Paper - Cleavage Stages of the Ova of the Horse with Notes on Ovulation
| Embryology - 28 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hamilton WJ. Cleavage stages of the ova of the horse, with notes on ovulation. (1945) J Anat. 79(3): 127–130. PMID 17104976 Historic Embryology Papers
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
| Online Editor Notes |
|---|
William James Hamilton (1903-1975) was originally at the St Thomas's Hospital Medical School and in 1936 became Professor of Anatomy in the University of London at St Bartholomew's Hospital Medical College. He was Regius Professor of Anatomy at the University of Glasgow from 1945 to 1948. Finally, he was Professor of Anatomy at Charing Cross Medical School and then Dean of the School from 1956 to 1962.
|
Cleavage Stages of the Ova of the Horse, with Notes on Ovulation
By W. J. Hamilton, Anatomy Department, St Bartholomew’s Hospital Medical College, London, and F. T. Day, School of Agriculture, Cambridge
Introduction
There are few observations on the early development of the horse, and up to the present no account has been given of the early cleavage stages. Hausmann (1840) recovered ova from large ovarian follicles, but it was not until almost one hundred years later that Amoroso, Griffiths & Hamilton (1939) recovered an unsegmented living tubal ovum from the uterine tube. This ovum was photographed in the living state and then sectioned; it showed evidence of degeneration. Later in the same year, Day (1939) recovered an unsegmented egg.
Follicular Growth and Ovulation
By rectal palpation of a mare’s ovaries it is possible to follow follicular growth and ovulation during the oestrous cycle; by daily examinations an accurate estimate of the time of ovulation can be made (1-6 hr.). The average length of oestrus is 6 days with great variations. As a rule one follicle gradually increases in size during oestrus and reaches an estimated diameter of 3~5—5 cm. just prior to ovulation. At first the follicle is quite tense, but within 12-24 hr. of ovulation becomes somewhat softened. At the time of ovulation the follicle bursts through the ovulation fossa and leaves a soft depression in the ovary. Within about 8 hr. the follicular cavity fills with blood clot, and for the next 10 hr. or so is soft and pliable to the touch until by 24-30 hr. after ovulation it is plum-like in consistency. It then becomes firmer and less conspicuous per rectum until by 4 or 5 days after ovulation it is no longer palpable, except that the ovary with the corpus luteum is normally about twice the size of the inactive ovary.
Insemination of mares with semen from a stallion of known good fertility gives equally good fertility results when insemination is carried out at any time from 0 to 72 hr. before ovulation.
The injection of pregnancy urine extract (B.D.H. ‘Gonan’) 500-1000 i.u. intravenously will cause ovulation (from the mare’s ovary) within about 30 hr. providing there is a follicle of 3-0 cm. diameter in the ovary at the time of injection.
With a series of eight Welsh ponies it was decided by utilizing the facts mentioned above, to obtain tubal ova at different cleavage stages. Additional unsegmented eggs were recovered from a Shire and two thoroughbred mares (see Table 1).
Method
The mares were examined daily until a fairly large follicle was found to be present in one ovary. They were then inseminated with 10 c.c. of sperm from a Shetland pony stallion of known good fertility. One mare was allowed to ovulate naturally, but, to save repeated examinations, the other mares were injected intravenously with lutealizing hormone on the same day as insemination. Rectal examinations were then made as often as was necessary to estimate reasonably accurately the time of ovulation. The mares were killed at definite times after ovulation.
The eggs were obtained by flushing the uterine tubes with Locke’s fluid into small watch-glasses. After being isolated they were transferred into several changes of Locke’s fluid in order to separate them from small pieces of epithelial detritus. The eggs were photographed in the living state with a Vickers projection microscope at a magnification of 480. After photographing they were fixed in either Bouin’s, Flemming’s or Susa’s fixatives, and then embedded by the agar technique as described by Samuel (1944) and sectioned at 7,1.
Unsegmented Eggs
There are six unsegmented eggs in our series, the measurements of which are given in Table 1. Three of the eggs were recovered from three ponies, the others from the Shire and two thoroughbred mares. From such a small series it is not possible to give a conclusive statement about differences in egg size in large and small breeds, but it may be noted that there is a striking similarity of size and so the results agree with what was found by Gregory (1930), who examined the living egg from three different genetic stocks of rabbit——giant strain, small Polish and mixed stock~and found that the eggs from all these rabbits were approximately of the same size and that no morphological differences could be detected.
