Book - The Frog Its Reproduction and Development 11
| Embryology - 22 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Rugh R. Book - The Frog Its Reproduction and Development. (1951) The Blakiston Company.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter 11 - The Germ Layer Derivatives - The Ectoderm and Its Derivatives
The Brain
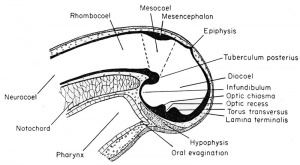
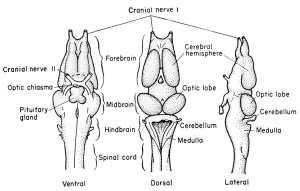
The primary embryonic brain of the frog has three main subdivisions. The most anterior of these, the prosencephalon, alone becomes further subdivided into two regions, the telencephalon and the diencephalon. In the higher vertebrates but not the frog, the rhombencephalon (hindbrain) is also subdivided. The adult frog brain then has four major divisions, arbitrarily determined, for they merge into one another structurally and functionally. Each of these divisions has a specific set of characteristics and derivatives, which will be described in sequence from the most anterior to and including the spinal cord.
The Prosencephalon
The Telencephalon (Secondary Forebrain). The most anterior division of the forebrain is the telencephalon with its original cavity, the telocoel. One may draw a line in a sagittal plane from a point just anterior to the epiphysis and extending through the brain cavity to the oosterior wall of the thickened torus transversus. This imaginary line separates the anterior telencephalon from the more posterior diencephalon of the forebrain. The telencephalon includes the fused anterior neural folds which comprise the lamina terminalis. This lamina will form a partition which will separate the paired cerebral hemispheres by a longitudinal groove.
The telencephalon is the embryonic cerebrum. Its cavity expands
laterally to give rise to the right (first) and left (second) lateral
ventricles and the surrounding thick-walled cerebral hemispheres, at
about the 12 mm. stage. These ventricles are laterally compressed.
In the frog the cerebral hemispheres are first differentiated at the 7
mm. stage but never become very large. The two telencephalic vesicles
are partially constricted off from each other but remain connected
by way of the tubular foramen of Monro, which opens into the
common (intermediate) third ventricle, known as the ventriculus
impar. The third ventricle overlaps and connects the telocoel and the
diocoel. The nervous tissue of the cerebral hemispheres is connected
transversely by the ventral anterior commissure, which is the original
torus transversus, and the anterior pallial commissure found posteromesially. The cerebral hemispheres become greatly thickened with
consequent enlargement and there is a parallel reduction in the size
of the contained ventricles. Antero-ventrally each grows out toward
the anteriorly placed olfactory placodes to establish connections
known as the olfactory lobes and nerves. The olfactory lobes arise as
a pair but subsequently become fused mesially. These are rather prominent brain structures in the adult frog. They seem to originate
together and become separated only at the point of origin of the
olfactory nerves. However, each originates from the telencephalon on
its side of the brain.
Dorsal to the anterior pallial commissure is the single median anterior choroid plexus which develops as a thin and highly vascular invagination from the roof just at the junction of the telencephalon and the diencephalon. It extends into the diocoel and into the two telocoels, and is therefore tri-radiate.
