Paper - A presomite human embryo with a neurenteric canal (embryo R.S.)
| Embryology - 28 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Odgers PN. A presomite human embryo with a neurenteric canal (embryo R.S.). (1941) J. Anat., 75(4): 381-388.3. PMID 17104868
| Online Editor |
|---|
| This historic 1941 paper by Odgers describes an early embryo development, later characterised as Carnegie stage 8.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
A presomite human embryo with a neurenteric canal (embryo R.S.)
By P. N. B. Odgers
Department of Human Anatomy, University of Oxford
THE paraffin block containing the embryo described in this communication was given to me by Dr Robb Smith, Pathologist to the Radcliffe Infirmary, who was also good enough to furnish me with the following information about its origin. He found it in a uterus which had been removed by hysterectomy the same day from a woman, aged 40, and which contained several intramural fibroids undergoing red degeneration. The whole of the endometrium was thickened, and a small cyst was noticed in the lower posterior quadrant; this was excised with the corresponding portion of the uterine wall. The excised specimen was fixed in 5 % formol saline and embedded in paraffin. When a few sections had been cut and stained, Dr Robb Smith discovered the presence of a blastocyst; he therefore handed the rest of the block to me.
The menstrual history was as follows. The last period began on 5 October, and the period which was due on 2 November was missed. The uterus was removed on 15 November. From these data the minimal age of the embryo must have been 22 days.
The block was cut into serial sections 10o in thickness, and these were stained with haematoxylin and eosin.They were found to contain the complete embryonal rudiment. Each section was photographed at a magnification of 150, and a reconstruction model of the embryonal disc was made at this magnification by the blotting paper and beeswax method. This latter has been used by myself for many years in this Department, and I consider that it is unrivalled for its accuracy and durability. From there construction the sections were found not to be truly transverse to the longitudinal axis of the embryonal plate. The angle which the plane of section makes with that transverse to the embryonal axis increases in the cranial half of the disc. It is 100 midway along the primitive streak, 150 at the level of the neurenteric canal, while it is 400 half-way along the chorda plate and almost 800 in the prochordal area. These differences are partly due to the angle which the chorda plate makes with the primitive streak at the site of the neurenteric canal (seebelow). Norarethe sections accurately vertical: they are cut with an obliquity which is most marked in the cranial end of the embryonal plate.
From the account of it which follows, the embryo will be seen to fall into the group to which the following four previously described specimens belong:
- Frassi (1907, 1908), length 1-17 mm.
- V. Spee, Embryo Gle (1889, 1896), length 15 mm.
- Triepel, Embryo Dy (1917), length 1.6mm.
- Boerner-Schwarzacher (1923), length 0.47 mm.
While a lot these are very similar to the present specimen in their size (with the exception of the Boerner-Schwarzacher embryo) and in the appearances they present, the Frassi embryo seems to be the most like, and many of his drawings might well illustrate, my description. The only substantial differences between the two are the length and shape of the embryonal disc, the Frassi disc being both shorter (1.17mm.) and narrower (0.6mm.) than mine. Hill & Florian (1931) have drawn attention to the two varieties of embryonal shield, the one small and comparatively narrow, the other large and comparatively broad. My embryo, R.S., evidently belongs to the latter type.
Description of Embryo
The germ disc (P1. 1, fig. 1 and Text-fig. 1). The embryonal area is roughly triangular in shape and its greatest breadth is not far short of its axial length. The following measurements of the disc and of the axial structures were made with a flexible tape measure on the reconstruction model (x 150) and reduced:
| Feature | Size (mm) |
|---|---|
| Length of disc from the cranial amniotic reflexion to the caudal end of the primitive streak | 1.5 mm |
| Greatest breadth of disc | 136 mm |
| Length of primitive streak | 04 mm |
| Length of chorda plate | 07 mm |
| Length of prochordal plate | 0.29 mm |
| Length of area in front of prochordal plate | 0.08 mm |
The embryonal disc exhibits a marked dorsal convexity in.either plane, the greatest height of this being about half-way along the chorda plate where it risesto037mm. above the baseline. While in the transverse plane this convexity forms an even curve, along the cranio-caudal axis of the disc (Text-fig. 2) it is definitely angulated midway along the chorda plate. In Triepel's embryo this angulation, which occurs at about the same situation, isincreased to a right angle.
There is no bend at the site of the neurenteric canal, as is seen in V. Spee's embryo Gle in which the streak area makes almost a right angle with the cranial portion of the plate at this point.
