Book - The Frog Its Reproduction and Development 4
| Embryology - 9 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Rugh R. Book - The Frog Its Reproduction and Development. (1951) The Blakiston Company.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter 4 - Fertilization of the Frog's Egg
- "The unfertilized egg dies in a comparatively short time, while the act of fertilization saves the life of the egg and allows it to give rise, theoretically at least, to an unlimited series of generations. . . . Fertilization makes the egg immortal." (J. Loeb, 1913.)
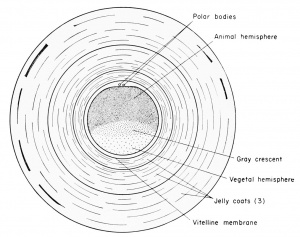
Fertilization generally is regarded as a two-fold process, involving first the activation of the egg. This can be accomplished by the sperm or by a variety of parthenogenetic agents. Second, there is the mixing of hereditary (nuclear and chromosomal) potentialities, a process known as amphimixis. Insemination means simply the exposure of the eggs to spermatozoa. The most accepted evidence for activation is an elevation of the vitelline membrane away from the egg and its transformation into what is then called a fertilization membrane. The vitelline membrane can be lifted from the ovarian eggs of the frog in distilled water or calcium-free Ringer's solution at pH 7.5 to 10.2. This purely physical change seems to be the result of progressive cytolysis or coagulation of the egg cortex, and the membrane itself has the consistency of thin cellophane. There are, of course, physiological changes in the egg accompanying fertilization which are so dynamic that even the microscopic surface pigment granules become violently active. The ultimate proof of successful fertilization is, of course, the diploid condition of the chromosomes of the zygote.
As the frog's egg is shed into the water, the male (in amplectic
embrace) simultaneously and by active expulsion movements sheds
clouds of spermatozoa over the eggs. Fertilization in Anura is therefore external. The jelly, deposited on each egg as it passed through the
oviduct, swells almost immediately thereafter, partially protecting
the egg against multiple sperm entrance. The swollen jelly appears to
be arranged in three layers, being twice the thickness of the egg diameter. Polyspermy, or multiple sperm invasion of the egg, occurs
naturally in some telolecithal eggs such as those of the bird. It is
also common in urodele eggs, but not so with the Anura (e.g., the
frog). More effective than the jelly in limiting fertilization to one
sperm is the negative chemical and/or physical reaction of the egg
toward any extra spermatozoa after one of them has made contact
with the egg surface. Simultaneously with sperm activation there appears an immune reaction of the egg to the invasion of any accessory
spermatozoon, even of the same species. In any case, one and only
one spermatozoon nucleus normally fuses with the egg nucleus. Any accessory spermatozoa which are successful in invading the egg attempt to divide independently of the egg nucleus and then degenerate. Extensive polyspermy, which may occur in aged eggs, interferes drastically with the cleavage mechanism and the eggs reach an early cytolysis and death.
The effective frog spermatozoon always enters the egg in the animal hemisphere. This does not deny the possibility of sperm entrance in the metabolically more sluggish vegetal hemisphere. The sperm nucleus which conjugates with the egg nucleus is one that enters by way of the animal hemisphere. The factors which limit effective sperm entrance to the animal hemisphere may be physical and/or chemical. In any case, the reaction is rapid and the entire egg becomes resistant to excess sperm entrance. In Amphioxus the sperm entrance is characteristically in the vegetal pole, and in other forms there may be no such polar restrictions.
