Book - The Nervous System of Vertebrates (1907) 3
| Embryology - 18 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Johnston JB. The Nervous System of Vertebrates. (1907) Blakiston's Son & Co., London.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter III. Development of the Nervous System
It is hoped in this chapter to bring to the attention of the student those features and processes in the development of the nervous system which are of the greatest morphological importance, and especially to bring forward certain facts which have not received full treatment in text-books of embryology. There will not be space for such a full and orderly treatment of the development as an elementary student in embryology would require. For this reason it will be assumed that the student is familiar with the more general facts in the development of some vertebrate such as the frog or chick.
It is commonly believed that all nervous functions are performed by structures derived from the ectoderm. In the early vertebrate embryo a certain area on the dorsal surface is to be recognized as the anlage of the greater part of the nervous system. This area is known as the neural plate. It becomes visible first as an area bounded on either side by slightly elevated ridges or folds, the neural folds. These folds are continuous in front and behind and enclose an area which is broader in front, in the region corresponding to the future brain (Fig. 18 A). This somewhat banjoshaped neural plate and the folds which bound it give rise to the brain and spinal cord,, the greater part of the sensory nerves with their ganglia, the motor nerves and the retina. The olfactory epithelium, the sensory epithelium of the ear and lateral line organs, and the ganglia and nerves of these organs are derived from parts of the ectoderm closely related to the neural plate. The organs of taste appear first in the embryo in the entodermal lining of the branchial cavities. In fishes they appear later in the outer skin also and their history requires further investigation.
In all vertebrates the neural plate becomes converted into a tube lying below the surface of the body. In all except cyclostomes and bony fishes this is accomplished by the neural plate rolling or folding up so that the neural folds meet above. The folds begin to fuse together at a point which later falls in the region of the mesencephalon or farther caudally, and the fusion continues forward and backward until a complete tube, the neural tube, is formed. The tube remains open for some time at the anterior end; the opening is called the neuropore. At the caudal end the tube remains for some time in connection with the archenteron by way of the neurenteric canal. The extent and relations of the neuropore will be important in later connections ; the neurenteric canal need not claim further attention. In the mean time the general ectoderm fuses over the dorsal surface of the neural tube and when the neuropore closes the tube lies wholly beneath the surface of the body. In cyclostomes and bony fishes the same results are reached by the neural plate thickening and sinking down as a solid cord of cells which comes to lie beneath the ectoderm. Within the cord of cells appear clefts which unite into a continuous canal.
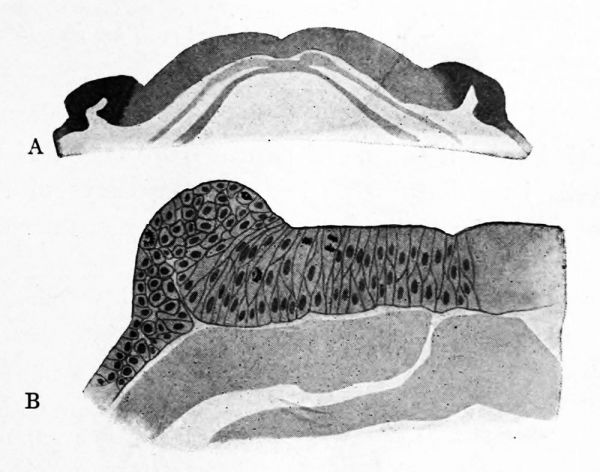
FIG. 13. Sections of the neural plate and folds in amphibia. A, the cephalic portion of the neural plate in Amblystoma tigrinum. The neural folds are more darkly shaded. It is evident that they are actual folds of the ectoderm. B, one side of the neural plate of Amblystoma punctatum at a slightly more advanced stage of development andat a higher magnification.
Roughly speaking the neural tube goes to form the spinal cord, brain and motor nerves. During its development, the material from which the sensory nerves are derived is taking on definite form in close relation with the neural tube. The accompanying figures illustrate the development of the neural tube and the ganglia of the sensory nerves in amphibia. As seen in Figure 13 B, the neural plate consists of columnar cells, while the neural folds which bound it consist of two or more layers of somewhat cubical cells irregularly arranged. As the edges of the neural plate close up to form the neural tube (Fig. 14), the neural folds connected with, and some actually enclosed within, the dorsal wall of the tube adjacent to the seam of closure. There results a pair of continuous flaps or ridges of cells connected with the neural tube at its mid-dorsal line and spreading laterally between the tube and the ectoderm (Fig. 15). These are known as the neural ridges or crests and contain the cells which enter into the formation of the spinal ganglia. This condition of the ganglionic material may be taken as typical for the embryos of vertebrates. In fishes a relatively large number of neural crest cells remain within the neural tube and there give rise to sensory fibers which run to the skin. These are the so-called giant ganglion cells of the spinal cord of fishes (Fig. 17). These facts suggest that the cells which give rise to sensory nerve fibers originally lay within the neural tube and have migrated to form the spinal ganglia. Already in Amphioxus, however, a large part of the spinal ganglion cells are situated in the roots of the nerves outside of the spinal cord. In the brain region the neural crest is formed in essentially the same manner as in the trunk, but two very noteworthy facts are to be pointed out. The first is that in the region opposite the ear the neural crest is entirely absent for a short distance. The second is that in front of the mesencephalon the crest is either very small or so modified that it can not be readily compared with that of the trunk.
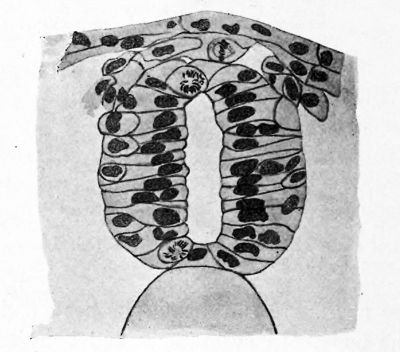
FIG. 14. Transverse section of the neural tube of Amblystoma punctatum just after closing. The cells which will form the neural crest are more darkly shaded. Mitotic figures are seen at all levels in the wall of the tube.
FIG. 15. Transverse section through neural tube, neural crest and ectoderm of Amblystoma punctatum at a later stage than that shown in Fig. 14.
form a sort of bridge connecting the tube with the ectoderm. Now the cells which originally formed the folds separate from the ectoderm and extend laterally on the surface of the neural tube, between it and the ectoderm (Fig. 15). Some of these cells remain
FIG. 16. Same as Fig. 15, later stage. The cells of the neural crest have migrated ventrally to form a spinal ganglion. Processes are seen upon neuroblasts within the tube and upon one spinal ganglion cell.
While these changes are taking place two special sense organs are making their appearance, the olfactory and auditory organs. Both are formed from parts of the ectoderm immediately adjacent to the neural plate. The olfactory organ appears as a pair of thickened patches of ectoderm at the cephalic border of the neural plate at either side of the neuropore. These patches eventually become depressed and form deep olfactory pits. The auditory organs arise on the dorso-lateral surface of the head opposite the region of the future myelencephalon. These are also at first thickened patches which sink in and form deep pits, which eventually separate from the ectoderm as closed sacs and may press against the neural tube on either side. These auditory sacs exercise a great influence on the form and course of development of other organs.
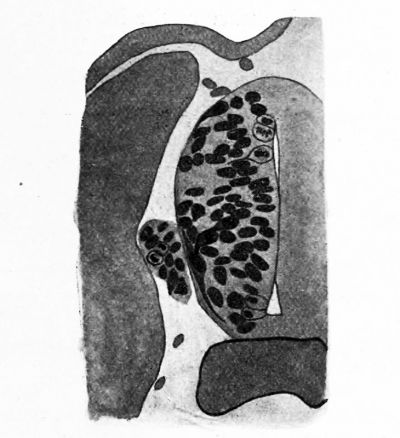
FIG. 17. A part of the spinal cord of an 18-day Catostomus embryo showing two giant ganglion cells. Golgi method.