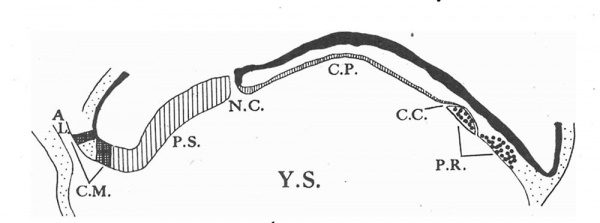
Text-Fig.1. A projection of the dorsal aspect of the embryonal shield on the flat (x 75), drawn to scale. C.M. cloacal membrane. P.S. primitive streak. N.C. neurenteric canal. C.P. chorda plate. P.R.(with coarse dots) prochordal plate.Thet at the cranial end of the chord plate indicates the site of the remains of the chorda canal. The arrows indicate the plane of section.
A dorsalview of the reconstruction model (PI. 1,fig. 1) shows at the junction of the caudal third with the anterior two-thirds the opening of the neurenteric canal,while behind this is the primitive groove. Immediately in front of the neurenteric orifice is a small projection which may be the remains of Hensen's node, and in front of this again the general dorsal convexity is broken for a short distance by the commencing medullary groove. Two further points may be observed in PI. 1, fig. 1 and in Text-fig. 1, in botb of which the different portions of the embryonal axis are mapped out. In the first place there is considerable asymmetry of the disc, so that roughly three-fifths to two-thirds of it, varying between these limits at different levels, lie to the left side of the embryonal axis when viewed from above. Secondly, it is apparent that the chorda plate and the primitive streak are not in the same straight line. The chordaplate is inclined to the left and makes an angle of 250 with the line of the primitive streak at the site of the neurenteric canal. This deviation is figured as being more marked stilinthe Boerner-Schwarzacher specimen and may be readily explained, I think, by the attachment of the massive connecting stalk to the caudal end of the plate. While the streak area is fixed in this way, the open neurenteric canal very possibly allows a certain amount of mobility to the anterior portion of the disc.
The primitive streak
The measurements of the extent of the primitive streak and the chorda plate (0.4 and 0 7 mm.) show that the former is of greater length than would be expected at this stage in comparison with most of the other embryos of about this age. In the Frassi embryo about half of the axial formation is represented by the streak so that the latter is even more extensive than in mine. In Triepel's embryo, however, only one-seventh of the embryonal area is stated to be caudal to the neurenteric canal, while in Rossenbeck's embryo Peh-Hochstetter (1923), which is younger than mine, the proportion between streak and head process is 2 to 5 and in Ludwig's specimen (1928), which is slightly older than mine, it is almost 1to5. Both these latter measurements are taken from Florian's. (1934) Schema der Entwicklung der Axial gebilde des menschlichen Embryos, in which the gradual diminution of the streak area in comparison with the steady elongation of the head processis graphically shown. Into such aschemeas this my embryo hardly fits.
PI. 1, fig. 2 is a microphotograph of a typical section of the streak area. A small point to which attention may be-directed is the condition here of the entoderm. While it generally takes no part in the fusion of the germ layers and forms a distinct layer throughout its extent, the cells composing it are crowded together and give the appearance of sharing in the cellular activity of the neighbourhood. In more than one place, moreover, particularly in the caudal end of the streak, there are connexions between it and the overlying mesoderm, which almost suggest that it may even stil be contributing to the formation of the latter. In earlier embryos fusion of the entoderm and of the mesoderm of the primitive streak has been frequently observed (Florian, 1932-3).
The neurenteric canal
(PI. 1, fig. 3). This is seen to be patent in two sections and is oval in shape, its longer axis being in the antero-posterior plane. The lips of the canal shelve more gradually on its ventral than they do on its dorsal aspect, this being particularly marked in the cranial lip. Fillingthelumenisan amorphous coagulum stained with eosin. The canal is a true neurenteric canal, being a vertical connexion between the amniotic and yolk sac cavities. Grosser (1924) insisted that the term should be confined to such a channel and should not be used synonymously with the chorda canal. The Boerner-Schwarzacher embryo showed a neurenteric canal with a larger lumen than mine: it was similarly oval in shape and measured 40 x,30p in its longitudinal and transverse diameters respectively. Sternberg(1927) suggested that such a widely open canal perpendicular to the disc was due to post-mortem changes and was only found in abortions (e.g. V. Spee's Embryo Gle.and Triepel's Embryo Dy), or in cases such as the Boerner-Schwarzacher embryo, in which 40 hr. had elapsed post-mortem before the specimen was found in the uterine wall, and he supposed,therefore,that it might not be truly characteristic of human embryos, He contrasted with these the oblique canals of the Frassi and M'Intyre (Bryce, 1925) specimens, but in the former of these the obliquity, as shown in the original drawings, is very slight, and Frassi stated, moreover, that he could not determine its patency owing to a damaged section. Again, the M'Intyre embryo is not comparable with the group under consideration, as this contains the remains of a chorda canal rather than the true neurenteric canal of Grosser's definition. On the other hand, both my own and Ludwig's Da I embryos were obtained by operation, and while the neurenteric canal in the latter had a narrower lumen it was, like mine,-perpendicular to the embryonal disc.
