File:G-protein coupled receptors.jpg: Difference between revisions
No edit summary |
mNo edit summary |
||
| Line 1: | Line 1: | ||
==Signal Transduction in G-protein-coupled receptors== | |||
Diverse signalling pathways regulated by the type 2 beta adrenergic receptor (2AR). | |||
The 2AR can activate two G proteins, Gs and Gi (part of the Gs and Gi heterotrimers, respectively), which differentially regulate adenylate cyclase. Adenylate cyclase generates cyclic AMP (cAMP), which activates protein kinase A (PKA), a kinase that regulates the activity of several cellular proteins including the L-type Ca2+ channel and the 2AR. cAMP second messenger levels are downregulated by specific phosphodiesterase proteins (PDEs). | |||
Activation of the 2AR also leads to phosphorylation by a G-protein-coupled receptor kinase (GRK) and subsequent coupling to arrestin. Arrestin is a signalling and regulatory protein that promotes the activation of extracellular signal-regulated kinases (ERK), prevents the activation of G proteins and promotes the internalization of the receptor through clathrin-coated pits. PKC, protein kinase C. The inset shows classification of ligand efficacy for GPCRs. Many GPCRs exhibit basal, agonist-independent activity. Inverse agonists inhibit this activity, and neutral antagonists have no effect. Agonists and partial agonists stimulate biological responses above the basal activity. Efficacy is not directly related to affinity; for example, a partial agonist can have a higher affinity for a GPCR than a full agonist. | |||
===Reference=== | |||
<pubmed>19458711</pubmed> | |||
====Copyright==== | |||
Reprinted by Permission from Macmillan Publishers Ltd: from The structure and function of G-protein-coupled receptors. Rosenbaum DM, Rasmussen SG, Kobilka BK. Nature. 2009 May 21;459(7245):356-63. Review. | |||
Original file name: Nature08144-f1.2.jpg | |||
http://www.nature.com/nature/journal/v459/n7245/fig_tab/nature08144_F1.html | |||
http://www.nature.com/nature/journal/v459/n7245/full/nature08144.html | |||
Revision as of 10:58, 23 May 2016
Signal Transduction in G-protein-coupled receptors
Diverse signalling pathways regulated by the type 2 beta adrenergic receptor (2AR).
The 2AR can activate two G proteins, Gs and Gi (part of the Gs and Gi heterotrimers, respectively), which differentially regulate adenylate cyclase. Adenylate cyclase generates cyclic AMP (cAMP), which activates protein kinase A (PKA), a kinase that regulates the activity of several cellular proteins including the L-type Ca2+ channel and the 2AR. cAMP second messenger levels are downregulated by specific phosphodiesterase proteins (PDEs).
Activation of the 2AR also leads to phosphorylation by a G-protein-coupled receptor kinase (GRK) and subsequent coupling to arrestin. Arrestin is a signalling and regulatory protein that promotes the activation of extracellular signal-regulated kinases (ERK), prevents the activation of G proteins and promotes the internalization of the receptor through clathrin-coated pits. PKC, protein kinase C. The inset shows classification of ligand efficacy for GPCRs. Many GPCRs exhibit basal, agonist-independent activity. Inverse agonists inhibit this activity, and neutral antagonists have no effect. Agonists and partial agonists stimulate biological responses above the basal activity. Efficacy is not directly related to affinity; for example, a partial agonist can have a higher affinity for a GPCR than a full agonist.
Reference
<pubmed>19458711</pubmed>
Copyright
Reprinted by Permission from Macmillan Publishers Ltd: from The structure and function of G-protein-coupled receptors. Rosenbaum DM, Rasmussen SG, Kobilka BK. Nature. 2009 May 21;459(7245):356-63. Review.
Original file name: Nature08144-f1.2.jpg
http://www.nature.com/nature/journal/v459/n7245/fig_tab/nature08144_F1.html
http://www.nature.com/nature/journal/v459/n7245/full/nature08144.html
File history
Yi efo/eka'e gwa ebo wo le nyangagi wuncin ye kamina wunga tinya nan
| Gwalagizhi | Nyangagi | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 10:57, 23 May 2016 | 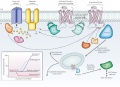 | 900 × 655 (109 KB) | Z8600021 (talk | contribs) |
You cannot overwrite this file.
File usage
The following page uses this file: