2014 Group Project 4: Difference between revisions
(→BOTH) |
m (Protected "2014 Group Project 4" ([Edit=Allow only administrators] (indefinite) [Move=Allow only administrators] (indefinite))) |
||
| (229 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{ANAT2341Project2014header}} | {{ANAT2341Project2014header}} | ||
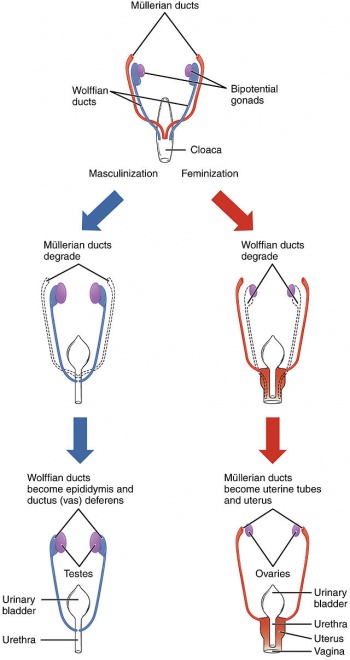
[[File:SexualDifferentation.jpg|350px|right|thumb|The stages in sexual differentiation of the female and male reproductive system]] | |||
=Genital= | =Genital= | ||
-- | ==Introduction== | ||
Genital system development occurs in both the embryonic and foetal phase of development. By the commencement of the foetal period, sexual determination and initial growth of the different gonads occur <ref name=Hill2014>Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development</ref>. It is within the foetal period that the internal and external genital organs develop <ref name=Hill2014>Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development</ref>. This page focuses on the foetal developmental processes, exploring the current and historical models and understanding, alongside the congenital abnormalities. | |||
Today there is an innumerable amount of research articles focused on foetal genital growth, with some addressing the system as a whole <ref name=PMID14641326><pubmed>14641326</pubmed></ref> and others investigating certain genital organs <ref name=PMID13362960><pubmed>13362960</pubmed></ref>. Current models and understandings have been obtained from both human <ref name=PMID11315960><pubmed>11315960</pubmed></ref> and animal populations <ref name=PMID13362960><pubmed>13362960</pubmed></ref>, and this page will outline some of the important current research. | |||
Research into foetal genital development can be traced to as early as the 16th century<ref name=PMID18462432><pubmed>18462432</pubmed></ref> with anatomists proposing theories and constructing models, proven to being critical in obtaining the in-depth information known today. This page will mention some of these historical findings in both the female and male genital system. | |||
This project will end with discussing some of the congenital abnormalities of the genital system, mentioning both the malformations that are most common <ref name=PMID16006950><pubmed>16006950</pubmed></ref> and those that are rare <ref name=PMID23635766><pubmed>23635766</pubmed></ref>. | |||
==System Development== | ==System Development== | ||
{| class="wikitable" | |||
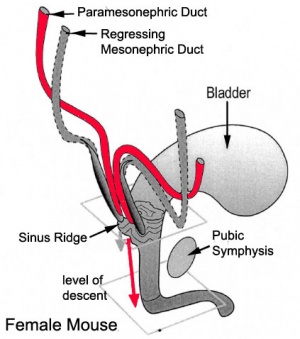
[[File:Paramesonephric duct.jpg|300px|thumb|Paramesonephric duct development]] | |||
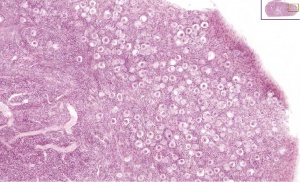
[[File:Infant ovary.jpg|300px|thumb|Histological image of primordial follicles in infant ovary]] | |||
'''Timeline of Embryonic, Fetal and Postnatal Genital Development'''<ref>Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development</ref> | |||
{| class="wikitable" | |||
|-bgcolor="purple"|align="centre" | |||
|'''Week''' || '''Development''' | |||
|-bgcolor="lavender" | |||
| 3-4 || Primordial germ cells migrate during gastrulation | |||
|- | |- | ||
| 4 || Intermediate mesoderm, pronephros primordium | |||
|-bgcolor="lavender" | |||
| 5 || Mesonephros and mesonephric duct | |||
|- | |- | ||
| | | 6 || Ureteric bud, metanephros, genital ridge | ||
|-bgcolor="lavender" | |||
| 7 || Cloacal divison, gonadal primordium - indifferent to first appearance of testis cords | |||
Female - Paramesonephric duct preservation or regression begins | |||
|- | |- | ||
| | | 8 || Paramesonephric duct, clear gonadal differentiation | ||
|-bgcolor="lavender" | |||
| 9 || Paramesonephric duct fusion in the female forming the uterus and lack of fusion laterally forming the fallopian tubes. Also forms vagina. | |||
|- | |||
| 15 || Primary follicles (ovary) | |||
From the 26th week (between week 4 and 5), the gubernaculum starts to pull the testes down and results in descent of the testes into the scrotal sac. | |||
Both male and female gonads undergo descent. | |||
|-bgcolor="lavender" | |||
| Puberty || Development of secondary sexual characteristics | |||
Female - start of menstruation, first egg released. | |||
Male - development of course voice, body hair and sperm formation. | |||
|- | |||
|} | |||
'''Related video''' | |||
<html5media>https://www.youtube.com/watch?v=MureNA-RSZM</html5media> | |||
---- | |||
===Development of Internal Genitalia=== | |||
{| style="width:100%" | |||
|bgcolor="lightskyblue"|'''MALE''' | |||
|bgcolor="violet"|'''FEMALE''' | |||
|- | |- | ||
| | |bgcolor="aliceblue"| The fetal development of internal genitalia is largely dependent on the endocrine functions of the fetal testes. The fetal testes produce masculinizing hormones such as '''testosterone''' which begins its release from the interstitial Leydig cells of the primitive seminiferous tubules during the 8th week of development, and also the release of '''Mullerian Inhibiting Hormone (MIS)''' which is released at the sixth and seventh weeks by the Sertoli Cells. Testosterone acts primarily on the mesonephric ducts to stimulate the formation of the male genital ducts, whereas the MIS acts on the paramesonephric duct to stimulate its regression. <ref name=PMID11315960><pubmed>11315960</pubmed></ref> <ref name=PMID24240231><pubmed>24240231</pubmed></ref> | ||
By the eighth week of fetal development in XY embryos, the testosterone produced in the testes results in the convolution of the proximal ends of the mesonephric ducts to form the '''epididymis''' <ref name=PMID24240231><pubmed>24240231</pubmed></ref>. The mesonephros begins to degenerate, however some of the mesonephric tubules remain and develop into efferent ductules, which then open into the duct of the epididymis. Distal to this end, the mesonephric duct begins to develop a thick lining of smooth muscle and progresses to become the ductus deferens.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
''Fetal Male Glandular Development'' | |||
* the seminal glands develop from lateral outgrowths from that caudal end of each mesonephric duct which nourishes the sperm and constitutes most of the fluid in the ejaculate | |||
* the endodermal outgrowths arise from the prostatic part of the urethra which grow into the surrounding mesenchyme. This acts as a base of the proliferation of the glandular epithelium of the prostate to differentiate- the associated mesenchyme will differentiate into dense stroma and the smooth muscle of the prostate. <ref name=PMID24240231><pubmed>24240231</pubmed></ref> | |||
* The bulbourethral glands are pea-sized and developed from paired outgrowths that originate from the spongy pat of the urethra. The adjacent mesenchyme gives rise to the stroma and smooth muscle fibres which will ultimately produce secretions which contribute to the semen. | |||
| bgcolor="lavenderblush"| The absence of testosterone production in XX embryos results in the regression of the mesonephric duct, and conversely the absence of MIH results in the development and progression of the paramesonephric ducts. The paramesonephric ducts give rise to most of the female internal genital system- the unfused cranial aspects of the ducts give rise to a primitive '''fallopian tubes''', whilst the caudally fused portions form the uterovaginal primordium which will develop into a '''uterus''' and '''superior vagina'''. The splanchnic mesenchyme gives rise to the endometrial stromal tissue and the myometrium.<ref name=PMID11315960><pubmed>11315960</pubmed></ref> | |||
Female genital development during the fetal period is not dependant on endocrine contributions from the foetus. Later in fetal development, maternal oestrogen's and oestrogen derived from the placenta contribute to the development of the fallopian tubes, uterus and the superior vagina. | |||
''Female Glandular Development'' | |||
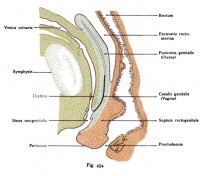
[[File:Kollmann454.jpg|thumb|200px|right|Internal genitalia in the female]] | |||
* | * The urethra develops outgrowths which form the mucus secreting '''urethral glands''' and paraurtehral glands. | ||
* | * outgrowths from the urogenital sinus form the '''greater vestibular glands''' in the lower third of the Labia Majora. These glands are also mucous secreting and are comparative to the bulbourethral glands in males. <ref name=PMID11315960><pubmed>11315960</pubmed></ref> | ||
|- | |- | ||
| | |- style="height:10px" | ||
| style="width:50%"| | |||
| | |} | ||
[[File:External genitalia current model.jpg|300px|right|thumb|Flow Diagram of the current model of embryonic and fetal development of the external genitalia]] | |||
===Development of the External Genitalia === | |||
{|class="wikitable mw-collapsible mw-collapsed" | |||
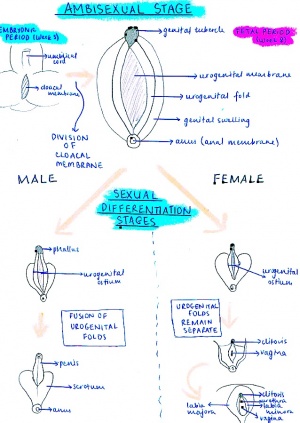
! ''Embryonic Period – fertilisation to end of 8th week (embryonic age) = AMBISEXUAL STAGE'' | |||
|- | |- bgcolor="lavender" | ||
| | | | ||
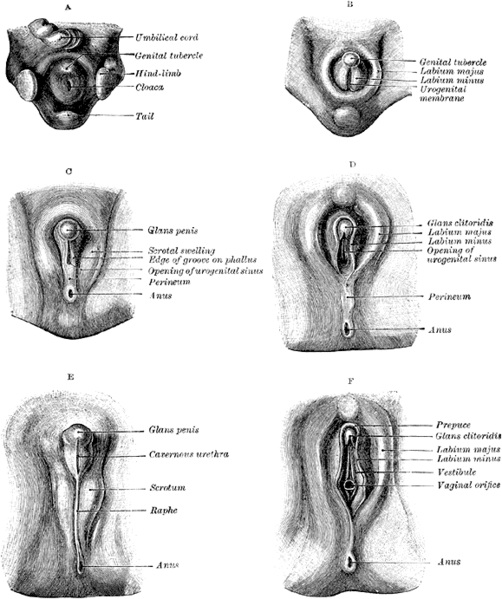
