ANAT2341 Lab 8 - Sex Determination
| ANAT2341 Lab 8: Introduction | Sex Determination | Early Embryo | Late Embryo | Fetal | Postnatal | Abnormalities | Online Assessment | Genital Quiz |
Introduction
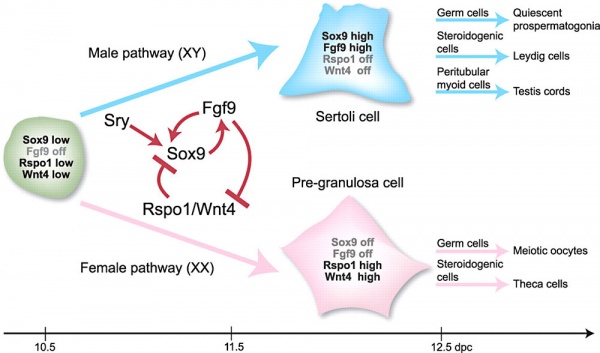
Male (XY)
Sry was discovered (1990) by studying a human XY female, resulting from a deletion in the Y chromosome that did not allow testis development. Subsequent mapping of this deletion allowed isolation and characterization of the SRY gene.
There is a suggestion that SRY may allow testes development by acting to inhibit DAX1, which is expressed in the indifferent gonad at the same time. The mechanism of an inhibitor inhibiting and inhibitor is seen in some other developing systems.
- encodes a 204 amino acid protein (Mr 23884 Da) that is a zinc-finger transcription factor.
- transcription factors bind to specific sites of DNA and regulates the transcription (expression) of other genes, we still do not know all the genes SRY regulates.
- expressed when testes begin to form, in gonadal tissue and does not require the presence of germ cells.
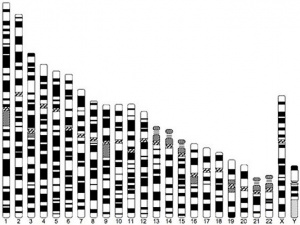
Notes: The Y chromosome is much smaller than the X chromosome and by definition, cannot contain important genes for other cellular functions.
Nomenclature, capital letters are used for human genes (SRY) and lower case letters are used for the equivalent genes in other species (sry).
Female (XX)
- In contrast to the Y chromosome, the X chromosome contains about 5% of the haploid genome and encodes house-keeping and specialized functions.
- The genetic content of the X chromosome has been strongly conserved between species.
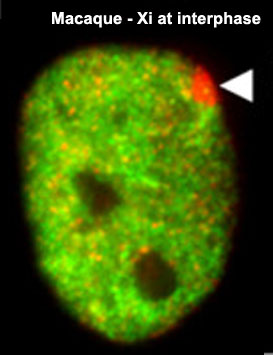
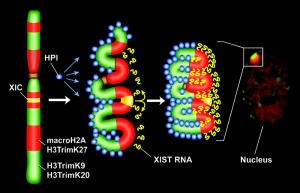
- Links: Signaling in genital development | Fig. 1 - image | X Chromosome Inactivation - Epigenetics 1
Primordial Germ Cells
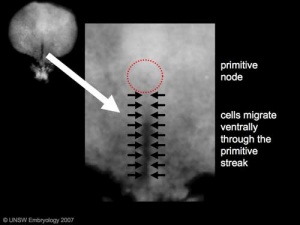
Human Embryonic Disc (Stage 7) |
|
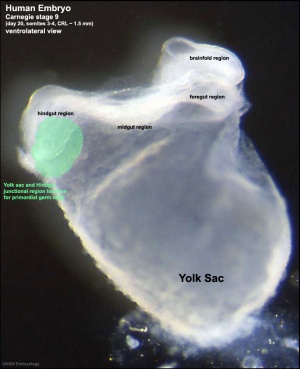
Human Embryo (Stage 9) primordial germ cell region |
|
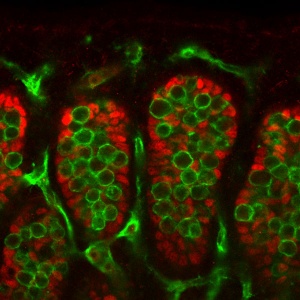
Mouse gonad Sertoli and Germ Cells |
|
References
- ↑ <pubmed>16700629</pubmed>| PLoS Biol.
