2011 Group Project 9
| Note - This page is an undergraduate science embryology student group project 2011. |
2011 Projects: Turner Syndrome | DiGeorge Syndrome | Klinefelter's Syndrome | Huntington's Disease | Fragile X Syndrome | Tetralogy of Fallot | Angelman Syndrome | Friedreich's Ataxia | Williams-Beuren Syndrome | Duchenne Muscular Dystrolphy | Cleft Palate and Lip
Williams-Beuren Syndrome
--Mark Hill 13:09, 8 September 2011 (EST) This is a good start in the way of sub-heading structure. The current problem is that many of these sub-headings contain no content. A previous individual assessment was to insert your text on the project page, someone has either not added their text or their are unallocated sections. Either way it makes the project look very unfinished in places. More importantly at this stage is the lack of any related images or student illustration.
- Introduction - should be more a brief text intro rather than bullet points here.
- History of the disease - "William-Beuren Syndrome is named after John C.P. Williams," perhaps you should start with this. Timeline, this sub-sub heading has no text.
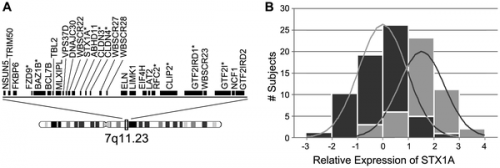
- Genetic factors and Etiology - could indicate the region deleted in an image.
- Diagnosis - there is a list of descriptions that should be illustrated. seems to be an overlap with the following section (Physical Characteristics).
- Physical Characteristics - there is no text in this section.
- Epidemiology - shod be linked earlier to diagnosis.
- Glossary - no descriptions. Should include all acronyms used (FISH, STR etc).
- Referencing - formatting seems to be applied well, I have not checked at this stage that all content is correctly cited.
Introduction
Williams-Beuren Syndrome, more commonly known as Williams Syndrome, is a congenital anomaly caused by the deletion of 28 neighbouring genes on chromosome 7q11.23.[1] [2]
This multisystem developmental genetic disorder implicates psychological, behavioural and medical defects, including diverse phenotypic characteristics such as distinctive facial deformities, cardiovascular abnormalities,intellectual disabilities/mental retardation, growth abnormalities, endocrine abnormalities and a unique personality and cognitive profile. [1] [3] Some or all of these features may be present in varying degrees, with the condition becoming apparent at the onset of birth or in early infancy. [4]
History of the disease
William-Beuren Syndrome is named after John C.P. Williams, a cardiologist from New Zealand, and Alois J Beuren, a German physician and cardiac researcher.
Initial investigation into Williams-Beuren Syndrome came from two apparently different disorders, idiopathic infantry hypercalcemia and Supravalvular Aortic Stenosis (SVAS). With further research, these abnormalities were identified as being aspects of this same syndrome. [5]
The first cases related to Williams Syndrome were in relation to Infantile Hypercalcemia. In 1957 Stapleton and colleagues studied the effects of hypercalcemia in a number of infants and noted several consistencies between them including abnormal facial features, failure to thrive, developmental delay, and systolic murmurs of the heart, all of which have now been associated with Williams Syndrome. [6] [1]
J.C.P Williams was one of the first to recognise some of the clinical factors associated with this syndrome. In a study conducted in 1961 of Supravalvular Aortic Stenosis (SAoS), an obstruction occurring in the left ventricular outflow tract (LVOT), Williams and his colleagues made the observation that patients suffering from this heart condition had strikingly similar unusual facial features that included broad foreheads, eyes set wider apart than normal, wide mouths with pouting lips and malocclusion of teeth, pointy chin and prominent pointed ears. As well as this they discovered their subjects also presented with mental retardation and a low IQ. Williams and his colleagues suggested that their findings might be indicative of a previously unrecognised syndrome. [5] [7]
In 1962 AJ Beuren and associates also studied the correlations between SAoS, mental retardation and distinctive facial features of a number of subjects and made similar observations to Williams, particularly pointing out the characteristic “elfin” features of Williams syndrome patients. [8] [9] Beuren also noted the behavioural traits of his subjects suffering from SAoS, describing them as all having a “friendly nature”, something which would later be recognised as one of the unique personality traits of people diagnosed with Williams-Beuren Syndrome. [8] [5]
In further studies conducted in 1964, Beuren and his colleagues detailed the possible association of Peripheral Pulmonary Stenosis and complex dental malformations with SAoS, mental retardation and certain facial appearance which they examined previously. They too came to the conclusion that these complications were representative of a new syndrome. [10] [5]
Timeline
--Mark Hill 13:16, 8 September 2011 (EST) No content here?