# The external genitalia initially begin in the perineal region as three primordia, being the genital tubercle in the midline and the bilateral genital swellings. These three primordia arise together with the differentiation of the cloacal part of the hindgut into the urogenital sinus, rectum and anal canal. The cloacal membrane extends from the perineum cranially to the root of the umbilical cord and during development, this bilayered cloacal membrane retracts into the perineum. This is due to cranial and medial migration of mesodermal cells into the ventral body wall between the ectoderm and endoderm of the cloacal membrane. These migrating mesodermal cells line around the membrane and accumulate, forming the three primordial swellings.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
# These external features are internally related to the cloaca, which becomes divided coronally by the urorectal septum into the urogenital sinus anteriorly, and the rectum and anus posteriorly. | |||
# This division of the cloaca occurs in a specific way so the allantois, Mullerian and Wolffian ducts and ureters all empty into the urogenital sinus. | |||
# When the cloacal membrane becomes divided into the urogenital and anal membranes, the urogenital membrane is bound cranially by the genital tubercle in the midline and laterally by the urogenital folds and genital swellings. <ref name=PMID11315960><pubmed>11315960</pubmed></ref> | |||
# The urogenital membrane degenerates to allow communication between the urogenital sinus and amniotic cavity.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
|} | |} | ||
<pubmed> | ---- | ||
<pubmed> | |||
<pubmed> | |||
'''The current model for fetal development of the external genitalia in humans and mice:''' | |||
{| style="width:100%" | |||
|bgcolor="lightskyblue"|'''MALE MODEL''' | |||
|bgcolor="violet"|'''FEMALE MODEL''' | |||
|- | |||
|bgcolor="aliceblue"|'''''Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION''''' | |||
# Initially, the female and male fetuses’ external genitalia are identical and include the genital tubercle in the midline, urogenital folds (forming the urogenital ostium) and genital swellings (laterally). | |||
# In males, the genital tubercle will eventually form the penis and the genital swellings migrate caudally and a fusion event in the midline occurs, thus forming the scrotum.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
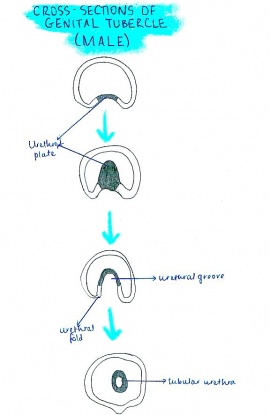
# As the genital tubercle elongates to form the penis, a groove forms on the ventral surface known as the urethral groove. The urethral folds that are continuous with the urogenital folds surrounding the urogenital ostium define the urethral groove laterally.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
# At first, the urethral groove and folds extend only part of the along the shaft of the elongating genital tubercle (known as the phallus at this stage). | |||
# Distally, the urethral groove terminates at the urethral plate, consisting of epithelial cells, and then extends into the glans of the penis, forming a channel. | |||
# As the phallus elongates, the urethral folds grow toward each other and fuse in the midline forming the midline epithelial seam, converting the urethral groove into a tubular penile urethra. The fusion of the urethral folds begins proximally in the perineal region and extends distally towards the glans of the penis.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
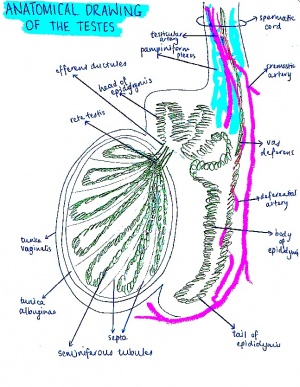
[[File:Cross section of genital tubercle male.jpg|270px|right|thumb|Cross section of the male genital tubercle]][[File:Anatomical diagram of testes.jpg|300px|left|thumb|Anatomical drawing of adult male testes]] | |||
* Hypospadias result from failure of formation or fusion of the urethral folds and this is the focus of current research. | |||
* The elongating phallus is covered externally by ectoderm that will eventually give rise to the penile epidermis. | |||
* Urethral epithelium has endodermal origins and the majority of the penis is derived from mesodermal cells. | |||
* During development, the mesoderm separates into connective tissues and dermis.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
* Dense areas of mesenchymal cells form within the shaft of the penis with the most superficial dense bodies forming the thick connective tissue capsule known as the tunica albuginae. | |||
* Mesenchyme surrounding the urethra forms smooth muscle of the urethral mucosa and submucosa. Erectile tissues such as the corpus spongiosum and corpus cavernosum then surround these two layers. | |||
* In some species, the mesenchyme of the genital tubercle also forms an os penis, comprised of bone and cartilage. | |||
* Genital tubercle development involves an outgrowth of somatic tissue from the body surface, similar to the development of the limb.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
* Development of the external genitalia is highly regulated by the endocrine system. Sexual differentiation of the external genitalia is determined by the presence or absence of androgen receptor signaling. The fetal testes produce testosterone, which travels to the genital tubercle via the bloodstream, where it is converted into 5a-dihydrotestosterone by the enzyme 5a-reductase. This formation of the highly potent 5a-dihydrotestosterone masculinizes the developing external genitalia, as binding of the 5a-dihydrotestosterone to its androgen receptor leads to the regulation of downstream signaling genes.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
*: Sonic Hedgehog (SHH) acts as an endodermal signal that normally regulates patterning of the hindgut and is expressed in the epithelium of the cloaca, urogenital sinus and urethral plate epithelium. However this has an important signaling pathway role in development of external genitalia. The SHH gene codes for a particular protein that has important roles in organogenesis as well as structures that are dependent upon mesenchymal-epithelial interactions, such as limbs, teeth and prostate.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
| bgcolor="lavenderblush"|'''''Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION''''' | |||
# Only minor changes occur from the embryonic ambisexual stage in the female, beginning with the minimal growth of the genital tubercle to form the clitoris. | |||
# The urogenital folds remain apart and unfused to form the labia majora.<ref name=PMID11315960><pubmed>11315960</pubmed></ref> | |||
# The genital swellings also remain apart and unfused to form the labia minora.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
* Together, these bilateral labial structures and the clitoris located proximally form the border for the urogenital ostium, thus forming the vestibule of the vagina, with vaginal and urethral openings. | |||
* The tubercle itself goes on to form the mons pubis. | |||
* Sexual dimorphism of the external genitalia in female humans is determined by the absence of androgenic pathways, however the female genital tubercle can be ‘masculinised’ as 5a-reductase and androgen recpetors are present. | |||
* The genital tubercle can not go on to form a penis as testosterone is not produced, however in some instances it is abnormally produced in excess by the suprarenal gland resulting in different degrees of masculinsation of the clitoris.<ref name=PMID11315960><pubmed>11315960</pubmed></ref> | |||
[[File:Clitoris.jpg|600px|thumb|center|Stages in the Development of the External Sexual Organs in the Male and Female]] | |||
|- | |||
|- style="height:10px" | |||
| style="width:50%"| | |||
|} | |||
===Descent of the Gonads=== | |||
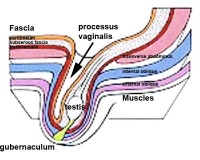
[[File:Testis-descent end.jpg|200px|thumb|right|Descent of testes]] | |||
'''Male:''' The testis undergo transabdominal and transinguinal descent. The testis lies in the subserous fascia. The processus vaginalis evaginates into the scrotum and the gubernaculum draws it into the scrotal sac. As it descends, it traverses past the superficial (external oblique fascia) and deep (transversalis fascia) inguinal rings of the inguinal canal. This begins in between the 4th and 5th week (usually around day 26) and spans over many days. It can occur unilaterally or bilaterally, more common in premature babies and can conclude postnatally.<ref>Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development</ref> | |||
'''Female''': ovaries undergo caudal and lateral shifts to be suspended in the broad ligament of the uterus. The gubernaculum does not shorten, attaches to paramesonephric ducts and causes medial translocation into the pelvis. The remnant in adult life of the gubernaculum is the ovarian and round ligament of the uterus, which suspend the ovaries and uterus respectively.<ref>Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development</ref> | |||
---- | |||
== | ==Current Research, Models and Findings== | ||
===Current Models=== | ===Current Models=== | ||