| Late development of the brain of the frog tadpole. |
|---|
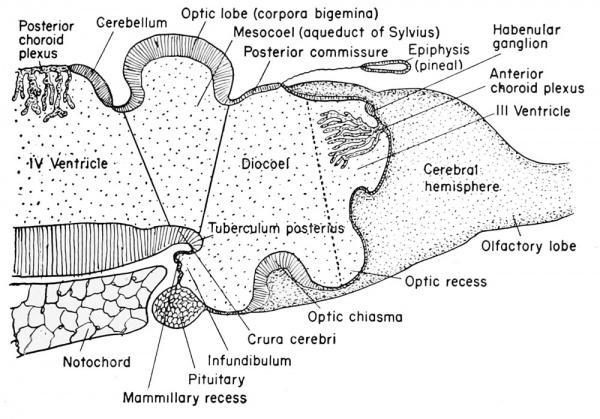
Pre-metamorphic stage. |
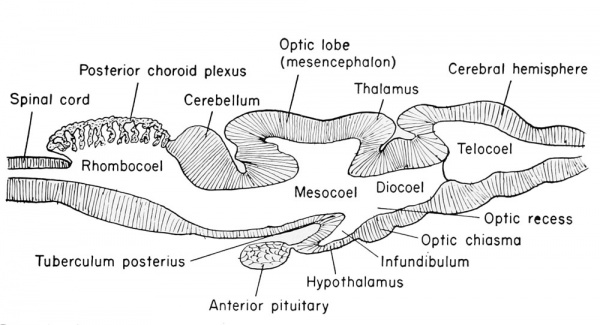
Adult brain, schematized, reduced in size. |
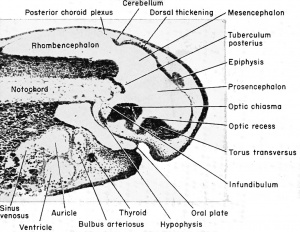
The Diencephalon
(Thalamencephalon or Between-brain).
The third ventricle is divided between the telencephalon and the diencephalon, but the enlarged portion found surrounded by the diencephalon is identified as the diocoel. This cavity extends antero-dorsally beneath the epiphysis, antero-ventrally into the optic recess, posteroventrally into the infundibulum, and laterally into the relatively large optic vesicles.
The structural derivatives of this diencephalon include the posterior commissure, just anterior to the dorsal limit of the mesencephalon. Anterior to this is the epiphyseal recess, and the dorso-mesial saccular outgrowth known as the epiphysis. This continues to grow forward and becomes separated from the brain as a small knob of cells which remain in the adult as the brow spot. It is presumably homologous to the pineal gland of higher vertebrates.
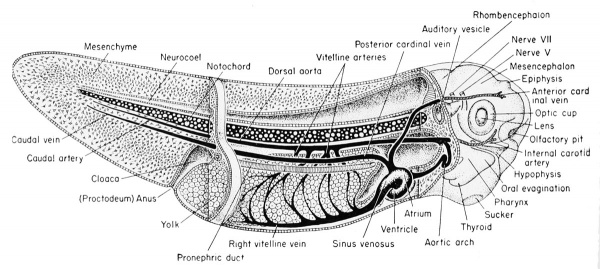
Anterior to the epiphysis, in the roof of the diencephalon and between it and the anterior choroid plexus, are the habenular ganglion and commissure. In front of this there later develops a dorsal outgrowth know as the paraphysis. In the floor just posterior to the optic recess is the very thick optic chiasma which will contain crossed fibers from the two optic stalks. Posterior to this chiasma is a thinwalled pocket or trough projecting beneath the anterior tip end of the notochord, known as the infundibulum. The cells of the infundibulum will combine with the approximated and pigmented cells of the ingrown hypophysis to form the pituitary gland of the adult. The infundibulum cells give rise to the posterior part of the pituitary gland and retain a hollow infundibular stalk connection with the brain. The hypophysis becomes the anterior part of the pituitary gland. During metamorphosis the individual lobes of the pituitary gland differ, both in gross morphology and in finer structure. The lateral and intermediate lobes show poor vascularization, a simple cellular structure, uniform staining reaction, and a rather static structural development. The anterior lobe becomes highly vascular, consists of several types of cells, and exhibits increasing complexity with further development. The acidophils of the anterior pituitary gland become highly differentiated and the basophils become poorly differentiated when the thyroid gland is either poorly developed or inactive. Conversely, thyroid activity is correlated with hyperactivity of the basophils of the anterior pituitary gland. This gland is formed from ectoderm, but some is epithelial and some is brain ectoderm. Between the infundibulum and the tuberculum posterius is a secondary and posteriorly directed pocket known as the mammillary recess.
