Paper - lnvolution of tissues in fetal life (1946)
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hertig AT. lnvolution of tissues in fetal life: a review. (1946) Anat. Rec. 94: 96-116.
| Online Editor |
|---|
| This historic 1946 paper describes early human development in relation to tissues that are lost during development. It includes several of the Carnegie Collection embryos in the figures.
See also:
Hertig AT. and Rock J. On a normal human ovum not over 7.5 days of age. (1945) Anat. Rec. 91: 281. Hertig AT. and Rock J. On a normal ovum of approximately 9 to 10 days of age. (1945) Anat. Rec. 91: 281. Hertig AT. and Rock J. On a human blastula recovered from the uterine cavity 4 days after ovulation. (1946) J Gerontol. 1(1): 96-117.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
lnvolution of Tissues in Fetal Life - A Review
Dr. Arthur T. Hertig graduated from the University of Minnesota in 1928 and received his M.D. degree from Harvard University in 1930. He has been assistant professor of pathology ‘at the Harvard Medical School since 1941. He is pathologist to the Boston Lying-in Hospital and to the Free Hospital for Women in Brookline. His work has been in pathology with relation to obstetrics and gynecology, and in the embryology and pathology of the early human ovum.
This paper is from the Pathological Laboratories of the Boston Lying-in Hospital, the Free Hospital for Women, Brookline, the Departments of Obstetrics and Pathology, Harvard Medical School, Boston, and the Department of Embryology, Carnegie Institution of Washington, Baltimore.
Introduction
Certain organs and tissues run a complete life cycle during pregnancy, appearing, fulfilling their function, aging, and disappearing within a short but variable period of time. The exocoelomic membrane, for example, is present in the human ovum from the ninth to the thirteenth day, reaches its peak at the twelfth day, then rapidly declines. The yolk sac has fulfilled its imperfectly known purpose by the end of the eighth week, while the placenta has a definite function lunar months, then is discarded at birth. Dr. Hertig discusses the process of aging in these organs and the relation of embryology to gerontology.
Ordinarily one does not associate the field of embryology with that of gerontology since the beginning and ending of life would seem, at first glance, to be diametrically opposed. If, however, one regards the fertilized human ovum as a whole, whose origin begins in the respective gonads of its parents and whose extrauterine life is largely a function of the original vitality with which it is endowed, then it becomes apparent that the problems of embryology and gerontology are more closely correlated. Such a point of View is admirably expressed by the eminent embryologist, George L. Streeter (1) who says, “The importance of quality of the egg, and of course I include both its maternal and paternal elements, is not limited to uterine life. Whether the infant survives its first year depends in considerable part on the original quality of the egg. If they withstand the usual Wear and tear of life until between fifty and sixty years they conform to the actuary’s expectation of life at birth—and to the embryologists’ expectation of the performance of an egg of average quality. It is only the extraordinarily good egg that is still going strong at 80 years, and we see him (or her) do this in the absence of any exquisite hygienic regime or environmental favor.”
Streeter further emphasizes that the total quality of such an egg is made up of the quality of the separate parts or organs and that any one of the latter may wear out or age prematurely and cause the individual’s death While the other organs are still capable of continued function. Thus this author goes on to state, “It is also probable that degenerative diseases of the heart and blood vessels are to be explained as well by the constitution of the germ-plasm with which the individual started life (that is, the inherited vulnerability of his Vascular system) as by the much blamed overwork, overeating, oversmoking and overdrinking, which in other individuals do not result in a comparable damage.”
One might infer from this point of view that the relation of embryology to gerontology lay entirely in the realm of quality of germ-plasm and its effect on the extrauterine life of the individual. That such is not the case is shown by the fact that many fetal organs, or portions thereof, involute or age during intra-uterine life.
Examples of such a process are seen in the development and involution of the pro- and mesonephros, both purely fetal organs, which play a functional role in the development of the embryo. That such a structure probably functions, albeit temporarily, is predicted by Flexner’s (2) biochemical studies on the metanephros, (successor to mesonephros) of the fetal pig. This author concluded that “1) In pre secretory stage, cytochrome oxidase is in low concentration and equally distributed between epithelium and stroma. In secretory stage, oxidase increases markedly in epithelium and disappears from stroma. 2) -In pre-secretory stage, redox potentials of epithelium and stroma are alike. With onset of secretion, the potential of epithelium rises and that of stroma falls so that a difference of 0.230 volt develops between them.” Whether similar studies on the proand mesonephros would show similar results or not is impossible to say, but studies such as the above do correlate biochemical changes with morphologic evidence of maturation and function. These biochemical techniques have also been applied by Flexner, Flexner, and Strauss (3) to the developing cerebral cortex of the fetal pig with results comparable to those obtained on the metanephros.
In discussing this matter with Louis B. ‘Flexner he defined aging as “the sum total of those processes in an individual which lead to death.” Furthermore, if one considers the rate of aging as including the rate at which morphologic, physiologic, and biochemical changes take place, it is apparent from the studies of Flexner and his coworkers that the individual ages most rapidly when he is the youngest, namely, during the embryonic stage.
The intra-embryonic coelomic tract is yet another structure that has a temporary but distinctly functional life span. Likewise, the cells which form the lens vesicle of the eye involute and are extruded once their function has been completed (Streeter, 4). Were it not for this fact, it would be impossible for light to be transmitted through the crystalline substance of the lens. Another temporary but probably functionally useful structure is the neurenteric canal of the late presomite embryo.
Such examples as the above are given merely to indicate that various structures in the embryo itself have a temporary but functional life span. None of these alterations are, however,/a/s striking as the involutionary processes which go on in the placenta and its associated structures—an organ whose entire life cycle takes place during the intra-uterine life of the individual.[1] It is the purpose of this review to describe and, insofar as is possible, to interpret the major involutionary changes that occur in the placenta and some of its associated structures. These will be considered under the following headings: (1) the exocoelomic membrane, (2) the yolk-sac, (3) physiologic aging, and (4) pathologic aging in the placenta.
I wish to acknowledge with gratitude the cooperation of my various colleagues in the preparation of this review: to Drs. George B. Wislocki, H. Stanley Bennett, and Edward W. Dempsey for allowing me to use the original plates of their respective publications some of which are shown in sections 3 and 4, to Drs. George L. Streeter and Louis B. Flexner for helpful discussion and suggestions with respect to aging in the fetal period, and to Chester Reather and Richard W. St. Clair for making some of the pictures here used. I am furthermore greatly indebted to W. B. Saunders Company of Philadelphia and to Dr. Thomas S. Cullen for their permission to reproduce Figures 1, 2, 3, 4, 5, 8, 11, 31 and 32 from the latter author’s book "The Umbilicus and its Diseases.” To Dr. E. J. Farris of the Wistar Institute of Anatomy and Biology I am grateful for permission to reproduce some of the illustrations from the various publications of Wislocki, Bennett and Dempsey (see bibliography for references and figures for specific illustrations used).
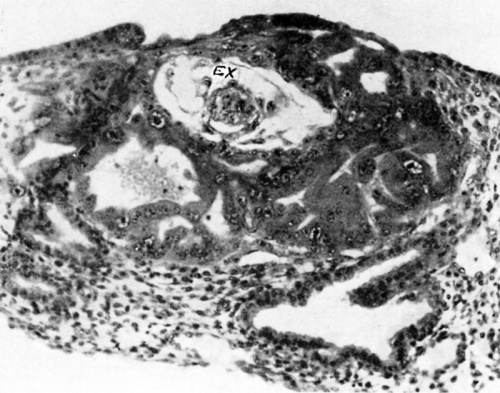
Fig. 1. A human ovum of approximately 8-9 days of age. It is barely implanted within the endometrium, the epithelium of which is attempting to repair the defect in its surface. The thick trophoblastic shell contains lacunae into which flows maternal blood. The central or chorionic cavity contains a globular, bilaminar embryonic disk to which is attached the recently developed exocoelomic membrane. The latter thus surrounds the small irregular exocoelom designated as EX. Carnegie 8215, section 12-5-1, X200.