Genetic factors and Etiology
Williams Syndrome is a multi-system genomic disorder that occurs due to a hemizygous deletion/nonallelic homologous recombination (NAHR). The sizes of deletion commonly range from 1.55 to 1.84 mega base pairs (Mb) on chromosome 7q11.23 which encompasses 28 genes.[1] [3]
The region associated with Williams syndrome contains a single copy gene region with repetitive sequences or Low Copy Repeats (LCR).The deletions that cause Williams syndrome are due to a misalignment of these repetitive sequences or gametes within the Williams-Beuren syndrome critical region. [11] This occurs during the process of meiosis and follows an unequal crossing over that is due to a high similarity of LCRs.
A genotype-phenotype correlation has been found for some of the genes within the deletion region. The most well known of these being elastin gene (ELN), whereby elastin haploinsufficiency is responsible for a number of abnormalities characteristic of Williams Syndrome, particularly connective tissue abnormalities and cardiovascular disease including arterial stenosis. [12] CLIP2, GTF2I, GTF2IRD1 and LIMK1 are some of the other genes that are most commonly deleted in individuals with Williams syndrome. Researchers have suggested that the deletion of these genes could help in the explanation of some of the characteristic signs of Williams syndrome including the unique behavioural characteristics and also some of the related cognitive difficulties.
In most cases, Williams syndrome occurs sporadically and the deletions can occur with no reference to the parental origin of the chromosome that transmits the disease. There have been, however, a very small number of cases where autosomal dominant inheritance of Williams syndrome has been reported.
| Gene | Normal Function | Relation to Williams Syndrome |
| Elastin (ELN) | Elastin is a main component of elastic fibres. It also contributes to the structure of connective tissue, particularly its flexibility and strength. | The loss of one copy of this gene reduces the normal production of elastin by half.
Elastin haploinsufficiency is responsible for a number of abnormalities characteristic of Williams Syndrome, particularly connective tissue abnormalities and cardiovascular disease including arterial stenosis. [12] |
| LIM domain kinase 1 (LIMK1) | Studies, such as Wang et al in 1998 [13] suggest that LIMK1 is involved in the area of the brain that is in control of the visualisation of an object as a set of parts.