The chorda plate
(P1.1, fig.4). This forms a flattened plate of varying thickness composed of round cels. It is thicker at its centre than at its periphery, one to three layers of cels being counted in the different portions in any one section. The plate is interpolated in the entoderm with which its edges are continuous, but in no case does any entoderm clothe its ventral aspect. Nor can,I see in any of my sections that the chorda plate is recessed and bounded by distinct lips of the entoderm as Bryce (1925) described in the M'Intyre embryo (the bourrelets16 citho-enteriques of Van Beneden). In some sections the plate is in direct continuity at its periphery with the streak mesoderm, and exceptionally the latter just overlaps the margin of the plate to separate it from the overlying ectoderm. For the most part,however, they are distinct from each other so that there is litle, if any, evidence in this embryo thatthechbrda plate contributes any thing to the formation of the mesoderm. One cannot but be impressed by the breadth of the plate. Allowing for the obliquity of the sections in this specimen, it seems impossible to think of it as being merely the remains of the dorsal wall of the chorda canal. As far as I know, Triepel is the only author who states that the plate increases in circumference after the disruption of the canal, but one must believe that this is the case. By the same token, as Bryce and others have insisted, the breadth suggests that it must yield something more than simply the notochordal rudiment, i.e. that it probably helps to form the entoderm of the digestive tract.
Caudally the plate becomes continuous with the ectoderm lining the cranial lip of the neurenteric canal. I cannot make out any certain evidence of either chorda plate or of the remains of the head process posterior to the neurenteric canal. Ludwig (1928) described in his embryo Da I twin anlagen of the plate joined in the midline, which he observed in the first section caudal to this opening.
In the sixth, seventh and eighth sections from the cranial end of the chorda platecan be seen a small collection of cels similar to those ofthe plate,which forms a short oval mass lying between its centre and the ectoderm and contiguous with both of these (P1.1,fig.5).This appears to be solid throughout its extent. Does this represent a premature attempt at the formation of a notchord? Triepel suggested that in his embryo Dy there was possibly an indication of a commencing notochord, but this consisted of a small narrow dorsally directed evagination of the chorda plate close to its junction with the entoderm and therefore not median in position, which occurred in some sections not far cranial to the neurenteric canal. If these appearances in my own embryoorin theembryoDy haveanysuchsignificance,they must be regarded as premature efforts. The earliest true evidence of the formation of a notochord from a chorda plate that has been recorded, so far as I am aware, is furnished by
Corner's (1929) embryo with ten somites.
The prochordal plate
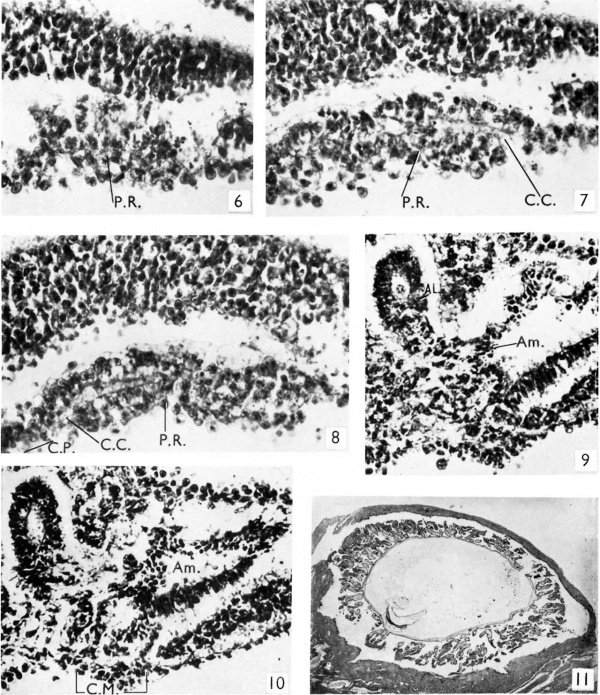
(PI. 2, fig. 6). The thickening of the entoderm with its characteristic chromatophilic granules (Bonnet, 1901) is seen to extend for 0-29 mm. cranial to the commencement of the chorda plate (PI. 1, fig. 1 and Text-figs. 1, 2'. As the latter appears to be more extensive on its left side, the prochordal plate overlaps it on the right for 0-13 mm. Hill & Florian (1931) described it in the embryo Dobbin as being prolonged backwards for 70/, on either side of the head process. In my embryo the prochordal plate does not reach the cranial margin of the embryonal disc but is separated from it by a region0-13mm. in extent in which the entodermis unaltered.The plate shows in four sections some irregular cavitation, and in its caudal portion is traversed in two sections by what appears to be a short canal. This channel, which is seen to be obliquely sectioned, may or may not be patent, but can be observed in PI.2, fig. 8 to be continuous by its dorsal wall with the chorda plate,whilein the next section cranial to this (PI. 2, fig. 7) it opens ventrally and its dorsal wall becomes continuous with the entoderm, the whole