Book - Physiology of the Fetus 2
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Windle WF. Physiology of the Fetus. (1940) Saunders, Philadelphia.
1940 Physiology of the Fetus: 1 Introduction | 2 Heart | 3 Circulation | 4 Blood | 5 Respiration | 6 Respiratory Movements | 7 Digestive | 8 Renal - Skin | 9 Muscles | 10 Neural Genesis | 11 Neural Activity | 12 Motor Reactions and Reflexes | 13 Senses | 14 Endocrine | 15 Nutrition and Metabolism | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter II The Fetal Heart
Initiation of the Heart Beat
While the embryo consists of an aggregate of relatively few cells there is no need of a special mechanism to circulate oxygen laden blood. Tissue respiration of the ovum is adequately supported by the gas tension gradients between maternal fluids and embryonal ce1ls at the site of implantationz nutritiona1 needs are slight. But with further growth in size the usefulness of a circulatory system becomes evident.
Until recent1y the earliest contractions ok the mammalian heart had not been seen. Although many investigatorsH have studied chick embryos incubated Iess than two days sabin4 and Johnstones appear to have been the lirst to watch the initiation of the beat at the ten somite stage. It was found that contractions begin on the right side of the ventricle at a point near its junction with the primordium of the atrium. It seemed to these observers that the earliest contractions occurred rhythmically. More recently others« have extended this worlc in a very painstalcing cinematographic study. They discovered that the first beats are arrhythmical fibrillations of a few cells located in the bulbo-ventricular region of nine somite embryos. The atrial myocardium shows no activity until three or four hours after contractions have started in the ventriclesz the sinus venosus begins to beat still later. synchronized rhythmical contractions of the entire ventricle result from coalescence of the early fibrillations of right and left sides. The earliest agitation of ventricular contents is simply tida1. Blood is not propelled directionally by the early cardiac activity but its movement begins before the beat has involved the still incompletely fused sinus venosus. When contractions of the atrium start they are faster than those of the ventricle. With coalescence of activity in the atrium and ventricle there results an acceleration of the rate of the ventricular beat. similarly the contractions of the sinus venosus, when added to those of the ventricle and atrium, bring about a second acceleration.
The genesis of contractions ok the mamma1ian heart has been observed in hanging drops cu1tures of whole ernbryonic vesicles of katsJ The beat begins at the three somite stage before the primitive myocardial tubes have fused. Thus the heart starts to tunction at an earlier time in the rat, relativelzz than in the chiclc Embryo. Three or four cells of the left ventricular primordium, near its junction with the future atrium, begin to contract rhythmically (Fig. 2, A) . About two hours later a s1ower rhythm starts in the right ventricular ruhe, retaining an independent beat until the two sides kuse (Fig. 2, B) . When this occurs the lekt side dominates the right and becomes the pacemalcer in establishing a single wavedike ventricular contraction; some variations were encountered Atrial contraxztionss begin. later than do those of the ventricular tubes. 1t was impossible to determine the existence of earlier, arrhythmical contractions lilce those seen in the chiclc embryo; perhaps failure to observe them can be explained on the basis of technical differences in the two investigations.
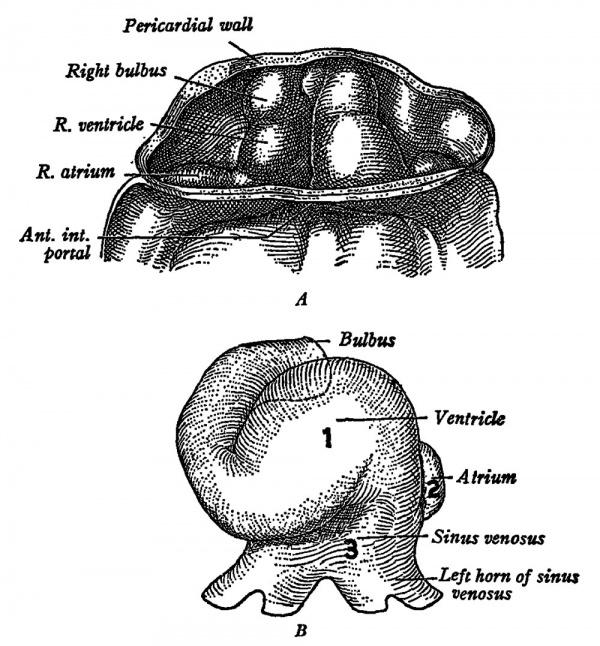
Fig. 3. The heart of human embryos of (A) 6 sornites and (B) 11 somites. The heart begins to beat in mamrnalian embryos eornparable with L. The order ok initiation ok the beat is shown in B by the figures 1 (ventricle) , 2 (atrium) and 3 (sinus venosus). (Arey: "Developmental Anatomy.")