It has also been implicated in other visual tasks such as drawing, making models and writing. |
Studies differ in regards to the contribution of LIMK1 deletions to the phenotype expressed in Williams syndrome. Some studies have suggested that the loss of this gene leads to the problems with visuo-spatial tasks that are common in Williams syndrome. Other studies suggest that it is involved with the characteristic progressive loss of hearing, [14] while other studies have not found these connections. |
| General transcription factor IIi (GTF2I) | This gene is involved in the production of the following two proteins:
• BAP-135: involved in the normal function of the immune system • TFII-I: helps in the regulation of other genes activity and is therefore active in many tissues of the body, particularly in the brain. |
It has been suggested that the loss of one copy of this gene may be responsible for the intellectual disability seen in Williams syndrome. It may also be involved in the social characteristics of those with Williams syndrome. |
Diagnosis
There are ways to clinically diagnose Williams Syndrome even though the phenotype varies between individuals. There is not a particular clinical feature that is able to ascertain the diagnosis alone, but there are particular features that individuals with Williams Syndrome may present with. These features include conditions and abnormalities discussed in the associated medical conditions sections, but most commonly include:
•Distinctive facial features
•Unique personality
•Intellectual disability
•Growth abnormalities
•Cardiovascular Disease
•Endocrine Abnormalities
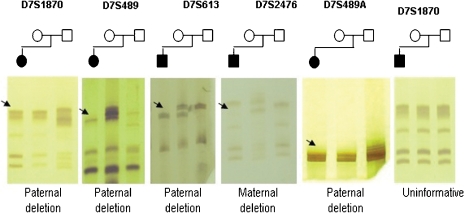
Although these clinical criteria for diagnosis are available, the detection of the contiguous gene deletion responsible for Williams Syndrome is the main form of diagnosis. It has been found that over 99% of individuals that meet the clinical diagnosis criteria for Williams Syndrome also have this contiguous gene deletion. This gene deletion is detected through the use of fluorescent in situ hybridisation (FISH) or through targeted mutation analysis. [15]
Fluorescent in situ hybridisation (FISH): FISH uses elastin probes for a specialised form of chromosome analysis. This method is commercially available and commonly used to determine if an individual showing the clinical signs of William’s syndrome does have the deletion of the genes in the Williams-Beuren syndrome critical region. If the individual only has one copy of the elastin gene, then a diagnosis of William’s syndrome is confirmed.
Targeted Mutation Analysis: these are non FISH methods that are also used to detect the contiguous gene deletion in order to determine whether or not an individual has Williams Syndrome.
•Real-time quantitative PCR: is used to establish the number of copies of three of the genes contained within the Williams-Beuren syndrome critical region. A deletion in this area would mean that only a single copy of one of these genes would be found.
•Genomic microarray analysis: makes use of array genomic hybridisation techniques which involved examining multiple genes simultaneously and comparing them in order to determine whether there are any abnormalities in the amount of chromosomal material.
•Heterozygosity testing: is mostly used to identify the size of the deletions, and involves the testing of short tandem repeats (STRs) from the Williams-Beuren syndrome critical region. If there is no deletion, than the STR sizes should be different at each of the markers determining heterozygosity. But if only one STR size is found at a marker, than this may indicate that there is a deletion, but may also indicate homozygosity. Quantitive PCR is then needed to determine whether or not the single STR size is dur to a deletion in the critical area or a non-abnormal finding of homozygosity.
Phenotype of Williams Syndrome
Most physical musculoskeletal abnormalities result from the absence of elastin due to the elastin gene deletion.
| Features | |
| Growth | • Transient hypercalcemia
• Developmental delay in height and weight. Often presenting smaller than others at the same gestational age and grow to have a short stature. • Poor coordination as well as a low IQ. The average IQ is 55 ranging from 40-90. This starts off with a failure to thrive as infants especially in language and motor development. • Some degree of mental retardation ranging from moderate to mild in the majority of cases. • Hypercacusis, an abnormal sensitivity to everyday sounds. |
| Facial Characteristics | • Lips are full and prominent. Often wide and help open.
• Teeth are often missing and/or hypoplastic, has thin enamel. • Long philltrum also present in the majority of cases. • Flat nasal bridge with a shorter, upturned nose and anterverted nares. • Eyes often have a stellate arrangement of the iris. • Eyebrows flare medially • Ears are lower than the normal height on the face and have prominent ear lobes. |
| Musculoskeletal Abnormalities | • Joint limitation
• Kyphoscoliosis, an abnormal corving of the spine in both the sagittal and coronal planes. • Hallux Vagus, a projection of the metatarsophalangeal joint inward to the innerfoot • hypoplastic nails, undergrown or incomplete nails. • Horse voice |
Cardiac Conditions
Stenosis
Other problems
Genitourinary Conditions
Renal Tract Abnormalities
18% of people with Williams Syndrome have some form of renal tract abnormality. This includes:
Renal Agenesis
Renal agenesis is a disorder involving the absence of one or both of the kidneys, categorised into unilateral or bilateral respectively.
