long axis of the organ. It was enveloped by glial fibres within which a cellular lining was noticed. Five examples of the pineal at puberty were examined. Each had distinctive features. All the glands had reached adult dimensions, the average anteroposterior diameter being 8-5 mm. One (18 years) contained a central cavity surrounded by a wide glial band. Lining the cavity distinct cells were observed. Posterior to the cavity were two irregular glial plaques. The rest of the organ consisted of typical glial meshwork and parenchyma.
Another pineal (12 years) had a very different appearance. The greater part of it was composed of large glial plaques, withglial network and parenchyma intervening. The parenchymal cells were considerably reduced in numbers, and each cell appeared to be isolated by glial fibres. A small cavity, devoid of cell lining, but containing cell debris, was situated within the largest glial plaque of this pineal.
fig. 5. Neuroglial network amongst pineal cells: full term. x 250.
fig. 6. Sagittal section of pineal of infant of 5 months. x 35. A, pineal reeeas lined by ependyma. B, strand of nenroglia. C, pinea1 peduncles. D, pineal mass.
fig. 7. Pineal parenchyma and neuroglial network. x 250. A, pineal oells with pale nuclei. B, neuroglial network.
fig. 8. Pineal of an infant aged 22 months. x 35. A, large cavity surrounded by concentric glia. B, pineal recess. 0, pineal parenohyma. D, pineal peduncle.
The preponderance of glia at this age and the marked reduction in the amount of parenchyma seem to contradict the former belief that the pineal cells are gradually replaced by glia as age advances.
The third pineal of puberty (15 years) was similar. There was one very large glial plaque, and a smaller one containing a cavity. Within the latter were some lining cells and coagulated fluid. Adjacent to this plaque was a larger cavity, devoid of cells within, but occupied by coagulated material. A narrow glial band enveloped the larger cavity.
Of two pineals from subjects aged 15 years, one contained five cavities of variable size, each surrounded by thick glial walls, and into one was invaginated a small mass of pineal parenchyma. The other gland was composed of a solid mass of pineal cells and supporting glial network, devoid of cavities or glial plaques.
An interesting feature in these glands was the appearance of the so-called calcareous bodies. These had no relationship to blood vessels, but occurred irregularly amongst the pineal cells or glial plaques. They were similar in size to the cells. From careful observations it was thought that these structures originated in the cytoplasm of the parenchymal cells by a deposit of calcareous material, the cell later disintegrating and liberating the calcareous mass (fig. 9). Like calcareous deposits elsewhere, those in the pineal may represent a retrogressive change. It was found, however, that they were very numerous, though small, at puberty, occurred also in adjacent parts, and were absent in some of the adult glands.
Finally the specimens from individuals of 15 years showed commencing “loculation,” but this appearance is more fully described below.
During the second decade many variations in the histology of the pineal body were encountered. The size was variable, the largest being fully a centimetre antero-posteriorly. Of the five specimens of this period examined, two contained cavities. In both cases the latter were surrounded by dense glial meshwork, and glial plaques also were observed. In none of the cavities were there any lining cells, but one contained cell débris. Small glial plaques were present in the remaining “solid ” glands. Calcareous bodies occurred in all but one of this group. The number was variable; one gland contained such quantities in all parts that sectioning was extremely difficult, due to the hard stonelike property of the deposit after histological fixation.
At this stage “loculation” was definitely established. Radiations of glia pervaded the organs from glial plaques, but in addition, fibroglial strands divided the glands into loculi in which lay the parenchymal cells with the intervening glial network (fig. 10).
Of two glands of the third decade, one contained a very small cavity within a glial plaque, whilst in the other, four large cavities were discernible with the naked eye. A thick wall of concentric glial fibres encircled each cavity, but no lining cells were present. The cavities, however, all contained coagulated material. The parenchyma and glial network called for no special description. There appeared to be no loculation in either case, whilst calcareous deposit was absent in one of the glands, and almost negligible in amount in the other.
The pineal bodies of the fourth decade conformed to type. Two of them contained a cavity enveloped by glia. In one case the cavity was large, but had no regular cellular lining, although many necrotic cells were observed around the periphery. In this gland small calcareous bodies were in process of formation.
In the other cavitated pineal from a subject aged 49 years, the space was surrounded by glia, and within this at one part was a definite row of cells lining the cavity.
Thirteen pineal bodies of the fifth decade were obtained. Of these seven were cavitated. Two contained- five separate spaces, another four, another two, and the remaining organs each had one (fig. 11). The gland in which there were two cavities was interesting, because a tongue-like projection of glia and cells was invaginated into one of the spaces (fig. 12). Reference to this will be made later (p. 43). A wall of concentric glial fibres encircled all the cavities. In no case could any lining of cells be discerned.