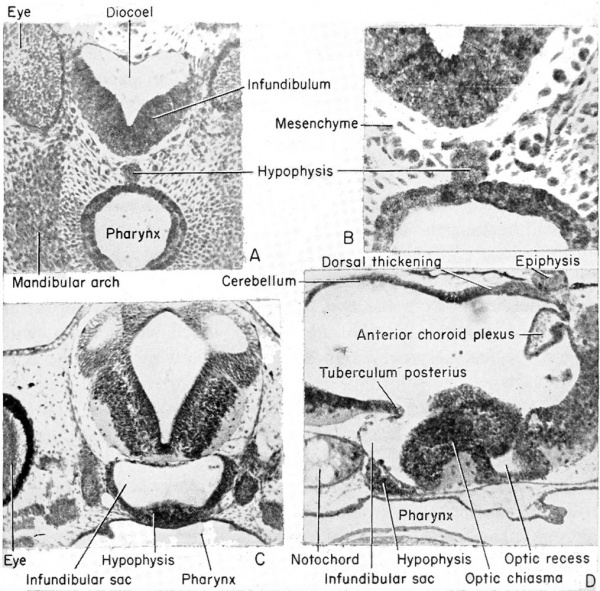
(A) Relationship of the hypophysis to the infundibulum as seen in a cross section through the 5 mm. frog tadpole. (B) Same as "A" but enlarged to show the pigmented hypophyseal cells as distinct from the gut endoderm. (C) The approximation of the hypophysis and infundibulum as seen in a cross section through the 11 mm. stage. (D) Sagittal section of the 11 mm. stage to show the relation of the hypophysis and infundibulum to other parts of the brain and pharynx.
The optic vesicles begin to develop very early as lateral outgrowths
from a slightly ventral level of the diocoel. The expansion of the
diocoel provides a temporary and slight thinning of the walls of the
optic vesicles. However, as these vesicles make contact with the lateral
head ectoderm, that portion of the vesicle in contact begins to thicken
and then invaginate to form a 2-layered optic cup. The most lateral
and invaginated portion of the cup will become the retina, the mesial
layer will become the pigmented layer of the eye, and the connecting
and somewhat constricted tube the optic stalk. The nervous elements
of this optic stalk will join in the optic chiasma which contains the optic
nerve fiber tracts from the two sides. The stalk will develop around
an inverted groove (the choroid fissure) which will contain, within
the groove, accessory nerves and blood vessels which feed the retina.
The Mesencephalon
(Midbrain)
This portion of the brain functions largely as a pathway of nerve tracts between the anterior prosencephalon and the posterior rhombencephalon. These tracts are found principally within the paired ventro-lateral thickenings of the walls and floor on either side of the tuberculum posterius. They are known as the crura cerebri.
The original dorsal thickening becomes subdivided by a median
fissure into paired dorso-lateral thickenings, at about the 1 mm. stage.
These are known as the optic lobes or corpora bigeniina. They do not
reach their full development until the time of metamorphosis. Anterior to these lobes is the posterior commissure. From the posterior
limits of the mesencephalon and optic lobes may be seen the valvulae
cerebelli and the fourth pair of cranial nerves (trochlear) which
emerge from the dorso-lateral wall. The original cavity of the midbrain (inesocoel) connects the rhonibocoel (fourth ventricle) with
the third ventricle which becomes narrow and is known as the aqueduct of Sylvius (also the iter e tertio ad quartum ventriculum).
The Rhombencephalon
(Hindbrain)
This portion of the brain is clearly marked off from the mesencephalon by a transverse constriction in the roof of the brain, at the posterior limit of the dorsal thickening. It is not clearly divided farther. There appears a slight transverse thickening in the roof of the rhombencephalon which corresponds to the metencephalon of higher forms and develops into the small cerebellum. Posterior to this the roof becomes broad, thin, and vascular, and folds into the rhombocoel (fourth ventricle) as the posterior choroid plexus. The ventral and ventro-lateral walls of the rhombencephalon are known as the medulla oblongata from which arise the cranial nerves V to X inclusive. The walls become thickened by fibers which form numerous pathways from the brain and cord.
