User:Z5014754
Uterine decidualisation (or uterine decidualization) is the process by which uterine stromal cells differentiate in response to both steroid hormones (progesterone) and embryonic signals into large epitheliod decidual cells. This process is essential for the progress of implantation and establishing fetal-maternal communication.
Introduction
The uterus is an organ of the female internal reproductive tract that houses the conceptus for the vast majority of development. Outside of pregnancy the uterus has an inner lining (endometrium) that undergoes cyclic changes in response to reproductive hormones. However, following implantation, these cycles cease and the endometrium must undergo morphological and functional changes in order to accommodate development of the conceptus. This process of changes, known as uterine decidualisation, causes the endometrium form highly specialised decidua. These changes are largely mediated by sustained high levels of maternal progesterone, and signals the beginning of placentation in the mother and child. [1]
Decidual changes
Processes
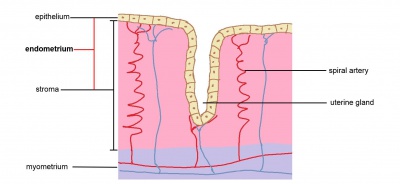
The endometrium consists of an epithelial layer lining the inside of the uterus, as well as an underlying layer of vasculature, cells and dense connective tissue (collectively known as the stroma). Uterine glands are simple invaginations of the endometrium that extend into the stroma. It is important to note that during pregnancy decidual changes occur only in the stroma and uterine glands.
Cells of the stroma are normally densely packed and small. They proliferate in the mid-late proliferative phase of the menstrual cycle, and slough off during menses (early proliferative phase) with closure of the spiral arteries. Uterine glands will dilate and secrete uterine milk within the secretory phase of the cycle. However, upon a viable implantation (during the mid-secretory phase), the trophoblast layers of the conceptus will push into the endometrium, releasing human chorionic gonadotropin (hCG). This prevents regression of the corpus luteum, the main source of hormones (especially progesterone) that keeps the endometrium in the secretory phase. Thus, the corpus luteum will continue to secrete, sustaining progesterone levels at a high level. This is detected by progesterone receptors (PRs) on the endometrial cells, mainly PRA receptors (the other isoform being PRB). Under this influence:
- stromal cells will expand and fill with glycogen and lipids, becoming large and polygonal (described as epitheliod decidual cells), and begin to secrete proteins [2]
- increased vasculature and bloodflow to the area [2]
- uterine glands will dilate, lengthen, become more coiled and wavy in shape, and actively secrete glycogen-rich uterine milk [1]
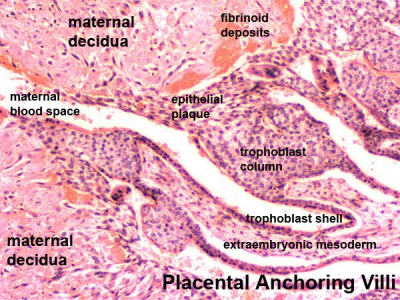
- deposition of glycogen, a fibrinoid layer and epithelial plaque at anchoring villi [3]
- immunological changes including an influx of specialised immune cells. [1]
These are the changes characteristic of decidualisation. While initially only occurring at the site of implantation, decidualisation then spreads through the entire uterus (but not the cervix), with the stroma layer becoming thick and edematous. Over the length of the embryonic period, the uterine glands slowly detract after their intial growth (as the embryo's nutrition begins to be given by the yolk sac)[4]; and distinct decidual structures begin to form around the conceptus.
Structure
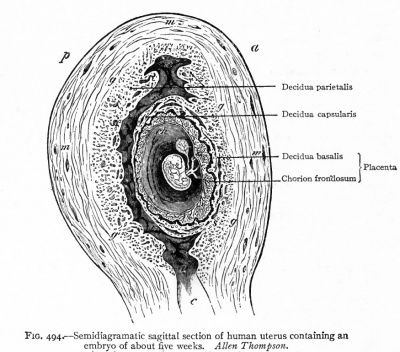
In the late embryonic period, the conceptus begins to bulge into the uterine cavity at the abembryonic pole: the decidua covering this portion of the conceptus is called the decidua capsularis, forming a thin layer that encircles the conceptus. At the opposite (embryonic) pole, the underlying decidua is known as the decidua basalis. The remaining decidua is known as the decidua parietalis, covering the rest of the uterus farther away from the conceptus and gradually becomes decidualised through the embryonic period. [3]
Changes in the decidual structures include: [3]
- The decidua basalis plays an important role in the formation of the maternal surface of the placenta, with the fetal surface being formed by the chorion frondosum, and the intervillous space between, together constituting the placenta.
- During the 4th and 5th months of pregnancy, wedges of decidua basalis known as decidual septa push into the intervillous space, separating the maternal surface of the placenta into cobblestone-like lumps called cotyledons.
- By the 3rd month of pregnancy, as the fetus grows large and fills the uterine cavity, the decidua capsularis is pressed against the decidua parietalis, causing the decidua capsularis to disintegrate by the 5th to 6th month.
- Due to pressure from the embryo and its associated membranes, the decidua parietalis becomes much thinner throughout the pregnancy.
Role
The changes of decidualisation are very important for the future development of the conceptus. Some of the roles performed by certain decidual changes are listed in the following table:
| Decidual Change | Role in Pregnancy |
|
Growth and accumulation of glycogen and lipids in stromal cells |
|
| Increased local vascularisation |
|
| Growth and secretion by uterine glands |
|
| Deposition of glycogen, fibrinoid layer and epithelial plaque at anchoring villi |
|
| Immunological changes including influx of specialised immune cells |
|
It should be noted that decidualisation is not a requirement for a successful implantation; a blastocyst can implant in areas that are not decidualised, as is often seen in ectopic pregnancy.
Decidualisation factors
Mediation of uterine decidualisation can be thought of to occur over three phases:[2]
- Days 8-10 post-ovulation (pre-implantation): Primarily mediated by estrogen and progesterone. The changes associated with this phase happen every secretory phase, whether or not implantation occurs within that cycle.
- During implantation: Combined effects of estrogen/progesterone along with blastocyst-derived signals such as preimplantation factor, Activin A and Prokineticin 1. Changes in this phase are unique to pregnancy.
- Post implantation: Combined effects of embryo-derived signals along with proteins released by decidual cells such as growth factors and cytokines. Changes during this phase are often triggered by autocrine and paracrine action.
Some Recent Findings
|
References
- ↑ 1.0 1.1 1.2 1.3 <pubmed>17960529</pubmed>
- ↑ 2.0 2.1 2.2 2.3 2.4 <pubmed>11900887</pubmed>
- ↑ 3.0 3.1 3.2 3.3 Gary C. Schoenwolf; William James Larsen (2009), Larsen's Human Embryology, Churchill Livingstone/Elsevier, ISBN 978-0-443-06811-9
- ↑ <pubmed>9647550</pubmed>
- ↑ <pubmed>23407384</pubmed>
- ↑ <pubmed>22679098</pubmed>
- ↑ <pubmed>11590404</pubmed>
- ↑ <pubmed>24086495</pubmed>
- ↑ <pubmed>24025400</pubmed>
- ↑ <pubmed>21173644</pubmed>