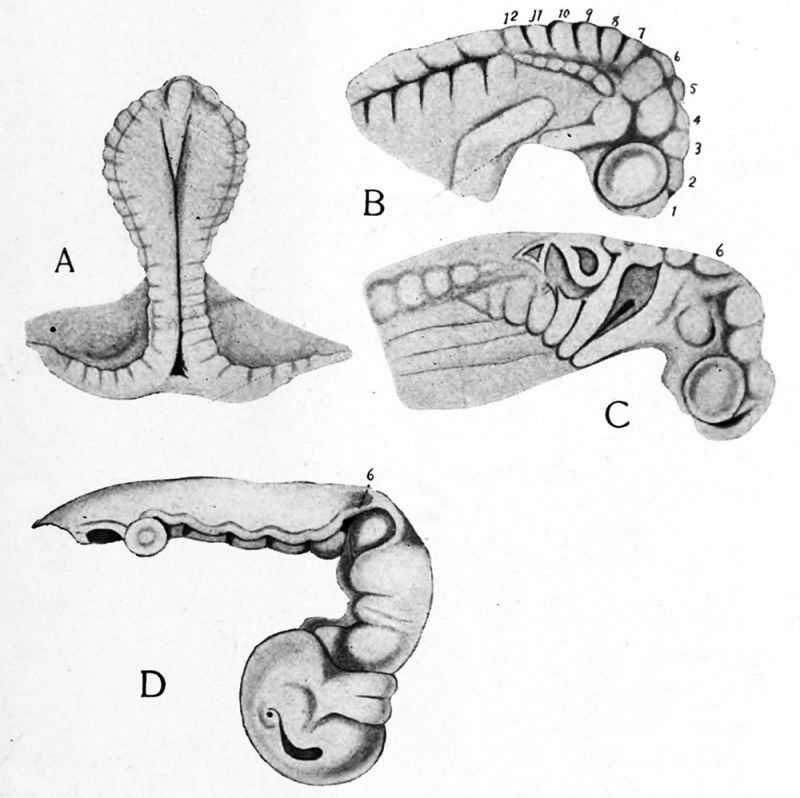
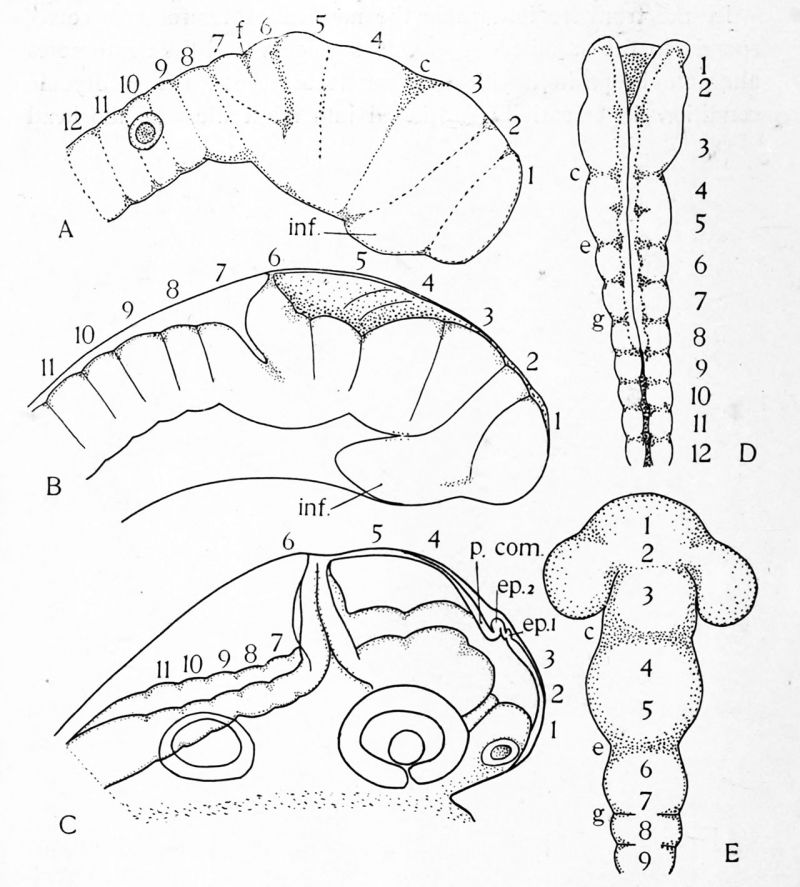
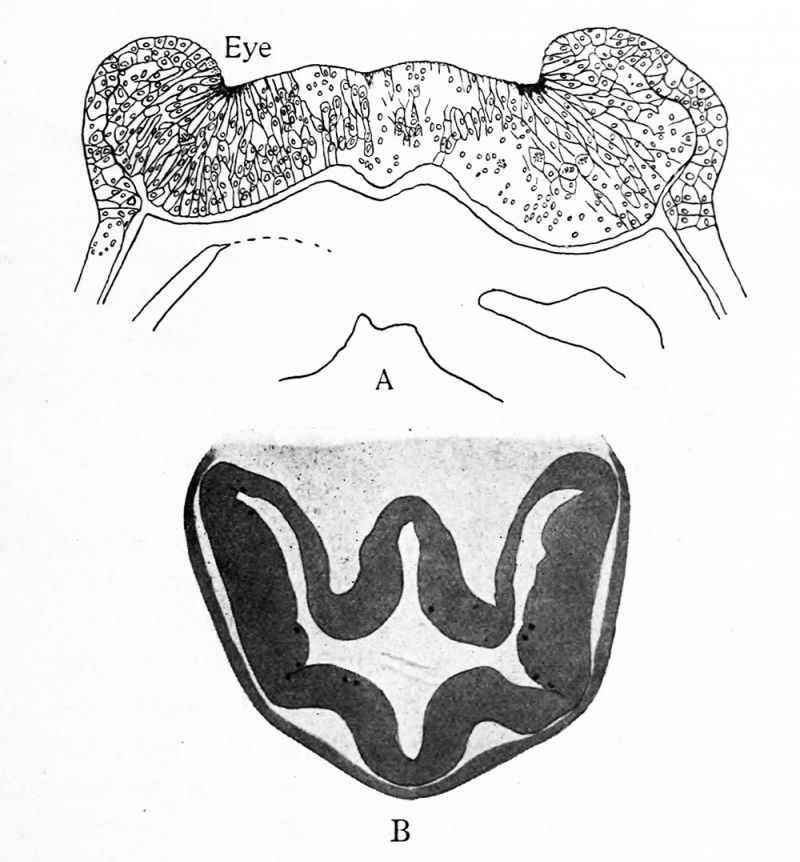
If now the neural tube be examined either in frontal sections or in embryos so dissected as to lay the tube bare, it will be seen that the tube is divided by slight transverse constrictions into successive segments approximately equal in length. These segments are called neuromeres. The neural crest is divided into corresponding segments and, as shown in Fig. 18, the segmentation of the crest is present in the neural plate stage in selachian embryos. While these neuromeres have been clearly seen and described in the region of the hindbrain and spinal cord in all classes of vertebrates, in the anterior portions of the brain they are of shorter duration and are more difficult to study. In selachians, bony fishes and the chick, however, they have been studied and described and some of the important stages in their history are shown in the accompanying series of figures. (Figs. 18, 19.)
In the trunk region where the number of neuromeres corresponds to the number of muscle segments and the neuromeres alternate in positon with the muscle segments, the embryonic condition is essentially continued into adult life. In the head region, where great changes take place during embryonic development, the neuromeres share in these changes and in the adult brain the embryonic neuromeres are lost from view. The causes for this are manifold, but perhaps the chief ones are the bending downward of the head, the crowding of the brain produced by its growing more rapidly than surrounding organs, and above all the rapid growth of those parts of the brain which are destined to become large and important in the adult. One result of these forces is the appearance early in embryonic life of certain bends or flexures. These are illustrated in the figures which show the neuromeres (Figs. 18, 19) and also in Figure 20. In the neck region, owing to the bending of the head, the brain is bent downward in relation to the spinal cord. Farther forward a sharper flexure in the opposite direction is found, called the pontial flexure, because the pons of higher vertebrates appears at this point. Then the front end of the brain is curved down again in such a way as to bring the ventral surface of the brain just behind the closed neuropore nearly into contact with the pontial flexure. The last bend, from its. position in the parietal region of the head, is called the parietal flexure. It corresponds in position to the future mesencephalon. These flexures are in themselves of no fundamental importance, except as the adult form of certain parts of the brain and the position of some of its nuclei are doubtless determined by the influence of the flexures in these early stages. As the head of the embryo straightens out and as the growth of the other organs relaxes the pressure upon the brain, the flexures tend to disappear and they are to a large extent obliterated in the brains of lower vertebrates (compare Figs. 2, n, 12, 158).
FIG. 18. Four stages in the development of the selachian brain to show the history of the neuromeres. After Locy. In A, B and C the whole head is shown, in D the brain alone. The Arabic numerals indicate the neuromeres.
FIG. 19. The history of the neuromeres in the bony fish and the chick. A, B, C, three stages in the development of the bony fish, as seen from the right side. D, E, the brain of the chick. After Hill. The letters c. e. g. mark corresponding furrows in the chick brain; ep. i, ep. 2, anterior and posterior epiphyses; in}, inferior lobe; p. com., posterior commissure. The neuromeres are indicated by Arabic numerals.
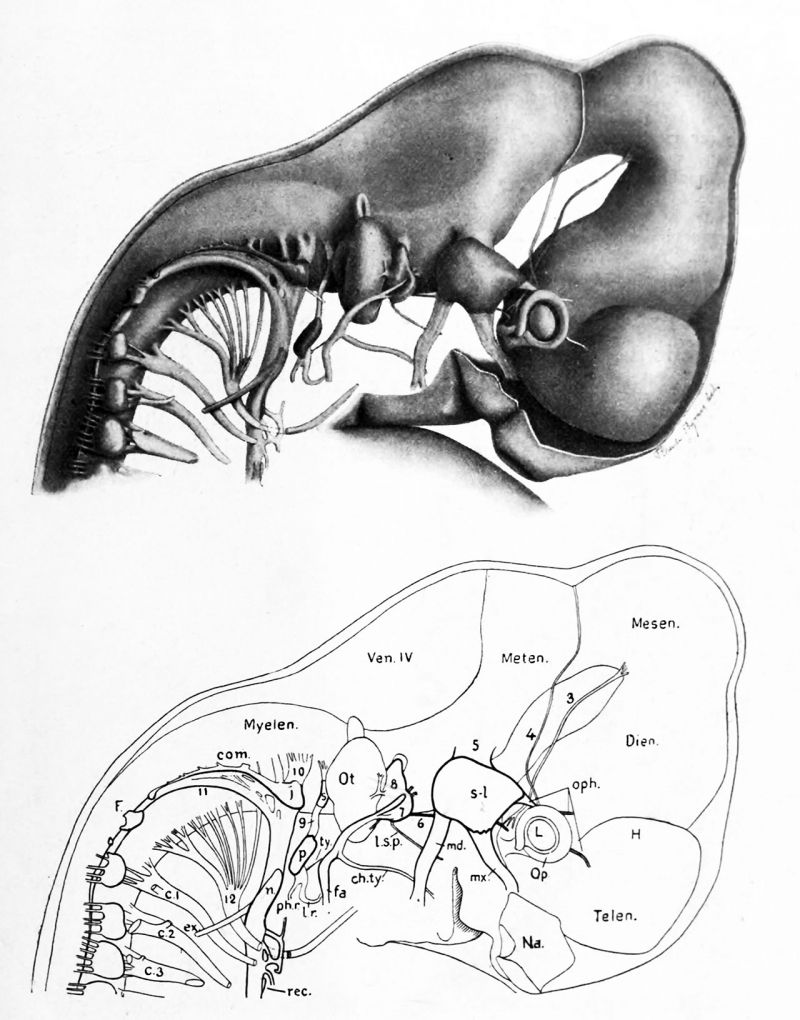
FIG. 20. Reconstruction of the brain and cerebral nerves in a 12 mm. pig embryo. (From Minot's " Laboratory Text-Book of Embryology." Drawrt and revised by F. T. Lewis.)
