2018 Group Project 3
| Projects 2018: 1 Adrenal Medulla | 3 Melanocytes | 4 Cardiac | 5 Dorsal Root Ganglion |
Project Pages are currently being updated (notice removed when completed)
Melanocytes
Introduction
Melanocytes are a type of neural crest-derived cells in the body [1], most commonly found in the stratum basale, the bottom layer of the epidermis of the skin, eyes [2], and to a lesser known degree, the meninges (membrane around the brain), the heart [3] and the inner ear [4].
Each melanocyte possesses one main cell body, where most of the cellular functions, including the production of melanin, take place. They also possess finger-like projections that extend into the surrounding tissue, similar to neurons, called dendrites. Melanin produced within the cell is packaged, as melanosomes, before being transported to the surrounding cells via dendrites, which stretch in between the surrounding tissue.
Their most commonly known function is the production of melanin (melanogenesis), of which there are two types produced; eumelanin (black) and pheomelanin (reddish yellow) [5], that are responsible for the pigmentation of various parts of the human body including the skin, hair and irises, etc. Melanogenesis has different levels - basal and activated. The basal level of melanogenesis determines an individual's skin colour and is determined by genetics, i.e. a light-skinned individual has low levels of basal melanogenesis. Activated levels of melanogenesis are usually due to external factors, such as exposure to UV-B radiation, resulting in increased levels of melanogenesis [6], which can be commonly seen as sunburn or tan after long exposure to sunlight. Other cells in the body also contain pigments, such as epithelium of the retina, some neurons and adipocytes, but melanocytes are the only cell of neural crest origin that can produce them.
While melanocytes have long been discovered, not much research has been done on many of the populations of melanocytes, excluding the skin. As such, this page, while still covering some aspects of these areas, will mainly focus on the skin-based melanocytes.
History
The location of pigmentation in the skin was examined and commented on by various scientists in the 1600s [7]. In 1618, Jean Riolan described pigmentation in the "outer layer" of skin, that was not present in lower layers, or "true skin"[8]. It wasn't until the early 20th century that the dark granules responsible for this pigmentation, nowadays termed melanosomes, were first identified using light microscopy [9]. The origin of melanosome synthesis within the cell was discovered later in the century using electron microscopy: in 1961, Moyer found that Stage One melanosomes (those containing matrix material, but no melanin), in pigmented retina, originate from the endoplasmic reticulum of epithelial cells. These findings were later confirmed by Turner, who studied melanosomes in goldfish [10] [11].
The idea that melanocytes originate from neural crest cells was first suggested by Ross Granville Harrison in 1910, when writing a paper on the outgrowth of the nerve fibre [12]. This was later proven by DuShane in 1935: He removed sections of neural fold in stage 15 Amphibia punctatum and tigrinum embryos, and observed a lack of pigment in their trunk region. He then placed the neural fold tissue explants in Holtfreter’s solution (a solution designed to facilitate amphibian development), and observed their differentiation into melanocytes [13]. In an article published in 1963, James Weston describes using tritiated thymidine labelling to track chick embryo neural crest migration in vivo - a new method of cell tracing. Weston was able to identify a dorsolateral stream of neural crest derived melanoblasts (precursors to melanocytes), migrating to enter the ectoderm [14]. Later, in 1992, Weston made another breakthrough in proving the existence of fate-restricted melanocyte precursors in the pre-migratory neural crest lineage cells of quail embryos. This provided new information regarding the timings involved in neural crest lineage restriction [15].
In the current century, more research from various scientific groups is expanding our knowledge of melanocytes. Aside from the melanocytes of the skin, information has been gained regarding those in the meninges, ears, eyes and heart, some of which will be discussed later in the page.
Embryonic Origins
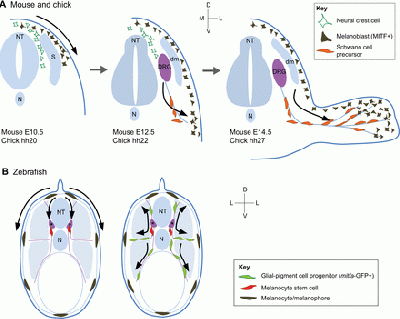
As mentioned earlier, melanocytes originate from the pluripotent cells in the neural crest specifically dorsolaterally migrating trunk neural crest cells in the dorsal ectoderm [1]. These eventually form part of the ears, eyes, skin and leptomeninges, therefore explaining the presence of melanin in these parts as a result of the development of melanoblast subpopulations during early migration [16].
Trunk Neural Crest
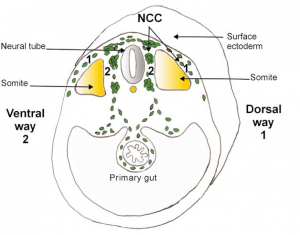
After formation of the neural tube, neural crest cells (NCCs) generated along the anterior-poster (AP) axis can be separated into four distinct segments: cardiac, cranial, vagal and trunk neural crest. It is from the trunk neural crest that melanoblasts, which will further differentiate into melanocytes, are derived. Trunk NCCs are derived from the caudal region of the embryo, and can migrate along three pathways [18]:
- Dorsalateral Pathway between the ectoderm and somites
- Ventro-lateral Pathway between and through the somites
- Ventro-medial Pathway between the neural tube (NT)
Dorsolateral Pathway
As the trunk NCCs can migrate in any of the 3 aforementioned pathways, specific signals are needed to prevent cells following one to suddenly enter a different pathway. NCCs which are destined to become melanoblast increase expression of EphB2 which are attracted to migrate along the dorsolateral pathway to the dorsal ectoderm by EphB1 [19] via minute holes in the basal lamina [20]. This route also contains Endothelins, preventing EdnrB-expressing neuronal and glial precursors from entering the pathway. It also stimulates EdnrB2-expressing melanocytic precursors to the migration onto this pathway, reinforcing the migration signal.
Differentiation
After settling in their respective sites, the NCCs receive environmental cues from their new location which will determine the cell fate of subsequent progeny cells. Kit signalling plays an essential role of the differentiation of NCCs into melanocytes, with MitfA and Sox10 signals being paramount to the specification of the NCCs into melanoblasts, the precursor to melanocytes. Eventually, MitfA-dependent activation of the histone deacetylase complex Hdac1 represses sox10 expression in melanoblasts, promoting both differentiation and fate commitment [21].
Latest Research
Cochlear Melanocytes of the Inner Ear
These melanocytes have recently been found to originate from neural crest cells delaminating from the rhombomere 6 region of the hindbrain, where the glossopharyngeal nerve and third pharyngeal arch are located [22].
Schwann Cell Precursors (SCPs) as Skin Melanocyte Origin
Some studies on the developmental process of Schwann cell precursors found along nerves are possible precursors of melanocytes found in the skin despite their different migration pathway from the trunk neural crest population [23]; nerve cells and the surrounding Schwann cells are derived from ventrolateral migrating trunk NCCs [24].
Development Time Course
Below summarises briefly the time course of the melanocyte development during the first trimester of embryogenesis (in particular between weeks 4 to 18) from formation of the neural crest, migration from the trunk NCCs to localising in the various subpopulations in the embryo. Skin melanocytes follow hair follicles closely and localise around them, but little is known about melanocytes in volar skin (skin of the palms and soles), but it is suggested that they localise around eccrine sweat glands. [25]
- Week 4: Visible neural plate where neural crest cells derived from.
- Weeks 6-8: Melanoblast (pre-cursor cell of melanocytes) migration along the dorsolateral pathway from the trunk neural crest cells. Melanoblast proliferation also takes place along the pathway, as the cells reach their areas of localisation, such as the eyes and the meninges. Melanoblasts have to migrate over incredibly long distances within the embryo, while at the same time proliferating and maintaining their chances of survival, so their development is a very dynamic process which is maintained by a combination of signal-receptor interactions.