canal passing dorsal to the prochordal plate. The site of this canal is marked by a cross in Text-fig. 1 and, while it is median in position, it is not at the extreme cranial end of the chorda plate which extends more anteriorly to the left of it. I am tempted, however, to regard it as the remains of the chorda canal. Such remains have been described by Eternod (1899) in the embryo Vull, while Rossenbeck (1923) in the earlier Peh-Hochstetter specimen, where there was a chorda canal throughout, found that this passed anteriorly immediately dorsal to the pro- chordalplate. Bryce (1925) described the chordaplate in the M'Intyre embryo as being continuous cranially with twin 'diverticula', which were dorsal to the prochordal area, but suggested that a cavity in the central mass ventral to these was the blind anterior end of the chorda canal. The channel in my embryo is similar in position to his diverticula, while I suggest that his ventral cavity may well be an instance of the irregular cavitation in the prochordal plate which has been frequently found in it by others. This interpretation would accord with the findings in the Eterjiod and Rossenbeck embryos.
The cloacal membrane
This begins directly at the caudal end of the primitive streak: there is no interval between the two as-in some earlier embryos. Two sections only show a real fusion of ectoderm with entoderm, which Florian (1932-3) insisted should be regarded as the true definition of the-cloacal membrane, but two sections on the cranial side and one on the caudal side of these show outgrowths of ectoderm which in places almost reach the entoderm. In one of the two sections (P1. 2, fig. 9) a-solid outgrowth of the amniotic ectoderm just dorsal to the junction of the amnion and the shield ectoderm is seen to reach and to fuse with the entoderm of the allantois. The other section (P1.2,fig.10) shows columns ofcelswhich, arisingfrom the shield ectoderm close to its junction with the amnion, fuse with the yolk-sac entoderm. These two outgrowths, the one entirely amniotic and the other from the shield ectoderm, are separated from each other by some stalk mesoderm.
Text-fig. 2. A vertical longitudinal section through the embryonal axis (x 75) drawn diagrammatically to scale and idealized to permit a representation of all the axial structures in the same section. AL. allantois. C.M. cloacal membrane. P.S. primitive streak. N.C.neurenteric canal. C.P. chorda plate. P.R. prochordal plate. C.G.remains of chorda canal. Y.S. yolk sac.
This is shown diagrammatically in Text-fig. 2. The cloacal membrane is less extensive here than in the earlier Heuser (1932) embryo in which it forms a continuous plate involving both allantoic and yolk-sac entoderm.This is to be expected, since it is generally agreed that the membrane is more extensive in the earlier stages and that its caudal portion gradually disintegrates later. It is, however, unexpectedly more extensive in the Sternberg (1927) embryo with four somites. Here there are three discrete contacts of amniotic ectoderm with the entoderm of the allantois together with one contact of shield ectoderm with the yolk-sac entoderm.
The mesoderm of the disc
There are a few small cavities in the peripheral portions of the streak mesoderm, some of which look more like vascular channels than commencing coelom. These are exceptional, and for the most part the mesoderm appears to be unsplit. In places quite apart from the streak area small bridges of mesoderm unite it to the underlying entoderm, which suggests that the latter may be contributing to its formation.
Yolk sac, body stalk and chorionic cavity
There is nothing in any of these structures that has not been fully described previously. Very marked angiogenesis is to be observed both in the mesoderm covering the collapsed yolk sac and in that of the body stalk. In the chorionic cavity is a very small cyst lined with a cubical epithelium. This is situated close to the chorion and 48 sections (480I) away from the cranial border of the yolk sac. Such entodermal cysts have been frequently described (Frassi, Grosser (1912-13) and Heuser) in embryos of about this stage, but, as a rule, they are close to the yolk sac wall.