The rhombocoel or cavity of the hindbrain is known as the fourth ventricle which communicates posteriorly with the central canal of the spinal cord and anteriorly with the aqueduct of Sylvius of the mesencephalon.
| Development of the brain and anterior structures of the frog tadpole. | |
|---|---|
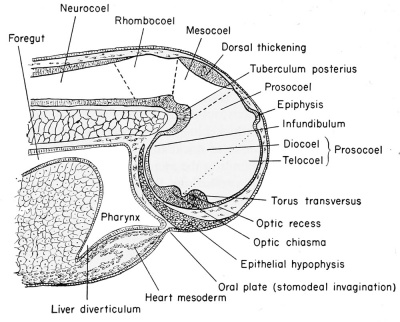
Median sagittal section of the 7 mm frog tadpole. |
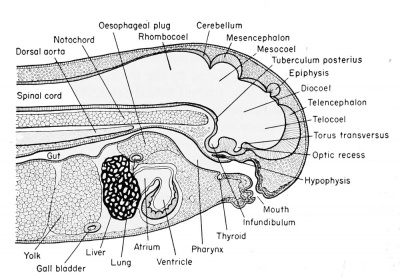
Median sagittal section of the 11 mm frog tadpole. |
The Spinal Cord
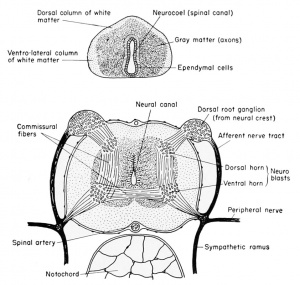
The neural or central canal (neurocoel) from the beginning of its development is laterally compressed by the thick lateral walls of the spinal cord derived from the original neural folds. The lining cells, coming from the dorsal epithelial ectoderm, retain both their pigment and their cilia, and are non-nervous in function. These cells, which continue to line the central canal of the adult, are known as the nonnervous ependymal cells. The thick lateral walls of the spinal cord are made up of the rapidly multiplying germinal neuroblasts (primitive or embryonic precursors of the neurons) and the supporting small and stellate cells known as the glia (neuroglia) cells. These cells have the function normally ascribed to connective tissue, namely support for the neuroblasts, but they are of ectodermal origin. The compact glia and neuroblasts close to the inner layer of ependyma comprise the gray matter of the cord. It is within this layer that the bulk of the cell bodies of neurons and the commissural fibers from one side of the cord to the other will be seen. As the cord develops further, the anterior and posteriorly directed fibers of the various neurons are concentrated more laterally so that only cross sections of axons will be seen. This peripheral area is then known as the white matter, to distinguish it from the more central and cellular gray matter of the cord. In addition to the lateral walls of the cord, which are thick, the dorsal wall is also thick because it consists of the fused neural folds.
The central canal or neurocoel is therefore displaced ventrally in the
embryo and larva, but, as the cord is developed and more neuroblasts
are formed, the neurocoel assumes a more central position. Within
the enveloping connective tissue membrane which surrounds the spinal
cord may be seen several blood vessels, principally the large spinal
artery located in the mid-ventral (inverted) groove of the cord.
The Peripheral Nervous System
The development of this variegated portion of the nervous system will be treated in the following sequence, and the description will be continuous to the final stage of development:
1. Organs of special sense: optic, auditory, olfactory, and lateral line organs.
2. Cranial nerves I to X.
3. Spinal nerves.
4. Other neural crest derivatives: dorsal root ganglia, sympathetic nervous system, chromatophores, parts of the visceral and cranial cartilage, and the medullary portion of the adrenal glands.