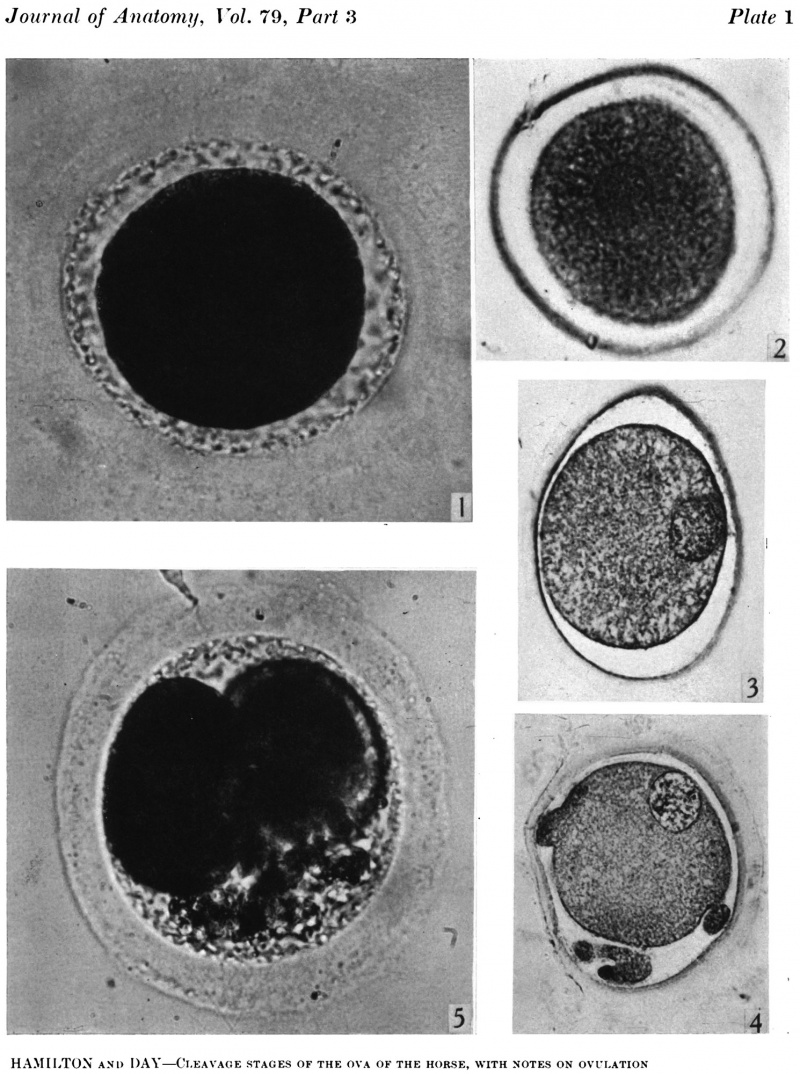
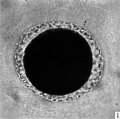
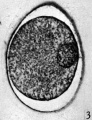
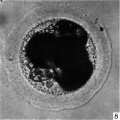
In the living state the vitellus is composed of very many fatty globules which are so closely packed together that it appears opaque when examined with transmitted light and hence no nuclear detail is visible (Pl. 1, fig. 1). In this respect the horse egg resembles more closely that o‘f the pig (Heuser & Streeter, 1929) than those of the other ungulates so far examined. It also shows a striking resemblance to the egg of the dog (van der Stricht, 1923), and to that of the ferret (Hamilton, 1934). The vitellus in the normal egg, as is usual with tubal eggs, does not completely fill the zonal cavity. The perivitelline space is filled with finely granular material. In one egg fatty globules are being extruded into the enlarging perivitelline space (Pl. 1, fig. 4).
In the three degenerating eggs the cytoplasm shows clumping, and in the uterine egg the perivitelline space is obliterated. The zona pellucida is homogeneous and cumulus cells are absent. In the tubal eggs there is a thin coat of material resembling the albumen of the rabbit egg.