Nerves." 3, oculomotor. 4, trochlear. 5, trigeminal, with its semilunar ganglion, s-l, and three branches, oph., ophthalmic; mx., maxillary; md., mandibular. The motor portion, which goes with the mandibular branch, is concealed in this view by the sensory portion. 6, abducens. 7, geniculate ganglion of the intermedius. Fibers from this ganglion mix with the motor fibers of the facial nerve, so that both sensory and motor fibers are found in the three branches, /. s. p., large superficial petrosal; ch. ty., chorda tympani; /a., facial. 8, acoustic, showing an upper vestibular portion, and a lower cochlear portion. 9, glossopharyngeal with its superior ganglion, s, above; its petrosal ganglion, p, below; and its three branches, ty., tympanic; /. r., lingual ramus; ph. r., pharyngeal ramus. 10, vagus, with its jugular ganglion, /, extending posteriorly as a ganglionic commissure, com.; and below, its ganglion nodosum, n. Its branches form the laryngeal plexus,beyond which is the recurrent nerve, rec. Just below the jugular ganglion is the auricular branch of the vagus. II, accessory, which joins the vagus; ex., its ramus externus. 12, hypoglossal. P., Froriep's hypoglossal ganglion. C. I, C. 2, C. 3, cervical nerves.
Brain and Sense Organs. Telen., telencephalon. Dien., diencephalon. Mesen. r mesencephalon. Meten., metencephalon. Myelen., myelencephalon. H., hemisphere. Ven. IV., roof of the fourth ventricle. Op., optic cup. L, lens. Na., nasal pit. Ot., otocyst.
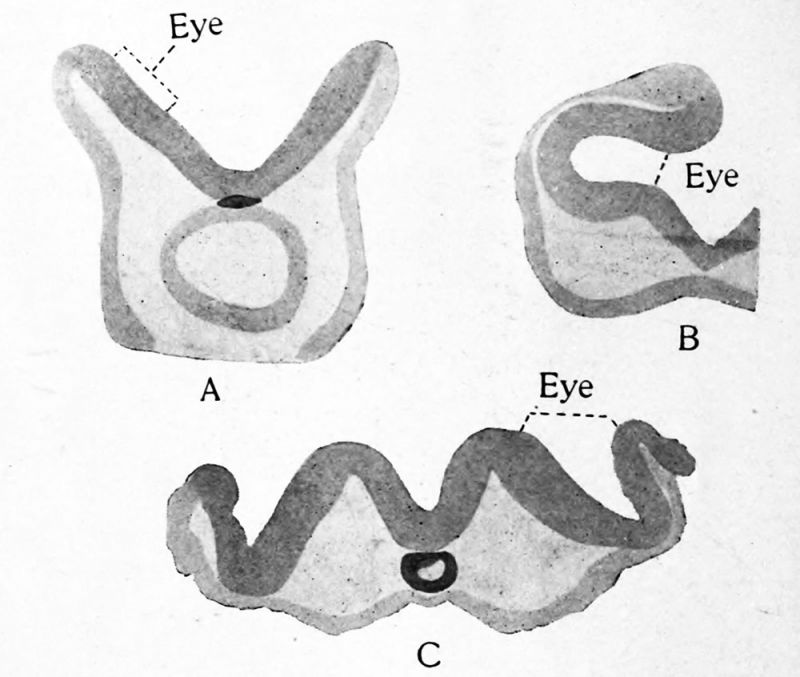
FIG. 21. Transverse sections through the region of the optic vesicles in selachian, avian and mammalian embryos : A, Torpedo ocellata ; B, Callus domesticus ; C, Cavia cobaya. After Froriep. These and the following figure show that the optic vesicles arise from the borders of the neural plate.
FIG. 22. The development of the optic vesicles in Amblystoma punctatum. Fig. 22 A, should be compared with a similar figure by Eycleshymer. In B the germinal cells in process of division are shown by small black figures.
In the meantime the rapid growth of certain parts of the brain while the flexures are still present, results in deepening the constrictions between certain of the neuromeres and the obliteration of others, and produces the prominent secondary segments which have been described in the previous chapter. Only a brief summary of these changes can be given here, since a full description would require much more space than .can be devoted to it. The forebrain is formed by the relatively enormous growth of the first neuromere, especially of its dorsal part. The optic vesicle, which will be treated more fully later (Chapter VIII), is formed from the dorsal part of the second neuromere. In the ventral portion of the second neuromere there is a great expansion which results in the formation of the inferior lobes and mammillary bodies. The floor of the second neuromere is depressed just behind the optic stalk and expanded backward beneath the third neuromere. The extreme ventro-caudal portion of the inferior lobes thus formed becomes thin-walled and is supplied with a great profusion of blood spaces. This is the saccus vasculosus which becomes closely related with the hypophysis in the pituitary body. The slight constrictions between neuromeres i and ii, and ii and iii quite disappear, while that between neuromeres iii and iv remains and is deepened. The formation of the parietal flexure is largely due to the great growth of the dorsal part of neuromeres iv and v, where the optic lobes are to be formed. The whole of neuromeres iv and v fuses into the mesencephalon and the constriction between neuromeres v and vi is deepened more than any other. This marks the boundary between the midbrain and cerebellum and is known in the adult as the isthmus. Neuromere vi forms the metencephalon and the dorsal part of it becomes greatly enlarged in many vertebrates as the cerebellum. The neuromeres from number vii caudally enter into the medulla oblongata and spinal cord, the exact number belonging to the medulla being uncertain, since no definite limit can be fixed between brain and spinal cord. It is possible that this limit varies in different vertebrates. As the result of these changes, the brain loses its primitive segementation and comes to be divided into five secondary segments of unequal size.
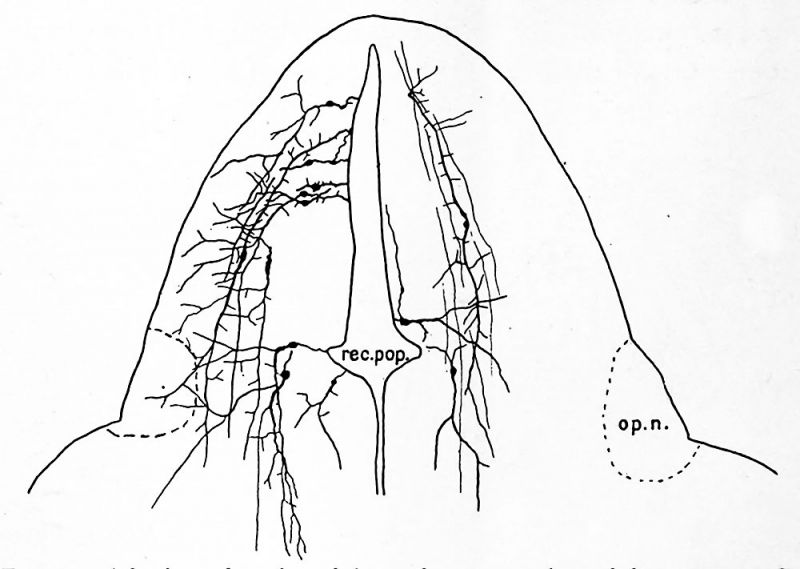
The optic vesicle has been mentioned as formed from the dorsal part of the second neuromere, the first of the two which constitute the diencephalon. At a very early stage while the nervous system is still in the form of a flat neural plate, the area which is destined to enter into the retina may sometimes be recognized in the broad cephalic part. The two retinal areas lie near the lateral borders of the neural plate. When the neural tube is formed these areas at once bulge laterally. They seem upon casual observation, for example in the chick, to involve nearly the whole lateral wall of the first two or three neuromeres. Careful examination shows clearly, however, that in fishes, amphibia, birds and mammals, these pouches belong strictly to the dorsal part of the brain, and they probably belong to a single neuromere, the second. The pouches grow out farther, retaining a portion of the brain ventricle, and finally come to be constricted next to the brain so that they remain connected with the brain wall only by a narrow stalk. The pouch is now known as the optic vesicle and the stalk as the optic stalk. Figures 18 to 20 show the optic vesicle in various stages of growth and Figures 21 and 22 represent it in transverse section. As the vesicle is formed it is crowded between the brain and ectoderm so that it is flattened. The flattening continues until the cavity becomes a narrow cleft and the vesicle becomes somewhat saucer shaped. The constriction from the brain takes place from above downward so that the stalk comes to be attached near the ventral border of the vesicle. As a result of the same process the attachment of the stalk to the brain has shifted ventrad to reach the position afterward occupied by the optic chiasma. In the meantime the two walls of the optic vesicle begin to be differentiated, the outer wall growing thicker to form the retina and the inner wall remaining thin. Finally the portion of the brain ventricle which was carried out into the optic vesicle is obliterated but in many lower vertebrates a vestige of the cavity is retained in the stalk. This lumen connects with the preoptic recess, a depression of the floor of the third ventricle in front of the optic chiasma.