The Exocoelomic Membrane
One of the most evanescent structures of the developing chorion (placenta) is the thin mesothelium-like membrane which is present in the human ovum from the 9th to the 13th days (Figures 1-3). This membrane lines the chorionic cavity (except where the latter is bounded by the embryonic disk) during this period of development. Conversely it forms the boundary of the exocoelomic cavity—hence its designation as the exocoelomic membrane. Its presence in the developing primate ovum (macaque) was first described by Heuser (5) in 1932 and more fully elaborated by the same author in 1938 (6). Its presence in the developing human ovum was first noted by Stieve (7) in 1926 in describing the “Werner” embryo although that author interpreted the structure as a yolk-sac. Hertig and Rock (8) in 1941 described two previous human ova of about eleven and twelve days of age, both of which showed extensive development of this membranous structure. In that paper, the authors discuss at some length the problem of whether or not this membrane constitutes the early phase of yolk-sac formation. In the opinion of Hertig and Rock it does not although Lewis (9) intimates that embryos of this age should have a large yolk-sac. Actually the human yolk-sac apparently does not begin to develop until late in the thirteenth or early in the fourteenth day as it does in the macaque monkey as shown by Heuser and Streeter (10) although the exact stage or stages in man have not yet been observed. Thus the yolk-sac appears to begin its development about the time the exocoelomic membrane has reached its zenith and is degenerating. For fuller discussion of this problem and the details of the membrane’s development see Hertig’s (11) recent paper.
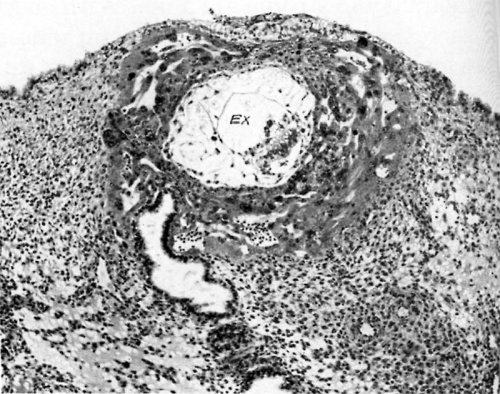
Fig. 2. A human ovum of approximately 11 days of age. The endometrial epithelium has superficially repaired the defect made by the implanting ovum. The exocoelom (EX) is now a much more definite space and is bounded below by the primitive entoderm of the bilaminar embryonic disk. Elsewhere it is surrounded by the sharply delineated exocoelomic membrane. Note the multiple gossamer attachments of the membrane by mesoblastic cells to the surrounding trophoblast from which it originally arose. Carnegie 7699, 8-5-3, X100.
For the purposes of this review the development and involution of the exocoelomic membrane may be treated very briefly. The first appearance of this structure is in ova of approximately 8 days of age or in their 9th day of development. Such a specimen is seen in Figure 1. It contains a thin, irregular and in places imperfect membrane within the central cavity (chorionic) of the ovum. Most of the cavity is occupied by the embryonic disk but to the edge of the latter is attached the exocoelomic membrane. It arises from primitive mesoblastic or mesenchymal cells which in turn delaminate in situ from the adjacent cytotrophoblast of the chorion. As the individual mesoblastic cells continue to differentiate, the older ones join with their fellows to form a membrane-like structure. This process tends to separate the central portion of the chorionic cavity—now called the exocoelom—from the remainder of the cavity. Since the exocoelomic membrane is never entirely perfect, the two portions of the cavity intercommunicate—especially in the earlier phases of the membrane’s existence.
Figures 2 and 3, of an 11- and a 12-day human ovum respectively, show later and more readily recognized stages in the development of the exocoelomic membrane.
These specimens have previously been described by Hertig and Rock (8). In the eleven day stage the membrane is well developed and sharply delimits the exocoelom from the surrounding chorionic cavity although the former is relatively small as compared to the latter. Even in this early stage of development, however, there appears to be a stainable difi"erence between the fluid in the exocoelom and that in the intercellular spaces of the chorionic mesoblast. Thus, the fluid within the exocoelom stains more darkly and with fewer precipitated granules—presumably of protein—than does the fluid outside. Whether this bespeaks a difference in function of the two fluids or not is impossible to say. However, the exocoelom is much more intimately associated with the embryonic disk—being bounded by the primitive entoderm of the latter—so that in all probability the exocoelomic fluid possesses some functional as well as structural significance.
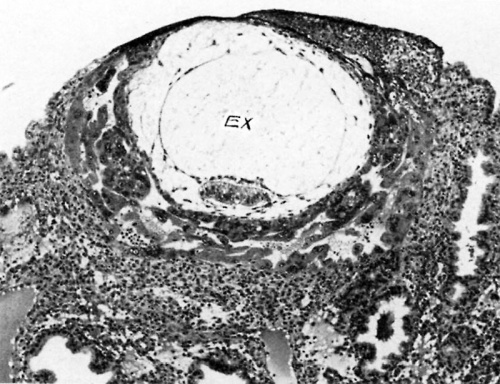
Fig. 3. A human ovum of approximately 12 days of age. The exocoelom (EX) now fills amost the entire chorionic cavity although its membranous envelope is being stretched to near the breaking point as a consequence of which it is becoming thinned out and attenuated. This stage precedes by some hours that of the 13% day ovum seen in fig. 4 at which stage the exocoelomic membrane has fragmented. Carnegie 7700, section 6-1-5, X100.
The peak in the development of the exocoelomic membrane is seen in the 12-day ovum (Figure 3). At this stage, the exocoelom is relatively enormous and occupies most of the chorionic cavity. There is still a stainable difference between the fluid within the exocoelom and that outside. In this respect, the present stage resembles that of the previous day (Figure 2). The exocoelomic membrane itself, however, begins to appear thinner and more frayed-out as though the growth of the ovum had stretched it nearly to capacity. That this could be the case is shown by the mesoblastic attachments of the membrane to the surrounding chorion from which it originally arose. Hence, with continued growth of the ovum as a whole, more and more stress is put upon the gossamer exocoelomic membrane. Whatever the mechanical or functional forces are, the wall of the exocoelom soon bursts—a good deal as though it were a tiny balloon whose stress had ex ceeded its strain. Unfortunately, no speci mens are available, either to the author or in the literature, that depict this cataclysm, although the ova showing early yolk-sac development usually contain remnants of the now defunct membrane. An example of this stage in human development is seen in Figure 4, an ovum about 13% days of age or its 14th day of development.[2]
|
|
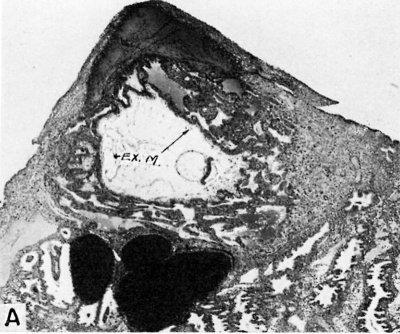
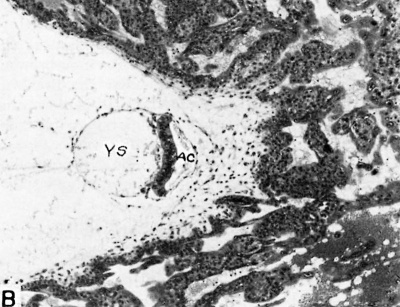
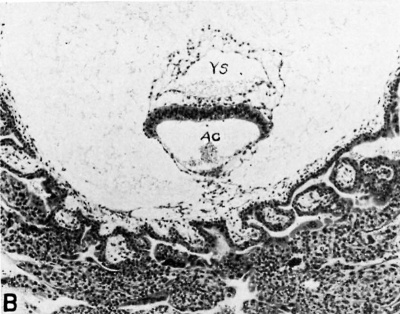
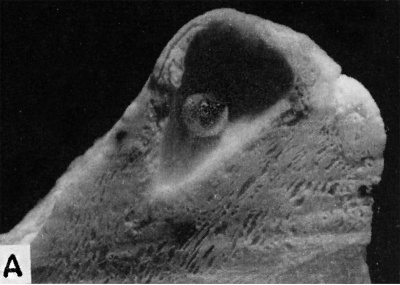
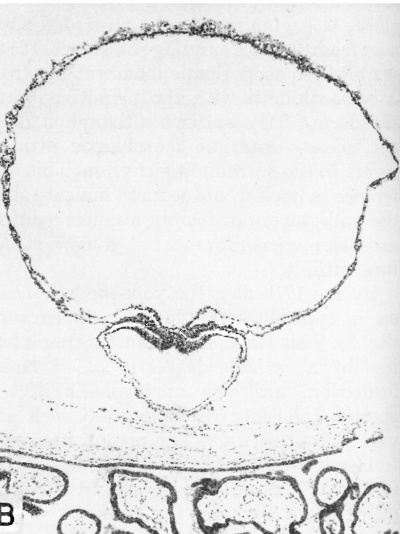
| Fig. 4.A. A human ovum of approximately 13.5 days of age. The hemorrhagic closing cap is seen above the ovum. The chorion possesses simple unbranched villi. The chorionic cavity contains an eccentrically situated embryo (see fig. 4B for details of the latter) and the remnants of the exocoelomic membrane (EX. M). The endometrium shows an early decidual reaction about the'ovum. The gland below the ovum contains extravasated blood. Note large sinusoid in the endometrium to the left of the ovum. Carnegie 7801, section 12-1-1, X35. | Fig. 4.b. Section through embryo of 13.5 day human ovum. Note the single-layered yolk-sac (YS) to the left of the embryonic disk and the amniotic caivity (AC) to the right. Note early connective tissue in base of simple chorionic villi. The blood at-the right is within the intervillous space of the early placenta. Carnegie 7801, section 12-1-3, X100. |
Thus does a temporarily useful structure develop, age, and die, all within a life span of 4 or 5 days. That it must perform some physiologic as well as structural function cannot be denied although its exact nature must await further study.