The initiation of the heart beat has been studied in amphibian embryos where, in generaL the observations on birds and mams mals have been coniirmedF The first activity occurs in different parts of the ventricle in different specimens. Most of the embryonic amphibian hearts exhibit rhythmicity in the earliest stages of beating.
It was held for many years that the cardiac beat has its origin in the sino-atrial region and that this region remains the pace— malcer thereafterks I» The recent studies in amphibian, bird and mammalian embryos demonstrate that this is not the case. By curting the embryonic heart between ventricle and sinus venosus it has been found that the beat of the ventricle remains unaItered unless contractions have already begun in the other parts, in which case the two pieces of tissue take up independent rhythmsks C» S« U The intrinsic beat of the sinus venosus is faster than that of the atrium, which in turn is faster than that of the ventricle. Each newly acquired contracting portion when added to older parts sets a faster pace for them. It may be concluded that although the heart beat has its genesis in the ventricular region and not in the sinus venosus, regulation of the ventricular beat is brought under control of the sino-atrial region very early in development.
The Fetal Electrocardiogram
A number of attempts have been made to record action currents of the human«fetal heart near term by means of the electrocardiograph, the investigators using leads placed on the mother’s abdomen«and in the Wagina or rectum.12—I9 similar studies have been undertaken in the horsesV The records obtained have been of little value for interpreting events of conduction in the fetal heart because the dellections were small. The fetal electrocardiogram at 9 months is saick to exhibit a· simple upright monophasszic curve.21 More recently however electrocardiograms have been obtained from younger human fetuses removed at operationjVs 23 ln the three conventional leads the records showed äll deliections of the adult. The most signiiicant difkerence between human electrocardiograms at birth and in the adult is that the newborn right ventricle exhibits as definite functional preponderanceFÅ Z« This is correlated with the fact that the right ventricle outweighs the left by 13 per cent in full term fetusesks Left ventricular preponderance begins to become evident at about the second or third postnatal month, and, by the sixth month of life one can scarcely see any difference between electrocardiograms of infants and adults. These observations signify that the left side of the heart becomes larger than. the right by the second or third month. It is doubtful if this comes about in those infants in which the ductus arteriosus remains patent for several weelcs or months after birth and consequently a1lows the blood pressure on the two sides to equalize.
The developing bird’s heart offers the best opportunity to examine action currents critically under controlled conditions. A number of investigations have been made in the chiclc but only two merit consideration here. In one series of experiments27 small holes were drilled through the shell of incubating eggs and electrodes were inserted without disturbing the embryos. The eggs were then placed in a special incubator and allowed to remain quietly for some time before electrical records were made. In the other series28 the eggs were opened, blastoderms removed and placed in a special chamber. A micromanipulator was used to place the electrodes upon the embryo. Ampliftcation was employed in both cases.
