Book - A Laboratory Manual and Text-book of Embryology 9
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Prentiss CW. and Arey LB. A laboratory manual and text-book of embryology. (1918) W.B. Saunders Company, Philadelphia and London.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
- (Text at early stage of editing, images to be added)
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter IX. The Development of the Vascular System
The Primitive Blood Vessels and Blood Cells
Both the blood cells and the primitive blood vessels arise from a tissue termed by His the angiobldsL Its origin has long been in doubt. According to Minot (in Keibel and Mall, vol. 2), Ruckert and others, the angioblast arises in the wall of the yolk sac from the entoderm.
Recent investigations by Maximow, Felix, Schulte, and Bremer point, however, to a mesodermal origin. In the body stalk of very young human embryos, Bremer (Am. Jour. Anat., vol. 16, 1914) has shown the direct origin of angioblast from splanchnic mesot helium. Moreover since this angioblast may antedate that of the yolk sac an entodermal origin is excluded. A further view, favored by Hertwig (Lehrb. d. Entw., 1915), derives the blood cells from entoderm, the vascular endothelium from mesoderm.
The angioblast consists initially of isolated solid cords and masses of cells which appear first in the splanchnic mesoderm of the body stalk and yolk sac. The solid cords of angioblast soon hollow out, the peripheral cells forming the endothelium of the primitive vessels, the inner cells, bathed by a clear fluid, persisting as the primitive blood cells or mesamoeboids of Minot. By the union of the isolated vascular spaces, the cellular network is soon converted into a vascular plexus which completely covers the human yolk sac. In the wall of the yolk sac this network is termed the area vasculosa, and here aggregations of blood cells form the blood islands (Figs. 33 and 79).
Hematopoiesis
Two sharply contrasted views are held as to the mode of origin {hematopoiesis) of the various adult blood elements. According to the monophyletic theory, a common stem- or mother cell such as the mesamoeboid gives rise to all types of blood elements, both red and white. The polyphyletic theory, on the contrary, asserts that the erythroplastids and the several kinds of white cells are derived from two or more distinct mother cells. The evidence seems to favor the monophyletic view, although Stockard (Amer. Jour. Anat., vol. 18, 1915) has recently advanced strong experimental evidence in support of the opposing theory.
The Primitive Blood Cells or Mesamoeboids
These show large vesicular nuclei surrounded by a small amount of finely granular cytoplasm (Fig. 250 a). They are without a cell membrane and are assumed to be amceboid. During embryonic life, the mesamoeboid ceUs multiply rapidly by mitosis and develop successively in the wall of the yolk sac, in the young blood vessels, in the liver, in the lymphoid organs, and in the red bone marrow.
Besides the mesamoeboids of extra-embiyonic origin, totipotent blood-forming cells appear to rise both from the mesoderm of the embryo and from the mesenchymal cells of adult connective tissue; such cells are believed by Maximow (Arch. f. mikr. Anat., Bd. 67, 1906; 73. 19f)8j to produce all tjrpes of blood elements.
Origin of the Erythrocytes (Red Blood Corpuscles)
These take their origin from the mesamoeboid cells of the embryo, and from the premydocytes of adult connective tissue and bone marrow, as erytkroblasts.
Fig.. 250. — Blood cells from embryos of 12 and 20 mm. X 1160. a, Primiti\*e mesamoeboid ceUs: h, ichthyoid cells or eiy'throblasts; f, d^ sauroid cells; f, cup-shaped nucleated cells; /, er>'throc)'tes. a, h and c are from a 12 mm. human embryo; d^ e and/, from a 20 mm. cmbr>'0.
1. Erythroblasts (ichthyoid blood cells of Minot, so-called because they resemble the t>pical red blood cells of fishes), are characterizedby the presence of hemoglobin in the homogeneous cytoplasm, which is thus colored red. The nuclei are vesicular, with granular chromatin (Fig. 250 b). There is a definite cell membrane. For the first six weeks of development (12 mm.) the erj-throblast is the only red blcKxl cell found.
2. XormoblastSy termed sauroid blood cells because they resemble the red blood cells of adult reptiles, are first formed in the liver from the er\-throblasts, and are predominant in embryos of two months. They are distinguished by their small, round nuclei with dense chromatin which stains so heavily that little or no structure can be seen (Fig. 250 c, d). The cytoplasm is larger in amount than in erythroblasts.
3. Erythrocytes (red blood corpuscles, erythroplastids) are developed in mammals from normoblasts which lose their nuclei by extrusion (Fig. 250/). The nucleus, extruded as several small granules or as a whole (Fig. 251), is ingested by phagocytes.
Fig. 252.— Human blood cells,
Emmel (Amer. Jour. Anat., vol. 16, 1914), studying cultures of blood cells from pig embryos, has observed the formation of bodies resembling erythrocytes by a process of cytoplasmic constriction. He suggests that this mav be their normal method of development in the embryo.
The first red blood corpuscles are spherical and are formed during the second month chiefly in the liver. During the third month the enucleated erythrocytes predominate (Fig. 250/). Although usually cuplike in preserved material, their normal shape is that of a biconcave disc (Arey, Science, vol. 44, 1916). During the later months of fetal life, the red blood corpuscles are developed in the liver, in the red bone marrow, and probably in the V«^ a
1 A J. . ^1 • r "%«" J. au l^iG. 251. — The development of
spleen. Accordmg to the view of Mmot, the red corpuscles in cat embryos (How++++cells from which they take their origin are mesa- ell), a, Successive stages in the de++++i.j I'vr ij j» .1 11 If velopment of a normoblast: b, the
mceboids which have lodged in the blood-form- . ® extrusion of the nucleus
ing organs and undergo cell division and differentiation there. In the bone marrow these cells are known as premyelocyies. They differentiate into both erythroblasts and myelocytes; from the former normoblasts and erythrocytes arise, from the myelocytes the granular leucocytes are developed. Soon after birth the red bone marrow is the only source of new red blood corpuscles.
Origin of the Leucocytes (White Blood Cells)
(Fig. 252). — These are divided into non-granular and granular types. According to the monophyletic view, it is assumed that both types are derived from the primitive mesamceboid cells of the embryo. I. Non-granular Leucocytes:
1. Small lymphocytes (22 to 25 per cent, of the leucocytes in adult blood) are regarded as immature leucocytes. They vary from 4 to 7.5 ^ in diameter and are developed in the lymphoid organs of the embryo and adult. The large nuclei, containing several connected masses of chromatin, stain darkly and are surrounded by a narrow zone of clear basic cytoplasm.
2. Liirgc mcnoHucUiir Icurocytts 1,1 to 3 per cent, of the leuoxi^'tes) are develo[xti from the endothelial celb lining the medullar}- sinuses of the lymph glands Kvaiis. .Ut-it. Rei:ord,voLS: 1914i.or from the reticulum of I>-iiiph ^ands (Kyes, 1115 , II. t<V„'(iM/jr or Polymo'phomuUiir Leucocytes:
Iho blood-forming cells lodged in the fed bcrae marrow are known as prett:\<'.i\\:ti. They give rise to myelocytes, cells with round or crescentic nuda and irrAr.uLir cvtopiasm. Similar celb are developed in the l^n^tuud organs. By '»;ii,:t rjroins; changi-s 1 1' in ihc lorm and structure of their nuclei. I2> in the size .-.r.i: >:-u:'.ing qualities ol their cytoplasmic granules, the mj-elocjtes gi^-e rise to thn.^ t>"iv> ot ixanuUr Icucvvytes:
It is commonly held that the eosinophilic granules differentiate endogenously (Downey, Anat. Rec, vol. 8, 1914). However, Weidenreich (Arch. f. mikr. Anat., Bd. 82, 1913) regards these granules as ingested fragments of red corpuscles or their hemoglobin derivatives. Badertscher (Amer. Jour. Anat., vol. 15, 1913) found numerous eosinophiles and free eosinophilic granules in the vicinity of degenerating muscle fibers in salamanders. Also during trichiniasis in man, when there is extensive degeneration of muscle fibers, the number of eosinophiles in the blood becomes greatly increased.