It was difficult to understand why some of these organs contained so many cavities. It has been suggested that they are a degenerative change, but the regular arrangement of concentric glial fibres around them appears to be rather an attempt, albeit abortive, on the part of the glia to encircle, constrict and obliterate the original cavum pineale. In some cases the invasion of the cavum by neuroglia appears to be irregular, so that the cave is separated into compartments, each of which persists with concentric glia surrounding it, thus giving origin to multiple cavities. Furthermore it was found that calcareous masses never occurred within those cavities surrounded by concentric glia, indicating that their formation is not dependent on “ brain sand” deposition (see p. 42).
Calcareous masses were present in all but three of these glands (fig. 13), and in some cases they were observed also in the pineal peduncles. The parenchyma and glia differed in no way from those at earlier periods of life. The cells appeared to be less numerous in some of these glands than in others, but this reduction may have been only apparent, depending on unavoidable post-mortem shrinkage, or slight variation in the thickness of the sections necessitated by the presence of “ brain sand.” Loculation occurred in the majority of the glands at this period of life.
Of the five pineal glands of the sixth and seventh decade, only one possessed a cavity surrounded by a glial wall. In all these cases were observed typical parenchymatous cells, glial network, loculation and calcareous deposits. The cells were present in considerable numbers in all the glands from aged individuals. This fact appears to throw doubt on the view (9) regarding the replacement of cells by glia in advancing age.
Four pineals from adults of uncertain age were included, since two of them each contained a very large cavity, enveloped by glia. Of great importance was the fact that distinct cells were observed lining the cavity inside the mural glia of the former case (fig. 14).
fig. 9. Pineal gland at puborty showing formation of calcareous bodies (A) amongst neuroglial network. x 250.
fig. 10. Adult pineal showing locuhtion by fibroglinl strands (A). x 60.
fig. 11. Pineal of adult of 53 years showing many cavities surrounded by glial walls. x 35.
fig. 12. Pineal during fifth decade showing tongue-like invaginntion into a. cavity. x 12. A, cavities with glisl walls. B, smell glisl plaque.
fig. 13. Masses of “brain send” in adult pineal. x 35.
Three features were common to all pineal bodies examined. first a fibrous capsule was present, being derived from the pia mater and entering the periphery here and there with blood vessels. Secondly, the blood supply of this gland was extremely poor in comparison with that of such organs as the pituitary gland. Thirdly, the glia appeared to be similar in all cases. The individual glial cells consisted of a small triangular body apparently filled ‘by a dark staining nucleus, and long delicate processes passing from the angles to envelop the parenchymal cells. With Cajal’s gold sublimate technique these glial cells were demonstrated as astrocytes (fig. 15).
From this brief survey of the pineal gland at different stages of life, the following conclusions are drawn:
1. The human pineal is composed of typical parenchymal cells enmeshed in a neuroglial network. At birth the cells are characteristic. The glia appears during early foetal life, and the network amongst the parenchyma is observed about mid term, having spread from the original glia (described below). The organ increases in size until puberty when adult dimensions are reached. Parenchymal cell multiplication occurs during childhood.
2. The primitive diverticulum pineale is reduced to the recessus and cavum pineale during foetal life. It is suggested, contrary to the belief of Krabbe(6), that neuroglia might be responsible for the division of the diverticulum pineale. The primitive cells lining the diverticulum arise from embryonic spongioblasts (1), which are capable of developing into either ependyma or neuroglia. In this way, then, certain of the spongioblasts become neuroglial cells which appear to be instrumental in reducing the diverticulum and perhaps obliterating the cavum pineale.
In the present investigation, however, it has been seen that it is the exception rather than the rule for the cavum to be completely obliterated. In these cases it appears as though the glia had spread circularly around the cavum in an attempt to “squeeze it out,” but had failed to do so completely, leaving a cavity, variable in size, surrounded by a wall of concentric glial fibres. The invasion by glia may be irregular so that the cavum pineale is divided into two or more compartments each of which becomes surrounded by glia and persists as a separate cavity.
In some cases, however, these particular spongioblasts may not all develop into glia, but some become what may be described as “pseudo-ependymal” cells. These line the cavity which has been surrounded by, but not obliterated by, glial fibres. The importance of these cells will be alluded to in the consideration of the pineal cysts. It is further suggested that, under some provocation, at present unknown, these “pseudo-ependyma ” cells may be concerned with the outpouring of fluid into the cavity.