The first deflections of the galvanometer were obtained from chicks of 15 somites (33 to 36 hours incubation) in which the heart consists almost entirely of ventricle. This is not much later than the time of initiation o·f rhythmical heart beaks. The first curves showed none of the deflections which characterize the adult but appeared as simple deflections first below, then above the isolec— tric line (Fig. 4) . In slightly older, 16 somite embryos a sharp downward deflection followed by a rapid return to or above the line appeared; this resembled the Q R s complex. The auricular deflection (P) did not appear until about 42 hours incubation, is» soon after the auricular beats had become established, and it was first seen as a downward deflection (Fig. z) . Later the P wave reversed. These results correlate nicely with what has been learned from direct- observations of. fhspe developing chick heart by the fourth day of incubation the embryonic electrocardiogram was practically identical with that of the adult hen. This is a remarlcable observation for there are no nerves in the heart at this time and the Special tissue of the cardiac conduction system is not as yet distinguishabla Time relationships taken from electrocardiograms of the chick will be found in Table 4.27
Fig. 4. Electrocardiogram from a 15 somite chick embryo (+/- 36 hours incubaction) . The diagram on the Iekt ok this and ok Fig. 5 shows the shape of the heart and position of the leads. (Hoff, et a1.: Am. Heart J. Vol. 17, 1939, C. V. Mosby Co)
Fig. 5. Electrocardiogram from a 20 somite chick embryo (+/- 42 hours incubation). Compare with Fig. 4. (Hoff, et a1.: Am. Heart J. Vol. 17, 1939, C. V. Mosby Co)
The Fetal Pulse Rate
The Pulse rate of chiclc embryos has been studied on a number of occasions, but most of the results are open to the criticism that temperature and mechanical kactorsjvere not controlled precisely during experiments The studies of Cohn and Wi1e29 and especially those of Bogue30 are the most significant. The latter recorded heart beats electrically from undisturbed eggs at a temperature which fluctuated no more than» o.250 C. It was found that the heart rate rises sharply in early stages of incubation, increases more s1ow1y from the tenth day onward to hatching, shows another rapid elevation at hatching and thereaftcr is maintained nearly at a constant 1evel throughout life. Average heart rates in chick embryos are illustrated graphically in Fig. 6. Bogue found that very slight temperature differences had a marked direct effect upon the embryonic pulse rate. He could find no relationship between the sex and pulse rate of chick embryos nor could a close correlation between metabolic rate and pulse rate be demonstrated. It may be signiiicant that a physiologic anoxemia is imposed upon the chiclc toward the end of incubationz this can have a direct inhibitory action upon the pacemalcer mechanism. The acceleration immediately after hatching may represent a release from the anoxemia.
| Age | Length of heart (mm) | Heart rate (/min) | P-R interval (sec) | Duration of R rise (sec) | Duration of R-T complex (sec) | Approximate conduction speed (mm/sec) |
|---|---|---|---|---|---|---|
| 40 hr. embryo | 0.8 | 120 | none | .009-.012 | 0.28-0.82 | 2.67 |
| 60 hr. embryo | 1.2 | 163 | 0.11 | .008-.010 | 0.20-0.21 | 3.80 |
| 120 hr. embryo | 2.0 | 230 | 0.09 | .010 | 0.21-0.24 | 6.85 |
| 5 day chick | 14.0 | 295 | 0.06 | .004-.006 | 0.24-0.33 | 40.58 |
| Adult hen | 40.0 | 320 | 0.06 | .009 | 0.15-0.18 | 117.77 |
Fig. 6. Heart rate during incubation of the chick. The solid line: Bogue’s data; brolcen linke: Cohn and Wile. (Bogue: J. Exp. Bio1., Vol. g. 1933.)
Because of its great importance in obstetrics, the human fetal heart rate has been studied extensively. It is well established that the average frequently at term is 130 to 150 beats per minute. Extremes of 110 and 180 are commonly observed but a rate of less than 1oo should be vieweLi with alarm. The average heart rate encountered in premature infants (about 120 per min.) is slower than that of infants at full term. The fetal heart slows during birth but accelerates when respirations begin and may be maintained at 17o to 180 beats per minute for iifteen to thirty minutes thereafter.92
Few systematic correlations of the human heart rate with fetal age have been made. In fact very little reliable information is available for any mammal. It is impossible to detect human fetal heart sounds accurately before about the fifth month. The human fetal heart beats faster in mid-fetal life than it does just before birth.33 A steady decline from a mean of 156 beats per minute at the fifth lunar month to 142 at the ninth, and a slight subsequent increase during the tenth month have been observed in one series of thirty subjects.
In rat embryos which had been exposed by opening the uterus it was found« that the heart rate fluctuated widely throughout pregnancy and was slower than but ran parallel to that of the mother. Only roughly did the data indicate an increase in frequency from early to late prenatal stages. During late fetal life of the dog, ox, goat and man the heart beats faster than it does in the young offspring after birth. A gradual decline is observed from infancy to maturity. This is illustrated in Table z, compiled from observations of several investigators.29-33, 47
The adult rate appears to be higher than the fetal rate during the latter part of gestation in the rat« and» monlceyks resembling the bird in this respect. The possibility of asphyxial depression of the fetal heart in the rat experiments should be borne in rnind, although most of the fetaj heart rates were not abnormally slow in comparison with other species of animals. The adult rat, being an example of a small animal with rapid pulse rate, may not actu— ally develop the inhibitory mechanism to the same degree found in larger mamma1s. The rapid maternal pulse of the adult monkey was attributed to the anima1’s excitement and exertion.