- Weeks 9-12: Beginning of melanoblast migration to developing hair buds.
- Weeks 12-13 The majority of melanoblasts are located in the epidermis and hair follicles. Melanoblasts differentiate into melanocytes, but a collection of stem cell melanocytes remain in the hair bulge [26].
- Week 18: Hair breaks through skin surface containing melanocytes. [27]
Tissue Organ Structure and Function
Skin
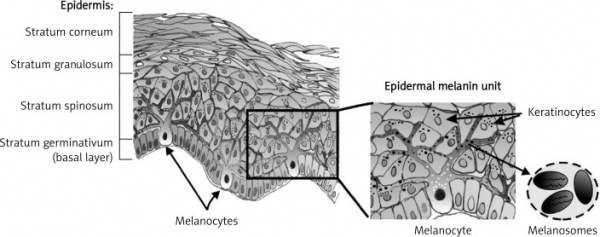
Melanocytes are located in the basal layer of the skin epidermis, amongst keratinocytes. Via dendrites, one melanocyte can communicate with around 30-40 keratinocytes [28]. The role of melanocytes is to produce melanosomes - granules containing the pigment melanin, which are then transferred to keratinocytes [29]. Melanocytes synthesise two types of melanin: pheomelanin, which is red/yellow; and eumelanin, which is brown/black. These are produced in varying proportions depending on the individual. Paler skinned individuals synthesise more pheomelanin, whilst darker skinned individuals synthesise more eumelanin [5].
Mature melanosomes travel from the cell body to the dendrites of the melanocyte, where they are transported to neighbouring keratinocytes. A recent study has shown that melanosome transfer occurs under the following process:
- Melanocyte dendrites release pigment globules that contain clusters of melanosomes into the extracellular space.
- The pigment globules are then phagocytosed by keratinocytes.
- The globule membrane degrades in the keratinocyte cytosol and the melanosomes are released to populate the perinuclear space [30].
In the skin, melanin provides photoprotection to keratinocyte nuclear DNA: By aggregating above the nuclei of keratinocytes, it absorbs ultra violet radiation, which would otherwise damage DNA [31].
We all have around the same number of melanocytes in our skin epidermis, but the quantity of melanosomes taken up into keratinocytes varies between individuals - this, as well as the pheomelanin-eumelanin ratio, is what causes variation in skin colour. The darkening of skin can occur due to two causes: Ultraviolet irradiation and locally produced α-Melanocyte-stimulating hormone (α-MSH), both of which stimulate melanogenesis [32][33][5].
Hair
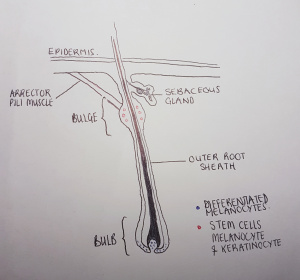
Though the hair follicle is initially formed from epithelial-mesenchymal interaction, neural crest derived melanocytes contribute to its completion and pigmentation[35]. Melanoblasts migrate from the epidermis into the developing hair follicles. Here they split into 2 populations: Differentiated melanocytes populate the hair matrix at the lower half of the hair bulb, whilst the quiescent melanoblasts, termed melanocyte stem cells (McSCs), reside in the hair bulge [26]. Like those of the skin, melanocytes secrete mature melanosomes to be taken up by surrounding keratinocytes, also populating the hair bulb. These melanin-containing keratinocytes form the cortex of the hair shaft[36]. Hair melanocytes differ from those in the skin, in that each melanocyte interacts with fewer keratinocytes. They also tend to be larger and have longer dendritic processes [37].
Hair growth follows a cyclic pattern in which there are 3 phases: Telogen (resting phase); Anagen (growth phase) and Catagen (regression phase). Pigmentation of the hair follicle by melanosomes only occurs in the anagen growth phase. During anagen, McSCs proliferate and differentiate into melanoblasts, populating the outer root sheath. Melanoblasts in turn migrate to the hair bulb, and differentiate into melanocytes [34]. Mature melanosomes are deposited into keratinocytes, as the keratinocytes proliferate to form the actively growing hair follicle. Cell proliferation ceases during the catagen phase, as both melanocytes and keratinocytes undergo apoptosis, the lower two thirds of the hair follicle regresses, leaving only their stem cell populations remaining in the hair bulge [38][26].
McSCs can migrate from the hair bulge into the epidermis of the skin, in response to skin lesions, or UVB irradiation. Once migration has occurred, they differentiate into functional epidermal melanocytes, capable of producing melanin that protects the damaged skin from UV irradiation. Thus McSC migration is one of the processes contributing to UV-induced skin tanning (mentioned in the skin section above) [39].
Ears
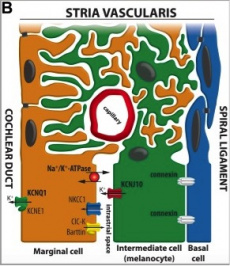
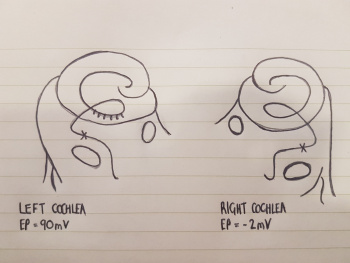
In the ear, melanocytes are mainly present in the cochlea, vestibular organ and endolymphatic sac. The cochlea of the inner ear functions as a transducer, by converting sound vibrations to electrical potentials in the auditory nerve via hair cells [40]. In the cochlea of humans, melanocytes are found in the vascularised epithelial tissue in the intermediate layer of the stria vascularis and the modiolus, between the marginal and basal cell layers [41](Figure 6). During development, marginal layer cuboidal epithelium develops processes which interdigitate with the intermediate melanocytes and basal cells [42].Based on studies performed on mice models, it is thought that melanocytes are important for the development of the endocochlear potential (EP), produced by strial cells. Intermediate melanocytes conduct K+, which plays an important role in sound conductance, as it flows from the endolymph into the ciliated epithelial cells of the ear via mechano-electrical channels. This influx of K+ is driven by a combination of the membrane potential of these ciliated epithelial cells, and the EP. A study on guinea pig models showed that blocking the K+ channels of melanocytes produces a lower EP [43]. This study agrees with evidence showing that mice deficient in cochlea melanocytes have a lower EP, requiring a greater sound stimulus to produce an action potential [44].
| Figure 6: Diagram of the Stria Vascularis, containing the marginal cells, intermediate melanocytes and basal cells. It shows marginal cell extensions intercalating with melanocytes, and the K+ ion channels involved in generating the EP [22]. | Figure 7: The cochlea of a Wv/Wv viable dominant spotting mutant - a mutation in both alleles encoding c-kit, an essential growth factor for formation, migration, proliferation and differentiation of melanoblasts. The distribution of melanocytes are shown as dots within the Stria Vascularis of the left cochlea [44] |
Eyes
Uveal melanocytes contain both eumelanin and pheomelanin. It has been speculated that eumelanin is photoreceptive, whereas pheomelanin is phototoxic. Uveal melanocytes produce growth factors, such as vascular endothelial growth factor that controls blood circulation around the eye, as well as extracellular matrix degrading enzymes. It was found that both of these roles may play a key part in the toxicity of uveal melanocytes. [45]The uveal tract (iris, ciliary body and choroid) and conjunctiva normally contains melanocytes derived from the neuro-ectodermal neural crest. Peri-orbital soft tissue is also derived from the embryonic neural crest [46]. On the other hand, pigmented melanocytes (such as those found on the retina) were derived from the neuroepithelium or from different layers of the optic cup. [47] Uveal melanocytes (like all other melanocytes) differentiate into melanin-producing cells, which in this case determines a person's eye colour, protects the eye from UV radiation [48] and can mutate to form many ocular diseases that can cause blindness, including age-related macular degeneration. [49] Melanocytes found in the eye are not able to participate in regeneration unlike epidermal melanocytes. [50]
Heart
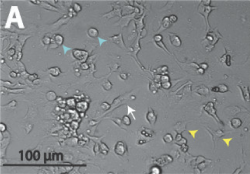
The term ‘cardiac melanocytes’ is used to describe the population of cells found in the human heart that express the melanin-synthesis enzymes. These cardiac melanocytes comprise less that 0.1% of all atrial cells. They also resemble the morphology of the melanocytes found at the cutaneous level [51].