8. In cases where obliteration of the cavum pineale has been complete, glial plaques are prominent, but they are also present in cavitated glands. These plaques, however, may be either tangential sections through the mural glia of cavities, or obliterated portions of the primitive diverticulum pineale formed by the irregular invasion of it by glia.
fig. 14. Cells lining cavity in an adult pineal. x 250. A, cavity. B, lining cells.
fig. 15. Aatrooyten demonstrated in an adult pineal body by 0.5.1’. gold hublimate method. x 260.
The presence of glial plaques does not appear to be associated with old age, since the glands at puberty were found to contain the largest glial masses. Glia usually forms a peripheral band underneath the pial capsule and surrounds the pineal recess. The glia appears to belong to the astrocyte group.
4. “Loculation” of the pineal body by fibroglial trabeculae is present in most adult pineals. This,may be part of the normal fibrosis consequent on advancing years, and is seen in other organs such as the pituitary body (3).
5. Calcareous bodies are discernible at puberty. It is suggested that they arise by a deposit of calcareous material within the parenchymatous cells which later disintegrate, freeing the mass. The small bodies later coalesce by further depositions on them.
6. The blood supply of the pineal body is not rich in comparison with that of the better known endocrine organs.
7. Judging from sections it appears as though the parenchymal cells were variable in numbers, but that a reduction is not necessarily associated with old age. Some organs at puberty were found to contain very few cells in comparison with glands from old people.
Discussion
In the course of this investigation it has been observed that the cellular types in the human pineal gland’ are two in number: (a) parenchyma, and (b) neuroglia. This is in agreement with the findings of Hortega (5), although it has not been possible to demonstrate the finer details of the cells depicted in the admirable researches of this author. Krabbe(6), on the other hand, believes that nerve cells occur in the pineal, but this investigation has not revealed any.
The glia appears to belong to the astrocyte group, as has been shown also by Hortega (5). Although it has been fully described by many authors, so far no function has been assigned to it. Hortega (5) suggests that the important function of the pineal is localised in the parenchyma, and an accessory one in the glia. After careful investigation of this gland through all stages of life, it has been concluded that the glia differentiates during foetal life for the purpose of reducing the original pineal diverticulum. Krabbe (6) assigned this function to the parenchymal cells. Later, according to the present investigation, the glia is believed to be instrumental in an attempt to obliterate the pineal cave. In the majority of cases, however, the attempts are abortive, and a cavity, or cavities, persists.
There does not seem to be any doubt, judging from the present research and that of Krabbe(6), that the cavities represent the unobliterated distal part of the original pineal diverticulum. Furthermore the presence of such a cavity serves as a foundation, in certain cases, for the production of cysts. On the other hand, Krabbe(6) believes that cavities in the pineal are a normal oc.currcncc, and in later life tend to close spontaneously, but this is questionable on examination of the present series of adult pineal glands (see fig. 12). Marburg, quoted by Krabbe(6), suggests that the cavities may result from necrosis of a glial plaque. Judging from the present investigation this theory cannot be maintained as an explanation of cavity formation in the pineal, but it is suggested that “ pseudo-cavities ” may result from the deposition and coalescence of calcareous deposits which are lost during the histological technique. Such cavities are never surrounded by concentrically arranged glial fibres (see p. 36). Krabbe (6) also concludes that cavities are never produced by the destruction or degeneration of the parenchyma.
In the. series under consideration, the wall of all cavities, irrespective of size, position, or age of‘ individual, consisted of concentrically arranged glia devoid almost entirely of enmeshed parenehyma. Krabbe(6) found the walls to be composed of parenchymal cells at first, but (hiring the second year a thin sheet of glia was noticeable between the parenchyma and the cavity. In old age he observed a thickening of the mural glia, the cavity in consequence becoming smaller. This was 11ot found to be the case in the present series.
An observation, to which great importance is attached, is the presence, in an appreciable number of cases under investigation, of a clearly demarcated cell lining inside the mural glia of the cavities. For convenience these cells have been called “ pseudo-ependymal ” and suggestions as to their origin have already been made (see p. 4-1).
As far as could be ascertained from the available literature, only Krabbe and Marburg [quoted by Krabbe](6) refer‘ to any cells lying within pineal cavities. Krabbe(6) describes them as necrosing pineal cells, and calls them “ependymal-like” cells. Marburg(6) believes them to be true ependyma. Neither of these authors mentions any special arrangement of these cells within the cavities. In the series under consideration, cell debris was present in the cavities in several of the pineal glands, but in others, distinct healthy cells in a. single layer were found around the periphery of the cavity (fig. 14).