Most current research uses mouse models and observes the development of their external genitalia, especially their penile development, which initially appears to be different to human development. However, more microscopic inspection shows that mice have very similar external genitalia and are therefore appropriate animal models for observing such fetal development. As a result, mutant mouse models can effectively be used in future research to observe molecular mechanisms underlying hypospadias and their aetiology. <ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
When observing the morphology and cell biology of the developing testis, it is important to note that most of the research conducted on the subject involves the use of mouse models as a result of a lack of human subjects. It can be assumed that events in the human embryo correspond to the same events in the mouse embryo, however there are some differences between the time course of certain events and anatomy. <ref name=PMID17237341><pubmed>17237341</pubmed></ref> | |||
Some examples of different animal models used in research involving fetal development of male and female genital systems are: | |||
* Sheep<ref name=PMID21223560><pubmed>21223560</pubmed></ref> | |||
* Mouse<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
* Dogs<ref name=PMID13362960><pubmed>13362960</pubmed></ref> | |||
* Porcine<ref name=PMID23571006><pubmed>23571006</pubmed></ref> | |||
* Rats<ref name=PMID22248293><pubmed>22248293</pubmed></ref> | |||
* Monkeys<ref name=PMID21710394><pubmed>21710394</pubmed></ref> | |||
* Bovine<ref name=PMID20347535><pubmed>20347535</pubmed></ref> | |||
* Goats<ref name=PMID22006251><pubmed>22006251</pubmed></ref> | |||
Different models are used for different research topics; for example, research involving Polycystic ovary syndrome (PCOS) uses murine models preferable due to the developmental time frame that allows studies of inherited PCOS to be examined within an appropriate time frame. The sheep model is also beneficial to use for PCOS research as it is cost effective and their size allows them to be subjected to certain procedures such as ultrasound and neurotransmitter measures. Primates are examples of an optimal model however are limited in their accessibility and long time frame of development.<ref name=PMID21710394><pubmed>21710394</pubmed></ref> | |||
It is important to note that any findings associated with an animal model should be translated appropriately to the human model, as animals differ in their anatomy and regulatory mechanisms, as well as placentation. That is, sheep, rats and mice display different placentation and ovarian development that occurs in utero in sheep, primates and humans differs to the ex utero development in murine models.<ref name=PMID21710394><pubmed>21710394</pubmed></ref> | |||
All animal experiments must be performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. [[http://grants.nih.gov/grants/olaw/Guide-for-the-care-and-use-of-laboratory-animals.pdf | Guide for the Care and Use of Laboratory Animals]] | |||
===Current Research and Findings=== | |||
====Male==== | |||
---- | |||
{| | |||
|-bgcolor="aliceblue" | |||
| | |||
[[File:Hypospadias.jpg|400px|thumb|right|Different types of hypospadias]] | |||
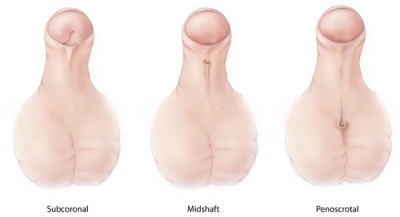
Extensive research into organogenesis of the external genitalia, mainly in males, is driven by the increasing incidence of hypospadias. Hypospadias are a result of the defect of fusion of the urethral folds of the lower part of the penis to fold and form the tubular penile urethra. The result of this in humans is the presence of an abnormal ventral urethral meatus, incomplete formation of the prepuce and an abnormal penile curvature.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
Development of the male external genitalia, which occurs in the fetal period of development, is androgen dependent and involves epithelial-mesenchymal interactions. Because of these interactions, which are very similar to limb development, research into the development of genital tubercle has utilised similar methods for both processes. A minority of hypospadias cases are a result of the androgenic pathways being impaired and causing this congenital defect. The cell-cell interactions that allow for the development of the male external genitalia are mediated by a broad range of signaling molecules and growth factors such as fibroblast growth factors (FGFs), Sonic hedgehog (SHH) and bone morphogenetic proteins (BMPs). Such signaling and growth factors are downstream of androgen receptor signaling and an understanding of the mechanisms that underlie normal penile development during the fetal period, will lead to a deeper understanding of the aetiology of hypospadias.<ref name=PMID14641326><pubmed>14641326</pubmed></ref> | |||
|} | |||
[http://npesu.unsw.edu.au/sites/default/files/npesu/surveillances/Congenital%20anomalies%20in%20Australia%202002-2003.pdf | Statistics regarding congenital abnormalities, including hypospadias and epispadias for 2002 and 2003 in Australia] | |||
---- | |||
{| | |||
|-bgcolor="aliceblue" | |||
| | |||
[[File:POPs and risk of hypospadias.jpg|400px|thumb|left|Table of levels of POPs in maternal serum samples and risk of hypospadias in infants]] | |||
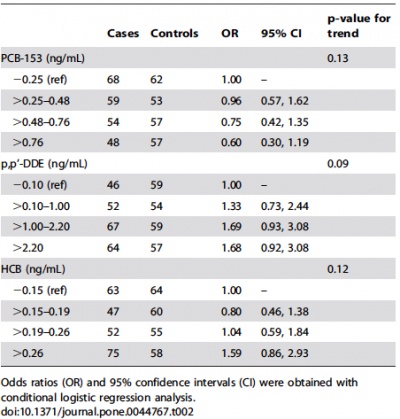
'' | '''A Nested Case-Control Study of Intrauterine Exposure to Persistent Organochlorine Pollutants and the Risk of Hypospadias (2012)'''<ref name=PMID23028613><pubmed>23028613</pubmed></ref> | ||
Hypospadias are a common congenital abnormality resulting from the failure of fusion of the urethral folds within the fetal period of development (8th-14th week of gestation). Whilst it is known that sexual differentiation of the male external genitalia depends on testosterone and its conversion into dihydrotestosterone, the risk factors of hypospadias are not thoroughly researched. This article aims to examine environmental exposure to endocrine disrupting chemicals (EDCs) and understand their potential to act as antagonists on androgen receptors, thereby disrupting the hormonal balance of the endocrine system ultimately leading to hypospadias.<ref name=PMID11469497><pubmed>11469497</pubmed></ref> | |||
More specifically, a major group of EDCs known as persistent organochlorine pollutants (POPs), for example, polychlorinated biphenyls, dioxins, pesticides (dichlorodiphenyl trichloroethane, DDT) and hexachlorobenzene (HCB). Such chemicals are lipophilic, resistant to biodegradation and are present throughout the hydrosphere and atmosphere. Although these chemicals were banned in the 1970’s and 1980’s, due to the potency of these chemicals, they are still found within humans<ref name=PMID22425898><pubmed>22425898</pubmed></ref> and are able to traverse the placenta, becoming exposed to the developing fetus<ref name=PMID6431068><pubmed>6431068</pubmed></ref>. | |||
Therefore, the aim of the present study was to investigate the linkage between exposures of the fetus to POPs and risk of developing hypospadias. | |||
This was a case-controlled study of the risk of hypospadias of single-born boys with regards to levels of POPs within the mothers’ blood during pregnancy. The study used 390 boys with hypospadias and controls were used. Boys with any cryptorchidisms, major malformations or even minor hypospadias were excluded from the study. | |||
== | This study concluded that EDCs such as PCBs, p,p’-DDE and HCB possess the potential to alter hormonal levels and affect the fetus, resulting hypospadias. It shows that in uterine exposure to HCB and possibly p,p’-DDE are risk factors and may affect androgen-signaling. | ||
|} | |||
---- | |||
{| | |||
|-bgcolor="aliceblue" | |||
| | |||
'''Paracetamol, aspirin, and indomethacin induce endocrine disturbances in the human fetal testis capable of interfering with testicular descent.''' '''(2013)'''<ref name=PMID24030937><pubmed>24030937</pubmed></ref> | |||
[[File:Male testosterone and AMH level graph.jpg|thumb|300px|right|Graph showing male testosterone and AMH levels]] | |||
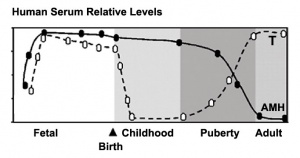
The differentiation of the gonads into male or female begins around week 6-8 and is linked to the sex-determining region of the Y chromosome. The formation and descent of the testis is determined by a number of hormones which are: | |||
*Anti-mullerian hormone (AMH) - produced by sertoli cells, which acts on the mullerian ducts. | |||
*Testosterone - produced by the fetal leydig cells, which ensures differentiation of wolffian ducts as well as the terminal phase of descent of the testis. | |||
*Insulin like factor 3 (INSL3) - is produced by the differentiated fetal leydig cells, which are involved in the transabdominal phase of descent. | |||
*Prostaglandins are also believed to be involved in the differentiation of the male genital tract and testis. | |||
The development of the male reproductive system requires the action of different hormones and is highly susceptible for development to be altered due to endocrine disruptions. | |||
Cryptorchidism is the failure of descent of the testis and is the most common congenital malformation in males. | |||