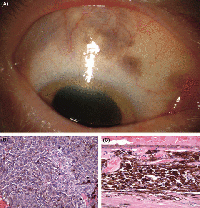
An experiment conducted by the Penn Cardiovascular Institute at the University of Pennsylvania resulted in the discovery of cardiac melanocytes being present at sites of the heart including the pulmonary veins, left atrium and foramen ovale where atrial arrhythmia triggers are believed to originate from [51]. The image below illustrates the results from this experiment where the cardiac melanocyte can be clearly seen and differentiated from the atrial myocyte.
Another study found an indicative experimental result that cardiac melanocytes and skin melanocytes depend on the same signalling molecules for development, further suggesting an origination from the same precursor cell population. Furthermore, these cardaic melanocytes were found in humans and mice but not in zebrafish or frogs and thereby establishing a potential association between cardiac melanocytes and four-chambered hearts [52].
However, limited research into cardiac melanocytes has been conducted as it is an upcoming topic of interest within the neural crest cell derivates. The gradually developing progress regarding cardiac melanocytes unfortunately restricts the amount of resources available for use outside of the scientific community.
Central Nervous System
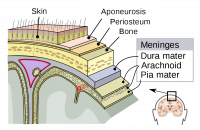
Melanocytes can also be found within the Central Nervous System, primarily in the leptomeninges [53], which consists of the inner two layers membranes of the meninges, the pia mater and arachnoid mater, which encapsulate the brain and the spinal cord. While its function within the meninges is unknown, they are essential to the health of the meninges, with their removal increases the risk of aseptic meningitis[54] (more detail can be found under Abnormalities). Furthermore, due to its common line of descent as nerve and Schwann cells, melanocytes possess receptivity to the same signalling molecules as neurons (though it may not trigger the same signalling pathways within the cell), scientists can study diseases afflicting the central nervous system using it as a model [55].
Molecular Mechanisms
As explained above, melanocytes are derived from the neural crest cells. The trunk neural crest gives rise to melanoblasts which then differentiate into melanocytes.
Through the study of a vast array of pigmentation disorders in recent years, several genes involved with melanocyte development have been identified. These genes regulate pigmentation in varying degrees and at different locations in the body. Mutations to these genes often result in abnormalities to their associated melanocytes which lead to the pigmentary disorders discussed later. Some of these genes include melanocortin 1 receptor (MC1R), tyrosinase-related protein 1 (TRP-1), protein tyrosine phosphatase, nonreceptor type 11 (PTPN11) among others [50] Melanocytes are diverse in their structure and functional purpose owing to the various anatomical regions within the body at which they are found and the role beyond melanin synthesis they play in that location.
However, at a molecular level melanocytes can be recognised through the identification of melanocyte-specific proteins like tyrosinase (TYR), tyrosinase-related protein 1 and 2 (TYRP1, TYRP2) or melanosomal matrix proteins Pmel117 and MART-1 or microphthalmia transcription factor (MITF) [27].
Factors affecting melanocytes
As seen above in Figure 4 of the histological image of the epidermis, melanocytes are surrounded by keratinocytes at a ratio of 1 : 10 which are necessary for the transferral of melanin. Paracrine factors derived from these keratinocytes are known to affect and influence the melanocytes’ biology and development. Some of these include;
- Basic fibroblast growth factor (bFGF): increase melanocyte proliferation
- Melanocyte-stimulating hormone (α-MSH): increase melanogenesis, melanosomal transfer and dendricity
- Endothelin 1 (ET-1): decreases proliferation and melanogenesis whilst increasing dendricity [27].
Genes involved in melanocyte development
MITF
The melanogenesis associated transcription factor gene (MITF) is crucial to melanocyte development as in encodes for an important transcription factor. MITF activates the pigment-producing genes dopachrome delta-isomerase, tyrosine-related protein 2 (dct) and tyrosinase (tyr). Melanoblasts are dependent on MITF for their survival. MITF is also vital in melanocyte specification where it is promoted by transcription factors like Sox10 and beta-catenin. It should be noted the MITF is dependent on Sox10 for transcriptional activation. [56].
Kit
The kit gene is a receptor tyrosine kinase required in the melanocyte development of vertebrates. It is heavily involved with pigmentation but also plays a role in melanocyte development, migration and survival in vertebrates especially. [56].
Endothelins
Endothelins plays a minor role in melanocyte development. Endothelins are a group of peptides that bind to G-protein coupled receptors. Of these receptors, EDNRB is important for melanocyte development as they assist in melanocyte survival and migration. [56].
Transcription
Microphthalmia transcription factor (MITF) is the key regulator of melanocyte identity without which melanocytes cannot be developed from the neural crest. The activation of MITF is a crucial stage in the formation of melanocytes. MITF have specific transcriptional targets, in particular genes which encode for melanosomes and the melanin synthesis pathway. Mutations to this transcription factor can lead to medical complications such as Waardernburg syndrome which has been discussed below. Furthermore, MITF is required to regulate the melanocyte lineage and subsequent melanocyte development. However, The over-expression of MITF during transcription has been shown to be a characteristic of melanomas.
MITF has been shown to be influenced by the up-regulation of WNT3A which induces melanoblast expression. [57]
Signalling Molecules
There are numerous signalling molecules and associated pathways involved in melanocyte development proliferation. These include MAPK-kinase signalling, α-MSH/cAMP/PKA and Endothelin/PKC (PKA protein kinase A, PKC protein kinase C). * *MAPK= mitogen-activated protein (MAP) kinases cAMP= cyclic adenosine monophosphate PKA= protein kinase A PKC= protein kinase C [27]
A particularly important signalling pathway in Wnt Signalling. It plays different roles across melanocyte development in vertebrates. Wnt signalling regulates melanocyte differentiation. Wnt signalling along with Sox10 drive the activation of MITF which then carries out its transcriptional function. Wnt signalling also protects MITF by preventing the degradation of beta-catenin levels. The image on the right illustrates how Wnt signalling stays active throughout melanocyte differentiation. Boxes B to G show the input of Wnt signalling as the melanocyte differentiates. [58].