The Yolk-Sac
As indicated in the section on the exocoelomic membrane, the exact time at which the human yolk-sac begins to develop and the manner in which it arises are unknown. That it undoubtedly develops when the ovum is about 13 full days of age, or in its fourteenth day of development, is shown by the fact that it has not yet appeared in the 12-day specimen (Figure 3) although it is a definite entity in the 13%day specimen seen in Figure 4. As noted before, Heuser and Streeter (10) have shown that in the macaque monkey the yolk-sac begins to form in the 12-day stage when the exocoelomic membrane is still a well developed structure. Whether the human exocoelomic membrane, by its disruption, assists in the formation of the yolk-sac or whether it forms in an independent manner, similar to that of the monkey, must await the securing of specimens of the late 13th and early 14th day.
The youngest human ovum to show a definitive yolk-sac is the famous specimen described by Linzenmeier (12) in 1914. Although this specimen is neither fixed nor stained as perfectly as some of the more recently acquired human ova, it still shows a tiny, cleft-like yolk-sac continuous with the ventral aspect of the primitive entoderm. Remnants of the exocoelomic membrane are present as they are in older speciments of 14-16 days of age. For a pictorial representation of early yolk-sac development the reader is referred to Hertig and Rock’s (8) paper on the 11~12 day human ova.
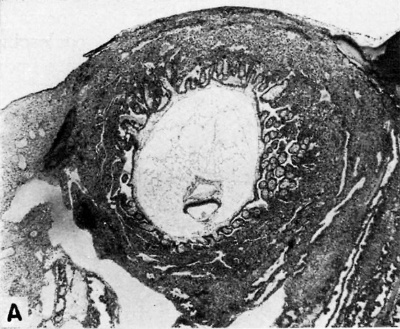
Figures 4 and 5 represent sections from the only normal specimens containing an early yolk-sac which are available to me for study. Complete descriptions of these ova have recently (1945) appeared in the paper by Heuser, Rock and Hertig (13).
The yolk-sac of the 14th day (fig. 4) possesses a single mesothelial layer which in places is beginning to take on added support from the surrounding mesoblast. It resembles the exocoelomic membrane in that it is continuous with the primitive (gut) entoderm of the embryo although it does not possess multiple mesoblastic attachments to the surrounding chorion. This difference in itself would seem to indicate that the yolk-sac is not merely a much-reduced exocoelomic membrane but a new structure entirely.
By the 17th day, the yolk-sac has taken on a typical double-layered appearance (fig. 5). This stage, in general, is characterized by a variable degree of blood vessel formation, although angiogenesis in the chorion (placenta) is moderately well advanced by this time. The reader is referred to Hertig’s (14) paper published in 1935 for further details on angiogenesis in the early primate chorion and for a review of the literature on blood vessel formation in the yolk-sac.
|
|
| Fig. 5.A. A human ovum of approximately 16%days of age. The chorionic villi are still simple, unbranched structures. The chorionic cavity contains a normally-situated embryo, the details of which are seen in fig. 5B. No trace of the exocoelomic membrane remains. Carnegie 7802, section 44-3-5, X30. | Fig. 5.B. Section through embryo of 16%-day human ovum. Note double-layered yolk-sac (YS) above and amniotic cavity (AC) below the embryonic disk. The amnion is lightly attached by mesoblastic tissue to the chorionic membrane whose simple villi are continuous with their trophoblastic cell columns. The latter are continuous with the surrounding endometrium although this feature is not seen in this picture. Carnegie 7802, section 44-3-5, X100. |
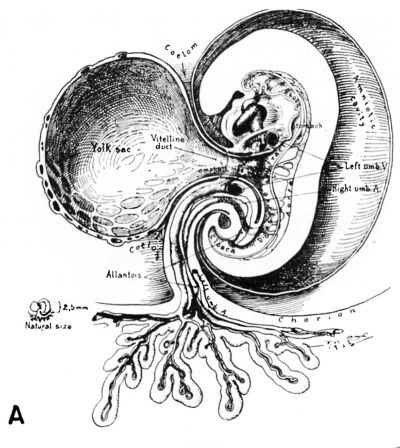
By the end of the third week, embryos of 17 to 21 days of age (Figures 6 and 7) possess a very prominent yolk-sac whose ventral wall is studded with small isolated blood islands. These structures are composed of thin, capillary-like structures containing nucleated red blood cells. Such discontinuous vessels are in the process of forming a vascular system by coalescing with those in the embryo proper, its body stalk, and chorion (placenta). The superb picture by the late Max Brodel, drawn for Cullen’s classic work on the umbilicus (15) shows the general features of such embryos (Figure 6). Figure 7, representing a 1.6 mm. embryo from The Carnegie Embryological Collection, shows an actual specimen of this general age. It is probably about 21 days of age and represents the stage of development just before the blood begins to circulate. Reconstructions of the chorionic vascular system at this stage done by Hertig (14) Show that the blood vessel primordia are discontinuous, devoid of blood cells and hence not yet capable of normal circulation.