Non-steroidal anti-inflammatory drugs (NSAIDs) and paracetamol are some of the most widely used drugs used. These drugs have recently been identified as potential endocrine disruptors (ED) in humans. A number of epidemiological studies have reported that exposure to NSAIDs and analgesics during pregnancy showed an increased risk of cryptorchidism. | |||
This study used 62 fetuses from the first trimester between 7-12 weeks in gestation (GW) from pregnant women who obtained an abortion legally and with were given information and verbal consent was obtained according to national guidelines. The terminations were not motivated by abnormalities. The testes were cut in approximately 1mm3 pieces and drugs were used in the same concentration compared to recommended dosages in the body. The drugs used were paracetamol, aspirin, indomethacin, ketoconazole (antifungal). | |||
Testicular cells were counted using histology and image analysis and the hormones were assayed in the medium. | |||
The results showed no changes in the architecture of the testis with the analgesic treatment whereas the ketoconazole caused the boundaries of the testis cords to become unrecognisable. The analgesics did not significantly modify the number of germ cells or sertoli cells. | |||
Ketoconazole reduced testosterone levels in contrast to indomethacin, which stimulated testosterone production. Paracetamol had no significant effect on testosterone while aspirin produced a dose response relationship with an increase in testosterone after 72 hours in the youngest fetuses (8-9.86 GW) but not the older testes (10-12GW). None of the analgesics significantly affect the number of interstitial cells. | |||
The results showed a consistent trend for lower INSL3 production after 48-72 hours of exposure to mild analgesics and ketoconazole. This was the first study to measure direct production of INSL3 by the testis. | |||
Aspirin strongly stimulated AMH production, whereas as paracetamol and indomethacin increased production but not significantly. The analgesics did not significantly alter the sertoli cells and Ketoconazole significantly inhibited AMH production. Aspirin and paracetamol showed significant inhibition of Prostaglandin E2 production while indomethacin had no effect. | |||
In conclusion, the study shows that painkillers have a direct effect on various hormones, which are crucial for endocrine function and development of the human testis. The study shows that there is a direct effect with the dosages which are currently found with most medications. | |||
|} | |||
---- | |||
{| | |||
|-bgcolor="aliceblue" | |||
| | |||
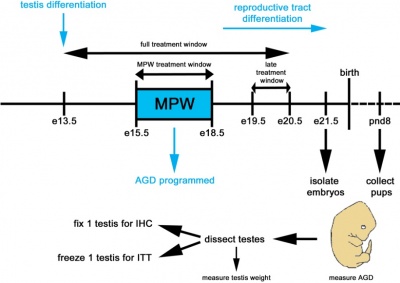
'''The Effect of Dihydrotestosterone Exposure During or Prior to the Masculinisation Programming Window on Reproductive Development in Male and Female Rats (2012)'''<ref name=PMID22248293><pubmed>22248293</pubmed></ref> | |||
[[File: | [[File:Schematic representation of the various treatment windows and experimental design..jpg|400px|thumb|left|Schematic representation of the various treatment windows and experimental design]] | ||
Whilst it is evident that some of the most common reproductive abnormalities in males occurs within the fetal stage, it is believed that disorders that affect young men later in life (such as low sperm count or testicular cancer) could also occur within this stage. Previous studies have demonstrated a critical period (masculinisation programming window - MPW) during fetal development that must transpire in order for masculinisation of the male fetus to occur. If the androgenic pathways do not occur in the correct manner, testicular dysgenisis (TD) may result. Although androgen production can be difficult to measure in humans, it can be measured by examining the anogenital distance, which is shorter in females versus males. | |||
Therefore, it can be established that the MPW is important in setting up normal male development and it is triggered to open by either the presence or absence of both androgens and their receptors. The aim of the present study was to investigate whether or not availability of excess androgens available to their receptors before or during the MPW could increase masculinisation and therefore, development of male and female rats. | |||
This study uses the highly potent dihydrotestosterone (DHT) androgen in comparison to other studies, which used testosterone, with the potential to convert to oestradiol and cause pregnancy disorders. The study used pregnant Wister rats and exposed them to DHT in two time frames: either before the MPW or during it. Relevant tissues were then extracted from the animals and certain procedures such as spectrophotometry, immunohistochemistry for AR proteins and RNA analysis were used to measure masculinisation and obtain results. | |||
Results showed that exposing male fetuses to a dosage of DHT that would result in masculinisation of the female fetus had no effect on the male. More specifically, the results indicated that exposure to DHT before or during the MPW did not stimulate any male reproductive development, and female masculinisation may begin much prior to the MPW. The results also support previous evidence demonstrating the potential for testosterone and oestrogen treatment to cause adverse pregnancy effects. | |||
Overall, the study concludes that genital development in male rats is not enhanced by DHT exposure before or during the MPW and that sensitivity of the female fetus to androgens can not only be refined to occurring during the MPW but also prior to. This has implications in that the female fetus is more susceptible to androgens and masculinisation of the genital system much before the male is, therefore suggesting a wider window. | |||
|} | |||
====Female==== | ====Female==== | ||
---- | |||
= | {| | ||
|-bgcolor="lavenderblush" | |||
< | | | ||
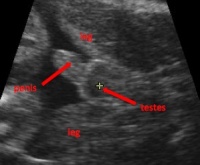
<pubmed> | '''Female External Genitalia on Fetal Magnetic Resonance Imaging (2011)'''<ref name=PMID21584884><pubmed>21584884</pubmed></ref> | ||
In the past, magnetic resonance imaging (MRI) has been used in conjunction with ultrasound to diagnose prenatal complications, including urogenital abnormalities. However, no previous research yielded any data regarding normal development of female external genitalia using MRI. As a result, this study investigated labial growth and correlated it with gestational age using prenatal MRI. | |||
[[File:Ultrasound male.jpg|200px|thumb|left|Ultrasound of male fetus]] | |||
This study included fetal MRI results from 197 female fetuses of Caucasian background, with either normal anatomy or minor congenital abnormalities. Fetuses with major congenital abnormalities, especially with urogenital abnormalities were omitted from the study. The MRI results were used to confirm if suspected anomalies during ultrasound screening were correct. Axial and coronal images of the fetus’ lower body were used to visualise the external female genitalia, including labia and clitoris and statistical analyses were performed on all MR images. | |||
Results showed a linear relationship between bilabial diameter and gestational age, and the morphology on the MRI showed a statistically significant difference between the 20-23 weeks age group and the rest (24-36 weeks) in the visual differentiation of the clitoris and the labial structures. Between 20-23 weeks, differentiation of the clitoris from the labia was not possible. Similar to what can be identified in ultrasounds, from 24 weeks onwards, in 12% of fetuses the clitoris and labia could be differentiated as 3-5 protuberances emerging from the pelvis, with the clitoris lying in the midline. | |||
= | The results are important as they demonstrate the MRI’s potential to be used in adjunct to ultrasound in order to assist in the diagnosis of certain genital abnormalities, such as hypospadias or micropenis. This is necessary as such conditions can mimic female external genitalia and so hypospadias especially should be determined on the basis of parallel labial lines and not exclusively on the direction of the genital tubercle.<ref name=PMID18431748><pubmed>18431748</pubmed></ref> | ||
In conclusion, this study examines the morphological development of the female external genitalia in utero using MRI, proving its effectiveness as a visualiser of the female phenotype and diagnosis of genital abnormalities, and should be used in conjunction with ultrasound. | |||
|} | |||
---- | |||
{| | |||
|-bgcolor="lavenderblush" | |||
| | |||
'''In humans, early cortisol biosynthesis provides a mechanism to safeguard female sexual development (2011)'''<ref name=PMID16585961><pubmed>16585961</pubmed></ref> | |||