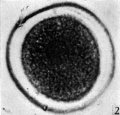
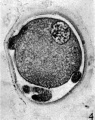
In the cleared agar whole mounts the cytoplasm has a granular appearance and, in normal eggs, the nucleus may be readily recognized (Pl. 1, fig. 2) exception to what has been found in most mammalian eggs. It could be argued that the first polar body had disintegrated and that the chromosomes had reconstituted themselves into a resting nucleus, but this is highly improbable. Another alternative might be that the administration of Gonan had prematurely caused ovulation. The observations of Hinsey & Markee (1933) indicate that the amount, of pituitary secretions necessary to activate the egg is less than that required to produce ovulation. In their experiments ovarian follicles did not rupture in hypophysectomized rabbits as the result of the injection of Prolan, but polar body formation did occur. It seems probable that sufficient Gonan was present to cause polar body formation in the horse.
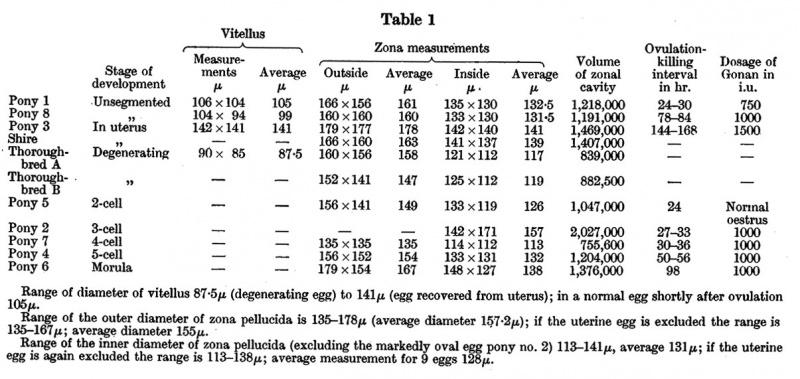
Table 1
- Range of diameter of vitellus 87.5 µ (degenerating egg) to 141 µ (egg recovered from uterus); in a normal egg shortly after ovulation 105 µ.
- Range of the outer diameter of zona pellucida is 135-178 µ (average diameter 157.2 µ); if the uterine egg is excluded the range is 135-l67 µ; average diameter l55 µ.
- Range of the inner diameter of zona pellucida (excluding the markedly oval egg pony no. 2) l13—141 µ, average 131 µ; if the uterine egg is again excluded the range is 113-138 µ; average measurement for 9 eggs 128 µ.
The zona pellucida has undergone a contraction. In the sectioned and stained material the cytoplasm is uniformly granular and shows no evidence of polarity. In each of the normal eggs there is a large vesicular nucleus lying at one side of the cytoplasm (Pl. 1, fig. 3). In none of the eggs were polar bodies found.
In all spontaneously ovulating mammals so far examined, with the exception of the dog, the formation and separation of the first polar body occurs when the egg is still in the ovary. The second polar spindle is then formed, but the separation of the second polar body is not effected unless fertilization supervenes. The ovum of the dog does not form the first polar body or even the chromosomes for the first maturation division until after i ’ = sled (Evans & Cole, 1931). They state: ‘ It would appear that the polar body is not indeed formed for a day or two after ovulation.’ They recovered eggs from the uterine tube, some with a vesicular nucleus and others with the first polar body and the second maturation spindle. The egg of the horse is also an since it caused ovulation in all our mares on which it was used.
Cleavage Stages
Two-cell stage. This egg was ovulated spontaneously; on 4 August 1944 a follicle of 3-5 cm. was felt in the right ovary, and the mare was artificially inseminated. As the result of rectal examination on 8 August 1944 it was thought that ovulation had occurred 24 hr. previously, the mare was then killed.