Animal Models
Zebrafish Melanoma Model Reveals Emergence of Neural Crest Identity During Melanoma Initiation
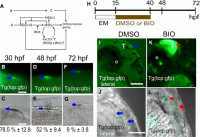
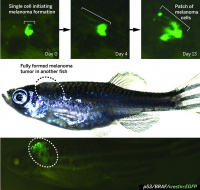
Melanoma is mainly driven by mutations in BRAF (particularly BRAFV600E) or RAS genes. Melanoma is considered to be easily treatable when it is localised and has not broken through the tissue boundary, this is known as in situ cancer. When the mutated cells begin to invade the underlying tissue, the cancer is now metastatic and cannot be treated effectively, even with newly developed kinase and immune checkpoint targeted therapies. BRAFV600E gene was placed under the influence of melanocyte-specific mitfa-promoter in transgenic zebrafish. When the fish were crossed with a p53 mutant loss-of-function background, they developed nevi, and after a few months the nevi became invasive cancer. The p53 gene is the main tumour suppressor gene that stops cell division when the cell undergoes damage. Tumours with mutations in the p53 gene are more aggressive, metastatic and have a poorer patient prognosis. It was found that the fish were prone to developing one to three melanoma tumours after several months, illustrating that molecular alterations are important for tumour initiation. PubmedParser error: The PubmedParser extension received invalid XML data. ()
Genetically Engineered Mice (GEM) Models For Melanocyte Development
The development of melanocytes in mice and humans are very similar, except they take longer to form in humans, so mice can be used to study melanocytes as they would occur in the human body. SCF (melanocyte growth factor) is essential for the maintenance and survival of melanocytes throughout life. The receptor for SCF is c-kit, and if it mutates, it can lead to sterility, anaemia and pigmentation disorders. If the receptor is completely lost, it results in loss of melanocytes, primordial germ cells and haematopoietic stem cells completely. In embryonic development however, ETRB (endothelin receptor) is more important to melanocyte developed compared to c-kit, as it regulates the number of precursor melanoblasts that will differentiate into melanocytes. [59]
Genetically Engineered Mice (GEM) Models For UV-induced Melanoma
UV-induced melanoma is explored in GEM as the skin of a mouse is similar to the skin of a human except that mice are covered in hair that undergo frequent cycles for follicular development, which in turn causes mature melanocytes to be shed. There is also a distinct difference in the levels of eumelanin and pheomalanin between mice and humans. These mice models allowed scientists to study the part UVR plays in melanomagenesis, in particular those that over-expresses HGF (hepatocyte growth factor). [60]
Current Research
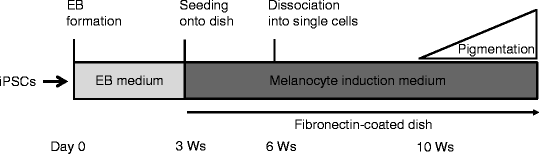
Generation of Melanocytes from Induced Pluripotent Stem Cells (iPSCs)
Melanocytes can be derived from autologous iPSCs, meaning the cells come from the one person. In this case the likelihood of the melanocytes being rejected by the immune system of the person they are being implanted in, is incredibly low. A benefit of autologous iPSCs from a patient with a pigmented disorder (such as albinism), is that the pathogenesis can be studied in order to find a suitable treatment for the disease, and possibly a cure. [61]
Altered Chromosome Expression of Uveal Melanoma in the Setting of Melanocytosis
Uveal melanoma seems to effect chromosomes 3p, 3q, 1p, 8p and 8q in particular as they show the most significant areas of DNA loss or amplification. However, the most important DNA alteration is monosomy on chromosome 3. In melanocytosis tissue, there is normalcy on chromosome 1, 3, 6 and 8, but there is exhibited disomy on chromosome 3. Ocular melanocytosis is predicted to impart a 1/400 lifetime risk of uveal melanoma in Caucasian individuals (which is 400x higher than the general population). It has been found that alterations in chromosome 3 does not always lead to the development of uveal melanoma, but it does take place sometime during the tumour development. [62]
Abnormalities
Skin
Abnormalities associated with melanocytes are often caused by disruptions to or loss of function of the pigmentation and its related factors. These pigmentary disorders are classified as hyperpigmentation disorder, hypopigmentation disorder, mixed hyperpigmentation disorder and mixed hypopigmentation disorder which are further subclassified into acquired and congenital [50].
Hyperpigmentation disorders refer to an array of usually benign skin conditions characterised by the darkening in colour of patches of skin. The darkening in colour is caused by the deposition of an excess of melanin in the skin, thus ‘hyper’ pigmentation [63].
LEOPARD Syndrome
LEOPARD syndrome is a congenital hyperpigmentation disorder that causes abnormalities to the appearance and functioning of the skin, heart, genitalia and inner ears. The name LEOPARD is an acronym which describes the characteristics of the condition;
L for Lentigines. These are pigmented macules (spots) on the skin. E for Electrocardiographic conduction defects. Aberration in the electrical activity of the heart due to factors including arrhythmia, intraventricular conduction delay etc leading to cardiac abnormalities. O for Ocular hypertelorism. An increased distance between the orbits within which they eyes lie resulting in widely spaced eyes. P for Pulmonary stenosis. Abnormalities of blood circulation caused by an obstruction to the pathway of blood outflow from the right ventricle to the heart. A for Abnormal genitalia. R for Retardation of growth. Slowed growth leading to short stature. D for Deafness. Sensorineural deafness (discussed below)
LEOPARD syndrome is autosomal dominant and can be inheritable. It can be caused by mutations to 3 different genes; protein tyrosine phosphatase, non-receptor type 11 (PTPN11), Raf-1 proto-oncogene (RAF1) or B-Raf proto-oncogene (BRAF) [64]
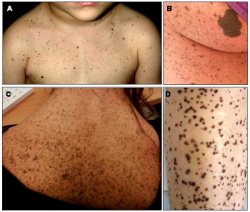
The lentigines in LEOPARD syndrome are caused by the development of additional matured melanosomes in the melanocytes and keratinocytes. An experiment conducted by the Department of Dermatology at Gunma University Graduate school of Medicine in 2015 used in vitro assays to study the melanocytes of patients with LEOPARD syndrome. The in vitro assay of a patient with LEOPARD syndrome caused by a mutation in the PTPN11 gene showed that melanin synthesis in the patient’s melanoma cells were higher than those of unaffected individuals due to the melanoma cells’ expression of SHP-2. SHP-2 is the protein tyrosine phosphatase encoded by the PTPN11 gene. This discovery suggested that the mutations to SHP-2 when associated with LEOPARD syndrome cause an increase in melanin synthesis by the melanocytes, thus resulting in the large pigmented spots [65].
As seen in the image to the right, the multiple lentigines that develop as a result of LEOPARD syndrome vary with age. Box A shows the development of lentigines as a result of a mutation in the PTPN11 gene in a 2 year old. Box C shows the neck and back of a 28 year old female patient with lentigines scattered throughout the displayed skin surface. Box D illustrates the lower leg of a 37 year old male with LEOPARD syndrome which multiple lentigines scattered throughout [66]. These images illustrate how the appearance of lentigines differs according to age and the manner by which LEOPARD syndrome has presented itself (the associated mutation).
Melasma
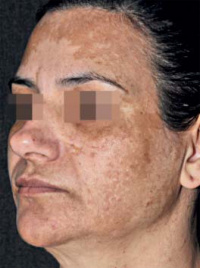
Melasma is an acquired hyperpigmentation disorder. It is a pigmentary condition occurring primarily on the face, characterised by brown patches usually found in three identified patterns; centrofacial , malar and mandibular. The most common pattern is centrofacial where pigmentation occurs on the forehead, nose, cheeks, chin and the upper lip. Malar pattern refers to the presence of melasma of the malar cheeks (cheek bones) on the face whilst the mandibular pattern features melasma on the mandible (jawline) and chin. Melasma occurring outside of the face in regions like the neck, sternum, forearms and other upper extremities is referred to as being of the extra-facial melasma pattern [67].
Melasma, previously known as chloasma, is ascribed to ultraviolet (UV) exposure and hormonal changes. UV exposure begins a cascade of events which ultimately results in excessive melanogenesis promotion. Exposure to UV light activates inducible nitric oxide (regulate of pathophysiological processes in the skin) [68] which in turn induces reactive oxygen species that increase oxidative stress resulting in inflammation that ends in post-inflammatory hyperpigmentation [67] [69]. The hormonal changes are especially prevalent in pregnant women, with the hormone oestrogen triggering melasma [70]. Women with darker skin types are at a higher risk of developing the condition.