As sexual differentiation of the external genitalia is an event that occurs within the fetal period, it is highly important to maintain the correct intrauterine environment in terms of hormonal content. In humans, differentiation and growth of the external genitalia is triggered by the release of androgens from the fetal testis such as dihydrotestosterone. Exposure of the female fetus to these androgens results in the female developing more male sexual characteristics at birth. This results from congenital adrenal hyperplasia, due to a deficiency of cytochrome P450 21-hydroxylase (CYP21), an enzyme involved in the biosynthesis of cortisol. | |||
A deficiency of CYP21 results in a decrease in cortisol levels, and this is believed to alleviate negative feedback at the fetal anterior pituitary. As a result, increased adrenocorticotropic hormone (ACTH) shifts steroid precursor formation towards androgen biosynthesis and therefore a balance between cortisol biosynthesis and androgen production is important for normal female external genitalia development. Therefore, this article illustrates the potential of utilising early cortisol biosynthesis to uphold normal female sexual development. | |||
The study used gas chromatography and mass spectrometry to observe a 9-18 fold increase in cortisol levels within the adrenal gland during the first trimester. The capacity of the adrenal gland in the fetus to secrete androgens was also determined using assays. | |||
By the time differentiation of the external genitalia occurs in the second trimester, the female fetus is well protected by high levels of placental aromatase enzymes, which convert androgens to oestrogens. Also, in order to prevent virilisation in CYP21 deficiency, dexamethasone needs to be administered at week 6. | |||
|} | |||
---- | |||
{| | |||
|-bgcolor="lavenderblush" | |||
| | |||
'''Fibrillin-3 in the Fetal Ovary: Can it Contribute to Polycystic Ovary Syndrome? (2012)'''<ref name= Abbott>Abbott David, H. '''Fibrillin-3 in the fetal ovary: can it contribute to polycystic ovary syndrome?'''. Expert Review of Endocrinology & Metabolism: 2012, 7(1); 31-34</ref> | |||
Fibrillin-3 contributes to microfibril formation within the extracellular matrix of many mammals and is predominantly expressed during fetal life<ref name=PMID20970500><pubmed>20970500</pubmed></ref>. The ovary continuously remodels its stroma in order to accommodate the constantly growing follicles from fetal life through to adult life, resulting in a continuously changing extracellular matrix, and therefore, fibrillin-3. Recent studies have discovered a linkage between an allele of the fibrillin-3 gene and polycystic ovary syndrome (PCOS), where hyperandrogenic interactions result in an enlarged and hyperstromal ovary with follicles that fail to mature and get released. | |||
This study obtained 29 samples of ovarian tissue from humans in first or second trimester fetuses and 6 non-PCOS adults. Fetal bovine ovaries were also obtained. mRNA expression analyses were performed, as well as PCR and indirect immunofluorescence immunochemistry. | |||
Results showed that in both human and bovine ovaries, fibrillin-3 mRNA is mostly expressed during the first trimester, with little to none being expressed in the adult ovaries. Localised expression of fibrillin-3 surrounding primordial and primary follicles results in fetal oocyte and adult follicle expansion within the stroma, as the ECM remodeling is necessary to support the growth of these follicles. Therefore, the fibrillin-3 gene in PCOS women displays potential for altering fetal ovarian follicle development, and since it is expressed in ECM throughout the fetus, it may result in altered development in non-ovarian organ systems in human fetuses. | |||
In conclusion, the study suggests that since the stroma within the ovaries is hyper developed in PCOS women, different alleles of the same fibrillin-3 gene could be expressed within the fetal ovary to overcommit the polycystic ovary to follicular growth that is unlikely to mature into preovulatory follicles. | |||
|} | |||
---- | |||
== | {| | ||
|-bgcolor="lavenderblush" | |||
| | |||
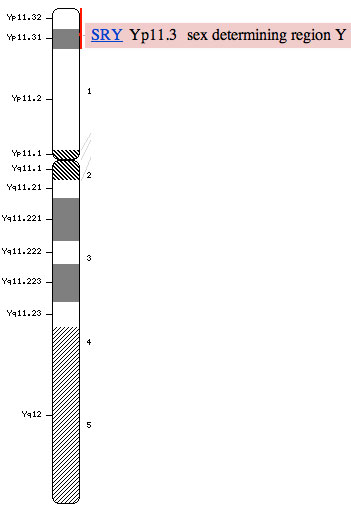
'''Expression of miRNAs in Ovine Fetal Gonads: Potential Role in Gonadal Differentiation (2011)'''<ref name=PMID21223560><pubmed>21223560</pubmed></ref> | |||
Genotype of sex is determined at the time of fertilisation, where a sperm carrying X or Y genetic material will fertilise an XX ova. This genotype that results then determines whether the genital ridge in the embryonic period will develop into the fetal testis (XY) or fetal ovaries (XX). The pathway involving testicular development includes a fine balance between genes that promote testis development and simultaneously genes that prevent ovarian development <ref name=PMID19027189><pubmed>19027189</pubmed></ref> | |||
[[File:Human Y chromosome SRY region.jpg|400px|right|thumb| Human Y chromosome showing SRY gene]] | |||
==== | Some critical genes involved in the testicular and ovarian pathways include: | ||
*SRY gene (sex-determining region of the Y-chromosome) <ref name=PMID2247149><pubmed>2247149</pubmed></ref> | |||
*Rspol gene (R-spondin homolog)<ref name=PMID18250098><pubmed>18250098</pubmed></ref> | |||
*Wnt4 – wongless-related MMTV integration site 4)<ref name=PMID18250097><pubmed>18250097</pubmed></ref> | |||
*Beta-catenin<ref name=PMID18250098><pubmed>18250098</pubmed></ref> | |||
Such genes are expressed in the support cells of the fetal gonads, for example, the Sertoli cells in the testis and the granulosa cells in the ovary.<ref name=PMID18250098><pubmed>18250098</pubmed></ref> | |||
Small non-coding RNA molecules, called miRNAs are RNAs that regulate gene expression and function within many different tissue types. Whilst studies have shown that miRNAs are important for growth and development of the gonads, none have yet indicated which miRNAs. | |||
Not much is known regarding the expression of miRNAs during fetal genital development in mammals and the purpose of this study was to identify this expression of miRNAs using the ovine as a model. Expression levels were examined and the importance of such research is to provide further understanding of human genital development on a genetic level, as well as the reproductive development of ovine, which may have economical implications as livestock. | |||
This study used sheep breeding methods and collected fetal gonads, which then underwent PCR genotyping. RNA was isolated, and miRNAs were treated with reverse transcriptase and then hybridised. These techniques were all used to detect expression levels of the relevant genes. | |||
From the study, it is evident that miRNAs are indeed present during fetal genital development in sheep. It is believed that miRNAs are important regulators of gene expression and function and based upon the results, the genes Let7 and miR-22 regulate oestrogen signaling during fetal genital development. Further, miR-22 may be needed for suppression of the oestrogen-signaling pathway during fetal development of the testes, as localisation of the gene in the testicular cords suggested that Sertoli cell development required such suppression of the oestrogen-signaling pathway. | |||
|} | |||
---- | |||
''' | {| class="wikitable mw-collapsible mw-collapsed" | ||
! '''Other current research findings and interesting reads:''' | |||
|- | |||
| * A recent study investigated the ability of in vitro cultures of female fetal mouse gonads to subsequently develop in vivo. It demonstrated that premeiotic germ cells in fetal gonads possessed the capability to develop into mature oocytes using this method. <ref name=PMID19379463><pubmed>19379463</pubmed></ref>Additionally, the study showed that the longer a culture of fetal gonads was kept (>14 days), follicular and development and oocyte growth in vivo was affected, as well as the maturation of the oocytes in vitro following transplantation into kidney capsules (the capsules are an ectopic site, however have all the necessary conditions for growth of the oocytes). <ref name=PMID8882299><pubmed>8882299</pubmed></ref> | |||
<pubmed>21584884</pubmed> | |||
<pubmed>18367374</pubmed> | |||
<pubmed>15086026</pubmed> | |||
<pubmed>14641326</pubmed> | |||
<pubmed>11684660</pubmed> | |||
<pubmed>22127979</pubmed> | |||
<pubmed>24631756</pubmed> | |||
<pubmed>23192465</pubmed> | |||
|} | |||
---- | |||
-- | |||
==Historic Findings== | |||
Click [https://embryology.med.unsw.edu.au/embryology/index.php/2014_Group_Project_9 here] for Historical Findings | |||
==Abnormalities== | |||
Click [https://embryology.med.unsw.edu.au/embryology/index.php/2014_Group_Project_9 here] for foetal genital abnormalities | |||
< | ==References== | ||
<references/> | |||
Latest revision as of 23:19, 24 October 2014
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Genital
Introduction
Genital system development occurs in both the embryonic and foetal phase of development. By the commencement of the foetal period, sexual determination and initial growth of the different gonads occur [1]. It is within the foetal period that the internal and external genital organs develop [1]. This page focuses on the foetal developmental processes, exploring the current and historical models and understanding, alongside the congenital abnormalities.