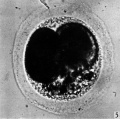
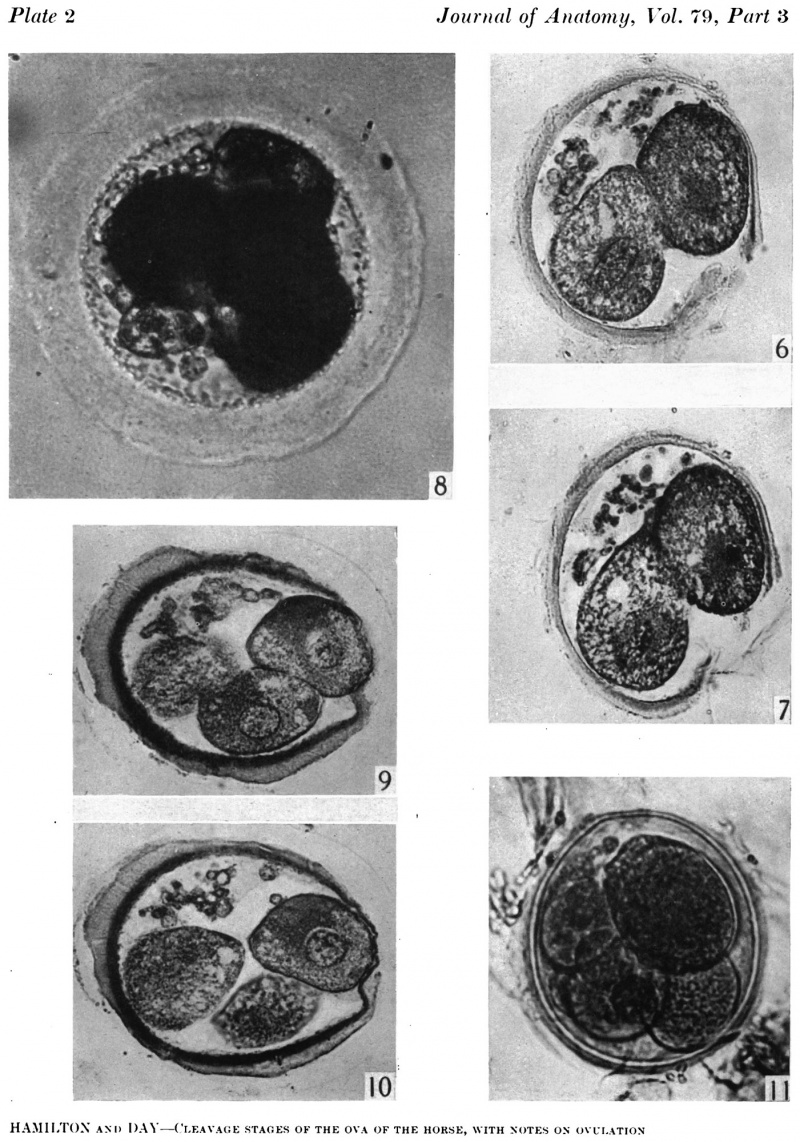
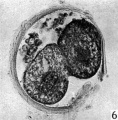
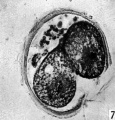
The blastomeres in the living state do not fill the zonal cavity (Pl. 1, fig. 5). In the space unoccupied by the cells there is a considerable amount of granular material which ha 1 lie-en eliminated from the egg as the result of deutoplasmolysis (see later). The cytoplasm of the blastomeres is similar to that of the unsegmented egg. There is a slight difference in’ the size of the cells but no difference in their texture. The sections show two blastomeres which are essentially similar in appearance (Pl. 2, figs. 6, 7).
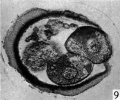
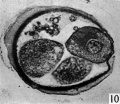
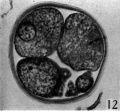
Three-, four- and five-cell stages. A three-cell stage Cleavage stages of the ova of the horse, with notes on ovulation was recovered at 27-33 hr. after ovulation, a four cell stage at 30-36 hr., and a five-cell stage 50—56 hr. after ovulation. In all of these specimens, in the living state, there is an extensive perivitelline space filled with granular material. At the four-cell stage the cells are approximately the same size and are arranged at the apices of a tetrahedron (cross arrangement) as is typical for most eutherian eggs (Pl. 2, fig. 8). Sections of the four-cell stage are seen in P1. 2, figs. 9 and 10. The arrangement of the cells at the five-cell stage is shown in P1. 2, fig. 11. The examination of the sectioned material of the five cell stage shows that the largest cell has a spindle of division (Pl. 3, fig. 12).