| Early fetus | Late fetus | Newborn | Adult | |
|---|---|---|---|---|
| chicken | 120—170 | 220-264 | 295 | 320 |
| rat | 96-114 | 123-248 | ... | 184—280 |
| Goat | ... | 120-246 | 145-240 | ... |
| Ox | ... | 161 | 141 | 50 |
| dog | ... | 120—170 | 160 | 100 |
| monkey | ... | 100—180 | ... | 140-240? |
| Man | 156 | l30-150 | 112 | 70 |
Nervous Control of the Fetal Heart and Circulation
It is thought that the progressive slowing of the pulse in postnatal life is associated with improvement of control of the heart by the vagus nerves. It has been pointed out that animals with small hearts have rapid pulse rates and those with large hearts have slower rates.37 control by the vagus is less pronounced in the former than in the latter. The higher metabolic rate of small animals is thought to make it necessary for the heart to beat about as fast as it can under normal circumstances; consequently there should be no call for an active inhibitory mechanism.
Perhaps the fetal heart rate is rapid because nervous control has not become well established or is inhibited in Some way. Most of the evidence points to the conclusion that vagus function is not nearly so marked in the fetus as it is in the adult, although considerable species variation is encountered. stimulation of the vagus nerves in the chick had no effects at 18 to 20 days of incubation, but it slowed the beat seven to eight hours after hatching.38 Inhibition wsas ekkected by direct stimulation of the heart itself in embryos of Ave days incubation.39 Muscarin produced the same result in even younger chick embryos; it slowed the heart of small rat embryos and was antagonized by atropine. These results in— dicate that the failure to obtain inhibition of the heart by vagus stimulation is not due to factors inherent in the myocardium.
Most investigators who have studied the phenomenon of vagus inhibition in young postnatal mammals have been able to produce slowing or cessation of the heart beat by stimulating the peripheral cut end of the nerve, but in many instances the ekkects seem to have been weaker than would have been obtained in adults.40-45 This was especially true in experiments with kittensJU The nearer to birth, postnatally, the less the chance of producing inhibition by stimulating the vagus nerves. In one newborn rabbit which showed no cardiac slowing upon stimulation of the peripheral vagus, respiratory inhibition was demonstrated by stimulating the central end, indicating that the nerve is capable of conducting and that conduction can take place through medullary centers at birth.45 In prenatal life a few successes as well as many failures have been reported in several species.40- 4I- 45- 43 It is probable that the negative results were obtained in many instances because the vagus nerves had been stimulated only after a maximum decline in the rate of the heart beat had occurred in consequence of asphyxia.
In experiments with rabbits Bauer« has found that stimulation of the vagus nerve increases the decline in heart rate which is brought about by asphyxia. This was not demonstrable how— ever until about the fourth day after birth. Clamping the umbilical cord at experimental Caesarean section led almost immediately to asphyxial bradycardia, b·ut the phenomenon was delayed in specimens which had breathed air and in which asphyxia was sub— sequently produced by occluding the trachea. The amount of oxygen available in the blood was greater in the latter than in the former instance. The asphyxial slo.wing of the fetal heart rate appeared to be due, not to inHuence of the central nervous system efkected through the vagus nerves, but. to a direct chemical action upon the pacemaker of the heart.
Various theories have been proposed to explain the slowing of the human fetal heart at the time of delivery. Compression of the skull of young rabbits produces bradycardia and some have held that the passage of the fetal head through the birth canal may bring about enough pressure to cause a similar cardiac depression. However, a declining heart rate is not infrequently encountered during 1abor before the head becomes engaged in the 1ower part of the pelvis.
lt has been held also that the human fetal heart rate varies with uterine contractions and relaxationssfss I! 1t is probable that a greater volume of blood is forced into the fetal heart by each uterine contraction and that the pulse rate consequently declines as the blood pressure rises in accordance with Mareyks law. Clark so demonstrated experimentally that the fetal blood pressure under— goes a brief rise followed by a prolonged fall during contraction of the cat’s uterus. He thought that the fall was due to a diminished venous return to the fetal heart which resulted from increased peripheral resistance set up in the placenta by the uterine contractions.