3. BasophileSy or Masl Leucocytes (Maximow), form only 0.5 per cent, of the leucocytes. Their nuclei are very irregular in form and may be broken down into several pieces which stain intensely. The granules are variable in number, size, and form, and often stain so heavily as to obscure the nucleus. The cytoplasm is clear and vacuolated. Basophiles have been regarded as degenerating granular leucocytes, but at present this view is not generally accepted.
Origin of the Blood Plates
In the bone marrow and spleen pulp are giant cells, or megakaryocytes, the cytoplasm of which shows a darkly staining granular endoplasm and a clear hyaline ectoplasm (Fig. 253). It has been shown by Wright (Jour. Morphol., vol. 21, 1910) that the blood plates arise by being pinched off from cytoplasmic processes of the giant cells. The granules of the plates are interpreted as portions of the endoplasm of the giant cells, for they stain in a similar manner. Genuine blood plates and giant cells occur only in mammals.
We have seen that the first blood cells and blood vessels take their origin in the angioblast, which develops in the wall of the yolk sac and chorion from the splanchnic mesoderm. The first vessels derived from the angioblast (see p. 243) are small isolated blood spaces which unite and form capillary networks. From these, endothelial sprouts grow out, meet, and unite until complete networks are formed. In human embryos of 1 mm., or less, these envelop the lower portion of the yolk sac, the body stalk, and chorion.
There are two views as to the manner in which the heart and the primitive vascular trunks of the embrycj originate. According to His and Rabl, and more recently Minot, Evans, and Bremer, all the blood vessels of the embryonic body arise as endothelial ingrowths from the extra-embryonic yolk sac angioblast. Kolliker, Rlickert, and Mollier (Hertwig's Handb., 1906), on the contrary, assert that the intra-embryonic vessels are formed by the fusion of discrete anlages in a way similar to that first occurring on the yolk sac. Corroborative investigations by Maximow, Huntington, Schulte, and others have shown that the apparent invasion of angioblast in reality represents a progressive fusion of isolated mesenchyniiil tissue spaces. Moreover, direct experimental proof on living chick embryos (Miller; Reagan, Anat. Rec, vol. 9, 1915) leaves little doubt of the correctness of the Ruckfrt-Mollier view.
The delicate injection methods of Mall and his students show that capillary plexuses precede the formation of definite arterial and venous trunks (Fig. 254), Only by the selection, enlargement, and differentiation of definite paths do the (Iflmitive vessels arise. Capillaries, from which the flow has been diverted, atrophy. The primitive paired aottse are formed from the medial margins of such plexuses. Exceptions to the general rule are the intersegmental arteries which arise as single trunks from the aorta (Evans).
Inheritance, as well as the hydrodynamic factors incident with the blood flow, doubtless participates in the selection of channels from the capillary bed.
Origin of the Tubular Heart
The heart of the lower fishes and of amphibians arises in the ventral mesentery of the fore-gut. A tubular cavity first appears, about which the cells diflferentiate directly into endo-, myo-, and epi-cardium.
In bony fishes, reptiles, birds, and mammals, the heart is formed, while the embryo is still flattened on the surface of the yolk, from paired anlages which later grow mosad and fuse. Aggregates of meso++++ells, which soon form thin-walled tubes, first appear between the ento++++1 splanchnic nu'S(xlerm; these are flanked by folds of splanchnic meso++++iili Inline laterally into the ca-lomic cavity (Figs. 255.4 and 35). Such llulnr masses (endothelial anlages) are present in the Spec 1,54 mm, nan embryo (lin. 77V .Vs the embryo grows away from the yolk and the fornieil. llic entoderm withdraws from between the endothelial tubes, wine ( liisf as well as Ihr mesodermal folds to fuse (Figs. 255 B. C, 36 and 37).
Fig. 254,— The caudal end of a chick embryo himinii the ptimao' capiHarj' p'etus i^liTliir liniU hulls Fr»ni which ihe sciatic ill liiniTnilinlc. .\orta; hnve formed from il miirKins of (he ]>lexuscs (Evans).
The heart is now an unpaired endothelial tube lying in the folds of the splanchnic mesoderm (Fig. 190 A). Soon the ventral attachment of the mesoderm disappears, leaving the heart suspended by a temporary dorsal mesocardium in the single pericardial chamber (Fig, 255 C). The endothelial tube forms the endocardium, the splanchnic mesoderm later gives rise to the epicardium and ^ myocardium (muscle layer of heart).
This type of heart occurs in human embryos of 2 mm. (5 or 6 somites, Fig, 256) and shows three regions: (1) the atrium, which receives the blood from the primitive veins; (2) the ventricle; (3) the bulb, from which is given off the ventral aorta.
Fig. 255. — Diagrams to illustrate the origin of Fic. 256. — The heart of a 2 mm. human the T jiarnmfflifl Tt heart. Ed., Ectoderm; End., endo- embryo in ventral view (Mall). X 65. Tbe thelial tubes; EM., entoderm; Fg., fore-gut; MscJ., open tube is the fore-gut, dor^l mesorardium; Mufl., splanchnic mesoderm (epi- and myocardium).
As the cardiac tube grows faster than the pericardial cavity in which it lies it bends to the right, the bulbus and ventricle forming a U-shaped loop (Fig. 257). Four regions may now be distinguished: (1) the sinus venosus; (2) the alrium, also thin walled and lying cranial to the sinus ; (3) the thick-walled ventricle or WmAjVentrad and caudad in position; (4) the bulbar limb, cranial to the ventricular limb and separated from it by the bulbo-ventricular cleft. Next, in embryos of 3 to 4 mm., the bulbo-ventricular loop shifts its position until its base is directed , caudadand ventrad (Fig. 257 B). At the same time the sinus venosus is brought dorsal to the atrium, which in turn is cranial with relation to the bulbo-ventricular loop, and the bulbar limb is pressed against the ventral surface of the atrium and constricts it (Fig. 258 ^4).
Fig. 257. — A, Heart of human embryo of 2,15 m atrial portion. B, Heart of human embryo of about .1 it (, primilLve ventricle (in front). Vtntrdl \iew5 (liis). I, Bulbus cordis; b, primitive i-entricle; c, I, Bulbug cordis; h, atrial portion (behind);
In embryos of 4 to 5 mm. the right portion of the sinus venosus grows more rapidly than the left, this being due to the fact that the blood flow of the left
FlC. 258. — A. Heart of human embryo of about 4.3 mm.; i. .\TMuni; '■, [■■irlinn of atrium corresponding to auricular appendage; c, bulbus cordis; d, atrial etnal; <■. primitive ventricle. B, Heart of human embryo of about 10 nun.: o, Left atrium; b, right atrium; c, bulbus cordis; d, interventriculoi groove; e, right ventricle;/, left ventricle. Ventral views (His).
umbilical vein is shifted to the right side through the liver. As a result, the enlarged right horn of the sinus opens into the right dorsal wall of the atrium through a longitudinally oval foramen, which is guarded on the right by a vertical fold.
