Talk:2018 Group Project 5: Difference between revisions
| Line 49: | Line 49: | ||
[[User:Z5229431|Z5229431]] ([[User talk:Z5229431|talk]]) 00:25, 28 August 2018 (AEST) | [[User:Z5229431|Z5229431]] ([[User talk:Z5229431|talk]]) 00:25, 28 August 2018 (AEST) | ||
[[File:Neural crest cell migration in erbb3b mutants.jpg]] | |||
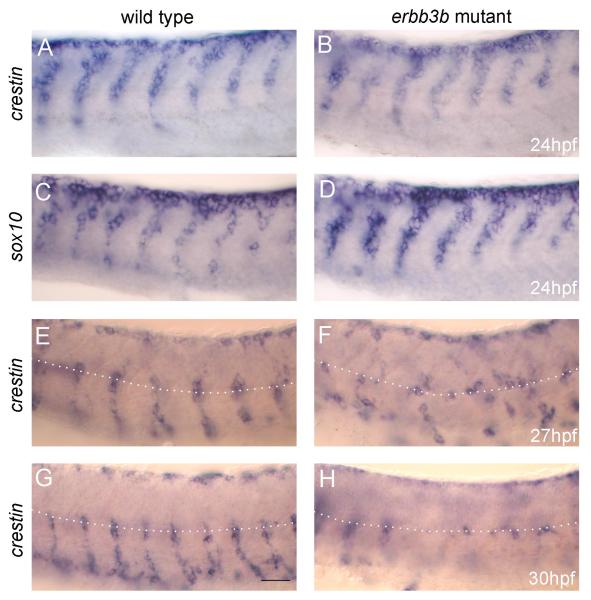
"Dorsal root ganglia (DRGs) arise from trunk neural crest cells that emerge from the dorsal neuroepithelium and coalesce into segmental streams that migrate ventrally along the developing somites. Proper formation of DRGs involves not only normal trunk neural crest migration, but also the ability of DRG progenitors to pause at a particular target location where they can receive DRG-promoting signals. In mammalian embryos, a receptor tyrosine kinase proto-oncogene, ErbB3, is required for proper trunk neural crest migration. Here, we show that in zebrafish mutants lacking ErbB3 function, neural crest cells do not pause at the location where DRGs normally form and DRG neurons are not generated. We also show that these mutants lack trunk neural crest-derived sympathetic neurons, but that cranial neural crest-derived enteric neurons appear normal. We isolated three genes encoding neuregulins, ErbB3 ligands, and show that two neuregulins function together in zebrafish trunk neural crest cell migration and in DRG formation. Together, our results suggest that ErbB3 signaling is required for normal migration of trunk, but not cranial, neural crest cells." | "Dorsal root ganglia (DRGs) arise from trunk neural crest cells that emerge from the dorsal neuroepithelium and coalesce into segmental streams that migrate ventrally along the developing somites. Proper formation of DRGs involves not only normal trunk neural crest migration, but also the ability of DRG progenitors to pause at a particular target location where they can receive DRG-promoting signals. In mammalian embryos, a receptor tyrosine kinase proto-oncogene, ErbB3, is required for proper trunk neural crest migration. Here, we show that in zebrafish mutants lacking ErbB3 function, neural crest cells do not pause at the location where DRGs normally form and DRG neurons are not generated. We also show that these mutants lack trunk neural crest-derived sympathetic neurons, but that cranial neural crest-derived enteric neurons appear normal. We isolated three genes encoding neuregulins, ErbB3 ligands, and show that two neuregulins function together in zebrafish trunk neural crest cell migration and in DRG formation. Together, our results suggest that ErbB3 signaling is required for normal migration of trunk, but not cranial, neural crest cells." | ||
Revision as of 01:44, 28 August 2018
| Projects 2018: 1 Adrenal Medulla | 3 Melanocytes | 4 Cardiac | 5 Dorsal Root Ganglion |
Project Pages are currently being updated (notice removed when completed)
About this Discussion Page
The project discussion page is where your group members can post discussion on the project topic. This will be demonstrated in the practical tutorial in week 3.
Please follow these 3 simple rules:
- Never identify yourself or any other students by name, use only your student number.
- Only edit your own student page or your own group project page.
- Only add content that is both correctly cited and you have permission to reuse.
| Group Assessment Criteria |
|---|
 Science Student Projects Science Student Projects
|
| More Information on Assessment Criteria | Science Student Projects |
|
|
|
Z5229399 (talk) 11:33, 14 August 2018 (AEST) Z5229597 (talk) 11:34, 14 August 2018 (AEST) Z5229431 (talk) 11:35, 14 August 2018 (AEST) Z5229549 (talk) 11:36, 14 August 2018 (AEST) Z5229438 (talk) 11:36, 21 August 2018 (AEST)
Introduction
- A good article for the overview of trunk neural crest cells
Vega-Lopez GA, Cerrizuela S & Aybar MJ. (2017). Trunk neural crest cells: formation, migration and beyond. Int. J. Dev. Biol. , 61, 5-15. PMID: 28287247 DOI.