Although there appears tobe no vagal tone before birth in sheep fetuses, ligating the umbilical cord results in an immediate elevation of the blood pressure and, in response to Marey’s law, an instantaneous bradycardia. When the vagi had been cut bradycardia followed occlusion of the cord, coming on gradually or suddenly as a 2:1 heart block. In either case it appeared only after an interval of about 25 seconds during which asphyxia had developed.
It has been reported that the fetal heart rate is entirely unalfected by changes in the oxygen and carbon dioxide contents of the mother’s blood.32 However, amyl nitrite, which has a great relaxing effect on uterine and other smooth muscle, has an immediate influence on the mother’s heart rate and within a few seconds a similar slowing of the fetal heart occurs. This delay sug·gests a comparable phenomenon encountered during asphyxia in rabbit fetuses.
The application of Marey’s law to explain· fetal bradycardia implies that ksascular reilex mechanisms are functioning in the fetus, at least near term. This is not borne out by experimental evidence in all species. Observations in cat and dog fetuses at experimental Caesarean section and in the young after birth led to the conclusion that pressor rellexes make their appearance about three or four days postnatally ksss No evidence of cardioaortic and carotid sinus reflexes was found before the fourth to sixth day in puppies and not before 1 1 days after birth in 1cittens. Faradic stimulation of the, depressor and carotid sinus nerves begins to elicit reflex inhibition ok the heart in 11 and 14 day old rabbits, respectively Asphyxiation fails to bring these reflexes into play unti1 about the kortieth day ok like because the blood pressure of -the young anima1 has not attained the necessary height until this time (Fig. 7) . The depressor reiiex hegins to be obtained when the systemic arteria1 pressure reaches 65 mm. Hg and the carotid sinus reliex appears at 8o mm. pressure. These are much higher pressures than are encountered in ketuses ok rabbits and other small anima1s. In the sheep and in man at the end of pregnancy ketal blood pressures are high enough to aikect responsive cardioaortic and carotid sinus mechanisms.
Fig. 7. Blood pressure of rabbits at different ages after birth. J, Maximum blood pressure attained in asphyxiaz B, normal blood pressure (Bauer: Jour. Physiol» Vol. 95, i939.)
The general conclusion reached from experimental studies and clinica1 observations is that the ketal heart pumps blood about as fast as it can without much control by the nervous System. The vagus nerves together with their endings and central connectionse are capable of conduction in some species near term, but in others which are born less completely equipped to cope with their en— vironment they may not be very well developed. Failure ok cardio-inhibitory and vascular reflex mechanisms to function at birth is related at least partially to low systemic blood pressures prevailing in the smalleiz less mature newborn animals. Asphyxia produces cardiac depression by direct chemical action upon the pacemaker.
An explanation of the slowing of the human fetal heart during birth may be fortheoming from these studies. It is inferred that elevation of the blood pressure caused by uterine contraction brings about a reflex bradycardia. A decline in the oxygen saturation of the fetal blood during labor may induce an anoxial cardiac depression.
The prompt but transient acceleration of the heart which occurs after breathing starts at birth may result from an awalcening, as it were, of sympathetic tone consequent upon the shower of new afferent impulses from the external environmenn Here too experimental evidence in· the sheep is available. The smooth muscle of the fetal spleen, which isinnervated by sympathetic neurons only, can be induced to contract reflexly by stimulating the central end of the cut vagus. splenic activity also follows ligation of the umbilical cord and is not related to the changes in blood pressure occasioned by this procedure. Furthermore, contraction of a smooth muscle sphincter of the ductus arteriosus occurs in the lamb at birth. In the chiclc sympathetic mechanisms are well formed at hatching time but vagus inhibitory function is deficient. 29
Arterial Blood Pressure
Determination of arterial blood pressure in fetuses of small animals involves diflicult technical problems and one can seldom be certain that results reflect the true condition in utero. Consequently few systematic studies have been reported. The force of the embryonic chiclc heart at two or three days incubation can lift a column of water only two centimeters during systole.34 , Pressures of 5 mm. to 20 mm. of mercury are necessary to obliterate How in the umbilical artery of rat fetuses 16.4 to 23.3 mm. 1ong.34 The variations in fetal carotid arterial pressure which accompany uterine contractions have been studiedfos I» Maximum estimations of zo mm. of mercury in the cat and 4o mm. in the dog fetus were reported during the latter part of gestation. The adult level was not reached in the dog until about 4o days after birth« and even Iater in the rabbit. 4
Haselhorstss has studied blood pressures in human umbilical arteries. At one Caesarean Section he found it 68 mm. of mercury before delivering the child. At normal delivery in eight other experiments pressures varied between 46 mm. and no mm. of mercury, averaging 75 mm. It was concluded that there is no signiiicant ditference between the arterial pressure at full term in utero and after delivery. An initial umbilical arterial pressure of 60 to 70 mm. of mereury was found in one newborn infant before respiration started; this did not change with the advent of respiration but was temporarily elevated to 1oo mm. at the first cry. This experiment is illustrated in Fig. 8.