Melanoma
Melanoma is basically a type of cancer developing from melanocytes [71] which usually occur in the epidermal melanocytes, but can also occur in the various subpopulations of melanocytes, which will be discussed later. It is the deadliest of the three major causes of skin cancer, with basal cell carcinoma and squamous cell carcinoma being the other two, though it is relatively rare, making up about 5% of the total skin cancer cases [72]. Despite its function in the production of melanin to protect cells from ultraviolet radiation, melanoma still primarily results from DNA damage caused by it [73], especially in individuals with low basal levels of melanin production. Genetic defects as such xeroderma pigmentosum[74], or family lines with increased mole development have a greater risk of developing melanomas. There are four major forms of melanoma including:
- superficial spreading
- nodular
- lentigo maligna
- acral lentiginous melanomas.
Of the four, superficial spreading remains the most common, accounting for about 70% of the cases, followed by nodular, representing approximately 15–30% of melanoma cases. The lentigo maligna and acral lentiginous forms represent less than 10% of the cases [71].
Development from Melanocyte to Melanoma
<html5media height="300" width="400">https://www.youtube.com/watch?v=aiARvdeXdKM</html5media> [75]
Studies on the possible causes shown 40–60% of all melanoma cases involve an activated BRAF mutation, which is a proto-oncogene that encodes a serine/threonine protein kinase as part of the RAS-RAF-MEK-ERK kinase pathway, which promotes cell growth and proliferation [76]. Possible causes for mutation could be:
- Ultraviolet Radiation: Exposure to ultraviolet (UV) radiation plays an important role in the development of melanomas later in life, with frequent and long periods exposure leading which lead to sunburns increasing the risk. This is due to mutations in DNA from damage done by UV which is known for its mutagenic capabilities, with the most commonly associated genes with melaonoma being PPP6C, RAC1, SNX31, TACC1, STK19 and ARID2 [77].
- Melanocytic Nevi: Also known as moles or birthmarks, nevi are benign concentrations of melanocytes and can form either a dark spot on the skin or lump, and can be acquired later in later in life or congenital. Having a large number of nevi is a possible indicator of greater potential risk of acquiring malignant melanomas later in life. When a nevus develops from a benign form to a malignant melanoma, a change in colour or texture serve a possible indicator, with approximately 81% of melanoma patients observing such a change in the location of melanomas [71].
Vitiligo
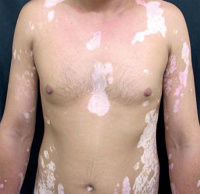
Vitiligo is an acquired hypopigmentation disorder. It occurs due to the loss of functional melanocytes resulting in chronic depigmentation of the skin in any region of the body in more than one location. It can also affect the mucous membrane inside the nose and mouth as well as the hair [78].
Symptoms include loss of skin pigmentation in blotches, premature greying of scalp, eyelash and eyebrow hair, loss of pigmentation within the retina of the eye and loss of colour in the mucous membranes lining the mouth and nose. [79] [80] These symptoms can begin to appear at any age but commonly before the age of 20.
The pattern in which the depigmented patches of skin appear in across the body allow for the vitiligo to be classified into three groups. Generalised vitiligo: Discolouration across most parts of the body. Segmental vitiligo: Depigmentation occurring on one side or region of the body. Focal vitiligo: Depigmentation in one area of skin only.
Vitiligo has an autoimmune basis, being driven by t cell-derived interferon-gamma, a cytokine who’s levels are found increased in patients. The increased levels of these cytokines and their associated chemokines affect the mleanocytes’ ability to synthesise melanin and thus results in the patches of depigmentation observed in vitiligo [50] [81].
Currently, there is no cure available to prevent vitiligo and its associated melanocyte loss. However, laser pigmentation treatment is available to treat the depigmented and discoloured patches of skin observed in patients.
Ears
Waardenburg Syndrome
There are varying forms of Waardenburg Syndrome, but they all result in deafness due to loss of melanocytes in the inner ears. As explained in the above section "Tissue Organ Structure and Function", melanocytes are necessary for sound transduction. The mutations that cause Waardenburg Syndrome, result in a lack of Melanogenesis Associated Transcription Factor (MITF) expression. MITF is a protein which works along side the transcription factor LEF-1 to regulate melanocyte gene expression [82]. These mutations may be in Sox10 and Pax3 genes, as these both regulate the expression of the MITF protein [83][84]. Hence, Waardenburg Syndrome can have varying genetic causes, but with the same outcome.
Eyes
Uveal Melanoma
Melanoma that occurs in the eye is known as uveal melanoma. This is an uncommon form of cancer that only accounts for about 3% of all melanomas. The risk factors of this cancer are light skin colour, red or blonde hair and blue or light irises. [46]. About 95% of uveal melanomas occur in the posterior eye (the ciliary body and choroid). Even though this is a rare from of cancer, it still contributes to a large percent of deaths and has a high chance of leading to distant metastases despite successful initial therapies in the local tumour or tumours of the eye. [85]
Oculocutaneous Albinism
Oculocutaneous albinism is a rare group of congenital hypopigmentation disorder affecting one in 17,000 people across most ethnic backgrounds. Oculocutaneous albinism (OCA) is caused by mutations to the genes required for melanin synthesis in melanocytes resulting in the lack of pigment. In OCA, this lack of pigment results in abnormal development of the eyes leading to complications with vision (photophobia, mis-routing of optic nerves, nystagmus etc.) and significantly lighter skin which exposes the skin to UV-radiation damage from the sun.
OCA can be caused by a mutation to several different genes including membrane-associated transporter protein (MATP), OCA2, Tyrosinase (TYR) and tyrosinase-related protein 1 (TYRP1) . There are various visual complications associated with OCA including nystagmus, reduction of iris pigment as seen in Figure 20 and reduction of retinal pigment. OCA also leads to poor visual acuity as it causes macular hypoplasia, where the macula lacks development leading to abnormal foveal development which negatively impacts visual acuity. It should be noted that visual acuity increases and improves with higher amounts of pigments [86]. [87]
There are seven identified types of oculocutaneous albinism; OCA1, OCA2, OCA3, OCA4, OCA5, OCA6, OCA7. Each of these types of the OCA disorder have differing degrees of impacts upon the eyes, skin and hair. Furthermore, the epidemiology of some of these disorders has shown higher prevalences within specific ethnic groups. For example, OCA3 is known to affect people of the African population at higher rates [88].
Central Nervous System
Harada Syndrome Related Aseptic Meningitis
Harada syndrome (AKA Vogt–Koyanagi–Harada syndrome, uveomeningitis syndrome and uveomeningoencephalitic syndrome), is a multisystem disease of a presumed autoimmune cause, that affects pigmented tissues, in other melanocytes, which have melanin. Aseptic meningitis that is observed with the disease is hypothesized to be due to the destruction of leptomeningeal melanocytes [54].
Melanomas
Melanomas or melanocytic tumours are ranked third in the causes of the central nervous system (CNS) related cancers. However, primary melanocytic tumours of the CNS still only make up 1% of the overall melanomas in the entire body [89]. To date, there are very few studies on melanomas originating from the leptomeninges in the CNS, and while a spectrum of primary melanocytic tumours is known to exist, the rarity of such cases pose a challenge to studying the disease in detail. Some examples include Primary diffuse leptomeningeal melanomatosis (PDLM) and primary thoracolumbar spinal melanoma [90].
Glossary
Autologous Cells or tissues obtained from the same individual
Epithelial-to-Mesenchymal Transition (EMT) A process by which epithelial cells lose their cell polarity and cell-cell adhesion, and gain migratory and invasive properties to become mesenchymal stem cells
Cross Talk When two pathways of signal transfer affect one another through unwanted communication
Haematopoietic Cells (often referring to stem cells) that commit to the differentiation into all blood cells (i.e. red/erythrocytes, macrophages, neutrophils, platelets/thrombocytes)
Lentigine Pigmented spots on the skin
Macule A discoloured patch of skin.
Meningitis An infection of the meninges, the membranous layers surrounding the brain and spinal cord.