The Organs of Special Sense
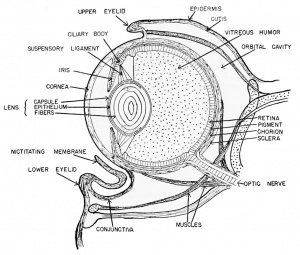
The Eye
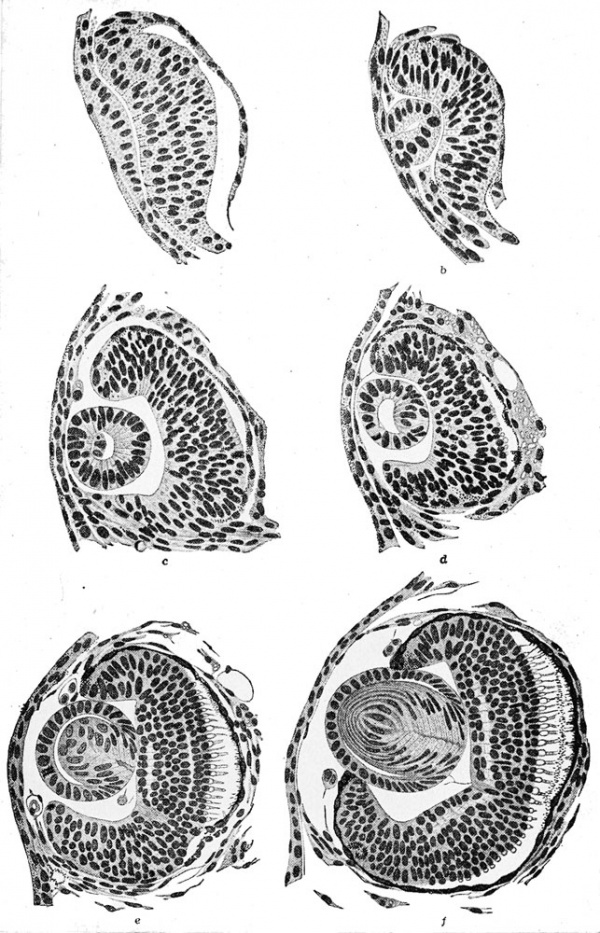
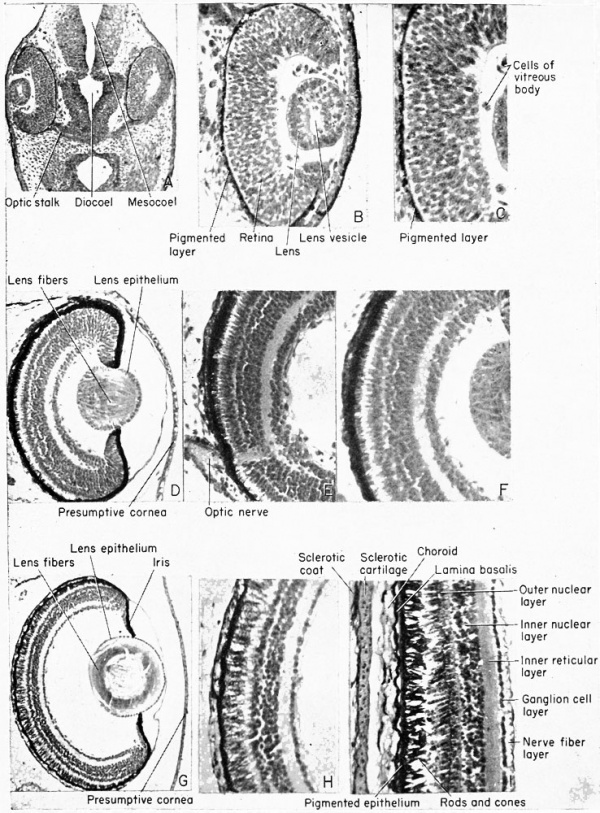
The optic vesicles arise early, by the tail-bud stage, as lateral diverticula from the ventro-lateral walls of the diocoel. The connection of the brain cavity with the optic vesicles becomes constricted, by the convergence of the surrounding mesenchyme, into a tube known as the optic stalk.
The dorso-lateral wall of each optic vesicle comes into contact with
the head ectoderm, as it expands laterally, and it is then flattened and
finally invaginated. This invagination begins ventro-laterally and is
continued obliquely mesio-dorsally. This process of invagination is
aided by a thickening of the vesicle wall between the dorsal and
ventral limits. This is the region which will later form the retina.
This thickening begins at the first point of contact (dorsal) and as a
result the connecting optic stalk is moved to a more ventral position.
