Paper - Development of the thyroid and parathyroid glands and the thymus (1950)
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Boyd JD. Development of the thyroid and parathyroid glands and the thymus. (1950) Ann R Coll Surg Engl. 7(6): 455-71. PMID 14790564
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Development of the Thyroid and Parathyroid Glands and the Thymus
by
Professor J. D. Boyd, M.A., M.Sc., M.D.
Anatomy Department, London Hospital Medical College
Lecture delivered at the Royal College of Surgeons of England on 30th June, 1950
Introduction
The thyroid and parathyroid glands and the thymus are derivatives of the embryonic pharyngeal region. A full description of their development, therefore, would entail a detailed account of the embryological history of the pharynx and related structures. Such an account is not possible in the present contribution which necessarily has had to be restricted to a consideration of the development in the human embryo of the three named structures. Further, discussion has had to be restricted to those stages in development, and to those details, that may have an interest to the surgeon or the surgical pathologist.
A study of the developmental history of the pharyngeal derivatives in the higher vertebrates is inevitably coloured by the phylogenetic background of the pharynx and of the branchial apparatus. The early position and nature of the endodermal pouches and the ectodermal clefts in these vertebrates and of the related skeletal structures, muscles, arteries and nerves, when compared with the gill region of fishes, furnish one of the most obvious examples of so-called “ recapitulation.” Nevertheless, it must be stressed at the outset that phylogenetic speculation in this regard has often outpaced reason and fact almost, or indeed actually, beyond hailing distance. With the registration of this caveat, however, it can clearly be stated that a knowledge of the phylogeny of the cervical region brightly illumines its topographical anatomy and throws much light, too, on developmental anomalies.
The Primitive Pharynx and the Pharyngeal Pouches
The pharynx itself is initially an endodermally lined cul-de-sac which forms the cephalic extremity of the foregut. The latter is derived from a part of the yolk sac by that process known as “ formation of the head fold.” This process results, amongst other fundamental changes (see Hamilton et al. 1945), in the production of the narrow endodermal foregut diverticulum which extends forwards, dorsal to the septum transversum and the region of the developing heart, and ventral to the notochord, to terminate blindly at the buccopharyngeal membrane. Here, the endoderm is fused with the ectoderm of the stomatodaaum or primitive buccal cavity. With the appearance of the primordium of the pulmonary apparatus as a small ventral outgrowth of the foregut diverticulum the latter can be divided into a more cranial pharynx and a more caudal foregut proper.
With rupture of the buccopharyngeal membrane the pharynx comes to communicate with the ectodermal stomatodaaum. In human embryos this rupture occurs at about the twenty somite stage, that is, when the embryo is about 26 days old. At approximately the same time, the lateral walls of the pharynx show growth alterationswhich result in a relatively marked increase in its breadth. The increase, however, is not uniform so that a series of five pairs of endodermally lined pouches are produced. These appear in order from before backwards, and extend towards the ectoderm where corresponding ectodermal clefts are developed. In lower vertebrates the intervening mesoderm between the endoderm of a pharyngeal pouch and the ectoderm in the -depths of a branchial cleft completely disappears, and the endoderm and ectoderm fuse. Finally, rupture of the fused layers occurs so that a gill cleft is produced. In the higher vertebrates the endoderm of the lateral extremities of the pouches, at least in the anterior members of the series, also fuses with the ectoderm in the depths of the corresponding clefts. These areas of endodermal-ectodermal fusion are often called the “ closing membranes.” With the exception of that of the first pouch in mammals, including man, the “ closing membranes ” are transient since a secondary separation of the endoderm from the ectoderm occurs. While the “ closing membranes ” are present, however, they separate the segmentally arranged masses of branchial mesoderm from each other. The temporary junction of ectoderm and endoderm in the pouches may also have an importance, as will be seen later, in the problem of the histogenesis of certain of the structures developed from the endodermal pouches.
From what has been stated it follows that, in these higher vertebrates, rupture of the “ closing membranes” does not occur and gill clefts, as such, are not normally produced. Indeed both endodermal pouches and ectodermal clefts are for the most part much reduced in the Amniota. Only the first pouch persists as a large hollow cavity to form at least the greater part of the middle ear cavity and tympanic tube. The second endodermal pouch almost completely disappears and there is still uncertainty as to how much of it, if any, is normally represented by the tonsillar fossa or sinus. The three remaining, more caudal, pouches (the last of which is small, if not vestigial) lose their cavities by proliferation of their walls. They are named. from their position in the series, the third, fourth and fifth pharyngeal pouches. As the fifth pouch is the last of the series it is frequently called the ultimobranchial body. It is intimately related in subsequent development to the fourth pouch. Consequently, the two pouches together are sometimes named the fourth pouch, or caudal pharyngeal, complex. It is from the third and fourth pouches that the human parathyroid and thymus glands develop and, as will be seen later, they may also contribute to the formation of the thyroid gland. The fate of the ultimobranchial body is still uncertain and will require special discussion later.