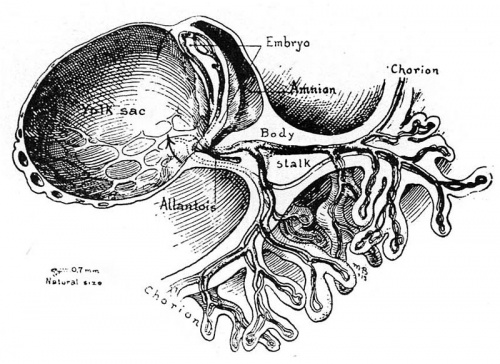
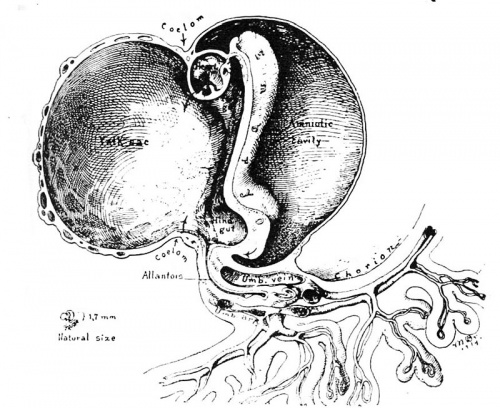
Fig. 6. A drawing of a 0.7 mm. human embryo, about 17 days of age taken from Cullen’s “The Umbilicus and Its Diseases,” W. B. Saunders Company. This figure, depicting the right half of the embryonic mass shows the circulation in the yolksac, embryo, body‘stalk and chorion as anatomically complete although later studies show that such a state does not actually occur until about 3 days later. Note the relatively enormous size of the yolk-sac with respect to the embryo and its amnion. Only the portion of the chorion or outer shell of the ovum near the body stalk is represented, the entire chorion at this stage measuring about 8.0 mm. in diameter.
|
|
| Fig. 7.A. A human embryo of approximately 20 - 21 days of age, measuring 1.6 mm in length. Fig. A. represents the chorion embedded within the endometrium and opened in such a way as to reveal the entire embryo when viewed from its left side. The yolk-sac is the most prominent feature of the embryo and is seen as a balloon-like mass whose distal surface is covered with blood islands. The embryonic disk itself together with its amnion is the thickened opaque mass where the embryo is attached to the chorion. See fig. B. for morphologic details of the embryo. Carnegie 7545, sequence 7, X5. | Fig. B. A cross section through the middle of a 20 - 21 day embryo. Note the enormous size of the yolk-sac containing blood islands Within its doublelayered wall. The embryo proper is the trilaminar mass between the yolk-sac and amniotic cavity and consists, from within outward, of primitive or gut entoderm, mesoderm and ectoderm. A portion of the chorion is seen below. Carnegie 7545, section 6-2-2, X60. |
According to Stieve and Strube (16), who studied the yolk-sac circulation by reconstructing two 17-somite embryos, the blood begins to circulate at about this stage of development, since one of their specimens showed a completed vascular system while one did not. Therefore, since Figure 8 represents a 1.7 mm. embryo possessing 7 somites, originally described by Dandy (17), it is clear that this specimen is just on the border line of possessing a functional circulation. Dandy was not able to show an anatomically complete set of blood vessels in the yolk-sac of his specimen,‘although it was complete in the embryo and chorion. It is generally agreed, during this phase of active vascularization of the yolk-sac the latter undergoes a complex process of growth and differentiation which results in a portion of its lining (the true gut entoderm—as contrasted to the yolk-sac entoderm) being transformed into the primitive embryonic gut. This series of events can best be followed in Figures 6-11. Figures 7 and 10, representing actual specimens, are best consulted to see the morphologic difference between gut entoderm, a portion of the original embryonic disk, and yolk-sac entoderm which lines the yolk-sac itself and is formed later.
5 therefore, that in man the circulation begins to function when the embryo possesses from 7 to 17 pairs of somites sometime dur ing its 4th week of development (6th week menstrual age). Several additional examples of this important stage of development are seen in Figures 9—11.
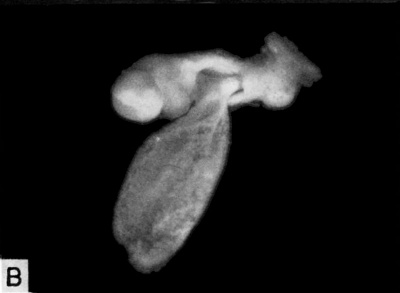
Fig. 8. A drawing of the right half of a 7 somite (1.7 mm.) embryo of the 4th week of development. At this stage, the blood vessels in the embryo, body stalk and chorion are anatomically complete although not quite so in the yolk-sac. The yolk-sac at its junction with the embryo is beginning to eonstrict and is associated with the invagination of the gut entoderm to form the fore and hind gut. (Fig. 2 from Cullen’s “The Umbilicus and Its Diseases,” W. B. Saunders Company.)
|
|
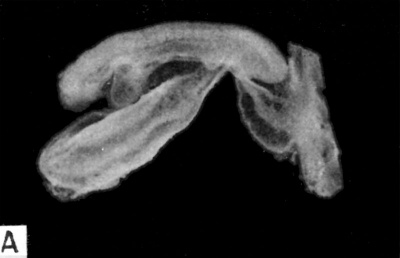
| Fig. 9.A. A drawing of the right half of a 2.5 mm embryo representing a stage in the middle of the 4th week of development. The circulation is complete and the heart functions at this stage. Note the constriction of the yolk-sac to form the omphalomesenteric or vitelline duct as the digestive tract becomes more mature. The growth of the embryo and its surrounding amnion have resulted in the partial approximation of the body stalk and the vitelline duct so that these structures are being combined to form an early umbilical cord. (Fig. 3 from Cullen’s “The Umbilicus and Its Diseases,” W. B. Saunders Company.) | Fig. 9.b. A 3.2 mm embryo with amnion removed as viewed from below and to the left showing the omphalo-mesenteric duct and its connection with yolk-sac and the primitive embryonic gut. Note the communication between the exocoelomic space around the yolk-sac and the coelom (body cavity) within the embryo. Carnegie 6488, sequence 2, X15. 104 |
|
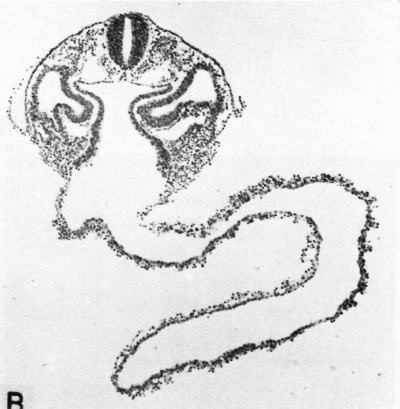
|
| Fig. 10.A. A 3.5 mm. embryo possessing 13 somites and representing the middle part of the fourth week of development. The amnion has been removed so that the approximation of the bodystalk and omphalo-mesenteric duct are less evident than in Fig. 11. This embryo is apparently a little longer than its developmental age would allow because its natural curvature is less, perhaps from having had the amnion removed. Carnegie 6344, sequence 4, X15. | Fig. 10.B. A cross section through approximately the middle of the embryo seen in Fig. A. Note the blood islands and vessels in the wall of the yolk-sac and the communication of the latter with the primitive gut of the embryo. The large spaces on either side of the gut are those of the coelom or body-cavity of the embryo. Carnegie 6344, section 3-7-11, X75. |
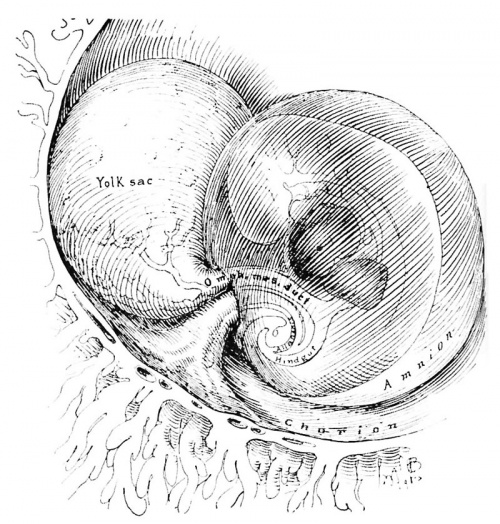
Accompanying the changes which result in the formation of the primitive gut are a series of changes resulting in the growth of the embryo, its assumption of embryonic form and the metamorphosis of the body stalk into a more definitive umbilical cord. The latter results in a gradual restriction and elongation of the connection between the yolk-sac and primitive gut known as the omphalo-mesenteric duct (Figures 9A11). With further "growth of the amnion, the omphalomesenteric duct is gradually incorporated into the early umbilical cord as shown in Figure 11.