Early Development of the Heart and Paired Blood Vessels
This fold, which projects into the atrium, is the right valve of Ike sinus venosus. Later, a smaller fold forms the left vaive of the sinus venosus (Fig. 260 B). The atrium is constricted dorsally by the gut, ventrad by the bulbus. It therefore must enlarge laterally and m so doing forms the right and le/l atria (Fig. 258 A, B) with the distal portion of the bulb between them. The deep external groove between the atria and the bulbo-venlricular part of the heart is the coronary sulcus/ As the bulbo- ventricular region increases in size, the duplication of the wall between the two limbs lags behind in development and finally disappears (Fig. 259), leaving the proximal portion of the bulb and the ventricular limb to form a single chamber, the primitive ventricle. In an embryo of S mm. the heart is thus composed of three undivided chambers: (1) the sinus venosus, opening dorsad into the right dilation of the atrium; (2) the bilaterally dilated atrium; opening by the single transverse atrial canal into (3) the primitive undivided ventricle. The three-chambered heart is persistent in adult fishes, but in birds and mammals a four-chambered heart is developed in which venous blood circulates on the right, and arterial blood on the left. In amphibians and reptiles transitional types occur.
Fig. 259. — Dugraros to show tbe reduction of the bulbo-ventricuUr fold (represented by diagonal lines) due to its letrded development. (Modified after Keith.)
The important changes leading to the formation of the four-chambered heart are: (1) the complete division of the atrium and ventricle, each into right and left chambers; (2) the division of the bulb and its distal continuation, the tnincus arteriosus, into the aorta and pulmonary artery; (3) the incorporation of the anus venosus into the wall of the right atrium; (4) the development of the semilunar and atrio-ventricular valves. The first of these changes is completed only after birth.
Endocardial Cushions and Atrial Septa
In embryos of 5 to 7 mm. there dividing the single atrial canal into right and left atrio-venlricular 266). The atrium is now partly divided into right and left atria, \ ever, communicate ventrad through the interatrial foramen. Next, in er of 9 mm. , the septum I thins out dorsad and cephalad and a second opening app
— Horizontal sertioni throuich the choinbcrs oi the bum"" hoartmm. (A and B are based on figures of Tandl
the foramen ovale (Figs. 260 and 261 B). The atria are now connected by two openings, the interatrial foramen and the foramen ovale. Soon (embryos of 10 to 12 mm.) the ventral and caudal edge of septum I fuses with the endocardial cushions, which have in turn united with each other (Figs. 260 and 261 C). The interatrial foramen is thus obliterated, but the foramen ovale persists until after birth. In embryos of 9 mm. the septum secundum (II) is developed from the dorsal and cephalic wall of the atrium, just to the right of the septum primum (Fig. 260 C). It is important, as it later fuses with the left valve of the sinus venosus and with it forms a great part of the atrial septum of the late fetal and adult heart.
Fig. 261. — Lateral dissections of the human heart viewed from the left side: ^,6 mm.; £,9 mm.; C, 12 mm. (B is based on a reconstruction by Tandler.) X about 38. Cor. sin.. Coronary sinus: P. aid. c, dorsal endocardial cushion; For. on., foramen ovale; Int. for., interatrial foramen; /. v. c, inferior vena cava; L. air., left atrium; L. m. i. p., left valve of sinus i-enosus; L. vtnl,, left \-entride; Pul. a., pulmonary artery; Pul. v., pulmonary vein; Sept. I, Stpt. II, septum primum, septum secundum; Sup. t. c, superior vena cava; V. end. c, ventral endocardial cushion.
Sinus Venosus and its Valves
The opening of the sinus venosus into the dorsal wall of the right atrium is guarded by two valves (Fig. 260). Along the dorsal and cephalic wall of the atrium these unite to form the septum spurium. Caudally the valves flatten out on the floor of the atrium, but, as stated previously, the left valve later fuses with the atrial septum II. In embryos of 10 to 20 mm. the atria increase rapidly in size and the lagging right horn of the sinus venosus is taken up into the wall of the right atrium. By this absorption the superior vena cava now opens directly into the cephalic wall of the atrium, the inferior vena cava into its caudal wall (Fig. 261 C). The transverse portion of the sinus venosus, persisting as the coronary sinus in part, opens into the posterior wall of the atrium.
The right valve of the sinus venosus is very high in 10 to 65 mm. embryos (fii^t to third month) and nearly divides the atrium into two chambers (Fig. 262). It becomes relatively lower during the third and fourth months. Its cephalic portion becomes the rudimentary crista lermiKalis (Fig. 263); the remainder is divided by a ridge into two parts, of which the larger cephalic division persists a the vaive of the inferior vena cava (Eustachian valve) located at the right of th opening of the vein, and the smaller caudal portion becomes the valve of the c nary sinus (Thebesian valve).
Fig. 262. — Utcn) (fesMiioa ot the hmti Idus lieavd b«m the ri^ tide. X I
The left xmIvt ot th*' sinus vtikisus becomes continuous with the septum se-1 cundum. utdv in cmhr\Ms of 20 to 23 mm. or larger, the two bound an oval inning (Figs. ItS lo 265). The bounding wall of tbc o\-al aperture is the limlms ontlis. m
Chnura of the Foramen Ovale. Iht- tiw eilge of septum 10 to 15 mm., directed dorsad and cephabd (F|g. 261 O- Cm stAgfs (Figs. 264 and 36S>\ its caudal and dorsal prokiagation giw wnt rad until its free edge is so diiected. OoiDddent with tUs dw II. with its free edge directed «l first vcntnd aikd oradad, sfcifts i ctlge is directed dorsad and cephalad. and ox'Vfl^is the septum I 264 and 265). The opening between these septa persists until after birth as the foramen ovale.
During fetal life the left atrium receives little blood from the lungs, so that the pressure is much greater in the right atrium. As a result, the septum I is pushed to the left and the blood flows from the right into the left atrium through the foramen ovale. After birth, the left atrium receives from the expanding lungs' as much blood as the right atrium, the septum I is pressed against the limbus of septum II, and soon fuses with it. The depression formed by the thinner walled septum I is the fossa ovaiis.
Fig. 263. — Lateral dbsection of the heart of a 105 nun. human fetus viewed (rom the right side. X 7.
The foramen ovale may fait to close soon after birth and the mixed blood prxjduces a purplish hue in the child which is known popularly as a "blue baby." This condition may be persistent in adult life. Incomplete closure occurs in about one in four cases, but actual mingling of the blood is rare, due to an approximation of the overlapping septal folds during atrial systole.
Pulmonary Veins
In embryos of 6 to 7 mm. a single vein (arising in the cat from a peripulmonary plexus, Brown, Anat. Rec., vol. 7, 1913) drains into the caudal wall of the left atrium at the left of the septum I (Fig. 261 C). This vein bifurcates into right and left pulmonary veins which divide again before entering the lungs. As the atrium grows, the proxima] portion of the pulmonary van is taken up into the atrial wall. As a result, at first two, then four pulmonary veins open into the left atrium.
Fig. 265. — Lateral dissecUoos of the human heart viewed from the left tide: A, from a : embryo; B, from a 105 mm. fetus, fitc. va., Bicusfnd valve; Cor. sin., oana "or. on., foi ovale; I.Ti.c, inferior vena cava; L. air. vent, c, left atrio-ventricular canal; ; ventricle, a., pulmonary artery; Sept. I, Sept. II, septum primum and septum
Fic. 266. — Venlral views of stages in tbe development of the heart to show the differentiation of the bulbus cordis into Che aorta and pulmonaiy tiunlt (KoUmao) ; A , Heart of a 5 mm. human embr>-o; B. of a 7.S mm. human embryo.