Today there is an innumerable amount of research articles focused on foetal genital growth, with some addressing the system as a whole [2] and others investigating certain genital organs [3]. Current models and understandings have been obtained from both human [4] and animal populations [3], and this page will outline some of the important current research.
Research into foetal genital development can be traced to as early as the 16th century[5] with anatomists proposing theories and constructing models, proven to being critical in obtaining the in-depth information known today. This page will mention some of these historical findings in both the female and male genital system.
This project will end with discussing some of the congenital abnormalities of the genital system, mentioning both the malformations that are most common [6] and those that are rare [7].
System Development
Timeline of Embryonic, Fetal and Postnatal Genital Development[8]
| Week | Development |
| 3-4 | Primordial germ cells migrate during gastrulation |
| 4 | Intermediate mesoderm, pronephros primordium |
| 5 | Mesonephros and mesonephric duct |
| 6 | Ureteric bud, metanephros, genital ridge |
| 7 | Cloacal divison, gonadal primordium - indifferent to first appearance of testis cords
Female - Paramesonephric duct preservation or regression begins |
| 8 | Paramesonephric duct, clear gonadal differentiation |
| 9 | Paramesonephric duct fusion in the female forming the uterus and lack of fusion laterally forming the fallopian tubes. Also forms vagina. |
| 15 | Primary follicles (ovary)
From the 26th week (between week 4 and 5), the gubernaculum starts to pull the testes down and results in descent of the testes into the scrotal sac. Both male and female gonads undergo descent. |
| Puberty | Development of secondary sexual characteristics
Female - start of menstruation, first egg released. Male - development of course voice, body hair and sperm formation. |
Related video
<html5media>https://www.youtube.com/watch?v=MureNA-RSZM</html5media>
Development of Internal Genitalia
| MALE | FEMALE |
| The fetal development of internal genitalia is largely dependent on the endocrine functions of the fetal testes. The fetal testes produce masculinizing hormones such as testosterone which begins its release from the interstitial Leydig cells of the primitive seminiferous tubules during the 8th week of development, and also the release of Mullerian Inhibiting Hormone (MIS) which is released at the sixth and seventh weeks by the Sertoli Cells. Testosterone acts primarily on the mesonephric ducts to stimulate the formation of the male genital ducts, whereas the MIS acts on the paramesonephric duct to stimulate its regression. [4] [9]
By the eighth week of fetal development in XY embryos, the testosterone produced in the testes results in the convolution of the proximal ends of the mesonephric ducts to form the epididymis [9]. The mesonephros begins to degenerate, however some of the mesonephric tubules remain and develop into efferent ductules, which then open into the duct of the epididymis. Distal to this end, the mesonephric duct begins to develop a thick lining of smooth muscle and progresses to become the ductus deferens.[2]
Fetal Male Glandular Development
|
The absence of testosterone production in XX embryos results in the regression of the mesonephric duct, and conversely the absence of MIH results in the development and progression of the paramesonephric ducts. The paramesonephric ducts give rise to most of the female internal genital system- the unfused cranial aspects of the ducts give rise to a primitive fallopian tubes, whilst the caudally fused portions form the uterovaginal primordium which will develop into a uterus and superior vagina. The splanchnic mesenchyme gives rise to the endometrial stromal tissue and the myometrium.[4]
|
Development of the External Genitalia
| Embryonic Period – fertilisation to end of 8th week (embryonic age) = AMBISEXUAL STAGE |
|---|
|
The current model for fetal development of the external genitalia in humans and mice:
| MALE MODEL | FEMALE MODEL |
Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION
|
Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION
|
Descent of the Gonads
Male: The testis undergo transabdominal and transinguinal descent. The testis lies in the subserous fascia. The processus vaginalis evaginates into the scrotum and the gubernaculum draws it into the scrotal sac. As it descends, it traverses past the superficial (external oblique fascia) and deep (transversalis fascia) inguinal rings of the inguinal canal. This begins in between the 4th and 5th week (usually around day 26) and spans over many days. It can occur unilaterally or bilaterally, more common in premature babies and can conclude postnatally.[10]
Female: ovaries undergo caudal and lateral shifts to be suspended in the broad ligament of the uterus. The gubernaculum does not shorten, attaches to paramesonephric ducts and causes medial translocation into the pelvis. The remnant in adult life of the gubernaculum is the ovarian and round ligament of the uterus, which suspend the ovaries and uterus respectively.[11]
Current Research, Models and Findings
Current Models
Most current research uses mouse models and observes the development of their external genitalia, especially their penile development, which initially appears to be different to human development. However, more microscopic inspection shows that mice have very similar external genitalia and are therefore appropriate animal models for observing such fetal development. As a result, mutant mouse models can effectively be used in future research to observe molecular mechanisms underlying hypospadias and their aetiology. [2]
When observing the morphology and cell biology of the developing testis, it is important to note that most of the research conducted on the subject involves the use of mouse models as a result of a lack of human subjects. It can be assumed that events in the human embryo correspond to the same events in the mouse embryo, however there are some differences between the time course of certain events and anatomy. [12]
Some examples of different animal models used in research involving fetal development of male and female genital systems are:
Different models are used for different research topics; for example, research involving Polycystic ovary syndrome (PCOS) uses murine models preferable due to the developmental time frame that allows studies of inherited PCOS to be examined within an appropriate time frame. The sheep model is also beneficial to use for PCOS research as it is cost effective and their size allows them to be subjected to certain procedures such as ultrasound and neurotransmitter measures. Primates are examples of an optimal model however are limited in their accessibility and long time frame of development.[16]
It is important to note that any findings associated with an animal model should be translated appropriately to the human model, as animals differ in their anatomy and regulatory mechanisms, as well as placentation. That is, sheep, rats and mice display different placentation and ovarian development that occurs in utero in sheep, primates and humans differs to the ex utero development in murine models.[16]
All animal experiments must be performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. [| Guide for the Care and Use of Laboratory Animals]
Current Research and Findings
Male
|
Extensive research into organogenesis of the external genitalia, mainly in males, is driven by the increasing incidence of hypospadias. Hypospadias are a result of the defect of fusion of the urethral folds of the lower part of the penis to fold and form the tubular penile urethra. The result of this in humans is the presence of an abnormal ventral urethral meatus, incomplete formation of the prepuce and an abnormal penile curvature.[2] Development of the male external genitalia, which occurs in the fetal period of development, is androgen dependent and involves epithelial-mesenchymal interactions. Because of these interactions, which are very similar to limb development, research into the development of genital tubercle has utilised similar methods for both processes. A minority of hypospadias cases are a result of the androgenic pathways being impaired and causing this congenital defect. The cell-cell interactions that allow for the development of the male external genitalia are mediated by a broad range of signaling molecules and growth factors such as fibroblast growth factors (FGFs), Sonic hedgehog (SHH) and bone morphogenetic proteins (BMPs). Such signaling and growth factors are downstream of androgen receptor signaling and an understanding of the mechanisms that underlie normal penile development during the fetal period, will lead to a deeper understanding of the aetiology of hypospadias.[2] |
|