Melanoblasts Melanocyte pre-cursor cells
Melanocytosis The presence of an excessive number of melanocytes
Melanoma A tumour of melanin-forming cells
Melanogenesis The production of melanin
Mutagenic A physical or chemical agent that alters the genetic material of an organism, increasing background mutation frequency
Nevus Chronic lesion of skin or mucosa
Nodular From the word nodule, which a growth of abnormal tissue on the body.
Nystagmus Rapid involuntary side-to-side movement of the eyes.
Pathogenesis The manner in which a disease develops.
Proto-oncogene A gene which, when mutated, becomes an oncogene contributing to cancer due to several effects (i.e. loss of cell cycle regulation, or shutting down of apoptotic signalling pathways)
Uveal Refer to something of the uvea, the pigmented middle layer of the three concentric layers forming the eye.
Volar Skin Skin of the palms of the hands and the soled of the feet (thick skin)
Xeroderma pigmentosum A genetic disorder which the individual has reduced ability to repair DNA damage such as those caused by sunlight, resulting in severe sunburns even with small amounts of UV exposure.
References
- ↑ 1.0 1.1 RAWLES ME. (1947). Origin of pigment cells from the neural crest in the mouse embryo. Physiol. Zool. , 20, 248-66. PMID: 20256541
- ↑ Westerhof W. (2006). The discovery of the human melanocyte. Pigment Cell Res. , 19, 183-93. PMID: 16704452 DOI.
- ↑ Theriault LL & Hurley LS. (1970). Ultrastructure of developing melanosomes in C57 black and pallid mice. Dev. Biol. , 23, 261-75. PMID: 5476812
- ↑ Markert CL & Silvers WK. (1956). The Effects of Genotype and Cell Environment on Melanoblast Differentiation in the House Mouse. Genetics , 41, 429-50. PMID: 17247639
- ↑ 5.0 5.1 5.2 Thody AJ, Higgins EM, Wakamatsu K, Ito S, Burchill SA & Marks JM. (1991). Pheomelanin as well as eumelanin is present in human epidermis. J. Invest. Dermatol. , 97, 340-4. PMID: 2071942
- ↑ Agar N & Young AR. (2005). Melanogenesis: a photoprotective response to DNA damage?. Mutat. Res. , 571, 121-32. PMID: 15748643 DOI.
- ↑ Klaus, S.N.N., 2007. A History of the Science of Pigmentation. In The Pigmentary System: Physiology and Pathophysiology: Second Edition. Blackwell Publishing Ltd, pp. 1–10.
- ↑ Riolan, J. Anthropographia. Paris: Hadrianum Perier, 1618.
- ↑ Quevedo, W.C.C. & Holstein, T.J.J., 2007. General Biology of Mammalian Pigmentation. In The Pigmentary System: Physiology and Pathophysiology: Second Edition. Blackwell Publishing Ltd, pp. 61–90.
- ↑ Moyer, F. H. Electron microscope observations on the origin, development and genetic control of melanin granules in the mouse eye. In: The Structure of the Eye, G. K. Smelser (ed.). New York: Academic Press, 1961, pp. 469–486
- ↑ Turner WA, Taylor JD & Tchen TT. (1975). Melanosome formation in the goldfish: the role of multivesicular bodies. J. Ultrastruct. Res. , 51, 16-31. PMID: 805261
- ↑ Granville Harrison, R. Archiv für Entwicklungsmechanik der Organismen (1910) 30: 15. https://doi.org/10.1007/BF02263801
- ↑ DuShane, Graham P. "An Experimental Study of the Origin of Pigment Cells in Amphibia." Journal of Experimental Zoology 72, no. 1 (1935): 1-31. https://onlinelibrary-wiley-com.wwwproxy1.library.unsw.edu.au/doi/epdf/10.1002/jez.1400720102
- ↑ WESTON JA. (1963). A radioautographic analysis of the migration and localization of trunk neural crest cells in the chick. Dev. Biol. , 6, 279-310. PMID: 14000137
- ↑ Marusich MF, Weston JA. 1992. Identification of early neurogenic cells in the neural crest lineage. Dev Biol 149:295– 306.
- ↑ Tolleson WH. (2005). Human melanocyte biology, toxicology, and pathology. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev , 23, 105-61. PMID: 16291526 DOI.
- ↑ Cichorek, M, Wachulska, M and Skoniecka, A. (2013). Heterogeneity of neural crest-derived melanocytes. Central European Journal of Biology, 8(4), 315-330.
- ↑ Richardson J, Gauert A, Briones Montecinos L, Fanlo L, Alhashem ZM, Assar R, Marti E, Kabla A, Härtel S & Linker C. (2016). Leader Cells Define Directionality of Trunk, but Not Cranial, Neural Crest Cell Migration. Cell Rep , 15, 2076-88. PMID: 27210753 DOI.
- ↑ Santiago A & Erickson CA. (2002). Ephrin-B ligands play a dual role in the control of neural crest cell migration. Development , 129, 3621-32. PMID: 12117812
- ↑ Gilbert SF. Developmental Biology. 6th edition. Sunderland (MA): Sinauer Associates; 2000. The Neural Crest. Available from: https://www.ncbi.nlm.nih.gov/books/NBK10065/
- ↑ Greenhill ER, Rocco A, Vibert L, Nikaido M & Kelsh RN. (2011). An iterative genetic and dynamical modelling approach identifies novel features of the gene regulatory network underlying melanocyte development. PLoS Genet. , 7, e1002265. PMID: 21909283 DOI.
- ↑ 22.0 22.1 Locher H, de Groot JC, van Iperen L, Huisman MA, Frijns JH & Chuva de Sousa Lopes SM. (2015). Development of the stria vascularis and potassium regulation in the human fetal cochlea: Insights into hereditary sensorineural hearing loss. Dev Neurobiol , 75, 1219-40. PMID: 25663387 DOI.
- ↑ Adameyko I, Lallemend F, Aquino JB, Pereira JA, Topilko P, Müller T, Fritz N, Beljajeva A, Mochii M, Liste I, Usoskin D, Suter U, Birchmeier C & Ernfors P. (2009). Schwann cell precursors from nerve innervation are a cellular origin of melanocytes in skin. Cell , 139, 366-79. PMID: 19837037 DOI.
- ↑ Vega-Lopez GA, Cerrizuela S & Aybar MJ. (2017). Trunk neural crest cells: formation, migration and beyond. Int. J. Dev. Biol. , 61, 5-15. PMID: 28287247 DOI.
- ↑ Nakamura M, Fukunaga-Kalabis M, Yamaguchi Y, Furuhashi T, Nishida E, Kato H, Mizuno T, Sugiura M & Morita A. (2015). Site-specific migration of human fetal melanocytes in volar skin. J. Dermatol. Sci. , 78, 143-8. PMID: 25818865 DOI.
- ↑ 26.0 26.1 26.2 Nishimura EK, Jordan SA, Oshima H, Yoshida H, Osawa M, Moriyama M, Jackson IJ, Barrandon Y, Miyachi Y & Nishikawa S. (2002). Dominant role of the niche in melanocyte stem-cell fate determination. Nature , 416, 854-60. PMID: 11976685 DOI.
- ↑ 27.0 27.1 27.2 27.3 27.4 Cichorek M, Wachulska M, Stasiewicz A & Tymińska A. (2013). Skin melanocytes: biology and development. Postepy Dermatol Alergol , 30, 30-41. PMID: 24278043 DOI.