Origin of the Right and Left Ventricles
In embryos of 5 to 6 mm. there appears at the base of the primitive ventricular cavity a sagittally placed elevation, the interventricular septum (Fig. 260 B). It later grows cephalad and dorsad toward the endocardial cushions, and forms an incomplete partition between the right and left ventricles, which still communicate through the persisting intercentricidar foramen (Fig. 266 B). Corresponding to the internal attachment of the septum there is formed externally the interventricular sulcus (Fig. 266 A) which marks the external line of separation between the large left ventricle and the smaller right ventricle.
Origin of Aorta and Pulmonary Artery from Bulbus
Coincident with the formation of the interventricular septum there arise in the aortic bulb (including its distal truncus arteriosus) longitudinal thickenings, four in the distal half, two in the proximal half. Of the four distal thickenings (Fig. 267), two, which may be designated a and f , are larger than the other thickenings, b and d. Thickenings a and r, which distally occupy left and right positions in the bulb, meet, fuse, and divide the bulb into a dorsally placed aorta and ventrally placed pulmonary trunk (Fig. 266). Traced proximally they pursue a clockwise, spiral course, a shifting from left to ventral, and c from right to dorsal, both becoming continuous with the proximal swellings. Thickenings b and d are also prominent at one point proximally; when the bulb in this region is divided by ingrowing connective tissue into the aorta and pulmonary artery, the aorta contains the whole of the thickenings b and half of a and r, while the pulmonary trunk contains the whole of d and half of a and r (Fig. 267). Distally the three thickenings now present in each vessel disappear, but proximally they enlarge, hollow out on their distal surfaces, and eventually form the thin-walled semilunar valves (Fig. 267). The anlagcs of these valves are prominent in embryos of 10 to 15 mm. as plump swellings projecting into the lumina of the aorta and pulmonary artery.
Fig. 267. — Scheme showing division of bulbus cordis and its thickenings into aorta and pulmonary artery with their valves. (Explanation in text.)
The two proximal bulbar swellings fuse and continue the spiral division of the bulb toward the interventricular septum in such a way that the base of the pulmonary trunk, now ventrad and to the right, opens into the right ventricle, while the base of the aorta, now lying to the left and dorsad, opens into the left ventricle close to the interventricular foramen, through which the two ventricles still communicate (Fig. 266 B).
Closure of the Interventricular Foramen
The interventricular foramen in embryos of 15 to 16 mm. is bounded: (1) by the interventricular septum; (2) by the proximal bulbar septum; and (3) by the dorsal portion of the fused endocardial cushions (Fig. 266). Soon these structures are approximated and fuse, thereby forming the septum membranaceum, which closes the interventricular foramen. The atrio-ventricular valves arise as thickenings of the endocardium and endocardial cushions of the atrio- ventricular foramina (Figs. 260 and 261). Three such thickenings are formed on the right, two on the left. The anlages of the valves are at first thick and project into the ventricles. Later, as the ventricular wall differentiates, the valvular anlages are undermined, leaving their edges attached to the ventricular walls by muscular trabeculcBy or cords. The muscle tissue of both the valves and trabeculae soon degenerates and is replaced by connective tissue, forming the chorda tendinete of the adult valves. Thus there are developed the three cusps of the tricuspid valve between the right chambers of the heart, and the two flaps of the bicuspid^ or mitral valve, between the left atrium and left ventricle.
Differentiation of the Myocardium
The myocardium, at first uniformly spongy, becomes compact at the periphery. The inner bundles remain trestle-like, forming the trabecule carna and the papillary and moderator muscles around all of which the originally simple endocardial sac is wrapped. The myocardial layers, at first continuous over the surface of the heart, become divided by connective tissue at the atrio- ventricular canal, leaving a small bridge alone. This connecting strand, located behind the posterior endocardial cushion, forms the atrio-ventricular bundle.
Descent of the Heart
At first the heart lies far cephalad in the cervical region, but it gradually recedes during development until it assumes its permanent position in the thorax.
Primitive Blood Vascular System
The first paired vessels of human embryos are formed as longitudinal anastomoses of capillary networks which, however, originate first in the angioblast of the yolk sac and chorion. In the Etemod embryo of 1.3 mm., in which the somites are still undeveloped the paired vessels are already formed (cf. Fig. 268). They are the umbilical veins which emerge from the chorion, fuse in the body stalk, then, separating, course in the somatopleure to the paired tubular heart. From the plexus of ventral vessels in series with the vitelline arteries. At this stage the vitelline circulation of the yolk sac is established.
In embryos of 15 to 23 somites (Fig. 270) the veins of the embryo proper develop as longitudinal anastomoses of branches from the segmental arteries. The paired anterior cardinal veins of the head are developed first, and, coursing back on either side of the brain, they join the vitello-umbilical trunk. In embryos of 23 somites the posterior cardinals are present. They lie dorsal to the nephrotomes. and, running cranially, join the anterior cardinal veins to form the common cardinal veins. Owing to the later enlargement of the sinus venosus, the proximal portions of the common venous trunks are taken up into its wall and thus three veins open into each horn of the sinus venosus; (1) the umbilical veins from the chorion; (2) the vilelline veins from the yolk sac ; (3) the common cardinal veins from the body of the embryo,
Fig. 270.— Di»Bfain of the blood vessels of human embryos of 2.6 mm.
The descending aortae have now fused caudal to the seventh intersegmental arteries and form the single dorsal aorta as far caudad as the origins of the umbilical arteries.
Of the numerous vitelline arteries one pair is prominent; these fuse into a single vessel which courses in the mesentery and later becomes the superior mesenteric artery. By the enlargement of capillaries connecting the ventral and dorsal aortse a second pair of aortic arches is formed at this stage (Fig, 270),
Development of the Arteries
Transformation of the Aortic Arches
In embryos 4 to 5 mm. in length five pairs of aortic arches arc successively developed, the first, second, third, fourth, and sixth (Fig. 271). An additional pair of transitory vessels which extend from the ventral aorta to the sixth arch appear later in embryos of 7 nun.,- but aood degenerate (Fig. 272 B). They are interpreted as being the fifth pair in the aexies. From each dorsal or descending aorta there devel(q> cranially the inlermal eanlid arteries. These extend toward the optic stalks where they bend dorsad and caudad, connecting finally with the first intersegmental arteries of each side (Fig. 271). The descending aortse are now fused to their extreme caudal ends and the umbilical arteries take tbeir origin ventially. Twenty-seven pairs tf dan
Firit lervical arlrry^ Pvimonary attrry
Inf. mtsmteric artery'
— Arteries and cardinal v Ingalls).
ins of the right side in a 4.Q mm. human embryo (modified ifter X 20. H, Heart; I-VI, aortk arches.
intersegmental arteries are present From the seventh cervical pair of these the subclavian arteries of the upper limbs arise. Of the ventral vitelline vessds three are now prominent, the ccsliac artery in the stomach-pancreas region, the vitelline or superior mesenteric in the small intestine region, and the inferior mesenteric of the large intestine region.