Summary
The embryonal disc described above measures 1-5x1-36mm. and is reckoned to be 22 days old. In it can be distinguished primitive streak, neurenteric canal, chorda plate and prochordal plate; in the latter area-there is apparently a remainder of the chorda canal. The cloacal membrane consists of two distinct portions, the first where amniotic ectoderm fuses with allantoic entoderm, and the second where shield ectoderm extends into yolk sac entoderm. The embryo belongs to the group of which the Frassi embryo is the best known example.
I am indebted to Mr W. Chesterman of this department for much skilled help in my reconstruction and for the preparation of the microphotographs.
References
BOERNER-PATZELT, D. & SCHWARZACHER, W. (1923). Z.ge8.Anat. 1.Z.Anat.EntwGesch.68,204.
BONNET, R. (1901). Anat.Hefte,IteAbt.16,23.
Bryce TH. Observations on the early development of the human embryo. (1925) Trans. Roy. Soc. Edinb. 53, 533-567.
Corner GW. A well-preserved human embryo of 10 somites. (1929) Carnegie Instn. Wash. Publ. 394, Contrib. Embryol., Carnegie Inst. Wash. 20: 81-102.
ETERNOD, A.C.F. 1899). Anat. Anz.16,131.
FLORIAN,J.(1934). Biol.Gen.10,Lief2,533. (1932-33). J.Anat.,Lond.,67,263.
FRASSt,L.(1907). Arch.mikr.Anat.70,492. (1908). Arch.mikr.Anat.71,667.
GROSSER,0. (1924). Z. ges. Anat. 3. Ergebn. Anat. Entw Gesch. 25, 391. (1912-13). Anat.Hefte,lteAbt.47,653.
Heuser CH. (1932). Contrib. Embryol., Carnegie Inst. Wash. 23: 251..
HEUSER, C. H. (1932). Contr. Embryol. Carneg. Inst. no. 138, 23, 251.
HILL, J. P. & FLORIAN, J. (1931). Philos. Trans. B, 219, 443.
LUDWIG, E. (1928). Jb. morphol. micro. Anat. 1te Abt. 59, 41.
ROSSENBECK, H. (1923). Z.ges.Anat. 1.Z.Anat. EntwGesch.68,325.
V.SPEE, F. (1889). Arch. Anat. Physiol. p.159. (1896). Arch.Anat.Physiol.p.1.
STERNBERG, H. (1927). Z. ges. Anat. 1. Z. Anat. EntwGesch. 82,142.
TRIEPEL, H. (1917). Anat. Hefte, ite Abt., 54,151.
Explanation of Plates 1 And 2
Plate 1
Fig. 1. Photograph of the dorsal aspect of a reconstruction model of the embryonal disc (x 75). The amniotic sac has been removed and the allantois dissected out of the body stalk, while the differentportionsoftheaxialformationsareportrayedonthesurface. P.S.primitivestreak. N.C.neurentericcanal. C.P. chorda plate. P.R.prochordal plate.
Fig. 2. A section (x 250) through the primitive streak. It shows the activity of the entoderm whichatX may be contributing to the mesoderm.
Fig. 3. A section (x250) through the neurenteric canal, N.C. C.P. indicates the commencement of the chordplate. Y.S. yolk sac.
Fig. 4. A typical section (x250) of the chordaplate,C.P.
Fig. 5. A section (x370) showing a mass of cels at X contiguous ventrally with the dorsal surface of the chord plate and dorsally with the ectoderm.
Plate 2
Fig. 6. A typical section through the prochordal plate, P.R. (x370) with its characteristic chromatophilic granules.
Fig. 7. Section showing the cranial end of the presumed remains of the chord canal, C.C. (x370). Its dorsal wall is seen to become continuous with the entoderm, while ventral to it is the prochordal plate, P.R.
Fig. 8. Section (immediately caudal to that shown in fig. 7) showing the caudal end of the same canal, C.C. (x370). Its dorsal wall is seen to be continuous with the chordaplate, C.P. Fig.9. Section 112 (x250) showing an outgrowth of the amniotic ectoderm, Am. fusing with the allantoic entoderm, AL.
Fig. 10. Section 113 (x250) showing strands from the shield ectoderm reaching and fusing with the entoderm to form the cloacal membrane, C.M.
Fig. 11. A section of the embryo in situ (x6).
Reference
Odgers PN. A presomite human embryo with a neurenteric canal (embryo R.S.). (1941) J. Anat., 75(4): 381-388.3. PMID 17104868
Cite this page: Hill, M.A. (2024, April 28) Embryology Paper - A presomite human embryo with a neurenteric canal (embryo R.S.). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_A_presomite_human_embryo_with_a_neurenteric_canal_(embryo_R.S.)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G