Sperm contact and penetration of the egg has two rather immediate effects. First, it seems to allow the superficial jelly to swell to its
maximum, by imbibition. The jelly on the unfertilized egg will expand,
but to a lesser degree. The expansion of the jelly is at quite a uniform
rate and can be observed under low power magnification against a
dark background. Within 5 minutes there has been about 30 per cent
swelling, in 15 minutes about 75 per cent swelling, and thereafter the
imbibition is slower. Eventually the thickness of the three jelly layers is
several times the diameter of the egg. Second, another effect of sperm
penetration is that there is an almost immediate loss of water from the
egg so that a space appears between the egg surface and the enveloping vitelline membrane now known as the fertilization membrane. This
is known as the perivitelline space, filled with a lluid, within which
the egg is free to rotate. Since the mature egg has a definite yolkcytoplasmic gradient, it slowly rotates within the gravitational field
(inside the fertilization membrane) until the black pigmented animal
hemisphere is uppermost and the heavier yolk-laden vegetal hemisphere is lowermost. When eggs are artificially inseminated with frog
sperm in the laboratory, this rotation will be complete within an hour
after insemination and the egg mass will present a uniform black appearance from above, provided there is adequate water coverage.
Completion of Maturation of the Egg
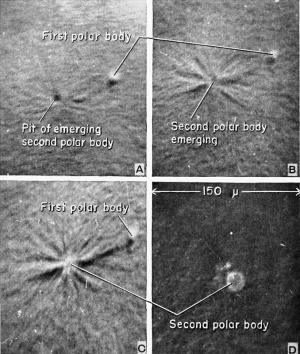
At the time of insemination the frog's egg nucleus is in the metaphase of the second maturation division, and the egg is provided with a vitelline membrane and a covering of unswollen jelly. The activation of the egg by the spermatozoon brings on a resumption of the maturation process so that the suspended mitosis is completed, and the second polar body is given off into the perivitelline space.
Occasionally at the time of insemination eggs will show a minute polar body pit or fovea, a surface marking indicating the point of previous emergence of the first polar body. The first polar body can be found generally as a translucent bead, floating freely within the perivitelline space in the general vicinity of the animal hemisphere. Within 7 to 10 minutes (at ordinary laboratory temperatures) a distinct pit will appear in the animal hemisphere, and this marks the position of the activated amphiaster. Apparently sperm activation causes the whole amphiaster to recede slightly into the egg, or such slight movement might be the result of cortical changes following activation, since the amphiaster is of quite different consistency from the balance of the egg. In any case, there appears a small pit measuring about 16 microns in diameter and often near the first polar body. Within another 15 to 20 minutes (a total of 25 to 30 minutes from insemination) the pit seems to disappear but only because there is seen emerging from it the second, and also translucent, polar body. Within about 1 minute this second body (nucleus) has emerged and its diameter measures about 26 microns, indicating that it was squeezed through an orifice somewhat smaller than its own diameter. There remains no evidence of any pit and, even though the egg nucleus is just below the surface, there is no superficial evidence of its presence. The egg nucleus now recedes into the egg, re-forms, and moves slightly in the direction of the approaching sperm nucleus. The path of the egg nucleus is not marked by any pigment as is the path of the invading spermatozoon.
Penetration and Copulation Paths of the Spermatozoon
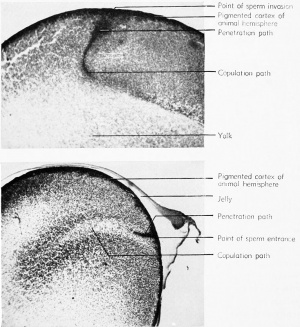
The sperm head generally makes a direct perpendicular contact with the egg cortex, through the unswollen jelly and the vitelline membrane. Usually the entire spermatozoon enters the egg, taking several minutes for the complete invasion of the cortex. The tail piece may be broken off, but the head and middle piece continue through the egg substance in the general direction determined by the direction of penetration, usually along the egg radius. This is therefore known as the penetration path. Almost immediately, however, the sperm head enlarges and loses its identity and is difficult to find by any known cytological method.
In the process of invading the egg, the spermatozoon takes along with it some of the pigment granules of the surface (cortical) layer so that a cone-shaped penetration path can be seen in sections cut in a parallel plane. This path is rather straight, indicating a certain amount of directional force on the part of the penetrating spermatozoon. The pigment path is the only evidence of the sperm movement, since the sperm nucleus is so indistinct that it cannot be located in sectioned eggs.