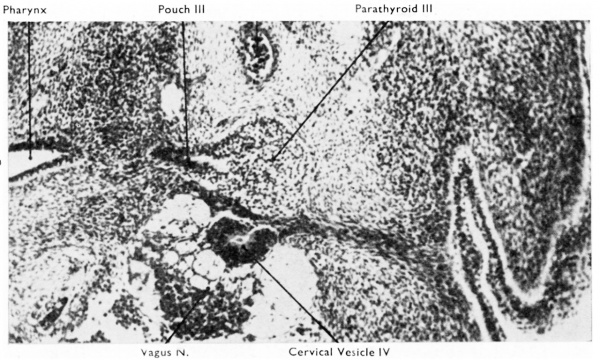
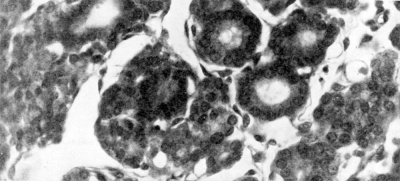
Fig. 1. Transverse section through right third pouch of a 10 mm human embryo (x 400). Parathyroid tissue already shows clear signs of differentiation. The cervical vesicle IV has separated from the surface endoderm of the region of the cervical sinus.
While these profound changes are occurring in the endodermal pouches, alterations, that are in part correlated, take place in the ectodermal clefts. These alterations result in the eventual disappearance of all the ectodermal clefts except a restricted portion of the first, and are a necessary prelude to the formation of a smooth contour to the neck region. These changes will receive brief attention at the end of this contribution.
Development of the Thymus Gland
The thymus of the human subject has a bilateral origin from part of each third endodermal pharyngeal pouch, the remaining portions of these pouches giving origin to the lower parathyroid glands of adult anatomy. Each third pouch becomes apparent in the pharyngeal wall at about the 10 somite stage and is soon in contact with the corresponding third ectodermal cleft. Growth changes result in the narrowing of the communication (ductus pharyngo-branchialis 111) between the pouch and the pharynx and in the extension of the pouch itself in a ventro-medio-caudal direction. At the same time contact with the ectodermal cleft is lost. A number of investigators, notably Norris (1938), have considered that the ectoderm of the third cleft donates cells to the endodermal pouch before separation occurs. The evidence for this viewpoint in the human embryo is not good. In certain other mammals (e.g., the pig), however, ectoderm from cleft III does contribute to the thymic rudiment and in yet others (e.g., certain marsupials) this ectoderm can give origin to a purely ectodermal thymus. This possibility of thymic tissue arising either from ectoderm or endoderm is puzzling, more particularly for those who hold rigid ideas on the germ layers and on their fates. Some further reference will be made to the problem later.
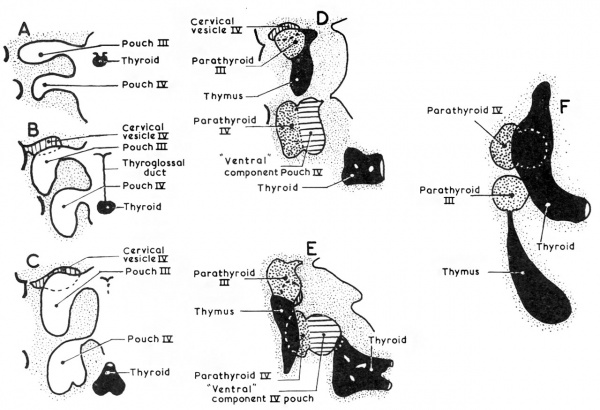
Fig. 2. Diagrams modified and combined from two figures of Norris (1938) to show schematically stages in the differentiation of the third and fourth pharyngeal pouches.
By the eight to 10 mm. stage of development a part of the third pouch begins to show a differentiation into parathyroid tissue (Fig. 1). The pouch is now thick walled with a cranial bulbous portion, still attached to the pharynx by the ductus pharyngo-branchialis III, and a caudal narrower portion. The parathyroid III differentiates from the wall of the antero-lateral portion of the bulbous upper portion (Fig. 2D). The remaining part of the pouch develops into the corresponding half of the thymus. A little later the ductus pharyngo-branchialis III becomes solid and then soon breaks (in my material it has broken in a 13 mm. embryo) so freeing the third pouch from the pharyngeal wall. The ductus may fragment in an irregular fashion, so leaving small isolated pieces which may later differentiate into accessory parathyroid or thymic rudiments or may even be the site of origin of epithelial cysts (see, for example, the discussion by Bréchet, 1938). The separation of the third pouch from the pharyngeal wall is accompanied by a very great increase in the rate of growth of the pouch and an increase in the thickness of its walls with gradual obliteration of its cavity. Weller (1933) has computed that the size of this pouch at the 16 mm. stage is six times that of the 14 mm. stage. At the same time as this great and rapid increase in size is occurring, the third pouch moves caudally, and its lower pole inclines medially. There is an accompanying, though gradual, change in the relative growth of the diflerent parts of the pouch so that the most caudal portion eventually becomes much thicker than the cranial portion. This change in the proportions has sometimes (e.g., by Hammar, 1911) been attributed to retrogressive changes in the more cranial part ; Sunder-Plassmann (1940) has given an elaborate account of the‘ fate of cells involved in these alleged changes. It seems more likely, however, that the changes in proportion are the result of differential growth rates and cellular mass migration.