Fig. 11. A drawing or a 6.9 mm embryo in its 4th week of development drawn somewhat diagrammatically—to show the further approximation of the omphalo-mesenteric duct and the bodystalk due to the expanding omnion. (Fig. 4 from Cullen’s "The Umbilicus and Its Diseases,” W. B. Saunders Company.) Note that the yolk-sac is now almost as large as the amnion, the former now approaching its period of maximum activity.
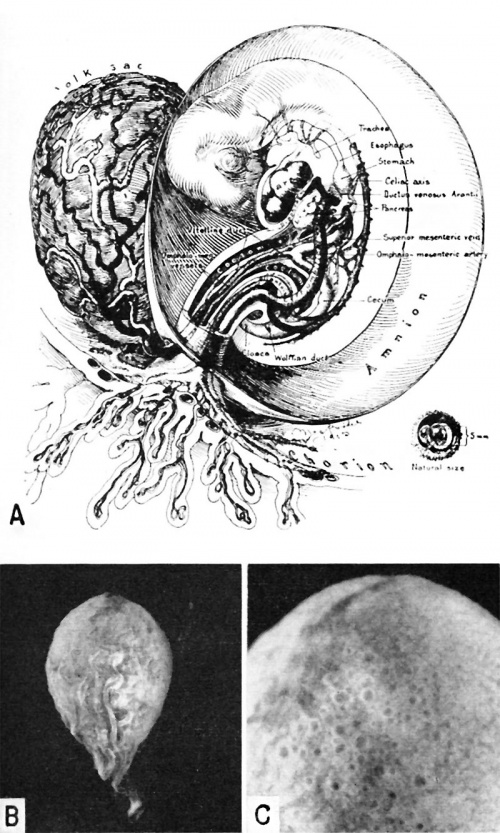
In embryos of the late 4th, 5th and 6th weeks of development (6th to 8th weeks menstrual age), the yolk-sac is a prominent structure with an active circulation and a prominent epithelial lining. The latter gives rise to tubular and cystic structures which were investigated by Meyer (18) in 1904. These gland-like spaces, best seen in Figure 12 C, begin to appear in embryos of 2-3 mm. and occasionally in younger specimens. However, they are most prominent and are usually present in embryos of 10— 20 mm. (5th and 6th weeks of development), two examples of which are seen in Figures 13 and 14. Their function is not understood but, since they are present during the period when the yolk-sac is largest and most prominently vascularized, one must assume that they possess some function. It is probable that they present a much larger surface area of yolk-sac epithelium to the rich vascular plexus than would the epithelial fining by itself. Whether the secretion of the yolk-sac has any specific function or not is unknown, but it probably does because of this mechanism for in creased absorption during the period of greatest yolk-sac activity.
Fig. 12. A. A drawing of a 5 mm. embryo (late 4th or early 5th week of development) showing further development of the umbilical cord. The yolk-sac is large, well vascularized and its wall contains numerous cystic and tubular spaces derived from the lining epithelium. (Fig. 5 from Cullen’s “The Umbilicus and Its Diseases,” W. B. Saunders Company.)
B. A photograph of the yolk-sac of a 6.7 mm. embryo. Carnegie 7522, seqtience 9, X8.
C. A higher power photograph of the same yolksac as seen in Fig. B. to show the numerous cystic and gland-like spaces in its wall. Carnegie 7522, sequence 7, X22.
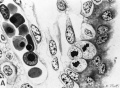
Fig. 17. A. A cytologic drawing of a section from a 30-day human chorionic villus (Fig. 4 from Wislocki and Bennett, American Journal of Anatomy). The thick syncytium is seen covering the actively growing Langhans epithelium..The loose immature connective tissue contains a thin-walled capillary within which are seen fetal blood cells. Note that the erythroblasts are frequently nucleated. Iron hematoxylin, X1600.
B. A histologic drawing of a mature human chorionic villus (Fig. 17 from Wislocki and Bennett, American Journal of Anatomy). The syncytium is now much thinner, especially overlying the dilated fetal capillaries. Langhans cells are absent in this section although mature villi still contain a few scattered ones. The stroma is dense, the capillaries numerous, the latter containing many non nucleated fetal erythroblasts. Mallory’s connective tissue stain, X1600.
Physiologic Aging of the Placenta
In any discussion of aging in a given tissue or organ, cognizance must necessarily be taken of the morphologic changes associated with normal maturation in the organ. Insofar as possible such changes should be correlated with function. The placenta lends itself to such studies and therefore the morphologic and function changes associated with physiologic aging of the placenta will be briefly considered.
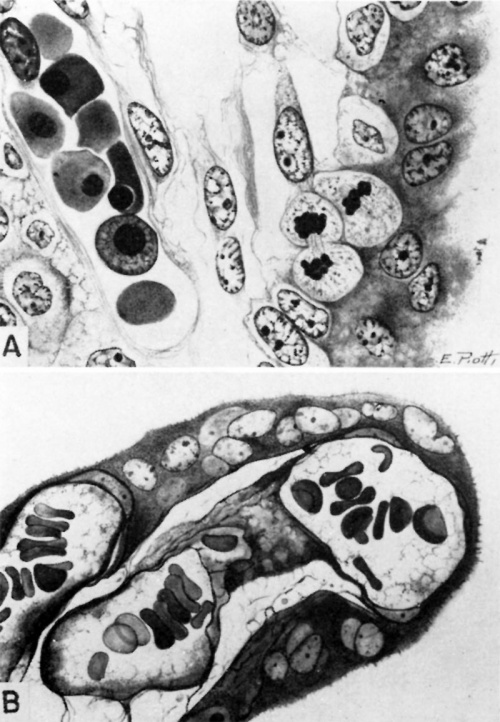
Fig. 18. Lipoid in the syncytium of human chorionic villi at various stages of development (figs. 9, 10 and 21 from Wislocki and Bennett, American Journal of Anatomy).
A. Villus at 30 days, fixed in Champy’s fluid but.otherwise unstained. Note large black lipoid droplets in the syncytium and their absence in the underlying Langhans cells. A vacuolated Hofbauer in the stroma is seen to the left. X1600.
B. Portion of chorionic villus from 13-weeks placenta fixed in Champy’s fluid but unstained. Note thinner syncytium, smaller ljpoid droplets and irregularly arranged Langhans cells. X1600.
C. Villus at term, fixed in Champy’s fluid but unstained, showing-thinned-out syncytium which still contains. lipoid although in. reduced amounts. X 1600.
Fig. 19. Syncytium from human placenta at various stages of gestation, stained by the Altmann-Kull method for mitochondria (figs. 13, 14 and 19, from Wislocki and Bennett, American Journal of Anatomy).
A. Villus from 30-day placenta. Note the large numbers of rod-like mitochondria in the syncytium and the lesser numbers in the Langhans cells. X1600.
B. Villus from 13-weeks placenta showing the continued abundance of smaller mitochondria in the thinner syncytium. X 1600.
C. Villus at term showing continued presence of mitochondria even though the syncytium is very thin. Note fuzzy or “stubble-like” border on surface of villus. X1600.
The most recent and complete histologic and cytologic studies on the maturation of the placenta are reported in the classic paper by Wislocki and Bennett (19). They have kindly allowed me to use some of their original illustrations for the purpose of this review (Figures 17—20) .
As a generalizing statement, it may be said that as the placenta matures it forms a larger number of smaller and thinnerwalled units, the chorionic villi, by means of which the maternal blood is made more efliciently available to the fetal blood within the placenta. Stated differently, the maturing placenta, in proportion to its volume, presents more of its surface to the maternal blood. These changes are vividly contrasted in Figure 17 which shows the relative thickness of the various layers through which any given substance must go in passing from the maternal to the fetal circulation. Thus in a 30 day placenta (Figure 17 A) a thick layer of syncytium and Langhans epithelium covers a relatively large villus‘ whose capillaries are often at some distance from its surface; whereas at term the villus (Figure 17 B) is much smaller, thinner-walled, more richly vascularized and present in larger numbers.