- ↑ FITZPATRICK TB & BREATHNACH AS. (1963). [THE EPIDERMAL MELANIN UNIT SYSTEM]. Dermatol Wochenschr , 147, 481-9. PMID: 14172128
- ↑ Nordlund, J. J., Boissy, R. E., Hearing, V. A., King, R. S., Oetting, W. P., & Ortonne, J. (2007). The Pigmentary System: Physiology and Pathophysiology: Second Edition. Blackwell Publishing: https://onlinelibrary-wiley-com.ezproxy.is.ed.ac.uk/doi/pdf/10.1002/9780470987100
- ↑ Ando H, Niki Y, Ito M, Akiyama K, Matsui MS, Yarosh DB & Ichihashi M. (2012). Melanosomes are transferred from melanocytes to keratinocytes through the processes of packaging, release, uptake, and dispersion. J. Invest. Dermatol. , 132, 1222-9. PMID: 22189785 DOI.
- ↑ Lin JY & Fisher DE. (2007). Melanocyte biology and skin pigmentation. Nature , 445, 843-50. PMID: 17314970 DOI.
- ↑ Tsatmali M, Ancans J, Yukitake J & Thody AJ. (2000). Skin POMC peptides: their actions at the human MC-1 receptor and roles in the tanning response. Pigment Cell Res. , 13 Suppl 8, 125-9. PMID: 11041369
- ↑ Eller MS, Yaar M & Gilchrest BA. (1994). DNA damage and melanogenesis. Nature , 372, 413-4. PMID: 7984233 DOI.
- ↑ 34.0 34.1 Mackenzie MA, Jordan SA, Budd PS & Jackson IJ. (1997). Activation of the receptor tyrosine kinase Kit is required for the proliferation of melanoblasts in the mouse embryo. Dev. Biol. , 192, 99-107. PMID: 9405100 DOI.
- ↑ Barsh & Cotsarelis. 2007 How Hair Gets Its Pigment. Cell. 130(5), pp.779–781. https://www.sciencedirect.com/science/article/pii/S0092867407010951#bbib6
- ↑ Weiner L, Han R, Scicchitano BM, Li J, Hasegawa K, Grossi M, Lee D & Brissette JL. (2007). Dedicated epithelial recipient cells determine pigmentation patterns. Cell , 130, 932-42. PMID: 17803914 DOI.
- ↑ Hirobe, T., 2014. Keratinocytes regulate the function of melanocytes. Dermatologica Sinica, 32(4), pp.200–204. https://www.sciencedirect.com/science/article/pii/S1027811714000238
- ↑ Fuchs E. (2007). Scratching the surface of skin development. Nature , 445, 834-42. PMID: 17314969 DOI.
- ↑ Chou WC, Takeo M, Rabbani P, Hu H, Lee W, Chung YR, Carucci J, Overbeek P & Ito M. (2013). Direct migration of follicular melanocyte stem cells to the epidermis after wounding or UVB irradiation is dependent on Mc1r signaling. Nat. Med. , 19, 924-9. PMID: 23749232 DOI.
- ↑ Purves D, Augustine GJ, Fitzpatrick D, et al., editors. Neuroscience. 2nd edition. Sunderland (MA): Sinauer Associates; 2001. The Inner Ear. Available from: https://www.ncbi.nlm.nih.gov/books/NBK10946/
- ↑ Meyer zum Gottesberge AM. (1988). Physiology and pathophysiology of inner ear melanin. Pigment Cell Res. , 1, 238-49. PMID: 3070525
- ↑ Steel KP & Barkway C. (1989). Another role for melanocytes: their importance for normal stria vascularis development in the mammalian inner ear. Development , 107, 453-63. PMID: 2612372
- ↑ Takeuchi S, Kakigi A, Takeda T, Saito H & Irimajiri A. (1996). Intravascularly applied K(+)-channel blockers suppress differently the positive endocochlear potential maintained by vascular perfusion. Hear. Res. , 101, 181-5. PMID: 8951443
- ↑ 44.0 44.1 Cable J, Huszar D, Jaenisch R & Steel KP. (1994). Effects of mutations at the W locus (c-kit) on inner ear pigmentation and function in the mouse. Pigment Cell Res. , 7, 17-32. PMID: 7521050
- ↑ Hu DN, Savage HE & Roberts JE. (2002). Uveal melanocytes, ocular pigment epithelium, and Müller cells in culture: in vitro toxicology. Int. J. Toxicol. , 21, 465-72. PMID: 12537643 DOI.
- ↑ 46.0 46.1 Schoenfield L. (2014). Uveal melanoma: A pathologist's perspective and review of translational developments. Adv Anat Pathol , 21, 138-43. PMID: 24508696 DOI.
- ↑ Baderca F, Solovan C & Boghian L. (2013). Epidemiological and morphological data of ocular melanocytic lesions. Rom J Morphol Embryol , 54, 77-83. PMID: 23529312
- ↑ Hou L & Pavan WJ. (2008). Transcriptional and signaling regulation in neural crest stem cell-derived melanocyte development: do all roads lead to Mitf?. Cell Res. , 18, 1163-76. PMID: 19002157 DOI.
- ↑ Hu DN, Simon JD & Sarna T. (2008). Role of ocular melanin in ophthalmic physiology and pathology. Photochem. Photobiol. , 84, 639-44. PMID: 18346089 DOI.
- ↑ 50.0 50.1 50.2 50.3 Yamaguchi Y & Hearing VJ. (2014). Melanocytes and their diseases. Cold Spring Harb Perspect Med , 4, . PMID: 24789876 DOI.
- ↑ 51.0 51.1 51.2 Hwang H, Liu F, Levin MD & Patel VV. (2014). Isolating primary melanocyte-like cells from the mouse heart. J Vis Exp , , 4357. PMID: 25285608 DOI.
- ↑ Brito FC & Kos L. (2008). Timeline and distribution of melanocyte precursors in the mouse heart. Pigment Cell Melanoma Res , 21, 464-70. PMID: 18444965 DOI.
- ↑ Gudjohnsen SA, Atacho DA, Gesbert F, Raposo G, Hurbain I, Larue L, Steingrimsson E & Petersen PH. (2015). Meningeal Melanocytes in the Mouse: Distribution and Dependence on Mitf. Front Neuroanat , 9, 149. PMID: 26635543 DOI.
- ↑ 54.0 54.1 Goldgeier MH, Klein LE, Klein-Angerer S, Moellmann G & Nordlund JJ. (1984). The distribution of melanocytes in the leptomeninges of the human brain. J. Invest. Dermatol. , 82, 235-8. PMID: 6699426
- ↑ Yaar M & Park HY. (2012). Melanocytes: a window into the nervous system. J. Invest. Dermatol. , 132, 835-45. PMID: 22158549 DOI.
- ↑ 56.0 56.1 56.2 Uong A & Zon LI. (2010). Melanocytes in development and cancer. J. Cell. Physiol. , 222, 38-41. PMID: 19795394 DOI.
- ↑ Mort, R., Jackson, I. and Patton, E. (2015). The melanocyte lineage in development and disease. Development, 142(4), pp.620-632.
- ↑ Vibert L, Aquino G, Gehring I, Subkankulova T, Schilling TF, Rocco A & Kelsh RN. (2017). An ongoing role for Wnt signaling in differentiating melanocytes in vivo. Pigment Cell Melanoma Res , 30, 219-232. PMID: 27977907 DOI.
- ↑ Herlyn M, Berking C, Li G & Satyamoorthy K. (2000). Lessons from melanocyte development for understanding the biological events in naevus and melanoma formation. Melanoma Res. , 10, 303-12. PMID: 10985664
- ↑ Day CP, Marchalik R, Merlino G & Michael H. (2017). Mouse models of UV-induced melanoma: genetics, pathology, and clinical relevance. Lab. Invest. , 97, 698-705. PMID: 28092363 DOI.
- ↑ Ohta S, Imaizumi Y, Akamatsu W, Okano H & Kawakami Y. (2013). Generation of human melanocytes from induced pluripotent stem cells. Methods Mol. Biol. , 989, 193-215. PMID: 23483397 DOI.