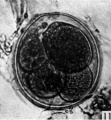
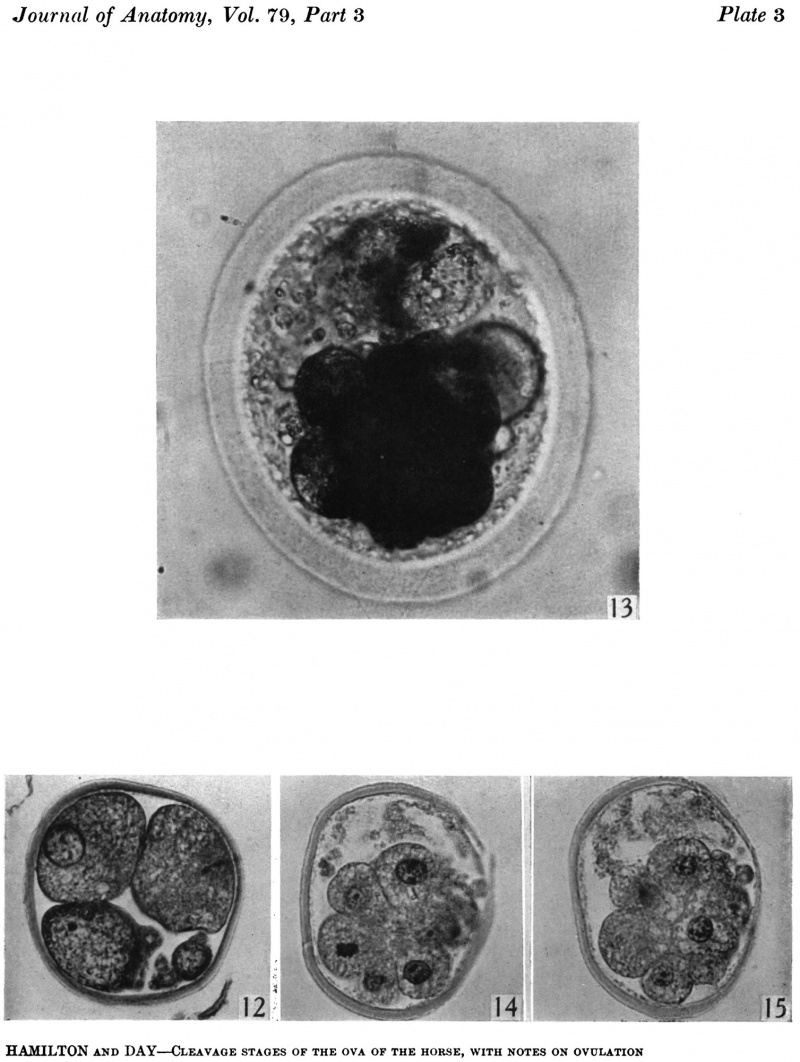
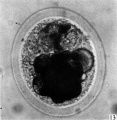
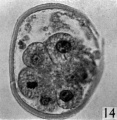
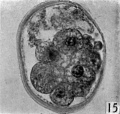
fifteen-cell stage. This egg was recovered from the uterine tube 98 (1 6) hr. after ovulation. The cells which were closely packed together lay at one side of the zonal cavity. The remainder of the cavity is occupied by granular detritus which is even more extensive than "at earlier stages (Pl. 3, fig. 13). When the egg was sectioned the cells were found to be similar in appearance except that one cell showed a spindle of division (Pl. 3, figs. 14, 15).
Remarks on Cleavage
The examination of the few stages at our disposal shows that the cleavage of the egg of the horse is essentially similar to that of most mammals. For a eutherian egg there is, however, an extensive amount of deutoplasmolysis (or yolk elimination). This phenomenon was first described by 0. van der Stricht (1909) in the egg of the bat (Vesperugo noctula), and since then has been described in other eggs: guinea-pig (Lams, 1913), dog (0. Van der Stricht, 1923), cat (0. Van der Stricht, 1923; Hill & Tribe, 1924), pig (Heuser & Streeter, 1929), and ferret (Hamilton, 1934). In the bat the process is found from the beginning of maturation, but becomes accentuated during fertilization and during the early cleavage stages. The yolk becomes de tached mainly at the vegetative pole. In the egg of the other types described the process occurs after ovulation. We have no evidence of deutoplasmolysis of the horse egg until after ovulation, after which material is extruded in that part of the egg which is farthest from the nucleus. Even in the later stages the material is segregated more towards one side of the zonal cavity while the blastomeres lie "more towards the other.
In Dasyums, Hill '(1910) found that the excess of yolk was eliminated at the upper pole prior to or simultaneously with the formation of the first cleavage furrow. In Didelphys, Hill (1918) found, owing to the absence of polar concentration of the yolk, that the surplus deutoplasm was eliminated from all the parts of the surface of the unsegmented egg and blastomeres.