Functional studies by Flexner and his associates on placentas of different animals (20), as well as in the human (21), have borne out this concept of increasing efficiency as term approaches. Thus Flexner and Gellhorn (20) state, “The rate of transfer of sodium per gram of placenta increases in each animal as gestation proceeds until just before term, at which time there is a sharp decrease. The variation in rate of transfer across the placentas has been correlated with morphologic change in the placenta.”
As gestation progresses, the syncytial covering of the villi becomes thinner and thinner, a fact long observed, although its cytological structure has not been fully studied until Wislocki and Bennett’s work (19). The syncytium throughout pregnancy contains lipoid droplets (Figure 18) although they become smaller and less numerous as this epithelial structure matures. Wislocki and Bennett (19) in their summary state, “The association in syncytium of fat droplets and birefringent substances soluble in acetone, and of acetone-soluble substances capable of reacting with phenylhydrazine to form yellow phenylhydrazones suggests that the syncytium is the site of formation of placental steroid hormones.” The latter include estrogen and progestin. The origin of the chorionic gonadotropic hormone, on the other hand, has been tentatively linked with the placental cytotrophoblast by these authors upon the basis of their own work and a review of the literature.
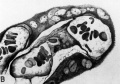
Fig. 20. Cytotrophoblast from human placenta (figs. 27, 40 and 42 from Wislocki and Bennett, American Journal of Anatomy).
A. The cytotrophoblast from a cell column of a 30-day placenta. Note close approximation of vacuolated cells with beginning formation of ground substance between. Iron hematoxylin, X1600.
B. A panchromatic photograph of a colored drawing made from a cell island of a 13-weeks placenta stained with Azan. The ground substance (originally blue) occupies most of the intercellular space but fibrin (originally red) appears as smaller darker masses throughout this substance. X1600.
C. A panchromatic photograph of a similar cell island from a 13-weeks placenta also stained with Azan. Note the lighter, larger homogeneous areas of fibrin (originally stained red) and the denser intercellular ground substance (originally stained blue). X225.
Fig. 21. Human chorionic villi of various ages showing the distribution of gradually increasing amounts of “fibrinoid” as the placenta ages. Note decreasing edema of villous stroma and size of villi as the placenta matures. All villi magnified 100 times.
A. Chorionic membrane and villi of 19-day ovum. Note the loose connective tissue stroma and double-layered epithelium. “Fibrinoid” is not yet present. Carnegie 5960.
B. Chorionic membrane and villi of 11-weeks placenta. Note beginning fibrinoid degeneration of trophoblast in large villus at right. (Figs. B. C. & D. from teaching collection at Free Hospital for Women.)
C. Villi from 16-weeks placenta. Note increasingly prominent dark masses of “fibrinoid” which project from the surface of villi in the upper portion of th picture.
D. Villi from term placenta showing the prominent dark masses of “fibrinoid” associated with the old villous branch running diagonally across the picture.
There has been considerable discussion in the literature as to whether the placental barrier is a pure semipermeable membrane or whether the placenta also has selective ability to pass certain substances and retard or reject others. Wislocki and Bennett (19) review the pertinent literature and conclude from that as well as their own work that the placental barrier is “more than a simple semipermeable membrane operating solely by diffusion and osmosis.” In keeping with such a concept is the presence of mitochondria in all stages of syncytial development—even at term (Figure 19) when the syncytium overlying the dilated capillaries has been regarded by Bremer (22) (quoted by Wislocki and Bennett) as being physiologically inert.
As the cytotrophoblast[3] of the placenta matures, certain invariable changes take place with the resultant formation of various amounts of "fibrinoid,” or ground substance and fibrin. Wislocki and Bennett (19) have discussed these changes fully, pertinentstages of which are seen in Figure 20. These authors believe that, “this ground substance is a protein-containing fluid of mixed origins, derived mainly from the decidua and maternal blood stream, but also substances released by the cellular trophoblast. This ground substance contains no argyrophile or collagenous fibers. Fibrin becomes variously deposited in it.”
This progressive physiologic aging of the placenta is discussed, not only for its intrinsic importance with respect to placental function but also because of its relation to changes of pathologic significance which will be considered in the closing section of this review.
Pathologic Aging in the Placenta
As was pointed out in the previous section, the development of “fibrinoid” or ground substance, together with the resultant deposition of fibrin, is a concomitance of physiologic aging of the placental cytotrophoblast. This substance occurs in the various places where such tissue either has been or persists throughout gestation. It occurs, for instance, as focal accumulations beneath the syncytium of older villi such as seen in Figure 21. Such a locus of deposition represents the site at which the Langhans epithelium had been present earlier, only to disappear with maturation of the villus. Hence, “fibrinoid” is usually found along the main villus stems or major branches thereof——the oldest part of the villus—but is much less common in the small terminal branches that continue to form throughout pregnancy.
Dempsey and Wislocki (23) have shown that as the placenta ages basophilia of the syncytium decreases whereas its alkaline phosphatase increases (Figure 22). Their studies prove that the basophilia is due to the presence of ribonucleic acid, and that such a substance (considered as a phos phoric ester) could be regarded as a possible substrate for phosphatase. These authors therefore suggested as a possibility, “-that an enzymatic hydrolysis of ribonucleic acid occurs in the syncytial trophoblast and that the decrease in basophilia as pregnancy advances is caused by its more rapid hydrolysis by the increased amount of phosphatase.”
Dempsey and Wislocki indicate further that phosphatase may play a subsidiary role in the calcification of certain portions of the placenta (Figure 23). Such areas follow closely the distribution of the “fibrinoid” deposits described above. Since such areas contain phosphatase as well as glycogen—the latter a readily available source for the production of glyco-phosphoric esters serving as a substrate for phosphatase in the calcifying process (Harris, 24) quoted — by Dempsey and Wislocki)—the basis is laid for calcification of placental “fibrinoid.”
Fig. 22. Chorionic villi from human placentas of various ages to show increasing amounts of alkaline phosphatase in the syncytium as the organ matures. (Figs. 10, 11 and 12 from Dempsey and Wislocki, American Journal of Anatomy.) All villi magnified 235 diameters.
A. Villi of 8 weeks. Note localization of phosphatase to outer portion of syncytium.
B. Villi of 16 weeks. Note rather uniform distribution of phosphatase in syncytium although it is somewhat more dense in the outer portion of the latter.
C. Villi at term. Note that the phosphatase is localized to the syncytium but tends to assume an inner and outer concentration even within that single layer of tissue.
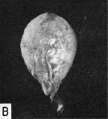
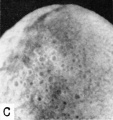
Fig. 23. Calcification in the mature human placenta. Material is from the Boston Lying-in Pathological Laboratory.
A. A portion of the maternal surface of a placenta, photographed under water, to show the fine, lace-like pattern of calcification. About half natural size.
B. A photomicrograph of the entire thickness of a placenta to show distribution of dark calcified “fibrinoid” associated with the larger villous stems. Note that the calcification is present throughout the placenta although its presence adjacent to the maternal surface (lower portion in picture) enables it to be seen and felt in the intact specimen as in Fig. A. X5.
C. A more highly magnified portion of the maternal surface from Fig. B to show the calcified fibrinoid. This picture represents the portion in Fig.3? enclosed in the small rectangle. X60.
Calcification is not a constant phenomenon in the placenta although its precursor —“fibronoid”—is always found, ‘albeit in variable but increasing amounts from about the 10th week until term. Since it involves the cytotrophoblast and the older supporting elements of the villus, calcification has no observable effect upon the function of the placenta as the small villi are usually spared.