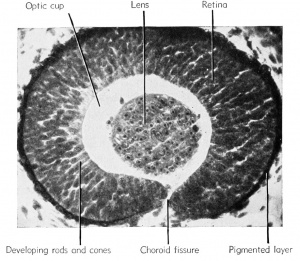
The newly inverted cavity thus formed by the invagination of the lateral wall of the optic vesicle is known as the optic cup. The rim of the cup now exposes the pupil. The cup will invaginate further and grow in thickness until it all but obliterates the original optic vesicle (opticoel). The lateral (retinal) and the inner or mesial (pigmented) layers of the optic cup are therefore brought into close proximity with only a slit-like space remaining between, the remains of the original opticoel. Ventrally this double-layered optic cup is connected with the optic stalk.
The three-dimensional aspect of these changes must be understood.
Transverse, sagittal, or horizontal (frontal) sections through the eye
will appear to be essentially alike. The cup is circular with only a
ventral cut-out where the inverted optic stalk is attached. If one could
look directly into such a developing eye, after removal of the lens, the
impression would be of a horseshoe-shaped cup, the pupil of the eye,
with a groove-like opening ventral. This central cavity is the optic cup
but ventral to it is the double-layered groove of the optic stalk, known
as the choroid fissure.
The lens of the eye is formed from the superficial ectoderm by invagination of the deeper or nervous layer of ectoderm opposite the opening of the optic cup. This is brought about under the inductive
influences emanating from the dorsal rim of the optic cup by the 4
mm. stage, some time before hatching. The lens originates as a placode
or thickening in the inner or neural layer of the head ectoderm. This
placode invaginates to form a (lens) vesicle by the 5 mm. stage. This
vesicle pinches off from the head ectoderm and comes to lie within the
ring of the optic cup and is supported by a suspensory ligament by
the 6 mm. stage. The outer wall of the lens vesicle remains as cuboidal
epithelial cells and the inner wall cells become elongated as lens fibers.
The cavity is ultimately obliterated. The head ectoderm then closes
over the lens to form a new covering which, in conjunction with the
head mesenchyme, forms the double-layered cornea. This cornea is
therefore derived from ectoderm and mesoderm and becomes a transparent covering of the lens by the 6 mm. stage.
After hatching, at about the 6 mm. stage, the outermost wall of the optic cup is seen as a pigmented layer which comes into contact with the thick inner retinal wall which thereupon begins to give rise to the rods and cones from its outer margin. These visually sensitive elements, the rods and cones, are fully developed by the 1 1 mm. stage. They point away from the light source, their posteriorly directed axons covering the exposed face of the retina. The inner margin of the retina, i.e., that toward the optic cup, is made up of neuroblasts and their jSbers (axons) which pass over the surface of the retina to leave the optic cup together by way of the choroid fissure. They then pass along the walls of the optic stalk, which acts as a guiding path to the fibers, to reach the diencephalon of the brain as the second cranial or the optic nerves. There is a junction and crossing of the paired optic nerve fibers in the optic chiasma which is seen in the floor of the diencephalon. The choroid fissure will eventually close around the blood vessels and nerves which supply the optic cup. These latter nerves have no visual function. The ventral margins of the optic cup, where the choroid fissure originates, eventually fuse to form the choroid knot and it is from this cluster of cells that the cells of the iris arise. Pigment very soon disappears from the outer superficial layer of the original optic cup.
The large cavity of the eye between the lens and the retina, designated as the optic cup, becomes filled with a viscous fluid known as the vitreous humor. This is derived from the cells of the retinal wall and lens and is therefore of ectodermal origin. Head mesenchyme gives rise to the connective tissue of the choroid coat that surrounds the pigmented layer of the eye. Outside of the choroid coat is the very tough sclerotic coat, also mesenchymal. The nervous (sensory) parts of the eye are therefore ectodermal in origin, but the blood vessels, connective tissues, cartilage, and parts of the cornea are all mesenchymal (mesoderm).
The Ear
The frog has no outer ear. The inner and the middle ear are developed much in the manner of all vertebrate ears, but to a less efficient and complicated degree.