Of the aortic arches the third pair is largest at 5 mm. (Fig. 272, A). From the sixth pair are given off the small pulmonary arteries to the lungs. At 7 mm. the first and second aortic arches are obliterated (Figs. 272 B and 273), but the dorsal and ventral aortse cranial to the third arch persist as parts of the internal and external carotid arteries respectively. The third arches form the stems of the internal carotids, while the ventral aorts between the third and fourth arches become the common carotids. In embryos of 15 mm. the bulbus cordis has been divided into the aortic and pulmonary trunks, so that the aorta opens into the left ventricle and the pulmonary trunk into the right ventricle. The dorsal aorls between the third and fourth arches disappear, but the fourth arch on the left side persists as the aortic arch of the adult. On the right side, the fourth aortic arch persists with the descending aorta as far as the seventh intersegmental artery and forms part of the rigid subclavian artery, which is thus longer than the left. The segment of the fourth arch proximal to the right common carotid becomes the innominate artery. On the right side, the sixth arch between the origin of the right pulmonary artery and descending aorta is early lost; on the left side, it persists as the ductus arteriosus and its lumen is only obliterated after birth. The proximal portion of the r^kt sixth arch fomu the stem of the rigkt ft^mmary artery, but the proximal portion of the l^t arch is incorporated in the pulmonary trunk.
Fig. 272. — Aortic arches of humui embryos: ^, of Sir gcal pouches. (after Tandler), /-/ V, phBryn
The aortic arches of the embryo ue of etpedal importuice ocwywimtivdy. Rve arches are formed in coonection with the gilli of adult fiihes. In adult tailed ■■"r*'"*^. three or four arches, and in some reptiles two arches, are represented on either nde. In birds the right, in mammals the left fourth arch persists as the arch of the aorta.
The different courses of the reewrmf laryngeal tunes arc easily explained. The vafui early gives off paired branches which reach the larynx by passing caudal to the primitive fourth aortic arches. When the Utter, through growth changes, descend into the chest, loops of both nerves are carried with them. Hence, after the transformation of thft.fourtb arches, the left recurrent nerve remains looped around the arch of the aorta, the right around the right subclavian artery (cf. Kg. 273).
Fig. 273.— Diagram diowing the aortic arches and thdr derivatives la human embryos.
Branches of the Dorsal Aorta
From the primitive aortie arise: (1) dorsal, (2) lateral, and (3) ventral branches (Fig. 274).
1. The dorsal branches are intersegmental and develop small dorsal and large ventral rami. From the dorsal rami are given off neural branches which tHfunate and form dorsal and ventral spinal arteries.
Origin of the Vertebral Arteries and Basilar Artery
As we have seen (Fig. 271), the internal carotids are recurved cranially in the 5 mm. embryo and anastomose with the first two pairs of dorsal intersegmental arteries. By longitudinal postcostal anastomoses (Fig. 274) of the dorsal rami of the first seven pairs of dorsal intersegmental arteries the vertebral arteries arise (Fig. 275). The original trunks of the first six pairs are lost so that the vertebrals take their origin with the subclavians Irom the seventh pair of inlersegmetilal arteries (Fig. 276). In embryos of 9 mm. the vcrtebrals in thr region of the mctenccphalon fuse to form a sin^
Fig. 274. — A dugmn of the trunk, in transvefse section, showing the amngement of the aortic biancbe.
Fig. 275. — The development of the vertebral and subclavian arteries and the coslo-cervical trunk ID a youni; rabbit embryo (modified after Hochatetler). /// ^B.-/F.1S.,Aorticarches:^^,pulDH>nary artery: A -t.e.b. and A .w.r.t.. cephalic and cervical portJMis of venebral artery: A j., ■ubcla^'ian artery; C.e., costocervical trunk; CJ. and C.r,, internal and external carotid arteries; ISpC, spinal ganglion.
2. The lateral (.visceral) branches of the descending aorta are not segmentally arranged. They supply structures arising from the nephrotome region (mesonephros, sexual glands, metanephros, and suprarenal glands). From them later arise the renal, suprarenal, inferior phrenic, and internal spermatic or ovarian arteries.
3, The ventral (splanchnic) branches are at first rather definitely intersegmental. Primitively they form the paired vitelline arteries to the yolk sac (Figs. 268 to 270). Coincident with the degeneration of the yolk sac the prolongations of the ventral vessels to its walls disappear, and the paired persisting arteries, passing in the mesentery to the gut, fuse to form unpaired vessels from which three large arteries are derived, the caliac artery, the superior mesenteric, and the inferior mesenteric (Fig. 271).
Fig. 278.— Reconstructions showinx tbe development of the umbilical and iliac arteries (after TandJer) : A, 5 mm. human embiyo; B, 9 nun. human embryo.
The primitive coeliac axis arises opposite the seventh intersegmental artery. Together with the mesenteric arteries, it migrates caudalward until eventually its origin is opposite the twelfth thoracic segment (Mall). This migration, according to Evans, is due to the unequal growth of the dorsal and ventral walls of the aorta. Similarly, the superior mesenteric artery is displaced caudad ten segments, the inferior mesenteric artery three segments.
The Umbilical and Iliac Arteries
As previously described, the umbilical arteries arise in young human embryos of 2 to 2.5 mm. from the primitive aortie opposite the fourth cervical segment. They take origin from a plexus of ventral
M s4ri. 270j. and are gradually shifted caudal ward until I lioin ihr ilorsal aorta opfxisite the twenty-third segment (fourth luml-.M • III S mill, nnhryos tlu* umbilical arteries develop secondary' l4Mieral coniM . iiMM- wiili ilic aoria M-i^. 27X A), The new vessels pass lateral to the mes..I.. |iliii. tlmi-. and, in 7 nun. embryos, the primitive ventral steni-arter>' has .li ..|.|.t .lie tl I \\v M'^mrni of ihis new trunk, proximal to the origin of the exter>/../ *//./. ./;/i;v wliiili Noc»n arises from it, lx»comes the common iliac. The rein. nmlii III \\\i' (intliiliial trunk constitutes the hypogastric artery. ^Tien theitn traMs at birth, the distal portion of the hjpogastric arteries, ii. II. |u 1\ I- ii. iiiubilii UN. atrophy, forming tlu* solid obliterated hypogastric arteries • I iitiili .iii.tiiintx
Development of Veins
carries blood from the liver sinusoids to the sinus venosus. The proximal portion of the left vitelline vein soon is largely absorbed into the sinusoids of the liver and shifts its blood flow into the right horn of the sinus venosus. In the meantime the liver tissue grows laterally, comes into contact with the umbilical veins, and taps them so that their blood flows more directly to the heart through the sinusoids of the liver (Fig. 280). As the channel of the right proximal vitelline is larger, the blood from the left umbilical vein flows diagonally to the right horn of the sinus venosus. When all the umbiUcal blood enters the liver, as in embryos of 5 to 6 mm., the proximal portions of the umbilical veins atrophy and disappear (Fig. 281). In 5 mm. embryos the vitelline veins have formed three cross anastomoses with each other (Figs. 280 and 281): (1) a cranial transverse connection in the liver, ventral to the duodenum; (2) a middle one, dorsal to the duodenum; and (3) a caudal one, ventral to it. There are thus formed about the gut a cranial and a caudal venous ring. In embryos of 7 mm. the left umbilical vein has enlarged, while the corresponding right vein has degenerated. Of the two venous loops, only the right limb of the cranial ring and the left limb of the caudal ring, together with the median dorsal anastomosis, persist. A new vein the superior mesetileric, develops in the mesentery of the intestinal loop and joins the left vitelline vein just caudal to its dorsal middle anastomosis with the right vitelline vein. Subsequently, with the atrophy of the yolk sac, the left vitelline vein degenerates caudal to its junction with the superior mesenteric vein. The persisting trunk between the superior mesenteric vein and the liver is the vena porta, and thus represents: (I) a portion of the left vitelline vein in the left limb of the caudal ring; (2) the middle transverse anastomosis between the vitelline veins; (3) the portion of the right vitelline vein which forms the right limb of the cranial ring.