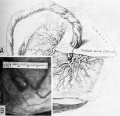
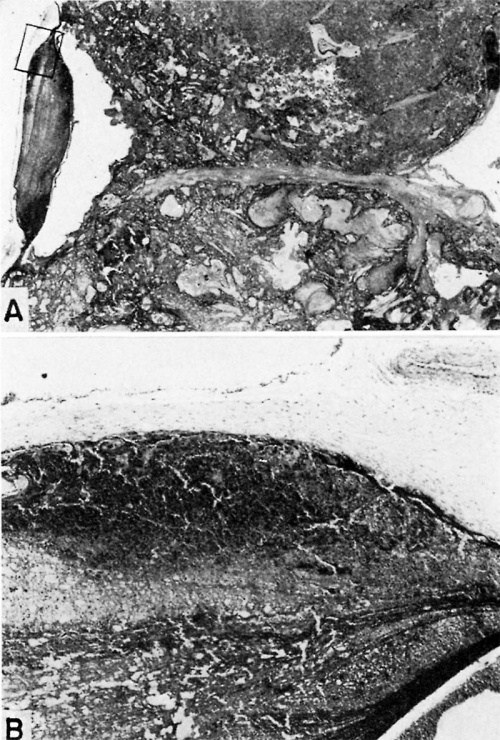
Fig. 24. A human placenta at term showing “fibrinoid” degeneration which has resulted in thrombosis of the intervillous space leading to infarction of the placenta. B. L-i. H., S-32-7'4.
A. The edge of a mature placenta—fetal surface on top and maternal surface on bottom—showing a laminated fibrin thrombus which appears to have been deposited as the result of “fibrinoid” degeneration of villi and trophoblast on maternal surface. Surrounding the thrombus and spreading out in a fan- or wedge-shaped area is an area of infarcted placenta whose blood supply has been cut off by the thrombosis of the intervillous space. Normal placental tissue is seen beyond. X5.
B. A higher power detail of rectangular area seen in A. Note “fibrinoid” degeneration in the center, thrombus on the right, necrotic villi at top left and normal villi bottom left. X60.
There are, however, pathological sequelae attendant upon the presence of “fibrinoid” which is apparently the etiological factor in the breakdown of the overlying syncytium. When this occurs maternal fibrin is deposited upon the denuded surface resulting in obliteration of that portion of the intervillous space. This is of frequent occurrence in term placentas although minimal in amount so that no observable harm is done the fetus. If the process is extensive, as it may be on occasion, widespread infarction[4] of the placenta may occur because the circulation through the intervillous space is impaired. A localized example of this sequence of events is seen in Figure 24.
A variant of this process, usually of no significance to the fetus, is the deposition of maternal fibrin on the under surfaceof the chorionic membrane (fig. 25). Such fibrin plaques are grossly observable as yellow opaque areas of somewhat increased density on the fetal surface of the placenta. On rare occasions this fibrin deposition— really a variety of mural thrombosis—is progressive and obliterates a significant portion of the intervillous space leading to placental infarction and fetal death.
The appearance of cystic foci in the “fibrinoid” of placental cytotrophoblast is another senile change which may produce lesions of pathologic significance. The placental septa, between adjacent cotyledons or lobes of the placenta, are structures composed of variable amounts of such degenerated trophoblast mixed with maternal decidua. Large maternal sinusoids drain the intervillous space of each cotyledon and are often situated at the margin of the latter in the base of the septum. These vascular structures have little intrinsic strength in their own walls but rely upon the surrounding decidua for their main support. Therefore, if a cystically degenerated septum becomes further weakened, with resultant rupture of the sinusoids, a hematoma results and a variety of placental separation follows (Figure 26). (This is an example of so-called “non-toxic” separation of the placenta because it is based upon a senile rather than toxemic basis.) With the disruption of the blood supply to the involved cotyledon, the latter becomes infarcted. It is unusual for this process to become extensive enough to endanger either the mother or fetus.
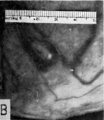
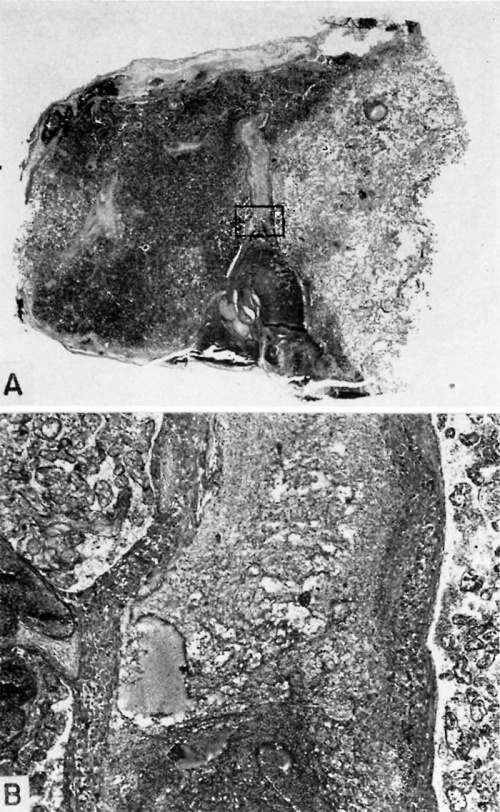
Fig. 25. A human placenta at term to show diffuse and discrete thrombosis of intervillous space. B. L.—i. H., S-32-217.
A. Laminated fibrin thrombus deposited on under surface of chorionic membrane at left of picture. Note septum running horizontally between two adjacent cotyledons, the lower of which is infarcted due to diffuse and discrete intervillous thrombosis while the top one is normal. X5.
B. A more highly magnified view of rectangle seen in A. The fibrous tissue of the chorionic membrane is at the top, beneath which is the cytotrophoblast whose “fibrinoid” degeneration has caused the discrete mass of fibrin to be deposited from the maternal blood. X60.
Fig. 26. A human placenta at term to show the cystic degeneration occurring in the “fibrinoid" of a septum separating two adjacent cotyledons. B. L.-i. H., S-34-581.
A. A low power view of two cotyledons separated by a cytotrophoblastic septum whose "fibrinoid” has undergone cystic degeneration. The resultant hematoma at the base has disrupted the blood supply of the cotyledon at the left and caused infarction but has spared the one to the right. X5.
B. A higher powered view of the rectangular area seen in A. Note degenerated septum with recent hemorrhage below. The villi at the left are congested and undergoing early necrosis whereas the ones at the right are normal. X60.
Discussion
Having attempted to point out that the placenta and various accessory portions thereof show evidence of physiologic and pathologic aging, it remains only to mention certain pathologic examples of delayed and accentuated aging in that organ.
The disease, erythroblastosis fetalis,[5] is an example of delayed aging of the placenta. Presumably the mechanism by which this occurs is the continued and abnormal stimulation of the fetal erythropoietic centers as the result of increased blood destruction. The placenta is a blood-forming organ, and as a result of this urgent need for fetal red cells the placenta continues, until term, a function it ordinarily loses in the first trimester. The placenta of a fetus suffering from the severest form of the disease —universal hydrops—is consequently large, pale, edematous, and friable with microscopic evidence of immaturity and intravascular erythropoiesis (25).
An example of premature aging of the placenta is found in the'hypertensive albuminuric toxemia of pregnancy—eclampsia. Tenny (26) called attention to the syncytial degeneration which occurs in the placenta of this disease. Since many of the involved placentas are actually premature, these syncytial changes might be considered as premature senility in that portion of the placenta because the syncytium tends normally to become thin and degenerate as it matures. In keeping with this morphologic alteration of syncytium in the eclamptic placenta are the studies by Smith and Smith (27) of the hormonal changes in this diseas . These authors concluded that their “results indicate that this disease of late pregnancy is closely associated with a progressive deficiency of progestin, a concomitant decrease of estrogen and a consequent changed metabolism of estrogen involving greater and more rapid destruction.” It has only to be recalled that Wislocki and Bennett (19) show the origin of the steroid hormones (estrogen and progestin) to be in the syncytium to appreciate the reason for the relative decrease of these hormones in the toxemic patient.