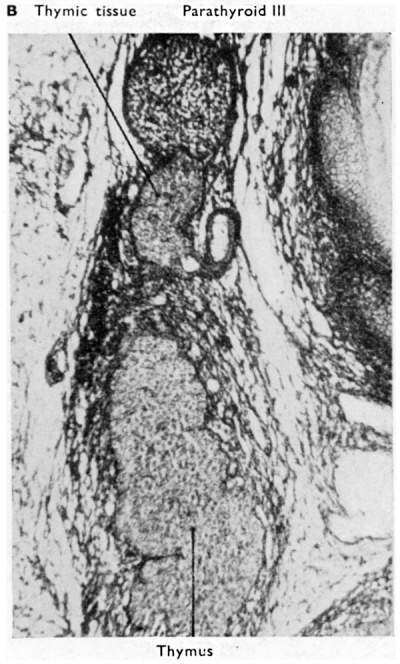
During the earlier stages of descent of the thymus the related parathyroid III moves down with the whole third pouch. This, of course, is the explanation of the normal lower position in later stages of this parathyroid, as compared with parathyroid IV, which, as we shall see, is derived from the fourth pouch. At a variable time, however, but usually soon after the 20 mm. stage, parathyroid III separates from the remainder of the third pouch so freeing completely. the thymic rudiment of the side concerned. Occasionally the separation may be much delayed (see Fig. 3B) and the parathyroid may then descend, or be drawn much further down the neck or even into the thoracic cavity (see, amongst others, the report by Gordon-Taylor and Handley, 1937).
|
|
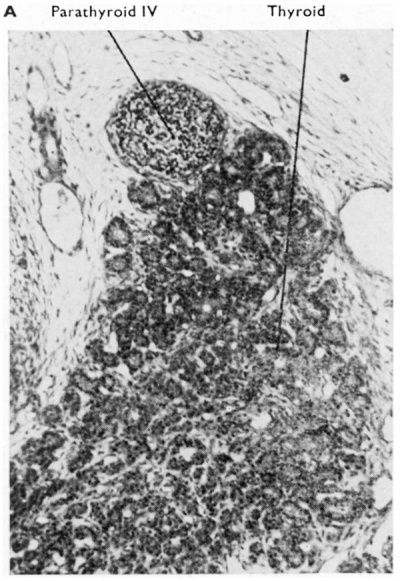
| Fig. 3A. Transverse section through thyroid and parathyroid IV of a 60 mm human embryo. (x 350.) The thyroid tissue shows developing follicles and commencing appearance of colloid in them. | Fig. 3B. Coronal section through parathyroid III and thymus of a 48 mm human embryo. The main bulk of the thymus has now separated from parathyroid II], but some thymic tissue is shown still connected with the parathyroid. On the right side of this aberrant thymic tissue a Kürsteiner canal is shown. |
After separation of the parathyroid gland each thymic rudiment possesses a thinner cranial and a broader caudal portion. The former becomes the cervical segment of the corresponding half of the thymus, while the caudal portion soon comes to be included in the upper part of the developing thoracic cavity. Here it meets, and eventually fuses with, its fellow of the opposite side, though connective tissue always separates the two halves. At first the intra-thoracic thymus lies dorsal to the upper part of the pericardium but as development proceeds, and the heart and pericardium “descend” yet further, this part of the thymus comes to lie ventral to the pericardium.
The cervical thymus shows much variation in subsequent stages, and the two sides frequently behave differently. They may extend as cords to the upper pole of the thyroid, or higher. They may also show fragmentation into separate portions which can persist as smaller or larger accessory thymic glands (Fig. 3B) ; this last point has to be kept in mind when we come to consider the possibility of thymic differentiation from the fourth pharyngeal pouch. In association with the rupture of the connection between the cervical part of the thymus and parathyroid III a variable number of small epithelial tubules arise. These are more intimately related to the thymic tissue than to the parathyroid gland. They extend for short distances into the surrounding connective tissue (Fig. 3B) and may persist into post-natal life as vesicular, canalicular or gland—like rudiments. They were first described by Kiirsteiner whose name is frequently associated with them. Gilmour (1937) gives a good account of them with a classification. He writes : “ They are rudimentary and of no apparent function but are important in that they persist into adult life and must be recognised if cystic structures in the neighbourhood of the parathyroids are to be interpreted correctly.” In my opinion the canals of Kiirsteiner have a further interest in that they may be serially homologous with the tubules frequently found in relation to the fourth endodermal pouch and often regarded as derivatives of the ultimobranchial body (see later).