Duplicated kidneys
Vesicourinary reflux
Nephrocalcinosis
Other Abnormalities
There are a number of other abnormalities associated with Williams Syndrome including a hoarse voice, inguinal hernias and joint abnormalities. These abnormalities vary in severity between different individuals and elastin haploinsufficiency is responsible for a number of these abnormalities characteristic of Williams Syndrome.[12]
Endocrine
Hypercalcemia
Diabetes Mellitus
Thyroid
Other Associated Medical Conditions
Joint Abnormalities : info
Inguinal Hernias : info
Auditory Abnormalities : [16]
Anxiety Disorders : It has been found that when compared to the general population, children with Williams syndrome have a significantly higher rate of anxiety related disorders. They particularly showed a higher occurrence of generalised anxiety disorder and specific phobia disorder. [17]
Vocal cord paralysis : info
Hoarse Voice : The hoarse voice is present in 98% of people with Williams Syndrome and it is due to a connective tissue abnormality, where the lamina propria in the vocal folds has a decreased amount of elastic fibres.
Cognitive, Behavioural and Neurological Problems
Speech impairment
Social use of language
Sociability
Musical ability
Epidemiology
Management
Currently, there is no cure for Williams-Beuren Syndrome as it is a complex multisystem medical condition. As symptoms of Williams-Beuren Syndrome involve multiple disciplines, treatment of those symptoms requires a large clinical team of doctors and nurses.
Treatment
Currently, there is no cure for Williams Syndrome as it is a complex multisystem medical condition. The treatment of conditions brought about by Williams-Beuren Syndrome can be difficult depending on the number of recognised conditions. The treatment of specific WS phenotypes such as hypercalcemia involves constant monitoring of the patients’ blood calcium levels and food intake. 15% of patients with Williams-Beuren Syndrome show signs of hypercalcemia and it is important to note as the condition contributes to the presence of extreme irritability, vomiting, constipation, and muscle cramps. Treatment of hypercalcemia may include diet modification, oral corticosteroids, and/or intravenous pamidronate.In any case, the WS patient should be referred to a nephrologist in order to treat renal abnormalities as they arise. [11]
Rate of Incidence
Williams-Beuren Syndrome occurs in less than 1 in 20,000 births. In the U.S, this figure is slightly more accurate with occurences of 1 in 7,500-20,000 births.
Specialised Facilities and Supportive Associations
Case studies
Interesting facts
Current research and developments
Glossary
Congenital anomaly:
Hemizygous:
Nonallelic homozygous recombination(NAHR):
Phenotype:
Hypercalcemia:
References
- ↑ 1.0 1.1 1.2 1.3 <pubmed>2042578 </pubmed> Cite error: Invalid
<ref>tag; name 'PMID20425781' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID20425781' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID20425781' defined multiple times with different content - ↑ http://omim.org/entry/194050
- ↑ 3.0 3.1 <pubmed>19568270 </pubmed>
- ↑ <pubmed>21107555 </pubmed>
- ↑ 5.0 5.1 5.2 5.3 <pubmed>21120465 </pubmed>
- ↑ <pubmed>13469755 </pubmed>
- ↑ <pubmed>14007182 </pubmed>
- ↑ 8.0 8.1 <pubmed>13967885 </pubmed>
- ↑ <pubmed>18941598 </pubmed>
- ↑ <pubmed>14136289 </pubmed>
- ↑ 11.0 11.1 <pubmed>20301427 </pubmed> Cite error: Invalid
<ref>tag; name 'PMID20301427' defined multiple times with different content - ↑ 12.0 12.1 12.2 <pubmed>20425789 </pubmed>
- ↑ <pubmed>9685409 </pubmed>
- ↑ <pubmed>21655442 </pubmed>
- ↑ <pubmed>7693128 </pubmed>
- ↑ <pubmed>20425785 </pubmed>
- ↑ <pubmed>20161441 </pubmed>