—Reconstruction of the blood vessels of a 4.2 m m. human embryo in ventral view (His).
Fig. 280.— Reeonsli human embryo (after Ingalls).
In the liver the portal vein through its cranial anastomosis between the vitelline veins is connected with the left umbilical vein. As the right lobe of the liver grows, the course of the umbilical and portal blood through the intrahepatic portion of the right vitelline vein becomes circuitous, and hence a new direct channel to the sinus venosus is formed through the hepatic sinusoids. This is the ductus venosus (Fig. 281), which is obliterated after birth and forms the Hgamentum venosiim of the postnatal liver.
According to Mall, ihe inlrahepatic portion of the right vitelline vein persists proximally as the riglil ramus of the hepatic vein, and distally as the ramus arcualus of the portal vein. The intrahepatic portion of the left vileliine vein drains secondarily into the right hom of the sinus venosus, and proximally forms later the hjl hepatic ramus. Distally, where it is connected with the left umbilical vein, it becomes the ramus angularis of the vena porta:. In this way two primitive portal, or supplying trunks, and two hepatic, or draining trunks, originate. Later there are differentiated first four, then six, such opposed trunks within the liver, and the six primary lobules suppUed and drained by these trunks may be recognized in the adult liver.
Of the umbilical veins the right disappears early; the left persists during fetal life, shifts to the median line, and courses in the free edge of the falciform ligament. After birth its lumen is closed and from the umbilicus to the liver it forms the ligamentum teres. In early stages veins from the body wall drain into the umbilical veins.
The Anterior Cardinal Veins and the Origin of the Superior Vena Cava
The anterior cardinal veins consist each of two parts (Fig. 271): (1) The true anterior cardinals, located laterad in the segmented portion of the head and neck and draining into the common cardinal veins; (2) the vena capitis medialis, extending into the unsegmented head proper and running ventro-lateral to the brain wall. In embryos of 20 mm. there has formed by anastomosis a large connection between the right and left anterior cardinals, which carries the blood from the left side of the head into the right vein (Fig. 282 O- Soon the left anterior cardinal loses its connection with the common cardinal on the left side (Fig. 282 D). The proximal portion of the left common cardinal, with the transverse portion of the sinus venosus, persists as the coronary sinus. The right common cardinal and the right anterior cardinal vein as far as its anastomosis with the left anterior cardinal become the superior vena cava. The anastomosis itself forms the left vena anonyma, while that portion of the right anterior cardinal between the left vena anonyma and the right subcla\ian vein is known as the rigkt vciia anonyma. The distal portions of the anterior cardinab become the internal jugular veins of the adult, while the external jugulars are new veins which develop somewhat later.
Fig. 281. — A diagram showing the development of the portal v about 7 mm. (modified after His).
human embryo of
Fig. 282. — Four diagrams showing the development of the superior and inferior vencCKVKaDdtlM fate of the cardinal veins (modiGed after Kollmann). X in A, anastomoais between hepatic and tubcantinal vein; *, anastomosis between subcardinal veins; X in C, anastomosis between anterioi cardinal veins which forms the left vena anonyma; * in C, cranial anastomosbi between the posterior cardinal veins; z, caudal anastomosis between the same veins; A', kidney; S, suprarenal gland; r, testb.
The tcna capitis medialis (Fig. 271) is the continuation of the anterior cardinal vein into ihe head of the embryo where at first it lies mesial to the cerebral nerves. Later it is partly shifted by anastomoses lateral lo Ihe cerebral ner\'esand forms the pen a capitis lateralis (Figs. 283 and 284). In 1 1 mm. embryos this emerges with the n. facialis, and. caudal to the n. hypoglossus, becomes the internal jugular. Cranially, in the region of the fifth nerve, the median vein of the head persists as the sinus caternosus and receives the ophthalmic vein from the eye and the anterior cerebral vein from the fore- and mid-brain regions (Fig. 284 C). Between the n. trigeminus and the facialis, the middle cerebral vein from the metencephalon (cerebellum) joins the v. capitis lateralis before it leaves the cranium. More caudally the posterior cerebral vein from the myelencephalon emerges through the jugular foramen and is drained with the others by the v, capitis lateralis into the internal jugular (Fig. 284 B). Soon the cerebral veins reach the dorsal median line (Fig. 284 O, and longitudinal anastomoses arc formed: (1) between the anterior and middle cerebral veins, giving rise to the superior sagittal sinus; and (2) between the middle and posterior cerebral veins forming the greater part of the transverse sinuses. In embryos of 33 mm. the v, capitis lateralis disappears and the blood from the brain passes through the superior sagittal and lateral sinuses and is drained by way of the jugular foramen into the internal jugular vein (Fig. 284 C, D). The middle cerebral vein becomes the superior petrosal sinus, but the inferior petrosal sinus is formed as a new channel median to the internal ear. The anterior cerebral vein becomes the superficial middle cerebral of adult anatomy. A more detailed account of these changes may be found in the original work of Mall (Amer. Jour. Anat., vol. 4. 1905).
Fig. 283,— Veins of the head ,.f 1 9 mm. human embryo (after Mall). X 9.
J.I ];l.i';j a<«/) 4/]^:ij kiU; Xbtt: cumffiuo caitfinal vcuk ilrig. 2BZ ^).
..I i.^huuii/ uim fiuBi tiMr imAksm qaimritirR.
ii*i'< >i<lii(/ aiid d^Mil imlet^gmiemial «bm» iram llr bodf *«■■ (E^ 283! J).
- ^>i'i. .lUil v^jiJi A'«u(ii *AiMt:r by «xu&sUimoecs veotzal to tbe aartm. Thn al lie l/Ji/< '1 ii'/fn < Im- iiiA'at;<i4yhf <ji , jiucicrior ertrefidfirs, aod donal body maM is ia cMJy . i»iK' 'i'liii^'^l i;y <Im' i^jtiUTTMr atrdinad vems alone.
i Im ill vi'l//j /iiu'iif of <ti«f ufi|jajred twna cava inferior bcgpns wImb aMmmm itannn I i i i.il^|j.^lu-il \fi'iwi^'ii the fifilU hepatic vein of the fiver and the wigM t ,uilhhil vthi n\ i\w nwmtw\}\\fm, primarily a tributaiy of the
Ilti livn I »iH lu' righl id'tt* becomes attached to the dorsal body waD and from M |iMini ifl Miiiun 11 rMgc, the* /tf^a v^mr coihb (Fig. 199), extends caudahrard. AiiHhlhiH lii llfivU (|V|0), itiplliaricM from the subcardinal vein mvade the pKca '•H.» »'t\u, iiimI, KiDwiiiK cninUlly, meet and fuse with capiUaries extending i.iMihitl IhUM \\w livi'i ninusioliU.
I liui* li llio Wm Fe f^ka vma: cawe (Fig. 282 A), which is already Ml III liMhUMi imihi\i^ til' In mm. (Kollmann). This vein rapidly enlarges, .) « .)l H titi ihv )>huimii«l(l Mmms i(imi U'twocn the subcardinab and postaior carM W UUhI (nun )^>th lower posterior cardinals is somt Un w^y 4 th light ;fuibcardinal and ligl&t hq[MUic veinsAnKna)» jwl cranial to their enlarged anasto;mvAn aiH) air intenupted (F. 282 C). niiauiKkate by across a n a ntwn o ri s Ans ai3np biBisijuwu of the"
vein. The corresponding portion of the new left vein with the transverse anastomosis becomes the lefi common iliac vein. The blood from these veins is now drained by the unpaired inferior vena cava which is composed of the following veins: (1) the common hepatic and right hepatic veins (primitive right vitelline) ; (2) the vein of the plica venae cavae; (3) an inter-renal portion of the right subcardinal vein with its great mesial anastomosis; (4) the new vein which replaces the right posterior cardinal.