Additional Information
The information shown below is not part of today's Practical.
Mouse - gonadal supporting cell development
Genes implicated in sexual development in mammals
Table below modified from Table 1. Genes implicated in sexual development in mammals in recent review article.<pubmed>17237341</pubmed>| Physiol. Rev.
| Gene | Protein Function | Gonad Phenotype of Null Mice | Human Syndrome | |
| Bipotential gonad | ||||
| Wt1 | Transcription factor | Blockage in genital ridge development | Denys-Drash, WAGR, Frasier syndrome | |
| Sf1 | Nuclear receptor | Blockage in genital ridge development | Embryonic testicular regression syndrome | |
| Lhx9 | Transcription factor | Blockage in genital ridge development | a | |
| Emx2 | Transcription factor | Blockage in genital ridge development | a | |
| M33 | Transcription factor | Gonadal dysgenesis | a | |
| Testis-determining pathway | ||||
| Gata4/Fog2 | Transcription/cofactor | Reduced Sry levels, XY sex reversal | a | |
| Sry | Transcription factor | XY sex reversal | XY sex reversal (LOF); XX sex reversal (GOF) | |
| Sox9 | Transcription factor | XY sex reversal | Campomelic dysplasia, XX sex reversal (GOF) | |
| Sox8 | Transcription factor | XY sex reversal in combination with partial loss of Sox9 function | a | |
| Fgf9 | Signaling molecule | XY sex reversal | a | |
| Dax1 | Nuclear receptor | Impaired testis cord formation and spermatogenesis | Hypogonadism | |
| Pod1 | Transcription factor | XY sex reversal | a | |
| Dhh | Signaling molecule | Impaired differentiation of Leydig and PM cells | XY gonadal dysgenesis | |
| Pgdra | Receptor | Reduction in mesonephric cell migration | a | |
| Pgds | Enzyme | No phenotype | a | |
| Arx | Transcription factor | Abnormal testicular differentiation | X-linked lissencephaly with abnormal genitalia | |
| Atrx | Helicase | ND | ATRX syndrome | |
| Insl3 | Signaling factor | Blockage of testicular descent | Cryptorchidism | |
| Lgr8 | Receptor | Blockage of testicular descent | Cryptorchidism | |
| Hoxa10 | Transcription factor | Blockage of testicular descent | Cryptorchidism | |
| Hoxal1 | Transcription factor | Blockage of testicular descent | Cryptorchidism | |
| Amh | Hormone | No Müllerian duct degeneration | Persistent Müllerian duct syndrome | |
| Misrl1 | Receptor | No Müllerian duct degeneration | Persistent Müllerian duct syndrome | |
| Pax2 | Transcription factor | Dysgenesis of mesonephric tubules | a | |
| Lim1 | Transcription factor | Agenesis of Wolffian and Müllerian ducts | a | |
| Dmrt1 | Transcription factor | Loss of Sertoli and germ cells | XY femaleb | |
| Ovary-determining pathway | ||||
| Wnt4 | Signaling molecule | Müllerian duct agenesis, testosterone synthesis, and coelomic vessel formation | XY female (GOF) | |
| FoxL2 | Transcription factor | Premature ovarian failure | BPES | |
| Dax1 | Nuclear receptor | XY sex reversal (GOF) | XY sex reversal (GOF) | |
|
a No mutations in human sexual disorders identified to date. b Candidate gene for 9p deletion, XY sex reversal. |
| ANAT2341 Lab 8: Introduction | Sex Determination | Early Embryo | Late Embryo | Fetal | Postnatal | Abnormalities | Online Assessment | Genital Quiz |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 7) Embryology ANAT2341 Lab 8 - Sex Determination. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/ANAT2341_Lab_8_-_Sex_Determination
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G