Histogenesis and Later Development of the Thymus
After its separation from parathyroid III and the pharynx, the thymus appears as a densely packed epithelial mass of cells in which frequent mitoses can be seen. After the 30 mm. stage these cells become more loosely arranged to form a cytoreticulum and at about the same time lymphocytes appear in the interstices of the reticulum. The question of whether the origin of the syncytial-like reticulum is causally related to the entrance of the lymphocytes has not been decided. There has been much discussion on the mode of origin of the lymphocytes, or thymocytes as they are frequently called. Neglecting older and discarded hypotheses (see Bargmann, 1943, in full discussion), there are two currently held opinions—that the thymocytes differentiate from the cytoreticulum or that they migrate into the thymus from the surrounding mesoderm either separately or by way of the invading blood vessels.
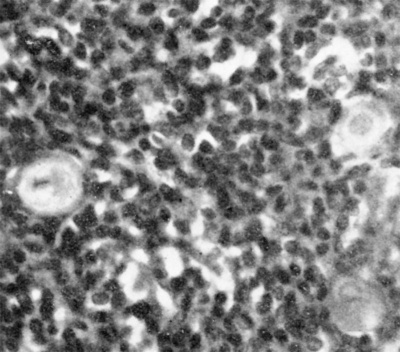
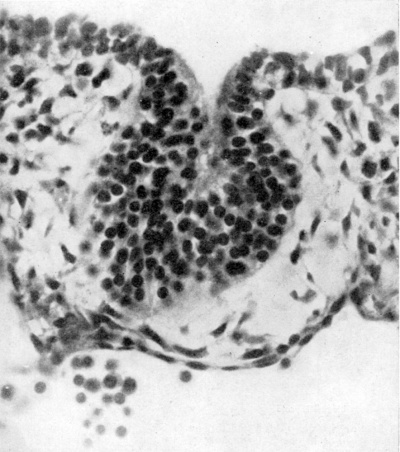
Fig. 4. Section of thymus of 140 mm. human foetus ( X 1000) to show three Hassall‘s corpuscles in the medulla.
At about the 40 mm. stage the cells of the thymus begin to show a rearrangement with the resulting production of marked lobulation and of a central medulla and a superficial cortex. The medulla appears to arise by a hypertrophy of the cytoreticulum and a decrease in the number of thymocytes, either by migration or degeneration. Shortly after the differentiation of medulla and cortex (50-60 mm. stages) the concentric corpuscles of Hassall (Fig. 4) commence to appear in the medulla. They are almost certainly derived from the cytoreticulum and can be regarded as epithelial pearls resulting from age changes in the epithelial cells as they migrate, or are forced, centrally from the more actively proliferating superficial cells. Norris (1938) has suggested that the Hassall corpuscles have an ectodermal origin, being derived from cells that have their origin from ectodermal cleft III of from the cervical vesicle (see later). The evidence for such an origin, at least in the human embryo, is far from satisfying.
The Parathyroid Glands
There are normally two pairs of parathyroid glands derived, respectively, from the endoderm of pharyngeal pouches III and IV (Figs. 1, 2, 3, 5, and 6). In association with their origins they are often called parathyroids III and parathyroids IV. As has been described above, each pharyngeal pouch III also gives origin to the corresponding half of the thymus. With the descent of this organ into the thoracic cavity the associated parathyroid III is also carried caudalwards until the time of its separation from the thymus and hence comes to be situated lower in the neck than parathyroid IV. This is, of course, the explanation of the paradoxical situation that the more caudal parathyroid of adult anatomy has a more cranial origin in the embryo. The close association of parathyroid III with the thymus has caused some investigators (e.g., Weller, 1933) to name it the parathymus.
Parathyroid tissue first becomes recognisable in the endodermal walls of the pharyngeal pouches concerned at about the 8 mm. stage. The differentiating parathyroid cells in each pouch multiply rapidly to form a small solid nodule (Fig. 1) and at the same time they increase in size and develop staining characteristics that distinguish them clearly from the endodermal cells of the remainder of the pharyngeal pouch. In particular the cytoplasm is less acidophilic and this feature, combined with the large size of the cells (about 12 microns or more in diameter), gives the developing parathyroid gland a less dense appearance than the other epithelial cells in its neighbourhood. This variety of parathyroid cell which is characteristic of these early stages has been called the “primordial” cell by Norris (1946). It can be noted that, notwithstanding their origin from separate pouches, parathyroids III and IV show very similar, if not identical, histogenesis.
At the time of separation of the parathyroid buds from the related pharyngeal pouches the “ primordial ” cells are the only ones to be found in the parathyroid glands. Norris (1946a) considers that it is not until the second half of gestation that other cell types become apparent. The first of these other types to appear are the “ vesicular ” cells in which, as compared with the “ primordial ” cells, there is some cellular enlargement, associated with a more or less extensive, and characteristic, vacuolization of the cytoplasm. There is some evidence to associate the “ vesicular ” cells with increased function of the parathyroid glands. Norris (l946b) states that they often predominate in the parenchyma of hyper-functioning parathyroid adenomas and Castleman and Mallory (1935) found a similar condition in secondarily hyperplastic glands. With yet further differentiation in the parathyroids, as development proceeds, Norris describes the appearance of three other cell types which he names “ clear,” “ dense,” and “ dark.” In my material it is possible to identify all the above-mentioned cell types in foetuses of 150 mm. C.R. length and older. It will, however, require careful investigation to determine the inter-relationships between these different cellular types and their functional significance. It can be noted, however, that similar cell types are found in post-natal parathyroid glands. Arguing from this fact Norris has made out a cogent case for pre-natal function of the parathyroid glands. Certainly the early histological differentiation of the parathyroids is a most marked feature of their development.