Fig. 8. Blood pressure in the human umbilical artery at birth. cannu1a in— serted at E and withdrawn at d. Respiration began at «! and the infant cried at s. (1-Iaselhorst: Ztschn Geburtsh. Gynälc.. Vol. g5, 1929.)
Fig. 9. Relation between blood pressure and fetal« age in the sheep. (Barcroft and lcennedzu Jour. Physiol» Vol. g5, 1g39.)
Barcroft and Kennedyw have made the most comp1ete series of observations correlating blood pressure with the fetal age in the sheep and their data are reproduced in Fig. g. 0ther records of 39 mm. to 51 mm. of mercury at approximately 1 1o to 12o days gestation and 84 mm. near full term are available in this species. 58
Print« Bsnoov Pnnsstrnn Nma Tnrku
Rat . . . . . . . . . . . . . . . . . . . . . . . . . . . . A) . · . . .
gäiåräea pig . . . . . . . . . . . . . . . . . . . . . . . . 5-10
it. . . . . . . . . . . . . . . . . . . . . . . . « . . . . .
Cis-l: . . . . . . . . . . . . . . . . . . . . . . . . . . . 80 7-I3
åog . . . . . . . . . . . . . . . . . . . . . . . . . . . W— X« . .
eep . . . . . . . . . . . . . . . . . . . . . . . . .
Mut! . . . . . . . . . . . . . . . . . . . . . . . . . cis-IN) 22-24
Left ventrieular systolie pressure.
A marked elevation of the blood pressure has been observed to accompany respiration at birth of the sheepfls 59 It is thought to be caused by respiration but not necessarily to be permanently maintained by it. How this comes about is illustrated in Fig. 10.
Fig. 10. Femoral arterial blood pressure ok the lamb at birtlx The Erst: and subsequent respirations are indicated by the Signal (lower line); time in seconds. A diagratnmatie interpretation ok the eikect ok respirations (R1, R» etc.) in elevating the blood presst-re is shown below the tracinz (Barcrokt: «The Brain and Its Environment," Yale Univ. Press.)
It was suggested that the cardio-accelerator center ok the brain is set into activity by afkerent impulses from the lungs or diaphragm vasoconstriction, like the contraction ok the splenic smooth muscle, may be initiated similarly.
Another investigator32 has suggested that an elevation of systemic arterial pressures and the increase in the rate of heart beat after birth are kactors which operate to overcome the apnea of ketal like and initiate respiration by causing more blood of high carbon dioxide content to reach the respiratory center in the brain. But the truly signilicant elevation ok bIood pressure follows res.piration. It is probabIe that any marked rise in arterial pressure appearing upon establishment of respiration signikies that the fetus was previousIy depressed by asphyxia. Some experiments have demonstrated prenatal arterial pressures nearly as high as those after breathing has begun.
By means of a special high—speed hypodermic manometer and photographic recording, Hamilton, Woodbury and Woodsso have obtained left ventricular pressures of 45X2 and zoxo in dog fetuses near term before breathing started. In the specimen having a pressure of 45X2 this increased to zoxo after the umbilical cord had been«clamped and breathing had begun. Pressures were i4Xo in a premature rabbit fetus, 2oX i at term before breathing began, 28Xo at the end of inspiration at birth and Zoxz at the end of ex— piration. The newborn rabbit’s pressures were »so-i at inspiration and 4oXi at expirationz those of the two day old were 27X2 and 47X11. It is evident that the left ventricular pressure rises and falls with expiration and inspiration but the mean pressure increases only gradually toward the adult level after respiration is established. Hamilton and his colIeagues found that right and left ventricular pressures talcen simultaneously were similar before breathing. Clamping the umbilical cord caused little or no change, but they point out that the umbilical circuit may have been obliterated before they clamped the umbilical cord. Upon establishment of air breathing the right ventricuIar pressure dropped in inspiration more than. the left because a negative intrathoracic pressure was established, resulting in decreased peripheral resistance in the lungs.