The significance of this layer of cells is not thoroughly understood. The presence of coagulated fluid in certain of the cavities associated with a cell lining suggests that the cells may be concerned with the outpouring of fluid. Absence of coagulated material in many of the sections did not necessarily indicate that during life such material was not produced.
Reference must be made to the presence of glial plaques in the pineal gland. Judging from the present observations, it is believed that these are formed during the efforts of the neuroglia to obliterate the early pineal cave. Krabbe (6) believes them to be produced by proliferation of the mural glia of a cavity, or less likely, by the enlargement of the tractus pineale. His former suggestion is readily acceptable, and the irregularity of these plaques could be explained either by his or the author’s theories.
Proliferation or hyperplasia of the glia has been sce11 to occur in the present series, and was particularly demonstrable in pineals at puberty. This proves almost conclusively that the hyperplasia is not dependent on, or concurrent with, advancing years. Furthermore in many pineal glands from aged individuals there did not appear to be any hypcrplasia of the glia. IIortega(5) refers to the glial h_vpcrplasia, but is unable to explain its initiation, as the amount is so variable. and the controlling factors are not known as yet. Ile found, however, that a number of diseases of the central nervous system were associated with an increase in the pineal glia. In the cases under investigation, there was nothing pathological in the central nervous system to explain the glial hyperplasia occurring in the two glands at puberty.
The significance of the lime concretions, or “ brain sand,” and the fibroglial strands in the pineal body is not yet fully understood. Krabbe (6) indicates that the former are related to nuclear excretion. In the present series, it was surmised that the lime salts were deposited in the parenehymal cells, which afterwards disintegrated and set f rec the calcareous mass. Formation of “ brain sand” and “ loculation ” of the pineal by fibroglial strands may be retrogressive changes consequent on advancing age. A similar invasion by fibrous tissue occurs in the pars anterior of the pituitary body (3). Lord(7) many years ago referred to the loculation of the pineal body by trabeculae derived partly from the pial covering, and partly from the peduneles. In the present research, too, these strands were found to be composed of fibroglial tissue.
Pineal Cysts
Four pineal glands with cystic enlargement were examined. No statistics regarding the incidence, and very few descriptions of this condition, could be found in the available literature. It is certainly not a very frequent condition, although it is probably overlooked at autopsies on occasions.
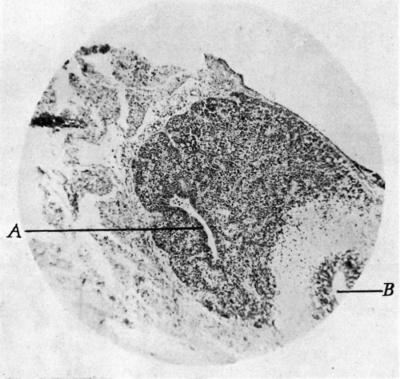
Three of the cases obtained were from females, aged 42 years (A.), 42 years (B.), and 73 years (1).). The fourth was from a male of 38 years (C.). All of the specimens were removed from asylum patients: this fact may have been only coincidence or the condition may have influenced the mental history. Each pineal was considerably enlarged, I). measuring 13-5 mm. antero-posteriorly. In A., C. and I). the cystic enlargement was posterior, but in B. the main one was anterior and smaller ones lay behind it. The very large cyst in D. seemed to
represent the coalescence of four smaller ones (fig. 16). The remaining parts of the glands were composed of typical parenehymal cells separated by glial fibres. The parenchyma was considerably reduced in all cases. Small glial plaques were noticeable in each gland.
All cavities, large and small, were encircled by a glial wall. In the larger cysts this was very attenuated and adjacent to and in contact with the pial covering.
In two cases A. and I). a tongue-like projection invaginated the cyst anteriorly (fig. 17). A similar feature occurred in a normal pineal from a subject of 51 years (fig. 12: see p. 38).
Fig. 16. Pineal cyst from female aged 73 years. Case D. x 4. A, cyst containing coagulated material. B, pineal tissue. 0, degenerated mass. D, tongue-like projection.
Fig. 17. Pineal cyst from female of 42 years (case A.), showing tongue-like projection into cyst (A). x 4.
Fig. 18. Cells lining the cyst in case D. x 250. A, cavity of cyst.‘ B, lining of cells. 0, fibroglial wall of cyst.