During their separation from the related pharyngeal endodermal pouches, the parathyroids frequently show slight fragmentation or little separated portions (Fig. SH). It is from these, undoubtedly, that accessory parathyroid glands usually arise. It is possible, however, that, in the separation, for instance, of the thymus from parathyroid III, some endodermal material, with the potentiality of becoming parathyroid remains attached to the thymus and descends for a variable distance with it—possibly even into the thoracic cavity. Thus can be explained the rare presence of parathyroid glandular material in this cavity. In all the human embryos available to me in serial sections I have been able to find at least four parathyroid glands. In five out of 35, there are five or six glands with, in each instance, the supernumerary glandules distinctly smaller than the others. Norris (1938) gives the incidence of supernumerary parathyroid glands as 2 per cent., and Gilmour (1938) as 6-5 per cent. My material rather favours the larger of these percentages.
Non-Parathyroid Portion of the Fourth Endodermal Pharyngeal Pouch
Such a circumlocution as is used in the heading of this section is forced upon one if the problem presented by the part of the fourth pouch, other than that which develops into parathyroid IV, is to be fairly posed. As was explained earlier the fourth pouch in the earliest stages of its development is followed by a fifth pouch—the “ ultimobranchial pouch.” Observations on this fifth pouch are meagre but it is generally assumed that it is either transient, and disappears completely, or is “taken up” into the fourth pouch. Adopting the latter explanation Grosser (1912) introduced the term “ caudal pharyngeal complex ” for the conjoined endodermal pouches IV and V and his concept has been widely adopted, though not always logically or rigorously followed in subsequent discussion by the adopters.
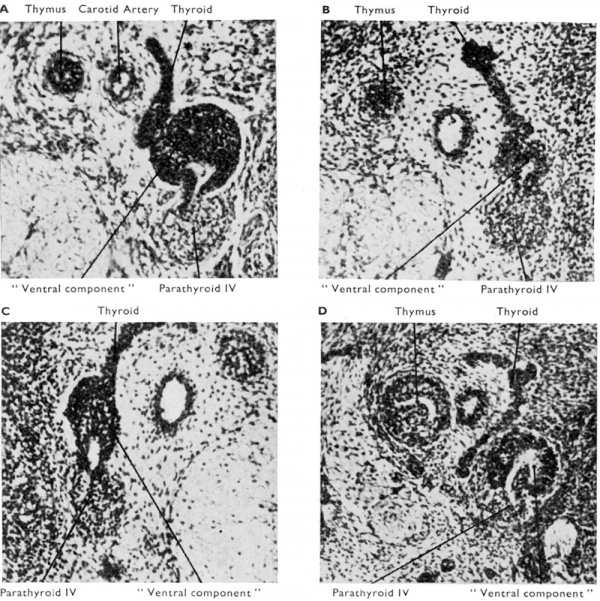
The derivation of parathyroid IV from the complex is universally accepted. In addition, however, there are recognised (1) a “ventral diverticulum ” IV which obviously corresponds in position to the thymus portion of pouch III, and (2) an “ ultimobranchial body” which, by the implication of its name, at any rate, is tacitly assumed to be derived from the fifth pouch. Many and diverse fates have been attributed to both of these additional parts of the caudal pharyngeal complex, and the confusion has often become worse by the occasional use of the terms as synonyms. Those interested will find a full description of the difficult terminological situation and of the plethora of hypotheses in van Dyke (1945). Here it is possible only to give the tentative opinion of the author and a brief survey of his personal findings. In the first place it can be stated categorically that more investigation is required to establish the assimilation by the fourth pouch of a fifth pouch in the human embryo. To avoid a tacit assumption on this issue, therefore, the non-commital term “ ventral component ” will be used for that part of the caudal pharyngeal complex which does not become parathyroid IV. Secondly, it can be stated that this ventral component of the caudal pharyngeal complex fuses with the dorsal aspect of the corresponding lateral lobe of the developing thyroid gland at about the l3 mm. stage (see Figs. 5 and 6). During this fusion the fourth pouch becomes solid by proliferation of its epithelial walls and the ductus pharyngebranchialis IV ruptures (Fig. 2) in much the same fashion as happens with that of the third pouch. The behaviour of the ventral component subsequent to its fusion with the thyroid has been much disputed and it should be pointed out that the different interpretations put forward are not all mutually exclusive and that there are doubtless species and even individual differences.