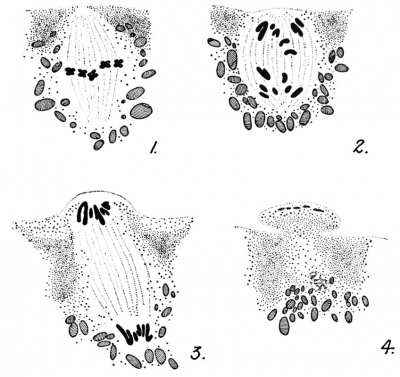
The middle piece of the spermatozoon, containing the centrosome, enters the egg behind and attached to the sperm head or nucleus. As this sperin head (nucleus) swells and loses its identity and establishes the penetration path, the associated centrosome divides. The resulting
centrosomes establish an axis at right angles to the direction of sperm
progression. Frequently, however, the sperm penetration path will not
lead the sperm nucleus directly to the egg nucleus, or to the final
position of the egg nucleus (which itself moves away from the cortex) .
In these cases the sperm path is diverted so that it will meet the egg
nucleus. This new direction is designated as the copulation path because it results in the copulation (or fusion) of the two nuclei. By the
time the two nuclei meet, the two sperm centrosomes are then well
separated and are ready to form the division spindle for the first
cleavage. It is probable that the chromosomes from the two sources do
not pair off before the first division, which occurs within 2Vi hours
after insemination at normal laboratory temperatures.
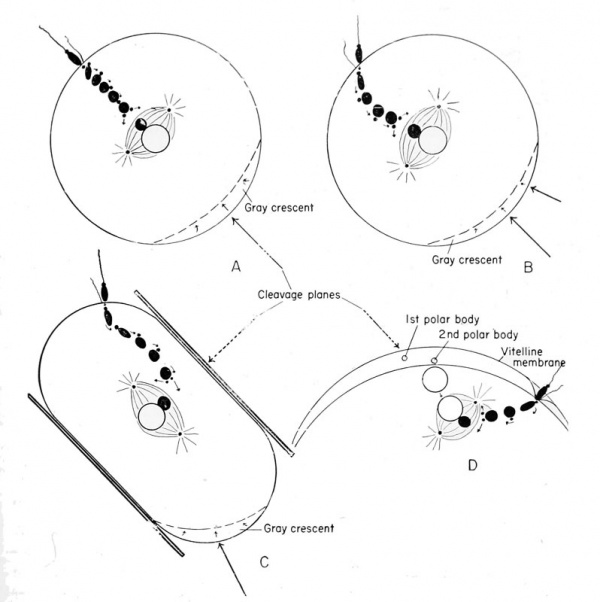
Symmetry of the Egg, Zygote, and Future Embryo
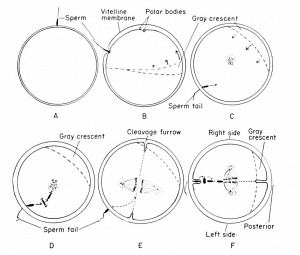
As has been emphasized, the egg polarity and the radial (rotatory) symmetry about its axis are established while it is in the ovary during the process of growth or yolk acquisition. One can draw an imaginary line from the center of the animal pole and through the nucleus which will pass through the geometrical center of the egg, and such a line will pass through the center of the vegetal hemisphere. This line represents the primary axis around which there is radial symmetry. The effective spermatozoon may enter the egg at any point within the animal hemisphere. This may be close to the egg nucleus (toward the center of the animal hemisphere), almost at the equator, or between these extreme positions. It is probably very seldom that penetration occurs just above the second maturation spindle, in the center of the animal hemisphere (i.e., in the egg axis). For this reason, it is safe to assume that the majority of penetration points will be at some other region of the animal hemisphere. This means that a third point is established (i.e., by sperm penetration), the other two being any two points on the linear axis of the egg. These three points will establish a plane, the penetration path plane, which is of major importance in establishing embryonic planes. The egg therefore loses its rotatory symmetry at the moment of sperm penetration.
