Developmental Mechanism - Autophagy
| Embryology - 23 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
- Lack of chromatin condensation
- Massive vacuolization of the cytoplasm
- Accumulation of (double-membraned) autophagic vacuoles
- Little or no uptake by phagocytic cells, in vivo
Autophagic cell death, defines cell death occurring with autophagy, though it may misleadingly suggest a form of death occurring by autophagy as this process often promotes cell survival.
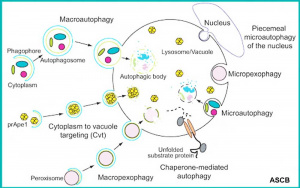
Autophagy Types
There are several different classifications of autophagy this is probably the simplest.
- macroautophagy - (also called autophagy) non-selective sequestration of cytoplasmic substrates
- microautophagy - lysosomal invagination and ingestion of minor portions of the cytosol
- chaperone-mediated autophagy - involves recognition of specific substrates by the heat shock cognate 70 (Hsc70) machinery
- to remove abnormal cytoplasmic organelles and components, it is also a stress response
- cellular self-catabolic process "eating oneself”
- initial sequestered in a structure called a phagophore
- which then closes into a double membrane vesicle the autophagosome
- some autophagosomes formed in a PI3P-enriched (phosphatidylinositol 3-phosphate) membrane compartment dynamically connected to the endoplasmic reticulum
- an autophagosome fuses with a lysosome
- Regulated process of the degradation and recycling of organelles and cellular components
- Resulting in organelle turnover and in the bioenergetics of starvation
- Could result in cell death
- through excessive self-digestion and degradation of essential cellular constituents
Is autophagy a cell survival or a cell death pathway?
Suggested that autophagic cell death is "cell death with autophagy rather than cell death by autophagy" see Figure 1
Autophagy Summaries
- Major cellular events during autophagy
- Autophagy in human disease
- p53 can be a positive or negative regulator of autophagy
- cell death with autophagy or by autophagy?
Some Recent Findings
Material below are new research findings on Autophagy and the origins of the membrane for autophagosome biogenesis.
Mitochondria Supply Membranes for Autophagosome Biogenesis during Starvation
- Autophagosomes are derived from mitochondrial outer membrane during starvation
- Lipids, but not most proteins, are transferred from mitochondria to autophagosomes
- Mitochondria-ER connections are required to form autophagosomes during starvation
- Mitochondrial contribution to autophagosome assembly is unique to starvation
Movies: JCB - Apg5 helps form autophagosomes Autophagosomes form as cup-shaped organelles that engulf large parts of the cytoplasm. As shown by Mizushima et al., Apg5, part of a ubiquitin-like conjugation system, localizes to the forming autophagosomes, and is essential for their formation.
Inner Ear
Magariños M, Pulido S, Aburto MR, de Iriarte Rodríguez R & Varela-Nieto I. (2017). Autophagy in the Vertebrate Inner Ear. Front Cell Dev Biol , 5, 56. PMID: 28603711 DOI.
Abstract
- "Autophagy is a conserved catabolic process that results in the lysosomal degradation of cell components. During development, autophagy is associated with tissue and organ remodeling, and under physiological conditions it is tightly regulated as it plays a housekeeping role in removing misfolded proteins and damaged organelles. The vertebrate inner ear is a complex sensory organ responsible for the perception of sound and for balance. Cell survival, death and proliferation, as well as cell fate specification and differentiation, are processes that are strictly coordinated during the development of the inner ear in order to generate the more than a dozen specialized cell types that constitute this structure. Here, we review the existing evidence that implicates autophagy in the generation of the vertebrate inner ear. At early stages of chicken otic development, inhibiting autophagy impairs neurogenesis and causes aberrant otocyst morphogenesis. Autophagy provides energy for the clearing of dying cells and it favors neuronal differentiation. Moreover, autophagy is required for proper vestibular development in the mouse inner ear. The autophagy-related genes Becn1, Atg4g, Atg5, and Atg9, are expressed in the inner ear from late developmental stages to adulthood, and Atg4b mutants show impaired vestibular behavior associated to defects in otoconial biogenesis that are also common to Atg5 mutants. Autophagic flux appears to be age-regulated, augmenting from perinatal stages to young adulthood in mice. This up-regulation is concomitant with the functional maturation of the hearing receptor. Hence, autophagy can be considered an intracellular pathway fundamental for in vertebrate inner ear development and maturation."
Cite this page: Hill, M.A. (2026, Mayıs 23) Embryology Developmental Mechanism - Autophagy. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Mechanism_-_Autophagy
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G