The permanent kidneys take up their positions opposite the great anastomosis between the subcardinals and at this point the renal veins are developed (Fig. 282 B) ; the longer left renal vein differs from the right in that proximally it represents a left portion of the anastomosis itself (Fig. 282 D), A cephalic portion of the left subcardinal vein persists as the left suprarenal veiny which thus opens into the left renal instead of joining the inferior vena cava as does the right suprarenal vein of similar origin. The left spermatic or ovarian vein early drains into the left caudal border of the great subcardinal anastomosis, which as we have seen contributes to the left renal vein. The right spermatic or ovarian vein opens into the right border of that portion of the subcardinal anastomosis which is incorporated into the inferior vena cava. Tlie lumoar vdns develop fioim the lune prevertebral plexus that gives rise to the caudal s^ment of the inferior vena cava.
Fig. 284. — Four diagrams showing the development of the veins of the head (after Mall). i4, At four weeks; B, at five weeks; C, at the beginning of the third month; D, from an older fetus.
Bcgg (Anat. Rec., vol. 10, 1916), on the contraiy, findi that in the At, where the mao nephrui arc diminutive, the posterior cardinal veins ue not incoiporatedinto the mcMoephrd to disappc&T with that organ, but the right posterior cardinal pcniita as the aegment ot the inrcrior vena cava below the lubcardinal contribution.
Fig. 285. — Four reconstructions ot the \-rina of the human right »tm (after F. T. I-ewis). X about IS. . /I, 10 mm. embryo; 8, It.S nun. embiyo; C, 16 mm. embryo; D, 22.8 mm. embiyo.
The Veins of the Extremities
The primitive capillary plexus of the upper and kiwer limb buds gives rise to a border vein (Figs. 285 and 321), which courses about the periphery of Lhe (lattennl IJmb buds (Hochstctter). In the upper extremity, the ulnar portion of the border vein persists, forming at different points the subclavian, axillary, brachial, and bantic veins. The border vein at first opens into the dorsal wall of the posterior cardinal vein (embryos of 10 mm.), but, as the heart shifts its position caudalward, it finally drains by a ventral connection into the anterior cardinal or internal jugular vein (Lewis). The cephalic vein develops secondarily in connection with the ulnar border vein; later, in embryos of 23 mm., it anastomoses with the external jugular and finally drains into the axillary vein as in the adult. With the development of the digits, the w. cephalica et basilica become distinct, as in embryos of 35 mm., but later are again connected by a plexus on the dorsum mani, as in the adult (Evans in Keibel and Mall, vol. 2).
In the lower extremity the fibular portion of the pnmitive border vein persists. Later the I', saphena magna arises separately from the posterior cardinal, gives off the w. femoralis and tibialis posterior, and annexes the fibular border vein at the level of the knee. Distal to this junction the border vein persists as the v. tibialis anterior and probably the v. saphena parva; proximally it becomes greatly reduced, forming the v. glutea inferior.
Anomalies. — Anomalous blood vessels are of common occurrence. They may be due: (1) to the choice of unusual paths in the primitive vascular plexuses; (2) to the persistence of vessels usually obliterated, e. g., double superior venae cavae; right aortic arch; permanent ductus arteriosus; (3) to incomplete development, e. g., double (unfused) heart or double dorsal aortac.
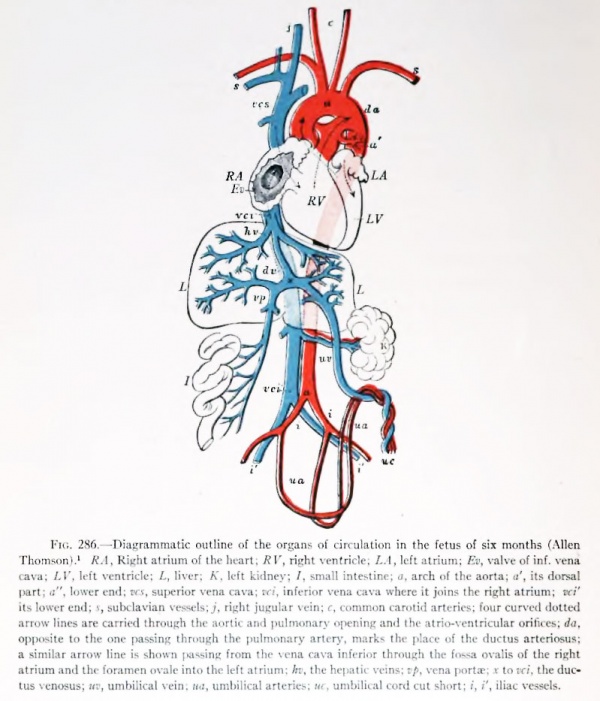
Fetal Circulation
During fetal life oxygenated placental blood enters the embryo by way of the large umbilical vein and is conveyed to the liver (Fig. 286). There it mingles with the small amoimt of venous blood brought in by the portal vein. It is carried to the inferior vena cava either directly, through the ductus venosuSy or indirectly through the liver sinusoids and hepatic vein. The impure blood of the inferior vena cava and portal vein affects but slightly the greater volume of pure placental blood. Entering the right atrium it mingles somewhat with the venous blood returned through the superior vena cava. It is said that the blood from the inferior vena cava is directed by the valve of this vein through \h& foramen ovale into the left atrium (following the path of the soimds in Figs. 262 to 264) which, before birth, receives little venous blood from the lungs. This purer blood of the left atrium enters the left ventricle, and is driven out through the aorta to be distributed chiefly to the head and upper extremities.
The venous blood of the superior vena cava, slightly mixed, is supposed to pass from the right atrium into the right ventricle, whence it passes out by the pulmonary artery. A small amount of this blood is conveyed to the lungs by the pulmonary arteries, but, as the fetal lungs do not function, most of it enters the dorsal aorta by way of the ductus arteriosus. Since the ductus is caudal to the origin of the subclavian and carotid arteries, its less pure blood is distributed to the trunk, viscera, and lower extremities. The placental circuit is completed by the hypogastric f or umbilical^ arteries by way of the umbilical cord.
Pohlman (Anat. Rec, vol. 2, 1908) interprets ...s experiments to indicate that, contrary to the generally accepted view, there is a mingling of the blood which enters the right atrium through the two caval veins. If this occurs there would be no difference in the quality of blood distributed to the various parts of the body.
Changes at Birth
At birth the umbilical vessels are ruptured and the lungs l>ecome functional. The umbUical arteries and veins, no longer used, contract and their lumina are obliterated by the thickening of the inner coat (tunica intima). The lumen of the umbilical artery is occluded after four days, that of the umbilical vein within a week. The cord-like vein is persistent as the ligamentum teres of the liver; the arteries become the obliterated hypogastrics.
Fig. 286. — Diagrammatic outline of the organs of circulation in the fetus of ail months (Allen Thomson).[1] RA, Right atrium of the heart; RV, right ventricle; LA, left atrium; Ev, valve of inf. vena cava; LV, left ventricle; L, liver; K, left kidney; I, small intestine; a, arch of the aorta; a', its dorsal part; a", lower end; TKi, superior vena cava; vci, inferior vena cava where it joins the right atrium; vet' its lower end; i, subclavian vessels; j, right jugular vein;c, common carotid arteries; four tnirvcd dotted arrow lines are carried through the aortic and pulmonary opening and ihe atrio-ventricular orifices; da, opposite to the one passing through the pulmonary artery, marks the place of the ductus arteriosus; a similar arrow line is shown passing from the vena cava inferior through the fossa ovalis of the right atrium and the foramen ovale into the left atrium; hv, the hepatic veins; vp, vena ports; i to vci, the ductus venosus; uv, umbilical vein; ita, umbilical arteries; ur, umbilical cord cut short; i, i', iliac vessels.