No discussion of placental aging would be complete without some reference to that clinically important condition of postmaturity-the situation in which the patient fails to go into labor at the expected time. Rathbun (28) has recently reviewed the situation at the Boston Lying-in Hospital and finds upon analysis of 250 cases that post-mature fetuses continue to gain weight until the 20th day after the expected date of confinement. After that time they begin to lose weight, suggesting that the postmature placenta may not be as efficient as it once was. Experimental evidence for this is found in the earlier-quoted work of Flexner and Gellhorn (20) who showed that in animals the peak of placental efficiency with respect to the passage of sodium occurred just before term was reached. It is not surprising, in the light of these and other studies, that nature has created in the placenta an organ whose efliciency does not last long beyond the expected onset of labor.
Footnotes
- ↑ For the benefit of the non-embryologist who may read this, it should be noted that the placenta, or after-birth, is just as much a part of the fertilized ovum as the fetus or baby even though it is a temporary organ and is cast off at birth.
- ↑ The age of these young specimens is given in terms of developmental or fertilization age rather than menstrual age since the material described up to now was recovered in uteri removed surgically prior to the first missed menstrual period. In older specimens, such as the 23 mm. embryo depicted in Figure 14, the age is expressed in weeks after the last menstrual period. Thus a 21-day ovum, developmental age, is equivalent to an ovum whose menstrual age is 5 weeks or 35 days, since embryonic development does not begin until the 14th day after the last menstrual period at which time ovulation usually occurs.
- ↑ The cytotrophoblast is the germinal bed of chorionic epithelium from which develops the syncytium, connective tissue, blood vessels and amnion. It is represented at different stages of placental development by: the inner epithelial shell of pre-villous ova (Hertig, 8), the Langhan’s epithelium, cell columns and trophoblastic shell of early placentas and the cell columns, cell islands and trophoblastic shell of maturing placentas.
- ↑ Such infarction, it must be emphasized, is not associated with the toxemias of pregnancy whose underlying pathologic anatomy lies in the precapillary arterioles throughout the body. Such involvement of the arteriolar sinusoids of the decidua leads to necrosis of the latter with attendant placental infarction of the toxemic variety. Toxic separation of the placenta is one variety of this pathologic picture.
- ↑ This results from the iso-immunization of the Rh negative mother by the Rh positive fetal red cells and the consequent destruction of the latter by the induced hemolytic antibody in the mother’s blood.
References
1. Streeter GL. Development of the egg as seen by the embryologist. (1931) The Scientific Monthly 32: 495-506.
2. Flexner, Louis B., “Biochemical Changes Associated with Onset of Secretory Activity in the Metanephros of the Fetal Pig,” Anat. Rec., 73, suppl. 2: 20, 1939.
3. Flexner, Josefa B., Louis B. Flexner and William L. Straus, Jr., “The Oxygen Consumption, Cytochrome and Cytochrome Oxidase Activity and Histological Structure of the Developing Cerebral Cortex of the Fetal Pig,” J. Cell & Comp. Physiol., 18: 355-368, 1941.
4. Streeter GL. Developmental horizons in human embryos. Description of age group XIII, embryos about 4 or 5 millimeters long, and age group XIV, period of indentation of the lens vesicle. (1945) Carnegie Instn. Wash. Publ. 557, Contrib. Embryol., Carnegie Inst. Wash., 31: 27-63.
5. Heuser, C. H., “An Intrachorionic Mesothelial Membrane in Young Stages of the Monkey (Macacus Rhesus),” Anat. Rec. 52: suppl. pp. 15-16, 1932.
6. Heuser, Chester H., “Early Development of the Primitive Mesoblast in Embryos of the Rhesus Monkey,” Carnegie Institution of Washington, Pub. 501, Cooperation in Research: 383-387, 1938.
7. Stieve, H., “Ein 13.5 Tage altes, in der Gebarmutter erhaltenes und durch Eingrifi‘ gewonnenes menschliches Ei,” Zeitschr. F. mikr-. anat. Forsch., 7: 295-402, 1926.
8. Hertig AT. and Rock J. Two human ova of the pre-villous stage, having an ovulation age of about eleven and twelve days respectively. (1941) Carnegie Instn. Wash. Publ. 525, Contrib. Embryol., 29: 127-156.
9. Lewis FT. The Minot pre-somite embryo and the Herzog pre-allantoic embryo as elucidating a still younger stage of human developments. (1939) Anat. Rec. 73: suppl. #2, 3637.
10. Heuser CH. and Streeter GL. Development of the macaque embryo. (1941) Contrib. Embryol., Carnegie Inst. Wash. Publ. 525, 29: 15-55.
11. Hertig, Arthur T., “On the Development of the Amnion and Exocoelomic Membrane in the Pre-villous Human Ovum,” Ferris Lecture, Delivered May 2, 1945 at Yale University, Yale J. Biol. & Med. In press.
12. Linzenmeier, G., “Ein junges menschliches Ei in situ,” Arch. F. Gynak,. 102: 1-17, 1914.
13. Heuser, C. H., John Rock, and Arthur T. Hertig, “Two Human Embryos Showing Early Stages of the Definitive Yolk Sac,” Carnegie Institution of Washington,’ Pub. 557, Contrib. Embryol., 31: 85-99, 1945.
14. Hertig, Arthur T., “Angiogenesis in the Early Human Chorion and in the Primary Placenta of the Macaque Monkey,” Carnegie Institution of Washington, Pub. 459, Contrib. Embryol., 25: 37-81, 1935.
15. Cullen, Thomas S., Embryology, Anatomy, and Diseases of the Umbilicus Together with Diseases of the Urachus, Philadelphia and London: Saunders, 1916.
16. Stieve, H., and I. Strube, “Uber die Entwicklung des Dottersackresilaufes beim Menschen,” Zeitschr. F. Mikr.-anat. Forsch., 32: 107-175, 1933.
17. Dandy, Walter E., “A Human Embryo with Seven Pairs of Somites, Measuring about 2 mm. in length,” Am. J. Anat., 10: 85-108, 1910.
18. Meyer, Arthur W., “On the Structure of the Human Umbilical Vesicle,” Am. J . Anat, 3: 155-166, 1904.
19. Wislocki, George B. and H. Stanley Bennett, “The Histology and Cytology of the Human and Monkey Placenta, with Special Reference to the Trophoblast,” Am. J. Anat., 73: 335-449, 1943.
20. Flexner, Louis B., and Alfred Gellhorn, “The Comparative Physiology of Placental Transfer,” Am. J. Obst. & Gynec., 43: 965-974, 1942.
21. Gellhorn, Alfred, Louis B. Flexner, and Louis M. Hellman, “The Transfer of Sodium Across the Human Placenta,” Am. J. Obst. & G_ynec., 46: 668-672, 1943.
22. Bremer, J. L., “The Interrelations of the Mesonephros, Kidney, and Placenta in Different Classes of Animals,” Am. J. Anat., 19: 179209, 1916.
23. Dempsey, Edward W., and George B. Wislocki, “Histochemical Reactions Associated with Basophilia and Acidophilia in the Placenta and Pituitary Gland,” Am. J. Anat., 76: 277301, 1945.
24. Harris, H. A., “Glycogen in Cartilage,” Nature, 130: 996-997, 1932.
25. Hellman, Louis M., and Arthur T. Hertig, “Pathological Changes in the Placenta Associated with Erythroblastosis of the Fetus, Am. J. Path., 14: 111-120, 1938.
26. Tenney, Benjamin, “Syncytial Degeneration in Normal and Pathological Placentas,” Am. J. Obst. & G_yrLec., 31: 1024-1028, 1936.
27. Smith, George Van S. and O. Watkins Smith. “Estrogen and Progestin Metabolism in Pregnancy,” J. Clin. Endocrin., 1: 470-476, 1941.
28. Rathbun, Lewis S., "An Analysis of 250 Cases of Postmaturity,” Am. J. Obst. & Gynec., 46: 278-282, 1943.
Submitted for publication November 5, 1945
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - lnvolution of tissues in fetal life (1946). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_lnvolution_of_tissues_in_fetal_life_(1946)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G