Histogenesis
The development of the microscopic structure, the histogenesis, of the brain, nerves and sense organs is going on at the same time with the development of the form of the brain. The neural tube consists at first of two incomplete layers of cells irregularly arranged (Figs. 13, 14), among which are some columnar cells which extend through the whole thickness of the wall. A little later the columnar cells become radially arranged around the lumen of the tube (Figs. 14, 15), and the shorter cells lie between them bordering upon the lumen or upon the external surface. The columnar cells retain their position throughout life, extending from the central canal to the surface. Since they give rise to a sort of meshwork or trellis which serves largely to support the nervous elements, these cells are called spongioblasts. The inner ends of neighboring spongioblasts fuse to form an internal limiting membrane bounding the central canal and the outer ends may form a similar external limiting membrane. The nuclei of the spongioblasts are situated in the middle and internal portions of the wall, the outer portion remaining relatively free from nuclei. Those spongioblasts whose nuclei occupy the internal ends of the cells become the ependyma cells of the adult brain.
Among the spongioblasts are cubical or rounded cells which are multiplying rapidly, so that many of them contain mitotic figures. They are known as germinal cells. While a few of these cells lie in the thickness of the wall or even upon the external surface, the great majority border on the central canal (Figs. 13 B, 14, 15, 1 6). The germinal cells are believed to give rise to all the nervous elements in the brain and spinal cord and to the stellate cells which with the spongioblasts make up the neuroglia. In general the history of both nerve cells and glia cells is the same. The cells produced by the division of the germinal cells migrate or are pushed toward the periphery of the cord or brain and then, supported by the meshwork of the spongioblasts, undergo development into specific nerve or glia cells.
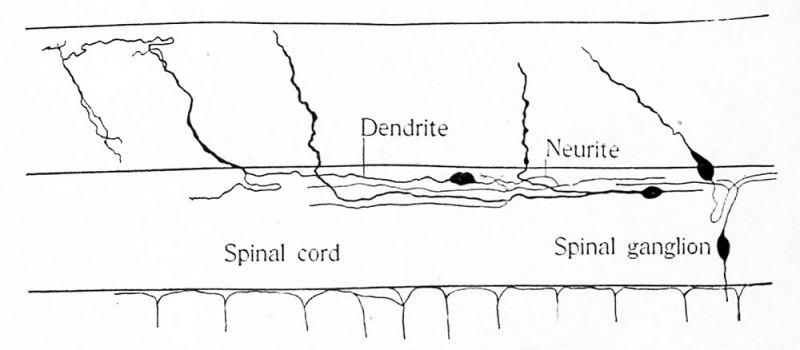
Those cells which are destined to form nerve cells are called neuroblasts. These continue to multiply rapidly by mitosis but the number of mitotic figures seen in the nervous system becomes rapidly less in later stages of development and in the adult the division of nerve cells is comparatively rare. The first step in the formation of a functional nerve cell from a neuroblast is the production of processes somewhat similar to the pseudopodia of an amoeba. First the neuroblast becomes drawn out at one end into a slender filament which is recognized as a nerve fiber (Fig. 1 6). The presence of this cell fiber is the only mark by which the neuroblast can be distinguished with certainty from spongioblasts or glia cells. The nerve fibers may collect into bundles as they are formed and in general the fibers run to the outer surface of the cord or brain, forming a peripheral fiber layer. Some time after the formation of the nerve fiber the neuroblast gives off other processes which branch more or less richly in the substance of the brain wall. The nerve fiber first formed remains always a relatively slender fiber of nearly uniform diameter. It is called the neurite. The processes later formed become more or less bush-like and are known as dendrites.
In most parts of the central nerrous system the greater number of nerve cells remain near the central canal and there form the central gray matter. But many cells migrate toward the surface and in some parts of the brain very rich layers of nerve cells are found at the outer surface. In fishes it is observed that in any given part of the brain those cells which are farthest removed from the central canal have undergone the greatest modifications of form as compared with the simple structure of the neuroblasts. The dendrites may become more richly branched, more numerous and more widely extended; they may be disposed parallel with the surface or otherwise modified to increase their functional efficiency. It is worthy of notice that in the central gray matter in lower vertebrates a large part of the cells have central processes which reach, and perhaps take part in, the internal limiting membrane. These cells seem to show that the bodies of cells formed from neuroblasts migrate toward >the periphery, but their inner ends remain connected with the internal limiting membrane and become drawn out into long central processes. Other cells situated at the extreme periphery in the fish brain are probably derived from peripheral cells of the neural tube such as are seen in Figures 14 and 15. Figures 23, 24, and 25, taken from the brain of the sturgeon, illustrate these relations.
FIG. 23. A part of a transverse section of the inferior lobe of the sturgeon, stained by the Golgi method. The cells all retain their primitive position adjacent to the cavity.
FIG. 24. A horizontal section of the nucleus praeopticus of the sturgeon. Golgi method. The cells have migrated toward the periphery but retain their connection with the internal limiting membrane by means of central processes.
While the histogenesis is in progress certain changes of form in the neural tube are taking place. The most important of these is the appearance of longitudinal zones. In the region of the spinal cord it is noticeable that the walls of the neural tube do not increase in thickness equally. The lateral walls thicken, while along the mid-dorsal and mid-ventral lines the tube remains thin. These thin bands have been named by His roof-plate and floor-plate, respectively. They remain relatively thin throughout life. As the spinal cord grows it expands laterally and ventrally at the sides of the floor-plate, so that the floor-plate comes to form the bottom of a deep groove which was mentioned in the last chapter as the ventral fissure. At the mid-dorsal line such an open fissure does not appear but it is represented by a septum of connective tissue. Two other zones which probably have much greater significance are marked by a groove which appears on the inner face of each lateral half of the cord. This pair of grooves causes a widening of the central canal and a thinning of the wall of the tube, so that each half of the cord is divided into a dorso-lateral and a ventrolateral zone or column. These zones were first brought to notice by His, who described them in the human embryo. There is reason to suppose that the dorso-lateral zone includes the two sensory columns to be described in later chapters and that the ventro-lateral zone includes the two motor columns.
FIG. 25. A part of a transverse section of the optic lobe of the sturgeon. The cells farther removed from the ventricle are more highly developed.
Observations on these zones in embryos of other vertebrates are much to be desired.
The Development of the Peripheral Nerves
The development of the sensory nerves is a very complex matter, especially in the case of the cranial nerves, and an account of its main outlines only can be given. It was said above that the neural crest shared in the segmentation of the neural tube. The cells of the crest multiply by mitosis and in the trunk region each segment moves latero-ventrad at the sides of the spinal cord, between it and the adjacent ectoderm and mesoderm. At the same time the segments separate more and more from one another until there are formed a series of flaps attached to the dorso-lateral surface of the cord and hanging down on its lateral surface opposite the interval between each two mesodermic somites. Each of these segments of the neural crest may now be spoken of as the anlage of a spinal ganglion. Each anlage moves further ventrad and may wholly lose its connection with the cord (see Figs. 15, 16). The cells are now elongated, spindle-shaped and it is soon noticed that each cell elongates further as if each end were being drawn out into a slender thread. In fact a process analogous to a pseudopodium grows out from each end of the cell. The process from the upper end of the cell, or central process, appears first and is from the first distinctly more slender than the opposite, or pheripheral process. The central process is therefore to be considered as the neurite, the peripheral process as the dendrite. Figure 26 is from part of a transverse section of an embryo of Amblystoma punctatum at the level of the glosso-pharyngeus ganglion. The central processes of the ganglion cells are growing upward, the peripheral processes are not yet formed. Figure 27 shows a few ganglion cells of the trigeminus nerve. The peripheral processes of the cells are seen but the central processes bend backward so that they do not fall in the same section.
The central processes soon reach the dorso-lateral surface of the spinal cord and grow into the cord where they form a bundle of afferent or sensory nerve fibers. The peripheral fiber grows also and with its fellows forms a sensory nerve or the sensory portion of a peripheral nerve trunk. The peripheral process sometimes divides, its branches entering two nerve rami. This is analogous to the branching of the dendrites of central nerve cells. The fibers continue their growth, following the path of least resistance between the muscles and other organs, until they reach the area of their ultimate distribution, for example in the skin, where they divide into terminal branches. The cells from which the fibers have grown out remain in a mass at the side of the spinal cord and form the spinal ganglion.
It is very important to notice that not all the cells of the neural crest give rise to nerve fibers, but as in the central nervous system, a part of the cells become supporting elements and a part go to form the sheaths of the nerve fibers. It has recently been experimentally proved that cells derived from the neural crest migrate out along the bundles of the peripheral processes and form the neurilemma or cellular sheath of Schwann of the nerve fibers. The fibers of the ventral nerve derive their sheath cells partly in this way and in part from the central nervous system, the cells wandering out directly along the ventral roots. The origin of the cells of this sheath shows their genetic relation with the neuroglia cells, and consistent with this is the fact that in the central nervous system the sheath of Schwann is absent, the myelinated fibers being surrounded by glia cells.
FIG. 26. The ganglion of the IX nerve in Amblystoma punctatum at the time of formation of the central processes.
FIG. 27. A few cells of the trigeminal ganglion in Amblystoma, punctatum with the fibers of the ramus mandibularis growing out from them.
In the region of the caudal part of the myelencephalon in all vertebrates the neural crest is relatively small. In cyclostomes and some selachians a complete series of segments of the neural crest in this region is formed. These represent the rudiments of nerves corresponding to the sensory nerves to the skin in the trunk region. In cyclostomes these rudiments give rise to permanent cutaneous nerves, while in all other vertebrates a variable number of these nerves fail to develop. (Compare Chapter VI. ") Jn front of this the neural crest is larger and is divided by a slight constriction into a longer caudal segment for the vagus nerve and a shorter cephalic segment for the glossopharyngeus. The development of the ganglia and fibers of these nerves proceeds in essentially the same manner as in the case of the spinal nerves, except that the ganglia migrate somewhat farther from the brain wall and come to lie outside the muscle somites and against the ectoderm above the gill slits. The ganglion of the glossopharyngeus lies over or nearly over the second gill slit. The vagus is connected with a series of ganglia equal in number to the number of gill slits from the third onward. In cyclostomes (Figs. 28, A and B) the ganglion of the vagus is formed first over the third slit and then grows backwards until a series of ganglia are formed, one over each gill slit. In bony fishes, amphibia and higher forms these ganglia tend to become consolidated and are not symmetrically placed over the gill slits. Where the ganglia reach the ectoderm over the gill slits the ectoderm becomes thickened by multiplication of its cells and at least in some lower vertebrates, cells proliferate from the ectoderm and join the ganglia. The thickening of ectoderm is called the epibranchial placode^ Each ganglion is now composed of two parts, a median part derived from the neural crest and a lateral part derived from the epibranchial placode. The ganglion may now be called the epibranchial ganglion. In man the ganglion petrosum on the IX nerve and the ganglion nodosum on the X nerve seem to represent the epibranchial ganglia of fishes, and ectodermal placodes are described in connection with these ganglia in man (Fig. 31), but no migration of cells from the ectoderm to join the ganglia has been made out. From each ganglion so formed the ramus posttrematicus described in the previous chapter grows downward behind the gill slit and the ramus pharyngeus grows forward and inward to the roof of the pharynx. From the ramus pharyngeus hi forms above the cyclostomes the ramus praetrematicus goes downward in front of the gill slit. It has been suggested that the epibranchial placodes represent ancestral sense organs whose sense cells have come to join the ganglia. In fact very little is certainly known as to the nature of these ectodermal thickenings. The presence of the placodes in man and the separation between the root ganglia and those formed in connection with the placodes suggest that the placodes probably have the same history and function in all vertebrates. It is of the highest importance for the morphology of the nervous system that the exact history and fate of the placodes should be traced in one or more classes of vertebrates and that the distribution, central connections and functions of the fibers formed from ectodermal cells, if any, should be ascertained. This can doubtless, be done by extirpation experiments on embryos.
FIG. 28 A. A diagram of the head of Petromyzon at a stage when two gill clefts are open and the neural crest is segmented into the anlages of the cranial ganglia. From the left side. The dorso-lateral placodes are indicated by vertical and horizontal cross-lines, the epibranchial placodes by oblique lines. After KoltzofT's diagram VIII.
FIG. 28 B. A diagram of the head of Petromyzon at a later stage when all the cranial nerves are to be recognized. After Koltzoff's diagram XIII.
FIG. 29. Three diagrams of the head of Squalus acanthias to show the differentiation of the neural crest into the cranial ganglia. After Neal. A, i, 2, 3, somites.
FIG. 30. Reconstruction of the peripheral nerves in a four weeks human embryo, 6.9 mm. long. Enlarged 16.7 diameters. From Streeter. Ot. v., audi tory vesicle; i, 2, 3, visceral arches; C. i, first cervical nerve; D. i, first thoracic nerve; L. i, first lumbar nerve; S. i, first sacral nerve.
FIG. 31. Reconstruction of the peripheral nerves in a four weeks human embryo, 7.0 mm. long. Enlarged 16.7 diameters. From Streeter.
FIG. 32. Reconstruction of the peripheral nerves in a six weeks human embryo, 17.5 mm. long. From Streeter.
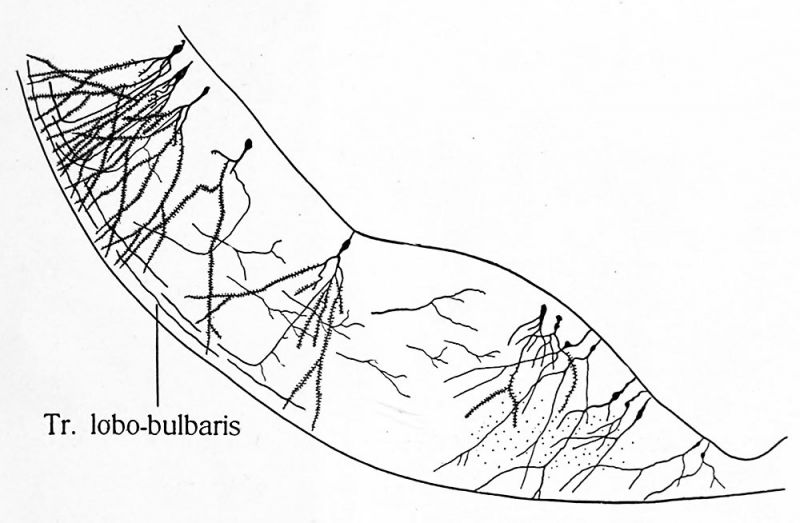
FIG. 33. Three stages in the development of the acustico-lateral system in the sea bass. From H. V. Wilson, a. s., auditory sac; a. s. t., anterior sensory tract; B. s. o., preauditory pit; g. s., gill slit; /. /., lateral line anlage; In., lens; m. con., furrow between midbrain and hindbrain: med., medulla oblongata; n. s., nasal sac; op. n., optic tract; op. s., optic vesicle; s. /., common sensory furrow.
In front of the anlage for the glossopharyngeus nerve there is a segment of the neural crest which gives rise to the sensory root of the VII nerve. This is deeply constricted from the segment for the IX nerve and is wholly separated from the portion belonging to the trigeminus group. While the ganglion of the IX nerve grows down wholly behind the auditory pit, that of the VII nerve comes into contact with the pit, in lower vertebrates in contact with its caudal surface, in higher vertebrates with its mesial wall.
The ganglion extends ventrally beyond the auditory pit and comes into contact with the ectoderm over the first gill slit (spiracle). An ectodermal thickening fuses with the ganglion and from the fused mass the rami praetrematicus and palatinus of the VII nerve and the visceral sensory portion of the ramus hyomandibularis take their origin. This mass is comparable to the epibranchial ganglion of the IX nerve, but the significance of the ectodermal constituent is equally unknown.
The auditory pit is itself a thickening of ectoderm and it plays a part in the development of certain cranial ganglia which gives it the name of a dorso-lateral placode. Where the neural crest anlage of the VII ganglion comes into contact with the auditory pit, cells undoubtedly wander from the walls of the pit and form the ganglion of the VIII nerve, no part of which is formed from the neural crest. In some selachians and bony fishes (Fig. 33) an ectodermal thickening continues for some distance forward and backward from the auditory pit, so that we may speak of a much elongated dorso-lateral placode. The cells which proliferate from the caudal portion of this placode continue to multiply by mitosis and form a large mass which grows and pushes backward along the inner surface of the ectoderm over the roots of the IX and X nerves. This mass grows caudally as a narrow band along the side of the body, ploughing through the deeper layers of the ectoderm. The cells derived from the placode behind the auditory pit form a ganglion lying over the ganglion of the IX or the first part of the X ganglion and send fibers toward the brain, forming the root of the lateral line nerve which is situated near that of the IX nerve as described in the last chapter. The growing placode continues backward along the line of division between the dorsal and the lateral body muscles and at intervals gives rise to sense organs at the surface of the ectoderm. These organs consist of high columnar supporting cells which form the thickness of the epidermis, and shorter pear-shaped sense cells which do not extend to the full depth of the epidermis but do send hair-like processes out beyond the surface. The organs later sink down beneath the surface so that they come to lie at the bottom of the ectodermal pits (pit organs). The pits then become enclosed as tunnel like canals and finally the successive canals unite to form a continuous canal, the canal of the lateral line. Meanwhile the cells of the ganglion send processes caudally beneath or imbedded in the growing placode, which come into relation with the sense organs as they are formed and constitute the nerve of the lateral line.
The cephalic part of the placode, in front of the auditory pit, grows forward and divides into two branches which continue forward and give rise to sense organs in the same way as does the lateral line placode. One branch extends fonvard above the eye and forms the supraorbital row of organs, the other goes ventral to the eye and forms the infraorbital line. Ganglion cells which proliferate from the placode in front of the auditory pit give rise to the rami ophthalamicus superfadalis and buccalis which supply these lines and form the one or two roots of these rami which have been described above (p. 20, 21).
Beneath and in front of the auditory pit a ventral projection of the common placode extends downward and forward on the mandibular arch. This develops in the characteristic manner the hyomandibular line of sense organs. The nerve fibers which supply this row of organs are also derived from the placode cells and form a part of the ramus hyomandibularis.
In this way the acustico-lateral system of sense organs and nerves in most fishes is derived from an extensive thickening of ectoderm on the side of the head, the dorso-lateral placode. Certain important differences in other forms must now be mentioned. The anterior part of the placode is not always directly continuous with the part which forms the auditory pit. Sometimes this part becomes invaginated to form another similar pit lying in front of the auditory pit and in front of the spiracular cleft. This pit may even become an enclosed sac and contain well formed sense cells (Wilson) similar to those of the lateral line organs. In embryos of certain amphibia (Gymnophiona) a deep pit is formed hi front of the auditory pit and independently of it. This gives rise to the nerve which supplies the supraorbital row of sense organs. In Amblystoma punctatum this pit appears earlier than the auditory pit. In cyclostomes (Fig. 28) two separate ectodermal placodes are formed in front of the auditory pit, one in front of the other, which give rise to the supraorbital and infraorbital lines of sense organs and whose ganglia are closely related to the trigeminus and profundus ganglia. It is evident that in the early history of vertebrates the acustico-lateral system occupied three segments and that pits were formed to contain the sense organs. It is probable that at first the portions of the system in the three segments were independent of one another.
In typical fishes the main rows of sense organs are enclosed in canals similar to the lateral line canal. In addition to these chief rows there are often auxiliary rows or groups of pit organs and in many cases the typical canals are incompletely formed, the organs remaining as pit organs. In cyclostomes and amphibia canals are not formed. In selachians there appear along the course of the main rows of organs depressions which sink in as pits and then become deep, narrow tubes and eventually extend beneath the surface to end in one of several masses of jelly-like tissue lodged in concavities of the skull. The deep ends of the tubes expand and develop sense organs which are related in structure to the lateral line organs. These are the ampullae of Lorenzini. The mode of development of the vesicles of Savi and of the nerve sacs of ganoids, both of which belong to the acusticolateral system of organs, is not known.
The complete hiatus in the neural crest in front of the anlage for the VII nerve is connected in some way with the development of the acustico-lateral system opposite this point. Probably this system actually uses up the material of this part of the neural crest. Forward from this, overlying the region of the cerebellum and midbrain, is a large segment of the neural crest which gives rise to the trigeminus group of nerves. It is necessary to say group of nerves, because the adult trigeminus is formed from two nerves which arise independently in vertebrate embryos.
From the crest covering neuromeres v and vi in selachians grow down two ganglionic anlages which remain connected peripherally so that they form an U-shaped loop, having two connections with the brain. The caudal of these two anlages forms the ganglion of the N. trigeminus proper and gives rise to the sensory portion of the rami maxillaris and mandibularis trigemini. The anterior arilage loses its connection with the brain and the fibers which grow centrally from its ganglion cells follow caudally over the trigeminal ganglion and enter the brain along with the root of the trigeminus. The peripheral fibers from this ganglion run cephalad through the dorsal part of the orbit and form the N. ophthalamicus profundus. Figure 29 shows the early stages in the differentiation of these two ganglia. While the above description applies to selachians it may be taken in a general way as typical for vertebrates. In cyclostomes the two nerves are more independent in their origin and adult relations, and in all vertebrate embryos the two ganglia can be recognized.
In certain selachians an anlage of a sensory nerve is formed from the neural crest between the mesencephalon and diencephalon. It is known as the nervus thalamicus (Fig. 29). The anlage is small, comes to be closely related to the profundus trunk and may furnish material for the ciliary ganglion. In the chick the ciliary ganglion is formed in part of cells which migrate out from the neural tube along the fibers of the oculomotor nerve and in part of cells which come from the ophthalamicus profundus ganglion.
FIG. 34. A transverse section through the nasal sac of an embryo of a bony fish at about the time of hatching, to show the origin of the fibers of the olfactory nerve.
In front of this point the neural crest does not give rise to any nerve which is found constantly in vertebrates. In selachians, however, the N. terminalis (see p. 31) possesses a ganglion and in Squalus acanthias develops in much the same way as do other sensory nerves. In the chick ganglion cells are found in connection with the olfactory nerve during its development which may bear some relation to this nerve in fishes.
The olfactory nerve differs from all other nerves in vertebrates in that its cells of origin arise and remain in the ectoderm. These are the sensory cells of the nasal epithelium. From the inner end of each of these cells arises a fiber (Fig. 34) which grows toward the forebrain and enters the bulbus olfactorius. These constitute the olfactory nerve. The fibers are never myelinated. Although so little is certainly known of the details of the part played by ectodermal placodes in the formation of the sensory nerves and ganglia, it is clear that the nervous ectoderm is not limited to the neural plate. It is probable that the limits of the ectoderm which give rise to the nervous system are indefinite and variable. All that can be said is that the neural plate gives rise to the spinal cord and brain with the neural crest, while the olfactory organ and nerve and the ganglia and nerves of the acusticolateral system are derived from ectoderm adjoining the neural plate. These ganglia and nerves, including the olfactory, are to be compared with those derived from the neural crest. All are derived from ectoderm which in the early embryo is closely adjacent to the lateral border of that which forms the central nervous system, and the neural crest is wanting in those segments in which the olfactory and acustico-lateral systems develop. The acusticolateral ganglion cells remain in the general ectoderm for a time, the olfactory permanently so. It is probable that the epibranchial placodes, which are much farther removed from the neural plate, give rise to a part of the visceral sensory nerves, but whether general visceral or gustatory fibers are so formed is unknown. In Amphioxus and cyclostomes the sensory ganglion cells remain throughout life bipolar, spindle-shaped cells such as have been described. During the course of development in true fishes a part of the cells become unipolar and in higher vertebrates most of them do so. This change is brought about by a sort of bending of the cell body by which the fibers at the two ends are brought together at one side (Fig. 35). Then by the growth of that part of the cell to which the fibers are attached both fibers come to be borne on a single process.
FIG. 35. Two figures representing the formation of unipolar cells in the spinal ganglion of the dog embryo. After Van Gehuchten. r., root; p., peripheral nerve.
The development of the motor nerves takes place somewhat later than that of the sensory nerves. In the ventral part of the spinal cord and brain, where motor nerves are to be formed, certain large neuroblasts send their neurites out on the ventral surface. At this period numerous cells wander out of the cord or brain, following the neurites in their growth, migrate and multiply and eventually form the sheath of Schwann (cf. p. 51 above). The neurites grow until they reach the muscles which they are to innervate, when they branch or expand on the surface of the muscle fibers in the form of special motor end organs. These ventral motor nerves innervate only those muscles which are derived from the mesodermic somites. It has recently been shown that in amphibia fibers of a single nerve are distributed to muscle fibers of two adjacent muscle segments. This fact is wholly inconsistent with the view held by some anatomists that from the earliest embryonic stages processes of nerve cells are in connection with their muscles by means of strands of protoplasm which become elongated to form motor nerve fibers.
The nerve fibers which innervate the muscles derived from the lateral mesoderm are developed in the same way from neuroblasts which lie in the lateral parts of the central gray. In lower vertebrates these fibers pass out of the cord or brain together with or close beneath the dorsal sensory nerve roots and pass through the sensory ganglia. In higher vertebrates a part of these fibers pass out with the ventral roots and in the cat, dog and monkey all of them are said to have this course.
Morphology of the Head
In order to understand the brain and cranial nerves it is necessary to take into account the morphology of the organs of the head. For this reason a summary of the more important conclusions of comparative embryology and anatomy will be given here.
At an early stage in the embryo the dorsal border of the sheets of mesoderm becomes divided into a series of segments, which soon separate from the undivided lateral mesoderm and lie as independent blocks or sacs at the sides of the neural tube. These are known as mesodermic somites. The first somites to be formed lie in the region which will form the neck and the segmentation continues from that point forward and backward. In front of this point there are formed in selachians and cyclostomes a definite number of somites which were first described by vanWijhe in 1882. These head segments bear constant relations to other organs and may be designated by numbers indicating their order from before backward. Somite i lies behind and below the optic vesicle and gives rise to those eye-muscles which are innervated by the III nerve (mm. rectus superior, rectus inferior, rectus internus, obliquus inferior). In front of somite i there is found in selachians a pair of lateral masses of mesoderm known as anterior he^d cavities. These are believed to represent a still more anterior pair of somites which disappear in vertebrates without forming any recognizable structure. Somite 2 is larger and is directly connected with the mandibular arch. It gives rise to the muscle innervated by the IV nerve (m. obliquus superior). Somite 3 is connected with the hyoid arch and gives rise to the muscle innervated by the VI nerve (m. rectus externus). Somite 4 lies mesial to or slightly behind the auditory pit and is known as the first postauditory somite. It gives rise to muscle on the dorsal surface of the head in cyclostomes; in all other vertebrates breaks down and disappears. Somite 5 in cyclostomes gives rise to a muscle lying next behind that of somite 4; in all other vertebrates disappears. In cyclostomes all the following somites form permanent myotomes, and myotome 10 together with several following it send muscle buds down behind and forward beneath the branchial apparatus to form the sub-branchial muscles corresponding to the tongue musculature of higher forms.
In selachians a variable number of postauditory somites may first form some muscle fibers and afterwards break down and only contribute to the formation of mesenchyme. The first somite to form permanent muscles is number 7 or 8 in most selachians.
Somite 8 or 9 and several following it send muscle buds ventrad and forward beneath the branchial apparatus to form the subbranchial muscles as in cyclostomes. In higher vertebrates the abortion of certain postauditory somites is more complete; and the number which disappear may be greater. In various classes of vertebrates more or fewer vertebral segments may be secondarily fused with the primordial cranium, entering into the occipital region of the skull. In mammals and perhaps man four such vertebrae are added to the cranium. In all vertebrates the sub-branchial or tongue musculature is formed from muscle buds derived from post-branchial somites.
In vertebrates above cyclostomes the breaking down of somites behind the ear produces a shifting of the relative position of the somites and nervous organs. The auditory sac is known to shift backward during development through the length of from one to three hindbrain neuromeres (Fig. 18). Also the permanent somites shift forward somewhat so that at every stage the existing somites lie close behind the auditory sac. The shifting forward of somites at the same time affects their position relative to the branchial apparatus. Also, the disappearance of somites leads to the disappearance of the ventral motor nerves which should innervate them, and apparently the shortening process in the region behind the ear leads to a decrease of the ectodermal area and the reduction or disappearance of one or more nerves of the skin. Finally, the development of the hypoglossal nerve is determined by the migration into the jmb-branchial region of the muscle buds which it is destined to innervate.
The lateral mesoderm in the meantime becomes segmented in a passive manner by the formation of the gill slits. In most vertebrates these slits seem to have no definite position relative to the somites. There is evidence, however, that the segmentation of the lateral mesoderm and pharynx in primitive vertebrates corresponded to that of the dorsal mesoderm. The mandibular and hyoid arches are connected with the second and third somites respectively. The third branchial arch is connected with the fourth somite, and in Petromyzon the total number of gill arches (eight) corresponds to the number of somites (somites 2 to 9) in front of that one which forms the first sub-branchial muscle bud. The space occupied by the branchial apparatus, however, is always greater than that occupied by the corresponding number of somites and there is apparently a lack of correspondence between muscle segmentation and gill segmentation. The reason for this is found first in the fact that the gills become voluminous structures which require more space than the corresponding number of somites, and second in the disappearance and shifting of somites in the postauditory region mentioned above.
The segment of the lateral mesoderm between each two . gill clefts is called a branchiomere and gives rise to the skeleton and muscles of the branchial arch. The mouth is regarded as representing a pair of gill slits and the mandibular arch as a branchial arch. The muscles of mastication which are attached to the lower jaw are derived from the lateral mesoderm and these together with most of the muscles of the branchial arches are visceral muscles, homologous with those in the wall of the alimentary canal in other segments of the body. In front of the mouth some rudimentary structures have been found in selachians which are thought to be vestiges of brancial arches which once existed anterior to the mouth. Possibly two such arches were present in primitive vertebrates.
The shifting of the auditory vesicle mentioned above changes its position with regard to the branchial arches also. From lying over the hyoid arch or second gill slit, it comes to lie over the first branchial arch or third gill slit. The bones of the middle ear in mammals are regarded as modified portions of the skeleton of the mandibular and hyoid arches. Recent researches show, however, that in the rabbit the stapes is a derivative of the auditory capsule.
It is believed that the ancestors of vertebrates had a mouth farther forward than the present mouth. The ancestral mouth is represented by a vestigeal structure formed in embryos and retained in the adult as a rudimentary organ, the hypophysis. The hypophysis makes its appearance as a median ectodermal pit slightly ventral to the olfactory pits. Opposite this pit there lies in selachians a median mass of entoderm connecting the two anterior head cavities mentioned above. This most anterior mass of entoderm is present in amphibia also (Fig. 36), although the anterior head cavities are not separately developed. In this is seen the vestige of a communication between the hypophysis and the archenteron. In many vertebrates an outgrowth of entoderm from this region, known as Seesel's sac, enters with the ectodermal pit into the formation of the adult hypophysis. In cyclostomes the ectodermal pit becomes deep and large, and in Bdellostoma (Kupffer) has an open communication with the archenteron at an early stage. This communication is afterward obliterated but the hypophysis remains throughout life as a large sac open to the exterior and extending beneath the brain. The communication with the archenteron has been described also for the sturgeon. Also in Amphioxus, where the permanent mouth is formed from a gill slit, a canal which opens externally farther forward communicates with the archenteron at an early stage of development (Legros). The relations of mouth and hypophysis in Amphioxus and Petromyzon are represented diagrammatically in the accompanying figure from Legros (Fig. 37). The ancient vertebrate mouth, or paleostoma, is to be thought of as lying very near the anterior end of the animal, just beneath the region of the olfactory organs which alone extend farther forward. In the sides of the canal which connected this mouth with the present pharyngeal region (paleopharynx) were one or two gill clefts and the present mouth has been formed from the second or third pair of ancestral gill clefts. Behind the mouth a variable number of clefts are present in existing vertebrates: in cyclostomes, 7 to 35; in selachians, 5 to 7; in amphibia and higher forms, 5 to 4. It is probable that the number in primitive vertebrates was large. The number of clefts has been gradually reduced from behind forwards (except in Myxinoids), probably because the greater development of a few anterior gills and the concentration of organs thus made possible is more advantageous.
FIG. 36. A median sagittal section of the head of an embryo of Amblystoma punctatum, to show the relations of epiphysis, velum, paraphysis, hypophysis, Seesel's sac, etc.
FIG. 37. Diagrams representing the development of the buccal cavity, hypophysis and nasal pit in Amphioxus and Petromyzon. After Legros.
At the same time that the disappearance of the more caudal gills reduces the number of aortic arches and tends to bring the heart farther forward, the expansion of the permanent gills actually pushes the heart caudally farther into the trunk. Thus in selachians it is shown that the opening of the duct of Cuvier into the sinus venosus moves back during development from a position in the second trunk segment to a position in the tenth trunk segment (Hoffmann).
We have to think of the development of the head as a gradual process which has been taking place during the phylogenetic development of vertebrate animals, the causes for which are to be found in the conditions of life, the food, habits, modes of locomotion, importance of the various senses, etc. The ancestors of vertebrates were very slightly specialized at the anterior end and the various specialized structures formed there make up the head. Special sense organs are formed in the head region (acusticolateral system, taste buds, ear, eye, olfactory organ) and each of these profoundly affects in a characteristic way the structure of the brain, and often modifies surrounding organs, determines the form of the skull, etc. The formation of the rigid cranium has come about largely for the protection of the sense organs and the existence of this cranium has led to the disappearance of the postauditory somites and to the modification of the preauditory myo tomes into muscles which move the eye-ball. Some of the most striking marks of the specialized head are due to the disappearance of various structures, the postauditory somites, the ancient mouth, certain gills, etc. The disappearance of these organs has led to the disappearance of their nerves and to great changes in the central nervous system. Furthermore, both the rise of new structures and the passing away of old ones have brought about shifting of organs which is of great importance in determining the structure of the nervous system. Of chief interest is the shifting of nerve roots due to these or other causes.
The ophthalamicus profundus nerve arises from the neural crest over the mesencephalon; its root has shifted back to join that of the trigeminus. The trigeminus arises over the metencephalon and in cyclostomes remains permanently attached to that segment. In all other vertebrates it moves back one segment to the first neuromere of the myelencephalon. The facialis has shifted back not only one segment, but two, leaving the neuromere between it and the trigeminus without any nerve root. The shifting of the VII is probably due to the shifting of the auditory sac which has in part pushed it back and in part moved back past it. By this movement the IX and X nerves are also affected. There is reason to believe that the IX has moved back one segment in cyclostomes, and that the IX and X have moved back two neuromeres in fishes and three in reptiles, birds and mammals.
A word should be said regarding the complex character of the X nerve. There is reason for thinking that each one of the epibranchial ganglia of the vagus represents an independent branchial nerve, and that these nerves one after another beginning at the caudal end of the gills have joined the nerve next anterior until all have united into a single root. In the myxinoids this process has gone one step farther than in other vertebrates, and the vagus itself is joined to the glossopharyngeus. The actual shifting forward and fusion of these nerves does not now take place during the ontogeny, so that it is to be regarded as a very primitive feature of vertebrate development.
It is impossible in this book to follow further the evolution of the vertebrate head. The accompanying tables and figures are intended to make more clear the brief statements that have been made. Figure 38 is meant to show the relations of the various structures which enter into the primitive head segments. The two tables give further facts which should be taken into account in connection with this diagram. Table A is abbreviated from the tables of Hoffmann showing the shifting of organs which takes place during the development of the selachian head. Table B is meant to show the elements present in each segment of the head and the more important reductions and changes of position which have taken place.
FIG. 38. A diagram of segmentation in a generalized vertebrate head. In its general features the diagram follows Petromyzon more closely than any other form. So, the number of the gill clefts, their position relative to the somites,' the position of the auditory vesicle, and the formation of hypobranchial muscles from myotome 10 and following, are taken from Petromyzon. The neuromeres, the nervus terminalis, the nervus thalamicus, the relation of the lateral lines of the head to the auditory vesicle, and the praeoral entoderm are taken from selachians. The sensory nerve roots are represented as retaining their attachment to the dorsal surface of the neural tube where they were formed from the neural crest. The segmental position of these roots is about that which they have in the embryo of Petromyzon, except the root of N. X, which has been shifted back a little farther than it is in Petromyzon. The general cutaneous nerve shown in dotted outline over somite 6 is the Vagusanhang of HATSCHEK in Ammocoetes and the nerve which unites with the vagus root in the embryo of selachians. This is the second dorsal spinal nerve in Petromyzon dorsatus and other primitive cyclostomes. The position of the viscero-motor nuclei somewhat caudal to the several roots is indicated. The viscero-motor nucleus of the vagus and accessorius is shown as a single' large nucleus extending through two segments. It might more properly have been continued caudally until it came into connection with the viscero-motor nucleus of the trunk nerve. The accessorius nerve is not shown. The somatic motor nucleus and root are shown for all the somites except somite 4, where they are shown in dotted outline. The nerve for somite 4 is absent in Petromyzon, but is present in Bdellostoma. It is possible that this root has joined with N. VI in gnathostomes.
The process of cephalization in vertebrates has consisted chiefly in (i) the development of special sense organs; (2) the consequent enlargement of the brain; (3) the formation of a rigid cranium to protect sense organs and brain; (4) the disappearance of certain muscle segments and the change of position and function of others (eye- muscles) ; (5) the reduction in number of gills, the formation of a new mouth and the expansion of the persistence gills; (6) the disappearance of various nerves owing to the reduction of somites and gills; (7) the shifting of position of various organs and nerve roots due to these changes; and (8) the great development in higher vertebrates of the so-called higher brain centers. At every step in the evolution of vertebrates these changes have left their impression on the nervous system so that the nervous system to a greater extent than any or all other organs preserves a record of the course of phylogenetic development. For this reason not only does a knowledge of the nervous system throw light upon the morphology of the head, but a knowledge of the evolution of the head is necessary for a true understanding of the nervous system.
Table A.
Shifting of organs during development of selachian embryo.
Length of embryo 12^-13 mm. 40 mm.
Duct of Cuvier shifts from trunk segment 2 to trunk segment 10
Omphalo-mesenteric artery " .... 4- 5 " 12-13
Sixth branchial artery, from occipital segment . 3 o
Ostium of Muller's duct, from trunk segment .. 5-6 " n
II
12
13
14
15
16
17
xi
xii
xiii
xiv
XV
xvi
xvii 13
7
8
9
10
ii
12
|r. 4
br. 5
br. 6
a larger number of branchial arches in lower cyclostomes and primitive vertebrates
,p. Cycl.
diments
rmanen miculi.
3 sp. Cycl.
in Gnath( t roots in i
4 sp. Cycl.
jstomes. [eptanchus
5 sp. Cycl. permanent root
6 sp. Cycl. s in Acanthias.
7 sp. Cycl. permanent root in Gnath.
8 sp. Cycl.
2Sp. ?
Gnath.
nucleus spinal is acustici.
Bdell.
V
motor ii
5 Bdell.
w
isciculus a
6 Bdell.
X
nd ventral
7 Bdell.
y
motor column.
8 Bdell.
z
9 Bdell. i sp. Fiirb.
10 Bdell.
2S P .
mp't > 'ct'd to . X.
comp't col'ct'd into N. X.
comp't col'ct'd into N. X.
comp't in r. br.-int. X
comp't in r. br.-int. X
comp't in r. br.-int. X
comp't in r. br.-int. X
us commissuralis of Cajal and visceral sensory column of spinal cord.
Is have not been fully distinguished fin any vertebrate.
Lot. N. bol'ct'd nto N.X
mot. N. col'ct'd into N.X
mot. N. col'ct'd into N.X
N. access.
N. access.
N. access.
N. access.
med as the lateral motor column of spinal cord.
Front edge of anterior fin (18 mm. embryo) ... 4 to trunk segment 9 Bark edge ... 14 i 7
Cloaca ...44-48 4 8- 49
Anterior end of coelom. from occipital segment, 3 n
Demonstration of Laboratory Work
- Study the development of the neural plate, neural tube and brain in embryos of the salamander, a selachian, pig or chick.
- In the same material study the neural crest and the formation of the spinal and cranial ganglia.
- Study the dorso-lateral and epibranchial placodes and the development of the auditory and lateral line organs, eye and olfactory organ.
- Study the relations of the chorda, the anterior end of the entoderm and mesoderm, hypophysis and olfactory pit and neuropore in selachian or salamander embryos.
- Notice the neuro meres in the hindbrain of any embryos and look for the shifting in position of the auditory vesicle and the trigeminus and other nerve roots.
- In any selachians or cyclostomes that are available make careful dissections of the nerves in the spino-occipital region, noting the number of dorsal and ventral roots present and their position relative to the vagus and glossopharyngeus. The smaller nerve roots can be brought to view by painting the surface of the brain with i or 2 per cent. osmium solution, which should be thoroughly w r ashed away with water as soon as the nerves are blackened.
- Study the formation of the neuroblasts in the central nervous system and the growth of central and peripheral processes from the sensory ganglion cells. This can be done well in amphibian embryos stained with haemalum and acid fuchsin.
Literature
Bardeen, C. R.: The Bimeric Distribution of the Spinal Nerves in Elasmobranchii and Urodela. Amer. Jour, of Anat, Vol. 3. 1904.
Brauer, A.: Beitrage zur Kenntniss der Entwickelung und Anatomic der Gymnophionen. Zool. Jahrb., Suppl. 7, 1904.
Braus, Herman: Beitrage zur Entwickelung der Muskulatur und des peripherischen N ervensystems der Selachier. Morph. Jahrb., Bd. 27, 1899.
Carpenter, F. W.: The Development of the Oculomotor Nerve, the Ciliary ganglion, and the Abducent Nerve in the Chick. Bull. Mus. Comp. Zool. Harvard College, No. 172. 1906.
Disse. T.: Riechschleimhaut und Riechnerv beiden Wirbelthieren. Merkel u. Bonnet's Ergebnisse. Bd. n. 1901.
Dohrn, Anton: Die Entstehung der Hypophysis bei Petromyzon planeri. Mitth. Zool. Sta. Neapel.
Eycleshymer, A. C.: The Development of the Optic Vesicles in Amphibia. Jour. Morph., Vol. 8. 1890.
Froriep, A.: Ueber ein Ganglion des Hypoglossus und Wirbelanlagen in der Occipitalregion. Arch. f. Anat. u. Physiol., Anat. Abth., 1882.
Froriep, A.: Ueber Anlagen von Sinnesorganen am Facialis, Glossopharyngeus und Vagus, ueber die genetische Stellung des Vagus zum Hypoglossus und ueber die Herkunft der Zungenmuskulatur. Arch. f. Anat. u. Physiol., Anat. Abth., 1885.
Froriep, A.: Die Entwickelung des Auges der Wirbelthiere. Hertwig's Handbuch der Entwickelungslehre. 1905.
Fuchs, H.: Bemerkungen ueber die Herkunft und Entwickelung der Gehorknochelchen bei Kaninchen-Embryonen u. s. w. Arch. f. Anat. u. Entvvick. 1905. Suppl.-Bd.
Fiirb ringer, Max: Ueber die spino-occipitalen N erven der Selachier und Holocephalen und ihre vergleichende Morphologic. Gegenbaur's Festschrift. Bibliography.
Gage, Susanna Phelps: A Three Weeks Human Embryo, with especial Reference to the Brain and Nephric System. Amer. Jour. Anat., Vol. 4. 1905.
Gaupp, E.: Zirbel, Parietalorgan und Paraphysis. Merkel u. Bonnet's Ergebnisse, Bd. 7. 1897.
Harrison, R. G.: Ueber die Histogenese des peripheren Nervensystems bei Salmo salar. Arch. f. mik. Anat. u. Entw., Bd. 57. 1901.
Hill, Charles: Developmental History of the Primary Segments of the Vertebrate Head. Zool. Jahrb., Abth. f. Anat. u. Ontog., Bd. 13. 1899.
His, W.: Zur allgemeine Morphologic des Gehirns. Arch. f. Anat. u. Physiol., Anat. Abth., 1892.
His, W.: Ueber das frontale Ende des Gehirnrohres. Arch. f. Anat. u. Entw., Anat. Abth., 1893.
His, W.: Anatomic menschlichen Embryonen.
His, W.: Die Entwickelung des menschlichen Gehirns wahrend der ersten Monate. Leipzig, 1904.
Hoffmann, C. K.: Ueber die Metameric des Nachhirns und Hinterhirns und ueber Beziehungen zuden segmentalen Kopfnerven bei Reptilien-Embryonen. Zool. Anz., Bd. 12. 1889.
Hoffmann, C. K.: Beitrage zur Entwickelung der Selachii. Morph. Jahrb., Bde. 24, 25, 27. 1896-1899.
Johnston, J. B.: The Morphology of the Vertebrate Head from the Viewpoint of the Functional Divisions of the Nervous System. Jour. Com. Neur. and Psych. Vol. 15. 1905. Bibliography.
Koltzoff. N. K.: Entwickelungsgeschichte des Kopfes von Petromyzon planeri. Bull, de la Soc. Imper. d. Natural, de Moscou, Annee 1901, No. 3 .-4. 1902.
von Kupffer, C.: Studien zur vergleichenden Entwickelungsgeschichte des Kopfes der Kranioten. Munchen u. Leipzig. 1894-1900.
von Kupffer, C.: Die Morphogenie des Centralnervensystems. Hertwig's Handbuch der Entwickelungslehre. 1905.
Legros, Robert: Developpment de la cavite buccale de 1'Amphioxus lanceolatus. Archive Anat. Micros., Tome i, 1898.
Locy, W. A.: A Contribution to the Structure and Development of the Vertebrate Head. Jour. Morph., Vol. n. 1895.
Minot, C.S.: A Laboratory Text-book of Embryology. Philadelphia. 1903.
Neal, H. V.: The Segmentation of the Nervous System in Squalus acanthias. Bull. Mus. Com. Zool. Harvard College, 31. 1898.
Neal, H. V.: The Development of the ventral nerves in Sdachii. I. The Spinal Nerves. Mark Anniversary Volume. 1903.
Platt, Julia B.: A Contribution to the Morphology of the Vertebrate Head based on a Study of Acanthias vulgaris. Jour. Morp., Vol. 5. 1891.
Price, G. C.: Some Points in the Development of a Myxinoid (Bdellostoma Stouti, L.) Verhdl. Anat. Ges. 10. Vers. Berlin. 1896.
van Wijhe, J. W.: Ueber die Mesodermsegmente und die Entwickelung der Nerven des Selachierkopfes. Amsterdam. 1882.
Wilson, H. V.: The Embryology of the Sea Bass (Serranus atrarius). Bull. U. S. Fish Com., Vol. 9. 1891.
Zimmermann: Ueber die Metamerie des Wirbelthierkopfes. Verhdl. d. Anat. Ges. 5 Versam. Munchen. 1891.
Johnston JB. The Nervous System of Vertebrates. (1907) Blakiston's Son & Co., London.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Cite this page: Hill, M.A. (2024, April 18) Embryology Book - The Nervous System of Vertebrates (1907) 3. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_The_Nervous_System_of_Vertebrates_(1907)_3
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G