The process of deutoplasmolysis appears to be related in some way to the amount of fat contained within the egg. In eggs which are particularly rich in fat, such as those of the horse, much material is eliminated during segmentation, in others, such as the mouse, where there is little fat, the process does not occur.
Van der Stricht (1923) states that though the ovum of the mammals is relatively poor in nutritive material it appears still to contain too much for the needs of the embryo, and so the excess is expelled into the perivitelline space.
Summary
- An account is given of living eggs of the pony and comparisons are made with eggs of other ungulates.
- Measurements of the eggs are also given.
- Cleavage stages (with 2, 3, 4, 5 and 15 blastomeres) are described and their approximate ovulation ages are recorded.
- Deutoplasmolysis was found to be extensive.
- The time of ovulation is discussed.
References
Amorcoso, E. 0., GRIFfiTHS, W. B. & HAMILTON, W. J. (1939). Vet. Rec. 51, 168.
DAY, F. T. (1939). J. Agrtc. Sci. 29, 459.
EvA.Ns, H. M. & COLE, H. H. (1931). Mem. Univ. Calif. 9, 65.
Gamoomr, P. W. (l93" Oortr. Embryol. Carney. Inst. 21, I41. -I
HAMILTON, W. J. (1934). 251. _
HAUSMANN, U. F. (1840). Ueber die Zeugung and Entatehung des wahren -weiblichen Eies bet‘ der Sdugetiere und M enschen. Hannover.
‘Trans. Roy. Soc. Edinb. 58,
Hansen, 0. H. &. Smsmna, G. L. (1929). Carma Embryol. Carney. I net. .20, 1.
HILL, J. P. (1910). Quart. J. Micr. Sci. 56, 1.
HILL, J. P. (1918). Quart. J. Mar. Sci. 63, 91.
HILL, J. P. & TRIBE, M. (1924). Quart. J. Micr. Sci. 68, 513.
HINSEY, J. C. & MABKEE, J. E. (1933). Amer. J. Physiol. 106. 48.
LAMS,‘H. (19i3lai flrch. Biol., Paris, 28, 229.
SAMUEL, D. M,-(1944). J. Anat., Lomi., 78, 173.
VAN DER Srmcnr, 0. (1909). -Mam. Aoad. R. Belg., 2nd series, 2.
VAN DER Srnrcirr, 0. (1923). Arch. Btol., Paris, 33, 229.
Explanation of Plates
- Links: Fig. 1 | Fig. 2 | Fig. 3 | Fig. 4 | Fig. 5 | Plate 1 | Fig 6 | Fig 7 | Fig 8 | Fig 9 | Fig 10 | Fig 11 | Plate 2 | Fig 12 | Fig 13 | Fig 14 | Fig 15 | Plate 3
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Plate 1
Fig. 1. Photograph of a living unsegmented ovum of the pony (P 1) in Locke’s fluid. Granular material is seen in the perivitelline space. X 480.
Fig. 2: Photograph of an unsegmented ovum of the pony (P 1) in agar. The nucleus is just visible. x 480.
Fig. 3. Section of an unsegmented ovum of the pony (P1) with a large vesicular nucleus. x 520.
Fig. 4. Section of an unsegmented ovum of the pony (P 8) showing deutoplasmolysis. x 520.
Fig. 5. Photograph of a living two-cell stage ovum of the pony (P 5). Granular material is seen in the lower part of the perivitelline space. x 480.
Plate 2
Figs. 6, 7. Two consecutive sections through the two-cell ovum shown in fig. 5. x 520.
Fig. 8. Photograph of a living four-cell stage ovum of the pony (P 7). x 480.
Figs. 9, 10. Two consecutive sections through the four-cell ovum shown in fig. 8. x 520.
Fig. 11. Photograph of a five-cell stage ovum of the pony (P 4) in agar. x 480.
Plate 3
Fig. 12. Section through the five-cell ovum shown in fig. 11. x 520.
Fig. 13. Photograph of the living morula. of the pony (P 6). it shows extensive deutoplasmolysis. x 480.
Figs. 14, 15. Two consecutive sections through the morula. shown in fig. 13. x 520.
Cite this page: Hill, M.A. (2024, April 28) Embryology Paper - Cleavage Stages of the Ova of the Horse with Notes on Ovulation. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Cleavage_Stages_of_the_Ova_of_the_Horse_with_Notes_on_Ovulation
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G