Venous Blood Pressure
Several investigators have reported venous pressures taken from the fetus at experimental hysterotomy. cohnstein and Zuntzss found pressures in the« lamb’s umbilical vein to vary between 16 mm. and 34 mm. of mercury at about Iio to 120 days gestation, and 33 mm. near term. The average of these values was about half the average of the arterial pressures. This gave the impression that thse venous pressure of the fetus is relatively much higher than that of the adult. Barcroft and KennedyM have estimated the pressure in the umbilical vein of the fetal sheep to be less than 1o mm. at 56 day gestation, about 1o mm. at 1 ro days and 18 mm. of mercury at 140 days (see Fig. g) . Blood passes through the umbilical veins of cat and guinea pig fetuses near term under pressures of 5 mm. to 13 mm. I-Ig, rising and falling with contraction and relaxation of the uterine musculatureJU Arterial pressures were not obtained in these animals but it is probable that they were considerably more than twice the venous pressures Because all the estimations of fetal venous pressures were made at experimental hysterotomy it is probable that many were higher than in -utero, being elevated by the force exerted upon the vascular bed of the placenta by the contracting uterus. Actua1ly a greater difkerential between systemic arterial and venous pressures is to be expected in the undisturbed fetus in utero than appears from the records obtained in the older experiments. It is diflicult to believe that there can be urine formation in the fetal lcidneys if the venous pressure is as much as half the arterial pressure (see Chapter VIII) .
Haselhorstw recorded the pressure in the human umbilical vein at Caesarean Sections in three instances while the uterus was quiescen»t. It varied between 22 and 34 mm. of mercury. In one case the pressure in the vein increased from 24 to 7o mm. when pituitrin was injected into the uterus. A possible placental or uterine function in maintaining an adequate venous return to the, fetus should not be overloolced.
References Cited
. Fano, G. 1885. Lo sperirnentale, i: 143 (cited by F. Bottazzi sc G. Fano, 19oo, in Richet«s Dict. Physiol» 4: ask» Alcan, Paris) .
I-Ioolcer, D. 1911. J. Exp. Zool» u: 159.
Lillie, F. R. 1919. The Development of the Chiclg Henry I-Iolt, N. Y. sabin, F. R. 192o. Contrib. Emb., g: arg.
Johnstone, P. N. 1925. Johns Hoplcins Hosp. Bull., 363 299.
. Farren, B. M. sc T. c. lcramen 1933. Am. J. Anat» 5z: 349.
Goss, C. M. 1938. Anat. Rec., 7o: gez.
. Copenhaven W. M. 193g. J. Exp. Zool» so: 193.
. Fano, G. 8c F. Bodano. 189o. Arch. Ital. Biol., is: 387.
Piclcering, W. 18g3. Physiol» 143 383.
II«
ZO Orts-wesse-
ii. Pakk, G. H. i9s5. Anat. Rec., 6s: sos.
is. Cremer, M. igo6. Mönch. med. Wochenschr., i: 8ii.
is. Foä, C. i9ii. A.i·ch. 1tal. Biol., zö- i45. i4. Nin-r, J. igsi. Ztschix Biol., 7s: iss.
is. sachs, H. i9ss. Pklügeks Arch., i97: 5s6.
i6. Hayna.l, E. sc D. Kellner. i9s4. Ztschn Hin. Mai» 98: s65.
i7. Maekawa, M. sc J. Toyoshima. i9so. Acta seh. Med. Univ. Imp. Kyoto,
is: 5i9.
is. strassmann, E. O.· i9s6. stakk Proc. Mayo Clin., ii: 778.
i9. Bell, G. H» i9s8. J. Obst. Gyn. Brit. Emp» 45: 8os.
so. stefkan, H. sc E. strassma»nn. i9ss. Zentralbl. Gynälc., 57: 6io.
si. Krurnbhaaiy E. B. i9i6. Am. J. Physiol» 4o: iss.
ss. Eashy, M. H. i9s4. Am. Heart J» io: us.
ss. Heard, J. D., G. G. Burlcley s: C. R. schadet. i9s6. Arn. Heart J., ii:
4i.