The cysts in C. and D. contained hyaline material coagulated by histological fixation which prevented examination of the fluid. Careful investigation revealed the presence of a clearly defined layer of cells lining the larger cysts in each case. The cells were flattened, probably due to distension. In specimen D. the cyst was divided into four loculi by fine fibroglial strands (fig. 16). The cell lining was complete around each of the four compartments (fig. 18). In A., B. and C. the cell lining of the cysts was not quite complete, but this may have been due to autolysis.
In cases A. and C. the post-mortem examination revealed a flattening of the superior quadrigeminal bodies and a slight excess of cerebrospinal fluid. The foramina of Munro were widely patent and the cerebral ventricles slightly dilated. There were no autopsy reports of cases B. and D.
After careful consideration of the histories, post-mortem findings and histology, of the four cases of pineal cyst, it is suggested that in each case death resulted from increased intracranial pressure. This is readily explained by the intermittent pressure of the enlarged pineal body on the vein of Galen. As pointed out by Stopford (11), a small subtentorial enlargement, especially in the neighbourhood of the pineal gland, compresses the vein of Galen as it curves upwards around the splenium of the corpus callosum to join the straight sinus. This results in the onset of symptoms dependent on increased intracranial pressure.
The next point requiring elucidation is the cause of the cystic enlargement of the pineal body. Bearing in mind the fact that in the human pineal the cavum pineale persists more frequently than it is obliterated, the initial factor, i.e. the presence of a cavity, is evident. Presence of a cellular lining and fluid content in the cavity, in some of these cases, contributes a second factor, that is, a medium possibly concerned in the outpouring of fluid into the cavity. How these lining cells function in relation to the production of fluid cannot be explained with certainty at the moment.
The final factor, i.e. the initiation of outpouring of fluid into the cavity and its distension into a cyst is merely a matter of conjecture. It is suggested, tentatively, that some increase in the general venous pressure may be responsible, but the brief histories and the absence of full post-mortem findings in the four cases make it impossible to pursue this conjecture further.
Many years ago Stanley(10), Garrod (4), Russell (8), Campbell(2) and Lord(7), each described cases of cysts of the pineal which had come under their care. Only Campbell described the microscopic appearance. He found “ a large cyst-like dilatation of the central cavity or ventricle of the pineal body. In places flattened cells could be seen along the inner lining of the cavity.” Apparently all these cases were similar to those alluded to in the present research, but none of these authors was able to state how the cystic enlargement of the pineal was responsible for the death of their patients.
In conclusion, it is suggested that cyst-like enlargement of the human pineal body is an exaggeration of a normal occurrence, but it cannot be stated dogmatically what factor is concerned in the initiation of the enlargement.
Acknowledgments
I wish to express my thanks to Prof. Stopford, of the Anatomy Department of the University of Manchester, for his kind help and valued criticism; to Dr R. Handley, of the County Mental Hospital, Prestwich, for providing me with three pineal cysts, histories of the patients, and also some normal glands; to Dr Dawson, Dr Susman, Dr D. Sheehan and Dr Henderson for many excellent specimens; to Mr H. Gooding, histologist in my department, for his skill and care in preparing the photographs.
Abridged References
(1) BAILEY, P. and Cusnmo, H. (1926). Turmmrs of the Gliorna Group. Lippincott & Co.
(2) CAMPBELL, A. W. (1899). Trans. Path. Soc. of London, vol. L, p. 15.
(3) COOPER, E. R. A. (1925). Histology of the Endocrine Organs at Various Ages. Oxford Medical Press.
(4) GARROD, A. E. (1899). Trans. Path. Soc. of London, vol. L, p. 14.
(5) HOBTEGA, R10 DEL (1929). Histological Structure of the Pineal. Reviewed by Hartman, Arch. Neurobiol. G. p. 26.
(6) KRABBE, K. (1916-17). Anat. Hefte, Bd. LIV, p. 187.
(7) Loan, J. R. (1899). Trans. Path. Soc. of London, vol. L, p. 18.
(8) RUSSELL, A. E. (1899). Trans. Path. Soc. of London, vol. L, p. 15.
(9) SCHAFER, A. E. (1926). The Endocrine Organs, 2nd edition, vol. 11, chapter xlv, p. 320. Longmans, Green & Co., Ltd.
(10) STANLEY, S. S. ( 1837-8). Larwet, i, p. 935.
(ll) Srorronn, J. S. B. (1928). Brain, vol. LI, Part 4, p. 485.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The Human Pineal Gland and Pineal Cysts. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_Human_Pineal_Gland_and_Pineal_Cysts
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G