A Nested Case-Control Study of Intrauterine Exposure to Persistent Organochlorine Pollutants and the Risk of Hypospadias (2012)[19] Hypospadias are a common congenital abnormality resulting from the failure of fusion of the urethral folds within the fetal period of development (8th-14th week of gestation). Whilst it is known that sexual differentiation of the male external genitalia depends on testosterone and its conversion into dihydrotestosterone, the risk factors of hypospadias are not thoroughly researched. This article aims to examine environmental exposure to endocrine disrupting chemicals (EDCs) and understand their potential to act as antagonists on androgen receptors, thereby disrupting the hormonal balance of the endocrine system ultimately leading to hypospadias.[20] More specifically, a major group of EDCs known as persistent organochlorine pollutants (POPs), for example, polychlorinated biphenyls, dioxins, pesticides (dichlorodiphenyl trichloroethane, DDT) and hexachlorobenzene (HCB). Such chemicals are lipophilic, resistant to biodegradation and are present throughout the hydrosphere and atmosphere. Although these chemicals were banned in the 1970’s and 1980’s, due to the potency of these chemicals, they are still found within humans[21] and are able to traverse the placenta, becoming exposed to the developing fetus[22]. Therefore, the aim of the present study was to investigate the linkage between exposures of the fetus to POPs and risk of developing hypospadias. This was a case-controlled study of the risk of hypospadias of single-born boys with regards to levels of POPs within the mothers’ blood during pregnancy. The study used 390 boys with hypospadias and controls were used. Boys with any cryptorchidisms, major malformations or even minor hypospadias were excluded from the study. This study concluded that EDCs such as PCBs, p,p’-DDE and HCB possess the potential to alter hormonal levels and affect the fetus, resulting hypospadias. It shows that in uterine exposure to HCB and possibly p,p’-DDE are risk factors and may affect androgen-signaling. |
|
Paracetamol, aspirin, and indomethacin induce endocrine disturbances in the human fetal testis capable of interfering with testicular descent. (2013)[23] The differentiation of the gonads into male or female begins around week 6-8 and is linked to the sex-determining region of the Y chromosome. The formation and descent of the testis is determined by a number of hormones which are:
The development of the male reproductive system requires the action of different hormones and is highly susceptible for development to be altered due to endocrine disruptions. Cryptorchidism is the failure of descent of the testis and is the most common congenital malformation in males. Non-steroidal anti-inflammatory drugs (NSAIDs) and paracetamol are some of the most widely used drugs used. These drugs have recently been identified as potential endocrine disruptors (ED) in humans. A number of epidemiological studies have reported that exposure to NSAIDs and analgesics during pregnancy showed an increased risk of cryptorchidism. This study used 62 fetuses from the first trimester between 7-12 weeks in gestation (GW) from pregnant women who obtained an abortion legally and with were given information and verbal consent was obtained according to national guidelines. The terminations were not motivated by abnormalities. The testes were cut in approximately 1mm3 pieces and drugs were used in the same concentration compared to recommended dosages in the body. The drugs used were paracetamol, aspirin, indomethacin, ketoconazole (antifungal). Testicular cells were counted using histology and image analysis and the hormones were assayed in the medium. The results showed no changes in the architecture of the testis with the analgesic treatment whereas the ketoconazole caused the boundaries of the testis cords to become unrecognisable. The analgesics did not significantly modify the number of germ cells or sertoli cells. Ketoconazole reduced testosterone levels in contrast to indomethacin, which stimulated testosterone production. Paracetamol had no significant effect on testosterone while aspirin produced a dose response relationship with an increase in testosterone after 72 hours in the youngest fetuses (8-9.86 GW) but not the older testes (10-12GW). None of the analgesics significantly affect the number of interstitial cells. The results showed a consistent trend for lower INSL3 production after 48-72 hours of exposure to mild analgesics and ketoconazole. This was the first study to measure direct production of INSL3 by the testis. Aspirin strongly stimulated AMH production, whereas as paracetamol and indomethacin increased production but not significantly. The analgesics did not significantly alter the sertoli cells and Ketoconazole significantly inhibited AMH production. Aspirin and paracetamol showed significant inhibition of Prostaglandin E2 production while indomethacin had no effect. In conclusion, the study shows that painkillers have a direct effect on various hormones, which are crucial for endocrine function and development of the human testis. The study shows that there is a direct effect with the dosages which are currently found with most medications. |
|
The Effect of Dihydrotestosterone Exposure During or Prior to the Masculinisation Programming Window on Reproductive Development in Male and Female Rats (2012)[15] Whilst it is evident that some of the most common reproductive abnormalities in males occurs within the fetal stage, it is believed that disorders that affect young men later in life (such as low sperm count or testicular cancer) could also occur within this stage. Previous studies have demonstrated a critical period (masculinisation programming window - MPW) during fetal development that must transpire in order for masculinisation of the male fetus to occur. If the androgenic pathways do not occur in the correct manner, testicular dysgenisis (TD) may result. Although androgen production can be difficult to measure in humans, it can be measured by examining the anogenital distance, which is shorter in females versus males. Therefore, it can be established that the MPW is important in setting up normal male development and it is triggered to open by either the presence or absence of both androgens and their receptors. The aim of the present study was to investigate whether or not availability of excess androgens available to their receptors before or during the MPW could increase masculinisation and therefore, development of male and female rats. This study uses the highly potent dihydrotestosterone (DHT) androgen in comparison to other studies, which used testosterone, with the potential to convert to oestradiol and cause pregnancy disorders. The study used pregnant Wister rats and exposed them to DHT in two time frames: either before the MPW or during it. Relevant tissues were then extracted from the animals and certain procedures such as spectrophotometry, immunohistochemistry for AR proteins and RNA analysis were used to measure masculinisation and obtain results. Results showed that exposing male fetuses to a dosage of DHT that would result in masculinisation of the female fetus had no effect on the male. More specifically, the results indicated that exposure to DHT before or during the MPW did not stimulate any male reproductive development, and female masculinisation may begin much prior to the MPW. The results also support previous evidence demonstrating the potential for testosterone and oestrogen treatment to cause adverse pregnancy effects. Overall, the study concludes that genital development in male rats is not enhanced by DHT exposure before or during the MPW and that sensitivity of the female fetus to androgens can not only be refined to occurring during the MPW but also prior to. This has implications in that the female fetus is more susceptible to androgens and masculinisation of the genital system much before the male is, therefore suggesting a wider window. |
Female
|
Female External Genitalia on Fetal Magnetic Resonance Imaging (2011)[24] In the past, magnetic resonance imaging (MRI) has been used in conjunction with ultrasound to diagnose prenatal complications, including urogenital abnormalities. However, no previous research yielded any data regarding normal development of female external genitalia using MRI. As a result, this study investigated labial growth and correlated it with gestational age using prenatal MRI. This study included fetal MRI results from 197 female fetuses of Caucasian background, with either normal anatomy or minor congenital abnormalities. Fetuses with major congenital abnormalities, especially with urogenital abnormalities were omitted from the study. The MRI results were used to confirm if suspected anomalies during ultrasound screening were correct. Axial and coronal images of the fetus’ lower body were used to visualise the external female genitalia, including labia and clitoris and statistical analyses were performed on all MR images. Results showed a linear relationship between bilabial diameter and gestational age, and the morphology on the MRI showed a statistically significant difference between the 20-23 weeks age group and the rest (24-36 weeks) in the visual differentiation of the clitoris and the labial structures. Between 20-23 weeks, differentiation of the clitoris from the labia was not possible. Similar to what can be identified in ultrasounds, from 24 weeks onwards, in 12% of fetuses the clitoris and labia could be differentiated as 3-5 protuberances emerging from the pelvis, with the clitoris lying in the midline. The results are important as they demonstrate the MRI’s potential to be used in adjunct to ultrasound in order to assist in the diagnosis of certain genital abnormalities, such as hypospadias or micropenis. This is necessary as such conditions can mimic female external genitalia and so hypospadias especially should be determined on the basis of parallel labial lines and not exclusively on the direction of the genital tubercle.[25] In conclusion, this study examines the morphological development of the female external genitalia in utero using MRI, proving its effectiveness as a visualiser of the female phenotype and diagnosis of genital abnormalities, and should be used in conjunction with ultrasound. |