- ↑ Horgan N, Shields CL, Swanson L, Teixeira LF, Eagle RC, Ganguly A & Shields JA. (2009). Altered chromosome expression of uveal melanoma in the setting of melanocytosis. Acta Ophthalmol , 87, 578-80. PMID: 18547285 DOI.
- ↑ Aocd.org. Hyperpigmentation - American Osteopathic College of Dermatology (AOCD). [online] Available at: https://www.aocd.org/page/Hyperpigmentation
- ↑ Genetic and Rare Diseases Information Center. (2016). LEOPARD syndrome. [online] Available at: https://rarediseases.info.nih.gov/diseases/1100/leopard-syndrome
- ↑ Motegi S, Yokoyama Y, Ogino S, Yamada K, Uchiyama A, Perera B, Takeuchi Y, Ohnishi H & Ishikawa O. (2015). Pathogenesis of multiple lentigines in LEOPARD syndrome with PTPN11 gene mutation. Acta Derm. Venereol. , 95, 978-84. PMID: 25917897 DOI.
- ↑ Sarkozy A, Digilio MC & Dallapiccola B. (2008). Leopard syndrome. Orphanet J Rare Dis , 3, 13. PMID: 18505544 DOI.
- ↑ 67.0 67.1 Ogbechie-Godec OA & Elbuluk N. (2017). Melasma: an Up-to-Date Comprehensive Review. Dermatol Ther (Heidelb) , 7, 305-318. PMID: 28726212 DOI.
- ↑ Cals-Grierson MM & Ormerod AD. (2004). Nitric oxide function in the skin. Nitric Oxide , 10, 179-93. PMID: 15275864 DOI.
- ↑ Denat L, Kadekaro AL, Marrot L, Leachman SA & Abdel-Malek ZA. (2014). Melanocytes as instigators and victims of oxidative stress. J. Invest. Dermatol. , 134, 1512-1518. PMID: 24573173 DOI.
- ↑ Aguirre, D. (n.d.). Melasma Unmasked. [online] Dermalinstitute.com. Available at: http://dermalinstitute.com/au/library/118_article_Melasma_Unmasked.html
- ↑ 71.0 71.1 71.2 Liu Y & Sheikh MS. (2014). Melanoma: Molecular Pathogenesis and Therapeutic Management. Mol Cell Pharmacol , 6, 228. PMID: 25745537
- ↑ Miller AJ & Mihm MC. (2006). Melanoma. N. Engl. J. Med. , 355, 51-65. PMID: 16822996 DOI.
- ↑ Kanavy HE & Gerstenblith MR. (2011). Ultraviolet radiation and melanoma. Semin Cutan Med Surg , 30, 222-8. PMID: 22123420 DOI.
- ↑ Azoury SC & Lange JR. (2014). Epidemiology, risk factors, prevention, and early detection of melanoma. Surg. Clin. North Am. , 94, 945-62, vii. PMID: 25245960 DOI.
- ↑ MelaFind MELASciences (2012, March 10) From Melanocyte to Melanoma [Video file]. Retrieved from https://www.youtube.com/watch?v=aiARvdeXdKM
- ↑ Kumar R, Angelini S, Czene K, Sauroja I, Hahka-Kemppinen M, Pyrhönen S & Hemminki K. (2003). BRAF mutations in metastatic melanoma: a possible association with clinical outcome. Clin. Cancer Res. , 9, 3362-8. PMID: 12960123
- ↑ Hodis E, Watson IR, Kryukov GV, Arold ST, Imielinski M, Theurillat JP, Nickerson E, Auclair D, Li L, Place C, Dicara D, Ramos AH, Lawrence MS, Cibulskis K, Sivachenko A, Voet D, Saksena G, Stransky N, Onofrio RC, Winckler W, Ardlie K, Wagle N, Wargo J, Chong K, Morton DL, Stemke-Hale K, Chen G, Noble M, Meyerson M, Ladbury JE, Davies MA, Gershenwald JE, Wagner SN, Hoon DS, Schadendorf D, Lander ES, Gabriel SB, Getz G, Garraway LA & Chin L. (2012). A landscape of driver mutations in melanoma. Cell , 150, 251-63. PMID: 22817889 DOI.
- ↑ Filipp, F., Birlea, S., Bosenberg, M., Brash, D., Cassidy, P., Chen, S., D'Orazio, J., Fujita, M., Goh, B., Herlyn, M., Indra, A., Larue, L., Leachman, S., Le Poole, C., Liu-Smith, F., Manga, P., Montoliu, L., Norris, D., Shellman, Y., Smalley, K., Spritz, R., Sturm, R., Swetter, S., Terzian, T., Wakamatsu, K., Weber, J. and Box, N. (2018). Frontiers in pigment cell and melanoma research. Pigment Cell & Melanoma Research, 31(6), pp.728-735.
- ↑ Mayo Clinic. (2018). Vitiligo - Symptoms and causes. [online] Available at: https://www.mayoclinic.org/diseases-conditions/vitiligo/symptoms-causes/syc-20355912
- ↑ Bastonini E, Kovacs D & Picardo M. (2016). Skin Pigmentation and Pigmentary Disorders: Focus on Epidermal/Dermal Cross-Talk. Ann Dermatol , 28, 279-89. PMID: 27274625 DOI.
- ↑ Faria AR, Tarlé RG, Dellatorre G, Mira MT & Castro CC. (2014). Vitiligo--Part 2--classification, histopathology and treatment. An Bras Dermatol , 89, 784-90. PMID: 25184918
- ↑ Yasumoto K, Takeda K, Saito H, Watanabe K, Takahashi K & Shibahara S. (2002). Microphthalmia-associated transcription factor interacts with LEF-1, a mediator of Wnt signaling. EMBO J. , 21, 2703-14. PMID: 12032083 DOI.
- ↑ Lee M, Goodall J, Verastegui C, Ballotti R & Goding CR. (2000). Direct regulation of the Microphthalmia promoter by Sox10 links Waardenburg-Shah syndrome (WS4)-associated hypopigmentation and deafness to WS2. J. Biol. Chem. , 275, 37978-83. PMID: 10973953 DOI.
- ↑ Potterf SB, Furumura M, Dunn KJ, Arnheiter H & Pavan WJ. (2000). Transcription factor hierarchy in Waardenburg syndrome: regulation of MITF expression by SOX10 and PAX3. Hum. Genet. , 107, 1-6. PMID: 10982026
- ↑ An J, Wan H, Zhou X, Hu DN, Wang L, Hao L, Yan D, Shi F, Zhou Z, Wang J, Hu S, Yu J & Qu J. (2011). A comparative transcriptomic analysis of uveal melanoma and normal uveal melanocyte. PLoS ONE , 6, e16516. PMID: 21305041 DOI.
- ↑ Grønskov, K., Ek, J. and Brondum-Nielsen, K. (2007). Oculocutaneous albinism. Orphanet Journal of Rare Diseases, 2(1), p.43.
- ↑ Genetics Home Reference. (2018). Oculocutaneous albinism. [online] Available at: https://ghr.nlm.nih.gov/condition/oculocutaneous-albinism
- ↑ Grønskov, K., Ek, J. and Brondum-Nielsen, K. (2007). Oculocutaneous albinism. Orphanet Journal of Rare Diseases, 2(1), p.43.
- ↑ Farrokh D, Fransen P & Faverly D. (2001). MR findings of a primary intramedullary malignant melanoma: case report and literature review. AJNR Am J Neuroradiol , 22, 1864-6. PMID: 11733317
- ↑ Trinh V, Medina-Flores R, Taylor CL, Yonas H & Chohan MO. (2014). Primary melanocytic tumors of the central nervous system: Report of two cases and review of literature. Surg Neurol Int , 5, 147. PMID: 25371847 DOI.