The Inner Ear
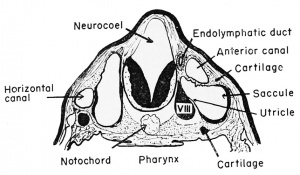
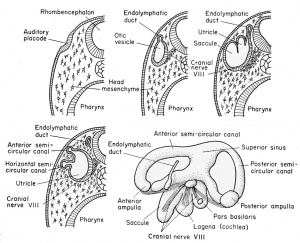
The superficially placed auditory placode develops from nervous ectoderm on the side of the head at the level of the rhombencephalon, prior to the time of hatching (at about the 2.5 mm. stage). This occurs under the inductive influences of the medulla and archenteric roof. It moves inwardly toward the brain and invaginates to form a vesicle with but a temporary external opening at the 3 mm. stage. This vesicle is the otocyst or auditory sac. After the head ectoderm closes over this invaginating vesicle at the 4 mm. stage, and the walls become continuous, there develops from its dorsomesial wall a small evagination. This soon (at the 7 mm. stage) becomes tubular and is known as the endolymphatic duct. The duct on each side grows dorsally to fuse with the duct of the other side of the brain. It finally loses its cavity and forms a vascular membrane which covers the rhombencephalon, having no auditory function whatever. The duct remains as a vestige even in the adult frog, originating between the membranous labyrinth (inner ear) and the hindbrain.
At about the 11 to 12 mm. stage there develops a vertical fold which divides the main cavity of the otocyst into mesial and lateral chambers. The more dorsal and mesial portion is the utricle and the more ventral and lateral portion is the saccule, both being joined by a small pore.
The utricle, by the 15 mm. stage, becomes further subdivided by three folds or ridges. These develop on the inside of the utricular wall. One is vertical and anterior (anterior semi-circular canal), one is horizontal and lateral (horizontal semi-circular canal), and finally there will appear a third fold which is vertical and posterior (posterior semi-circular canal). These ridges fuse with one another to form three loop-like tubes, each of which opens at both ends into the utricular cavity with which it retains connection throughout the life of the frog. These are known collectively as the semi-circular canals which later become free from the utricular wall and continue to grow and later function as organs of equilibration in the adult. The junction of each canal with the utricle causes the local enlargement of the utricle to form an anipuHa.
The saccule and the endolymphatic duct remain continuous. The
endolymphatic duct, however, has no auditory function but joins its
bilateral homologue to form the fused endolymphatic sacs which remain only as the vascular covering of the hindbrain. The saccule,
however, gives rise (at the 15 mm. stage) to two evaginations or sacs.
One of these is posterior and ventral, known as the cochlea or lagena,
and the other is slightly more dorsal (but posteriorly directed) and
is known as the pars basilaris (basilar chamber) of the ear. These
auditory structures never develop to the extent that they do in higher
vertebrates. However, in all vertebrates the lining epithelial walls of
the utricle (1), saccule (1), cochlea (3), and the various ampullae
(3) develop sensory patches (as numbered) which are connected
functionally with the eighth cranial or the auditory nerve.
The sensory (auditory and equilibratory) parts of the ear are
therefore purely ectodermal in origin. However, the entire ear is encased in mesenchyme which gives rise to cartilage that is finally
displaced by bone. This makes up the auditory capsule which is
mesodermal. Between the capsule and the inner ear (membranous
labyrinth) is the perilymph space which is filled with the perilymph
fluid, derived from mesenchyme.
The Middle Ear
This portion of the ear grows as an endodermally lined tube extending dorso-laterally from the original hyomandibular pouch and developing a terminal chamber. The chamber comes to lie between the inner ear and the head ectoderm. That portion of the endoderm in contact with the head ectoderm (original hyomandibular plate) becomes the tympanic membrane (ear drum), later to be invaded by mesenchyme which forms blood vessels and connective tissue. This membrane is supported by a cartilaginous ring known as the annulus tympanicus (tympanic ring) which is believed to arise from the palatoquadrate bone. These latter two structures are presumed to cause the development of the tympanic membrane by induction.
This endodermally lined chamber expands inwardly to make contact with the inner ear capsule at the site of the foramen ovale, an aperture in the inner ear capsule and extending through the perilymph space. This aperture is obstructed by a cartilaginous plug known as the operculum. This operculum later becomes ossified into bone. A cartilaginous rod arises from the roof (dorsal wall) of this cavity and extends from the operculum of the inner ear to the tympanic membrane and across the middle ear cavity, and is known as the columella or plectrum. This columella is mesodermal, arising from the inner stapedial plate (part of the otic capsule) and a cartilage from the palatoquadrate bar. The columella becomes partially ossified and conveys to the inner ear the auditory vibrations which impinge upon the outer tympanic membrane. It is derived presumably from part of the second or the hyoid arch.