44. Kruinbhaaiz E. B. sc H. H. Jenlcs. i9i7. Heim, S: i89.
45. Leu-is, T. i9i6. .Phil. Trans. Roy. soc» Lond. B» so7: ssi.
46. Patten, B. M. igss. In A. H. Curtis« Obstetrics and Gynecology, i: saunders, Philadelphia
s7. Bogue, I. Y. i9ss. J. Exp. Biol» io: s86.
s8. HolL E. C» T. C. Kramer, D. DuBois sc. B. M. Farren. i9s9. Anker.
Heart J., i7: 47o. Cohn, A. E. s: E. L. Wile. i9s5. J. Exp. Med., 4s: s9i.
Bogue, I. Y. i9ss. J. Eikp. Biol., g: s5i. Blaclckan, K. D. igss. Growth and Development ok the Childz White
House Conkerence Reports sect. I, Pt. i: P. 5s. Century Co» N. Y.
. Kraklca, J. i9ss. Am. J. Dis. Child» 45: ioo7.
sontag, L. W. and T. W. Richards i9s8. Monog.. soc. Res. Child
Des-ei» Vol. s, No. 4.
Corey, E. L. i9ss. Am. J. Physiol» ioi: so4.
Hartman, C. G» R. R. squier s: O. L. Tinlclepaugh i9so. Proc. soc. Exp. Biol. s: Med., s8: s85.
. Barcrokh J. i9s6. Physiol. Reis» is: ios. . Clarlc, A.· J. i9s7.
Comparative Physiology ok the Heart, Cambridge Univ. Press.
Botta2zi, F. s: G. Fano. i9oo. In C. Richet’s Dictionnajre de Physiologia 4: s5s. AIcan, Paris.
Pickeiinz W. i896. Physiol» so: i65.
. soltmann, O. i877.« Jahrb. Kinderhllc» ii: ioi.
. Anrep, B. i88o. Pfliigeks Arch» si: 78.
. Heinrichs, G. i89o. «Ztschr. Biol., s6: i·97.
. Meyer, E. i89s. Arcli. Physiol. Norm. et Path» z: 475. . Buglia, G. igs6. Archq Fisiol» s4: 448.
. Kellogg, H. B. . Clark, G. A.
. Bauer, D. i9s8. Ibid» 9s: gox 95: i87. . LeiL M. i9ss. Arn. J. Obst. Gyn., s4: 898. . Wissen, C. J.
i9s7. Pi·oc. soc. Exp. Biol. s: Med» s4: 8s9. i9s4. J. Physiol» As: ss9.
i9s4. In I. A. Abiks Pediatrics, 4: i98, saunders Phila— delphia.
. C1ar1c, G. A. i9ss. J. Physiol» 74: s9i.
Hi. Barcrokh J. 1938. The Brain ancl Its Environmenr. Yale Und. Frass,
New Hatten. «
se. Bach, W. 193i. Arclh Gynäk.,·i47: se.
53. Tayloxz D. B. sc T. Gotsem 193s. citecl by J. Bat-Gott, 1938.
54. Bill, L. sc Y. A2uma. 1927. Physiol» 62: 27P.
55. Mark, G. A. sc H. E. Ho11ing. 1931. 1bicl., 73: 3o5.
56. Haselhorsh G. Weg. Ztscluc Geburt-h. Gynä1c., 95: 4oo.
M. Bekannte, J. sc J. A. Icenneclzn 1g3g. J. Physiol» gzc 173.
58. cohnsteirh J. sc N. Zuntz 1ss4. Pilügerk Arm» 34: i73.
59. cohnsteirh J. sc N. Zuntz 1ss8. 1bicl.. 42: 342.
6o. Hamilton, W. F» R. A. Wooclbury sc E. B. Woods. 1937. Am. J. Physiol»
119: 2o6. Si. DeMarslh B. sc W. F. Winde. 193g. Unpublishech
Cite this page: Hill, M.A. (2024, April 26) Embryology Book - Physiology of the Fetus 2. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Physiology_of_the_Fetus_2
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G