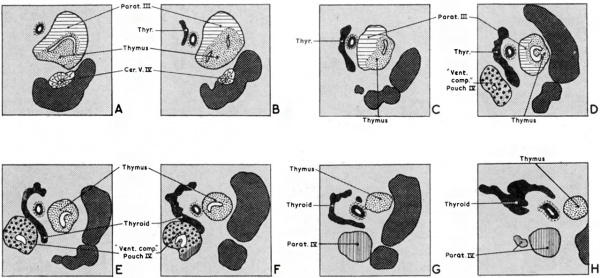
Fig. 5. Sections at intervals of 30 microns through the third and fourth pharyngeal pouch derivatives of a 19 mm. human embryo. The general relations of the derivatives to the vagus nerve and the carotid artery are shown. Note the intimate relationship in E. and F. between the “ventral component” of the fourth pouch and the developing thyroid. H. shows a small accessory parathyroid in relationship to parathyroid IV.
Fig. 6. Sections through developing fourth pouch of human embryos to show relationship between “ ventral component ” of this pouch and the differentiating thyroid tissue. (x 450.) (a) Left side 13 mm. human embryo : (b) Right side 17 mm. human embryo; (c) Left side l7 mm. human embryo ; (a') Left side 19 mm. human embryo. Note different appearance presented by the “ventral component” of the fourth pouch. In (b) and (c) it appears to be more intimately related to median thyroid tissue than in the other sections.
The alleged fates for the ventral component of the fourth pouch, after fusion with the thyroid gland, are (1) that it disappears completely in subsequent development, melting, as it were, and resolving itself into an ontogenetic dew; (2) that it differentiates, in its own right, into thyroid tissue; (3) that it is “induced ” by the proximity of thyroid tissue to convert itself into such tissue; (4) that it, at least occasionally, develops into thymic tissue, constituting a thymus IV; (5) that it forms a cyst or a series of tubules, possibly comparable to the canals of Kiirsteiner developed in relation'to the third pouch derivatives ; and (6) that its cells scatter in the thyroid gland to form the “ parafollicular ” cells (“interfollicular” cells or “ macrothyrocytes”). Now the work of Gilmour (1938) and of van Dyke (1941) make it quite clear that this tissue can occasionally, possibly even frequently, become thymic in nature.and this transformation, with some certainty, can be regarded as one possible fate of the fourth pouch. Other observations show that it can, at least occasionally, give origin to an epithelial endodermal cyst or a series of tube—like epithelial spaces.
Whether thyroid tissue ever differentiates from the derivatives of the ventral part of the fourth pouch complex cannot, in my opinion, be decided on the available evidence. The problem, however, has more than an academic embryological interest, as a number of investigators (see Dunhill, 1931 ; Ward, 1940 ; Lahey and Ficarra, 1946) have suggested that aberrant thyroid tissue, possibly derived from the fourth pouch, may be the site of origin of the so-called lateral aberrant thyroid tumours. Van Dyke (1945) has also described the behaviour of post-natal ultimobranchial tissue in the sheep. He has shown that thyroid-incorporated “ ultimobranchial ” tissue does not ordinarily undergo complete transformation into thyroid-like parenchyma. It usually persists in the postnatal thyroid gland as a “ central canal” or as multiple cysts lined by stratified squamous epithelium. If, however, there is an apparent degree of thyroid hyperactivity, cells of a glandular character arise from the epithelium of the cysts and come to be situated in the adjacent thyroid tissue. Here they may be transformed into thyroid-like parenchyma or may even form conspicuous adenoma-like masses. Investigation of human late foetal and post-natal specimens is urgently required to determine if equivalent conditions obtain in man.
Development of the Thyroid Gland
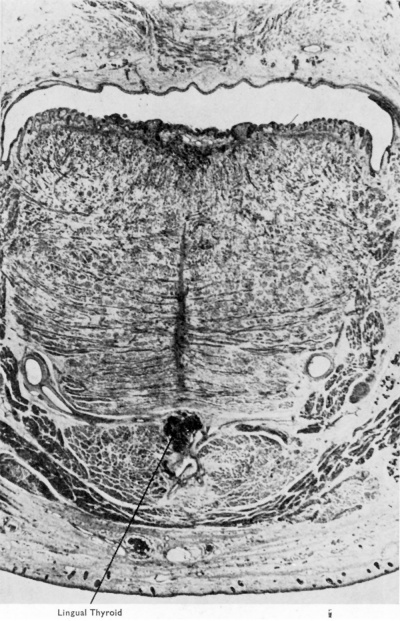
The origin of the thyroid gland as a median endodermal diverticulum (Fig. 7) from the floor of the pharynx is a well-established fact of vertebrate ontogeny and, indeed, indicates quite clearly the probable manner of its phylogenetic origin. There remain however (and the discussion given above on the enigmatic behaviour of the ventral component of the fourth pouch is a good example) many problems for study of the detailed development of the gland. In this contribution only a few of these problems can receive attention. The thyroid gland first appears in 17-I8 somite embryos as a median diverticulum of the floor of the pharynx between the elevations there caused by the first and second branchial arches. The evagination is formed from a plaque of specially difierentiated endoderm. Underlying it is the primitive aortic sac (Fig. 7). Growth of the diverticulum and the associated development of the neck region and “ descent ” of the heart results in the production of the thyroglossal duct. As is to be expected from the situation of the initial site of origin this duct passes ventral to the developing hyoid bone. The occasional inclusion of the duct within this bone is undoubtedly secondary (see Sgalitzer, 1941, for discussion). During the caudal migration of the gland the duct becomes longer and thinner and the lumen disappears. At about the 10 mm. stage the attenuated solid duct fragments, usually at about its mid-point. The pyramidal process is a late thyroid differentiation of the distal part of the duct. After rupture of the thyroglossal duct the proximal portion, called by Sgalitzer the lingual duct, shortens and thickens and its epithelium comes to resemble that of the tongue and frequently re-develops a lumen from which there grows out a more or less extensive system of branches. These may become cystic in nature but do not usually develop into thyroid tissue. As is well known, however, any part of the thyroglossal duct may persist and differentiate frank thyroid tissue. Such diflerentiation is shown in Fig. 8 which is a coronal section of the mouth and tongue of a 145 mm. human foetus.
Fig. 7. Section through median thyroid primordium of a 3.8 mm (30 somite) human embryo (x c.1800). Note the intimate relationship between the thyroid primordium and the aortic sac in which primitive blood cells can be seen.
Fig. 8. Section through mouth and tongue of 145 mm. human foetus to show lingual thyroid. (X 8.) The general anatomy of the tongue and floor of the mouth are well shown. Lingual artery and hypoglossal nerve, hyoglossus, mylohyoid, geniohyoid, genioglossus and anterior belly of digastric muscles can all be identified. To the right, superficial and deep pans of the submaxillary gland are shown. In the lower part of the picture, superficial to the mylohyoid, is seen a diflerentiating submental lymph gland.
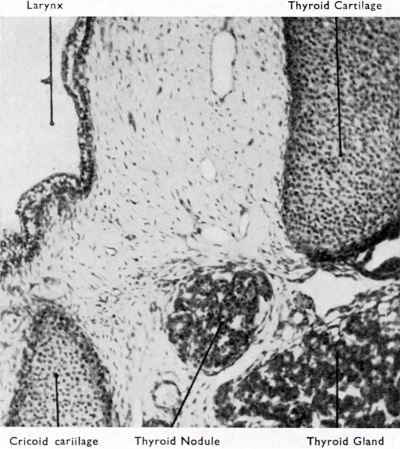
Fig. 9. Section to show aberrant portion of thyroid tissue lying between thyroid and cricoid cartilages. This nodule might have become an intratracheal thyroid. (X 450, 145 mm. foetus.)
In addition to thyroid tissue differentiating along the course of the thyroglossal duct such tissue may be found inside the larynx or trachea, forming the so-called struma intra-laryngotrachealis. I have never observed this rare condition in the fully developed state, but one of my embryos (Fig. 9) shows what may be the explanation of an anomaly that is otherwise difficult to explain. Hiickel (1932) has also suggested that this condition is to be explained by the migration of isolated fragments of developing thyroid tissue through the crico-thyroid or, less probably, thyro-hyoid gaps before the connective tissue has condensed to form the corresponding membrane.
Fig. 10. Section through developing thyroid tissue in 145 mm. human foetus. Colloid in well-established follicles is well shown. The larger spaces lined by endothelium are lymphatic in nature.
After rupture of the thyroglossal duct the cells of the bulbous lower end of the developing thyroid gland proliferate and extend laterally as thin epithelial layers which drape themselves in front of the trachea. Further growth lateralwards brings these plates of cells into contact with the “ ventral component ” of the fourth pouch (see Figs. 2, 5 and 6) at about the 13 mm stage. The question of the possibility of the conversion of this component into thyroid tissue has already been discussed. Later differentiation of the epithelial plates results in the production of the thyroid follicles (at the 40-50 mm. stage) in which colloid can be observed as early as the 60 mm. stage (Fig 3A). During the histogenesis the gland becomes extremely vascular and also shows, which may be of considerable importance, a very rich lymphatic system (Fig. 10).
The Cervical Vesicles
As was indicated in the earlier part of this lecture profound changes take place in the ectodermal clefts which result in their disappearance and in the establishment of a smooth contour to the developing neck. In the course of these alterations, which occur between the 5 and 15 mm. stages, the so-called “cervical sinus,” which is the depression found between the second arch (hyoid) elevation and the region of the developing upper thoracic wall, disappears. In the course of disappearance of the cervical sinus two little ectodermal cysts are normally cut off from the surface ectoderm and come to lie in the depths of the developing neck. These are the cervical vesicles. One of them, which has its origin from the second ectodermal cleft, and can be named cervical vesicle II, and which is closely related to the glossopharyngeal nerve, is transient and normally disappears by the time of “ closure ” of the cervical sinus.