- ↑ In this diagram the arteries are conventionally colored red and the veins blue, but these colors are not intended to indicate the nature of the blood conveyed by the reflective vessels.
The ductus venosus atrophies because after birth only the blood from the portal vein enters the liver, and this is all drained into the liver sinusoids, forming the portal circulation. The ductus venosus is persistent as the fibrous ligamentum venosuMf embedded in the wall of the liver.
The ductus arteriosus ceases to function after birth, as all the blood from the pulmonary arterial trunk is conveyed to the expanded lungs. The ductus becomes impervious from ten to twenty days after birth and persists as a solid, fibrous cord, the ligamentum arteriosum.
After birth, the large amount of blood now returned to the left atrium from the functional lungs equalizes the pressure in the two atria. As a result, both during diastole and systole, the septum primum, or valve of the foramen ovale, is pressed against the septum secundum^ closing the foramen ovale. Eventually the two septa fuse, though they may be incompletely united during the first year after birth, or even longer (p. 255).
The Lymphatic System
The lymphatic system originates in a plexus of lymphatic capillaries distributed along the primitive main venous trunks. By the dilation and coalescence of this network at definite regions five lymph sacs appear (Fig. 287). Paired jugular sacs arise in 10 to 1 1 mm. anbryos lateral to the internal jugular veins. In embryos of 23 mm. the unpaired rdroperitoneal sac develops at the root of the mesentery adjacent to the suprarenal bodies, and the cisterna chyli also appears. Paired posterior sacs arise in relation to the sciatic veins in embryos 24 nmi. long. These sacs at first contain blood which they soon discharge into neighboring veins, thereupon losing their venous connections. Vfith relation to the lymph sacs as centers, the thoracic duct (at 30 mm.) and the peripheral lymphatics develop. The jugular sacs alone acquire with the internal jugular veins secondary connections which are later utilized by the thoracic and right lymphatic ducts. The various sacs themselves are eventually transformed into chains of lymph nodes.
Two discordant views exist as to the origin of the lymphatics. According to Sabin (Amer. Jour. Anat., vols. 1, 1901 ; 9, 1909) and Lewis (Amer. Jour, Anat., vol. 5, 1906), sprouts arising from the endothelium of veins form the single and paired sacs already described. From these five sacs the thoracic duct and peripheral lymphatics develop as endothelial outgrowths. Thus lymphatic vessels grow to the head, neck, and arm from the jugular sacs; to the hip, back, and leg from the posterior sacs; and to the mesentery from the retroperitoneal sac Accnrding to this view, then, endothdium can arise ordyfrom pre-€xisting endotkeUum.
Other investigators (Huntington, Amer. Jour. Anat., vtA. 16, 1914; Mem. Wistar Inst., 1911 ; and McClure) hold that the lymph sacs are formed in situ by the fusion of discrete mesench>-mal spaces which become lined with an endothe
Fig. 287. — Flat reconstruction of the primitive lymphatic system in a human embryo 30 mm. long (Sabin). X about 3.5. C.e., Cistema chyli; Lg., lymph gland; SJ.jug., jugular lymph sac; SJ.me!., retioperitoneal lymph sac; SJ.p., posterior lymph sac; SJ,i,, subclavian lymph sac; V.e., cephalic vein; V,c.i., inferior vena cava; V.f., femoral vein; Vj.i., internal jugular vein; VJ.p., V.T., renal vein; Vj., sciatic vein; V.u.ip.), prinil
Hum of transformed, bordering mesenchymal cells. Venous connections are purely secondary. The thoracic duct and the peripheral vessels develop similarly by the progressive fusion of separate clefts; hence endothelium can diJerenluUe conlinually from young mesenckyma. The further growth of endothelium, already formed, is not denied.
Fig. 289. — Diagrams representing four stages in the development of lym|)h glands. The earlier stages are shown on the left side of each figure (I^wis ami Stiihr).
Rapidly accumulating evidence seems to favor the latter view. The method of injection, upon which Miss Sabin and her followers have largely relied, has its limitations, for it obviously can furnish no information regarding discrete spaces prior to their linkage into continuous channels. Concise summaries of these views are given by Sabin (Anat. Rec, vol. 5, 1911) and by McClure (Anat. Rec, vob. 5, 1912; 9, 1915).
Lymph Glands
Paired lymph glands appear during the third month, first in the axillary, iliac, and maxillary regions. Those from the lymph sacs develop later. Plexuses of lymphatics first form either as ordinary networks of peripheral vessels or as secondary networks produced by a connective tissue invasion of the primitive lymph sacs. In either case a capillary plexus, with simple connective tissue septa, marks the first stage of development. Next (Fig. 289 A), lymphocytes collect in the connective tissue, forming follicles which become associated with blood capillaries. Finally, the lymphoid tissue is channeled by sinuses formed from lymphatic capillaries. The peripheral sinus develops afferent and efferent vessels. The central sinuses cut the lymphoid tissue into cords. In the larger lymph glands (Fig. 289 B) the connective tissue forms a definite capsule from which trabeculse dip into the gland.
— Two stages in the early development of the embryo oF 10.5 ram. (KoUinanD); B, from a an spleen (dorsal is below). 10 mm. embryo (Tonkoff ) .
Hemolymph glands, according to Schumacher (Arch. f. mik. Anat., Bd. 81, 1912), begin their development like lymph glands, but soon after the formation of the peripheral sinus the lymphatic connections degenerate and blood escapes from the blood capillaries into what are henceforth blood sinuses.
Spleen
This appears in embryos of about 10 mm. as a swelling on the left side of the dorsal mesogastrium near the dorsal pancreas (Fig. 288 A). The thickening is due to a temporary proliferation and invasion of mesotlielial cells into the underlying mesenchyme, which, meanwhile, has also undei]g!cme local enlargement and vascularization. Contrary to the older view, the cells from the peritoneal epithelium probably give rise to a large part, if not all, of the future spleen. The splenic anlage becomes pinched off from the mesogastrium (Fig. 288 B) with which it is ultimately joined by a narrow band only.
At first the blood vessels constitute a dosed system. The peculiar adult' circulation is acquired relatively late. Lif schitz has shown that, in human fetuses between 150 and 500 nun. long, red blood cells are actively formed in the qplenic pulp as clusters around the giant cells. The lymphoid tissue of the ^leeu first appears as ellipsoids about the smallest arteries in fetuses of four mcmths. At seven months the ovoid splenic corpuscles appear as lymphoid nodules about the larger arteries.
Glomus Coccygeum
The coccygeal gland is present in 150 mm. (C H) fetuses as an encapsulated cluster of polyhedral cells at the apex of the coccyx. Later it becomes lobtilated by the ingrowth of connective tissue trabecule and receives a rich vascular supply. According to Stoerck (1906) its tissue at no rit«#> resembles the chromaffin bodies, although this has commonly been believed.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Prentiss CW. and Arey LB. A laboratory manual and text-book of embryology. (1918) W.B. Saunders Company, Philadelphia and London.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
- (Text at early stage of editing, images to be added)
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 27) Embryology Book - A Laboratory Manual and Text-book of Embryology 9. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_A_Laboratory_Manual_and_Text-book_of_Embryology_9
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G