|
In humans, early cortisol biosynthesis provides a mechanism to safeguard female sexual development (2011)[26] As sexual differentiation of the external genitalia is an event that occurs within the fetal period, it is highly important to maintain the correct intrauterine environment in terms of hormonal content. In humans, differentiation and growth of the external genitalia is triggered by the release of androgens from the fetal testis such as dihydrotestosterone. Exposure of the female fetus to these androgens results in the female developing more male sexual characteristics at birth. This results from congenital adrenal hyperplasia, due to a deficiency of cytochrome P450 21-hydroxylase (CYP21), an enzyme involved in the biosynthesis of cortisol. A deficiency of CYP21 results in a decrease in cortisol levels, and this is believed to alleviate negative feedback at the fetal anterior pituitary. As a result, increased adrenocorticotropic hormone (ACTH) shifts steroid precursor formation towards androgen biosynthesis and therefore a balance between cortisol biosynthesis and androgen production is important for normal female external genitalia development. Therefore, this article illustrates the potential of utilising early cortisol biosynthesis to uphold normal female sexual development. The study used gas chromatography and mass spectrometry to observe a 9-18 fold increase in cortisol levels within the adrenal gland during the first trimester. The capacity of the adrenal gland in the fetus to secrete androgens was also determined using assays. By the time differentiation of the external genitalia occurs in the second trimester, the female fetus is well protected by high levels of placental aromatase enzymes, which convert androgens to oestrogens. Also, in order to prevent virilisation in CYP21 deficiency, dexamethasone needs to be administered at week 6. |
|
Fibrillin-3 in the Fetal Ovary: Can it Contribute to Polycystic Ovary Syndrome? (2012)[27] Fibrillin-3 contributes to microfibril formation within the extracellular matrix of many mammals and is predominantly expressed during fetal life[28]. The ovary continuously remodels its stroma in order to accommodate the constantly growing follicles from fetal life through to adult life, resulting in a continuously changing extracellular matrix, and therefore, fibrillin-3. Recent studies have discovered a linkage between an allele of the fibrillin-3 gene and polycystic ovary syndrome (PCOS), where hyperandrogenic interactions result in an enlarged and hyperstromal ovary with follicles that fail to mature and get released. This study obtained 29 samples of ovarian tissue from humans in first or second trimester fetuses and 6 non-PCOS adults. Fetal bovine ovaries were also obtained. mRNA expression analyses were performed, as well as PCR and indirect immunofluorescence immunochemistry. Results showed that in both human and bovine ovaries, fibrillin-3 mRNA is mostly expressed during the first trimester, with little to none being expressed in the adult ovaries. Localised expression of fibrillin-3 surrounding primordial and primary follicles results in fetal oocyte and adult follicle expansion within the stroma, as the ECM remodeling is necessary to support the growth of these follicles. Therefore, the fibrillin-3 gene in PCOS women displays potential for altering fetal ovarian follicle development, and since it is expressed in ECM throughout the fetus, it may result in altered development in non-ovarian organ systems in human fetuses. In conclusion, the study suggests that since the stroma within the ovaries is hyper developed in PCOS women, different alleles of the same fibrillin-3 gene could be expressed within the fetal ovary to overcommit the polycystic ovary to follicular growth that is unlikely to mature into preovulatory follicles. |
|
Expression of miRNAs in Ovine Fetal Gonads: Potential Role in Gonadal Differentiation (2011)[13] Genotype of sex is determined at the time of fertilisation, where a sperm carrying X or Y genetic material will fertilise an XX ova. This genotype that results then determines whether the genital ridge in the embryonic period will develop into the fetal testis (XY) or fetal ovaries (XX). The pathway involving testicular development includes a fine balance between genes that promote testis development and simultaneously genes that prevent ovarian development [29] Some critical genes involved in the testicular and ovarian pathways include:
Such genes are expressed in the support cells of the fetal gonads, for example, the Sertoli cells in the testis and the granulosa cells in the ovary.[31] Small non-coding RNA molecules, called miRNAs are RNAs that regulate gene expression and function within many different tissue types. Whilst studies have shown that miRNAs are important for growth and development of the gonads, none have yet indicated which miRNAs. Not much is known regarding the expression of miRNAs during fetal genital development in mammals and the purpose of this study was to identify this expression of miRNAs using the ovine as a model. Expression levels were examined and the importance of such research is to provide further understanding of human genital development on a genetic level, as well as the reproductive development of ovine, which may have economical implications as livestock. This study used sheep breeding methods and collected fetal gonads, which then underwent PCR genotyping. RNA was isolated, and miRNAs were treated with reverse transcriptase and then hybridised. These techniques were all used to detect expression levels of the relevant genes. From the study, it is evident that miRNAs are indeed present during fetal genital development in sheep. It is believed that miRNAs are important regulators of gene expression and function and based upon the results, the genes Let7 and miR-22 regulate oestrogen signaling during fetal genital development. Further, miR-22 may be needed for suppression of the oestrogen-signaling pathway during fetal development of the testes, as localisation of the gene in the testicular cords suggested that Sertoli cell development required such suppression of the oestrogen-signaling pathway. |
| Other current research findings and interesting reads: |
|---|
| * A recent study investigated the ability of in vitro cultures of female fetal mouse gonads to subsequently develop in vivo. It demonstrated that premeiotic germ cells in fetal gonads possessed the capability to develop into mature oocytes using this method. [33]Additionally, the study showed that the longer a culture of fetal gonads was kept (>14 days), follicular and development and oocyte growth in vivo was affected, as well as the maturation of the oocytes in vitro following transplantation into kidney capsules (the capsules are an ectopic site, however have all the necessary conditions for growth of the oocytes). [34]
<pubmed>21584884</pubmed> <pubmed>18367374</pubmed> <pubmed>15086026</pubmed> <pubmed>14641326</pubmed> <pubmed>11684660</pubmed> <pubmed>22127979</pubmed> <pubmed>24631756</pubmed> <pubmed>23192465</pubmed> |
Historic Findings
Click here for Historical Findings
Abnormalities
Click here for foetal genital abnormalities
References
- ↑ 1.0 1.1 Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 <pubmed>14641326</pubmed>
- ↑ 3.0 3.1 3.2 <pubmed>13362960</pubmed>
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 <pubmed>11315960</pubmed>
- ↑ <pubmed>18462432</pubmed>
- ↑ <pubmed>16006950</pubmed>
- ↑ <pubmed>23635766</pubmed>
- ↑ Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development
- ↑ 9.0 9.1 9.2 <pubmed>24240231</pubmed>
- ↑ Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development
- ↑ Hill, M.A. (2014) Lecture - Genital Development. Retrieved October 24, 2014, from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Genital_Development
- ↑ <pubmed>17237341</pubmed>
- ↑ 13.0 13.1 <pubmed>21223560</pubmed>
- ↑ <pubmed>23571006</pubmed>
- ↑ 15.0 15.1 <pubmed>22248293</pubmed>
- ↑ 16.0 16.1 16.2 <pubmed>21710394</pubmed>
- ↑ <pubmed>20347535</pubmed>
- ↑ <pubmed>22006251</pubmed>
- ↑ <pubmed>23028613</pubmed>
- ↑ <pubmed>11469497</pubmed>
- ↑ <pubmed>22425898</pubmed>
- ↑ <pubmed>6431068</pubmed>
- ↑ <pubmed>24030937</pubmed>
- ↑ <pubmed>21584884</pubmed>
- ↑ <pubmed>18431748</pubmed>
- ↑ <pubmed>16585961</pubmed>
- ↑ Abbott David, H. Fibrillin-3 in the fetal ovary: can it contribute to polycystic ovary syndrome?. Expert Review of Endocrinology & Metabolism: 2012, 7(1); 31-34
- ↑ <pubmed>20970500</pubmed>
- ↑ <pubmed>19027189</pubmed>
- ↑ <pubmed>2247149</pubmed>
- ↑ 31.0 31.1 31.2 <pubmed>18250098</pubmed>
- ↑ <pubmed>18250097</pubmed>
- ↑ <pubmed>19379463</pubmed>
- ↑ <pubmed>8882299</pubmed>