The other cervical vesicle (IV) has its origin from the epithelium of the fourth ectodermal cleft. It is larger and persists longer than the other (see Figs. 1, 2 and 5). It was to this cervical vesicle that Norris (1937) attributed a part in the development of the human thymus. While this suggestion appears to have no real basis in fact, cervical vesicle IV is closely related during the stages when it is present to the dorsal surface of the third endodermal pouch. It has also intimate relations with the vagus nerve (Figs. 1 and 5) and may indeed contribute cells to its ganglion inferius. Normally, however, cervical vesicle IV has disappeared completely by the 25 mm. stage. Persistence of the vesicles can explain certain ectodermal cervical cysts (see Malcomb and Benson, 1940). For further details on the cervical vesicles reference should be made to Frazer (1926) and Garrett (1948).
References
BARGMANN, W. (1943) Thymus. In von Mollendorff Handbuch der mikroskopischen Anatomic des Menschen. 6 Bd., 4 Abteilung, Berlin, Springer.
BRECHET, J. (1938) Uber eine Cyste des Ductus thymopharyngeus. Zbl. Path. 69, 353.
CASTLEMAN, B., and MALLORY, T. B. (1935) The pathology of the parathyroid glands in hyperparathyroidism. Amer. J. Path. 11, 1.
DUNHILL, T. P. (1931) Carcinoma of thyroid gland. Brit. J. Surg. 19, 83.
Frazer JE. The disappearance of the precervical sinus. (1926) J Anat. 61(1): 132-43. PMID 17104123.
Garrett FD. Development of the cervical vesicles in man. (1948) Anat. Rec. 100: 101.
Gilmour JR. Embryology of the parathyroid glands, the thymus and certain associated rudiments. (1937) J. Path. and Bact. 45(3): 507–522.
(1938) The gross anatomy of the parathyroid glands. J. Path. and Bact. 46, 133.
GoRDoN-TAYLOR, G., and HANDLEY, R. S. (1937) An unusual case of hyperthyroidism : anterior mediastinal parathyroid tumour removed by trans—sternal approach. Brit J. Surg. 25, 6.
Grosser O. The development of the pharynx and of the organs of respiration. In: F. Keibel, F.P. Mall (ed) Manual of human embryology. (1912) Philadelphia, Lippincott, pp 446-497.
Hamilton WJ. Boyd JD. and Mossman HW. Human Embryology. (1945) Cambridge: Heffers.
HAMMAR, J. A. (1911) Zur groberen Morphologie und Morph. genie der Menschenthymus. Anat H. 43, 201.
HUCKEL, R. (1932) Die Entwicklungsstorungen der Schilddrijse. In Schwalbe-Gruber, Die Morphologie der Missbildungen des Menschen und der Tiere. 3 Teil, 15 Lieferung, 3 Abteilung. Jena, G. Fischer.
LAHEY, F. H., and FICARRA, B. J. (1946) The lateral aberrant thyroid. Surg. Gynec. Obsr. 82, 705.
MALCOMB, R. B., and BENSON, R. E. (1940) Branchial cysts. Surgery 7, 187.
Norris EH. The morphogenesis and histogenesis of the thymus gland in man: in which the origin of the Hassal's corpuscles of the human thymus is discovered. (1938) Contrib Embryol Carneg Instn 27: 191-207.
Norris EH. The parathyroid glands and the lateral thyroid in man. (1938) Contrib. Embryol., Carnegie Inst. Wash. No. 159.
Norris EH. Anatomical evidence of prenatal function of the human parathyroid glands. (1946) Anat. Rec. 96, 129.
Norris EH. Primary Hyperparathyroidism - a report of five cases that exemplify special features of this disease. (1946) Arch. Path. 42, 261.
Sgalitzer KE. Contribution to the study of the morphogenesis of the thyroid gland. (1941) J Anat. 75(4): 389-405. PMID 17104869
SUNDE3R-PLAssMAN, P. (I940) Zum Basedow-Thymus Problem. Dtsch. Z. Chir. 253, 4 S.
Van Dyke JH. On the origin of accessory thymus tissue, thymus IV: The occurrence in man. (1941) Anat. Rec. 79: 179–209.
4 (1945) Behaviour of ultimobranchial tissue in the post-natal thyroid gland: epithelial cysts, their relation to thyroid parenchyma and to “new growths” in the thyroid gland of young sheep. Amer. J. Anat. 76, 201.
WARD, R. (1940) Relation of tumours of lateral aberrant thyroids to malignant disease of the thyroid gland. Arch. Surg. 40, 606.
Weller GL. Development of the thyroid, parathyroid and thymus glands in man. (1933) Contrib. Embryol., Carnegie Inst. Wash. 24: 93-139.
Cite this page: Hill, M.A. (2024, April 24) Embryology Paper - Development of the thyroid and parathyroid glands and the thymus (1950). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Development_of_the_thyroid_and_parathyroid_glands_and_the_thymus_(1950)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G