Z5229431 (talk) 12:25, 24 August 2018 (AEST)
History
Embryonic Origins
Developmental Process
Adult Function
- Differentiation process
- When they start to function
Tissue / Organ structure
Molecular Mechanisms / Factors / Genes
"Blocking of CXCR4 by morpholino or shRNA in premigratory chick trunk neural crest cells leads to significantly fewer cells that reach the dorsal aorta and instead populate the dorsal root ganglia"
Kasemeier-Kulesa JC, McLennan R, Romine MH, Kulesa PM & Lefcort F. (2010). CXCR4 controls ventral migration of sympathetic precursor cells. J. Neurosci. , 30, 13078-88. PMID: 20881125 DOI.
Z5229431 (talk) 12:18, 24 August 2018 (AEST)
Abnormalities / Abnormal development
- Environmental factors
- Genetic mutations
- Images of how they look like
Animal Models
"In zebrafish, trunk NCCs start migrating along a medial pathway in-between the somites and the NT. These NCCs align to and are affected by slow muscle cells in the middle part of the somite"
Honjo Y & Eisen JS. (2005). Slow muscle regulates the pattern of trunk neural crest migration in zebrafish. Development , 132, 4461-70. PMID: 16162652 DOI.
Z5229431 (talk) 12:16, 24 August 2018 (AEST)
"Hedgehog (Hh) signal transduction is directly required in zebrafish DRG precursors for proper development of DRG neurons. Zebrafish mutations in the Hh signaling pathway result in the absence of DRG neurons and the loss of expression of neurogenin1 (ngn1), a gene required for determination of DRG precursors. Cell transplantation experiments demonstrate that Hh acts directly on DRG neuron precursors. Blocking Hh pathway activation at later stages of embryogenesis with the steroidal alkaloid, cyclopamine, further reveals that the requirement for a Hh signal response in DRG precursors correlates with the onset of ngn1 expression. These results suggest that Hh signaling may normally promote DRG development by regulating expression of ngn1 in DRG precursors."
Hedgehog signaling is directly required for the development of zebrafish dorsal root ganglia neurons. Josette M. Ungos, Rolf O. Karlstrom, David W. Raible. Development 2003 130: 5351-5362; doi: 10.1242/dev.00722
Z5229431 (talk) 00:25, 28 August 2018 (AEST)
"Dorsal root ganglia (DRGs) arise from trunk neural crest cells that emerge from the dorsal neuroepithelium and coalesce into segmental streams that migrate ventrally along the developing somites. Proper formation of DRGs involves not only normal trunk neural crest migration, but also the ability of DRG progenitors to pause at a particular target location where they can receive DRG-promoting signals. In mammalian embryos, a receptor tyrosine kinase proto-oncogene, ErbB3, is required for proper trunk neural crest migration. Here, we show that in zebrafish mutants lacking ErbB3 function, neural crest cells do not pause at the location where DRGs normally form and DRG neurons are not generated. We also show that these mutants lack trunk neural crest-derived sympathetic neurons, but that cranial neural crest-derived enteric neurons appear normal. We isolated three genes encoding neuregulins, ErbB3 ligands, and show that two neuregulins function together in zebrafish trunk neural crest cell migration and in DRG formation. Together, our results suggest that ErbB3 signaling is required for normal migration of trunk, but not cranial, neural crest cells."
Honjo Y, Kniss J & Eisen JS. (2008). Neuregulin-mediated ErbB3 signaling is required for formation of zebrafish dorsal root ganglion neurons. Development , 135, 2615-25. PMID: 18599505 DOI.
Z5229431 (talk) 00:37, 28 August 2018 (AEST)
"cdon is expressed in developing premigratory NCCs but is downregulated once the cells become migratory. Knockdown of cdon results in aberrant migration of trunk NCCs: crestin positive cells can emigrate out of the neural tube but stall shortly after the initiation of migration. Live cell imaging analysis demonstrates reduced directedness of migration, increased velocity and mispositioned cell protrusions. In addition, transplantation analysis suggests that cdon is required cell-autonomously for directed NCC migration in the trunk."
Powell DR, Williams JS, Hernandez-Lagunas L, Salcedo E, O'Brien JH & Artinger KB. (2015). Cdon promotes neural crest migration by regulating N-cadherin localization. Dev. Biol. , 407, 289-99. PMID: 26256768 DOI.
Z5229431 (talk) 00:45, 28 August 2018 (AEST)
Current research (Labs)
--Z5229438 (talk) 19:27, 27 August 2018 (AEST)
Glossary
- Brief, clear and concise