The significance of the sperm entrance point is brought out when it is realized that it immediately establishes the antero-posterior axis or bilateral symmetry of the future embryo. That side of the animal hemisphere which is toward the sperm entrance will be toward the anterior, the opposite will be toward the posterior, of the future embryo. This antero-posterior plane (formerly the sperm penetration path plane) separates the future embryo into right and left halves.
Immediately upon invasion by the spermatozoon tiie egg substance
becomes more labile and there is a streaming of the protoplasm toward
the animal pole and of the yolk (deutoplasm) toward the vegetal pole.
Polarity is therefore accentuated. The movement of the egg contents
is reflected in the very violent activity of the microscopic pigment
granules on the surface. These granules appear motionless in the unfertilized or the dead egg. These facts have been established by cytological and experimental investigations, and observed in high magnification motion pictures. However, since the sperm penetration involves the loss of pigment from the surface of the egg, there appears
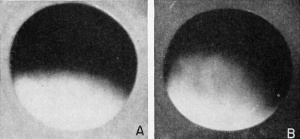
a compensatory marginal region between the animal and the vegetal
hemispheres which is neither black nor white, but is gray. This intermediate shading is due to the partial loss of surface pigment and the
consequent greater exposure of the underlying yolk, so that it is known
as the gray crescent. It establishes the gray crescent plane, and is
known as the plane of embryonic symmetry. The gray area has a
crescentic shape, due to the spherical surface involved. The surface
pigment is largely in a separate non-living coat, but one which is
necessary for the maintenance of cell integrity and is actively involved
in the subsequent cleavage process. But this sheet of pigment is pulled
slightly in the direction of the penetrating sperm which carries part of
the pigment into the egg. The opposite side of the sheet of pigment
is therefore pulled away from the underlying yolk, leaving it partially
exposed. The gray crescent is entirely a surface phenomenon and has
nothing whatever to do with syngamy (the fusion of gametes). Even
if the sperm aster formation is experimentally inhibited, the gray
crescent will form, it being a response to sperm penetration. The region
of the gray crescent will become the posterior side, and the opposite
(region of sperm entrance) will become the anterior side of the future
embryo. If we now take any two points on the egg axis, and the center
of the gray crescent as a third point, this plane represents the median
sagittal plane of the future embryo. It must be pointed out that the gray
crescent is not always readily apparent, and yet such eggs will develop
normally.
It is presumed that the closer to the equator that the sperm makes its entrance into the egg, the more extensive will be the opposite gray crescent. And the nearer it is to the animal pole the less apparent will the gray crescent be. This gray crescent appears in about one hour after insemination. It may develop earlier and be more extensive in aged eggs. While it will move somewhat, by the process of epiboly (e.g., downgrowth of surface materials), it will ultimately be found in the region of the blastopore and the anus. This is the region of origin of most of the mesoderm. It can be seen readily with the naked eye or under low magnification, but it is difficult to detect in the sectioned egg because the pigmented layer is relatively very thin. These descriptions of egg polarity and rotatory symmetry, and of the secondarily imposed bilateral symmetry, are of fundamental importance in understanding the development of the normal embryo. However, factors of unequal pressure on the egg in any clutch of eggs may so alter the position of the nucleus and the distribution of yolk and or cytoplasm that the bilateral symmetry of the fertilized egg and the early cleavage planes may not be so causally related. Even so, normal embryos will develop.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Reference
Rugh R. Book - The Frog Its Reproduction and Development. (1951) The Blakiston Company.
Cite this page: Hill, M.A. (2026, Haziran 9) Embryology Book - The Frog Its Reproduction and Development 4. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_The_Frog_Its_Reproduction_and_Development_4
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G