The original connection of the pharynx and the middle ear cavity, the dorso-lateral tubular growth from the hyomandibular pouch, remains in the adult as the tubo-tympanic cavity or the Eustachian tube and is lined with endoderm.
The Olfactory Organs
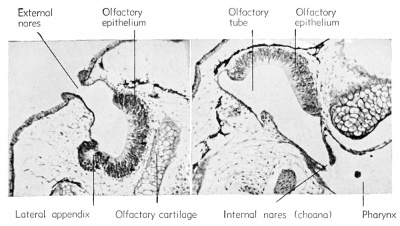
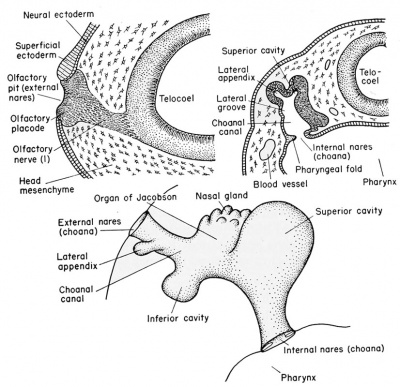
The external olfactory placodes arise as thickenings in the neural ectoderm of the sense plate, dorso-lateral to the stomodeum, at about the 2.5 mm. body length stage, long before the time of hatching. The overlying superficial epithelial ectoderm disappears so that the nervous layer of the placode becomes* the exposed lining of the olfactory pit.
After hatching (6 mm. stage) a solid rod of ectodermal cells grows ventro-laterally from the olfactory pit to become attached to the pharynx just dorsal to the oral plate (i.e., stomodeum). By the 11 mm stage this core of cells acquires a lumen which is continuous from the external nares (olfactory pits) to the internal nares (internal choanae) which open into the pharynx. The major part of this olfactory tube is lined with non-sensory epithelium. It develops a dorsal and a ventral chamber (sacs), each of which acquires glandular masses which are known as the organs of Jacobson.
The neuroblasts of the olfactory placodes send extensions posteriorly and give rise to the fibers which form the olfactory or first cranial nerve. These grow toward the brain and are guided in their directional development by the outgrowth of the telencephalon known as the olfactory lobes.
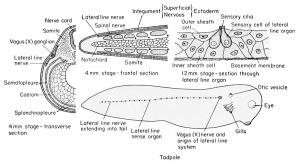
The Lateral Line Organs
The most posterior or fourth cranial placode (cranial nerve X) sends a growth posteriorly beneath the lateral body epidermis, on either side, beginning at about the 4 mm. stage. It grows posteriorly to the tip of the tail. Along these cords arise groups of sensory cells which grow through the epidermis to become exposed along the sides of the body as the lateral line system. The exposed cells are ciliated, and are therefore sensitive to vibrations in the surrounding aquatic medium. They are protected by inner and outer sheath cells and a basement membrane and are connected functionally with branches of the lateral line nerve. At the head level the extensions of this system seem to be innervated by cranial nerves VII, IX, and X, principally the latter. This structure is no doubt a vestige of the aquatic ancestry of the Anura, for, as in lower forms, these organs are innervated by a branch of the vagus (X) ganglion, known as the lateral line nerve or ramus lateralis. In the Anura this system disappears by the time of metamorphosis.
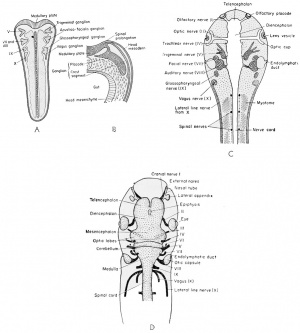
The Cranial Nerves
These nerves, as in the case of practically all nerves of the embryo, are developed in pairs. Many of them are both sensory (afferent) and motor (efferent). Each nerve will be dealt with as a separate entity.
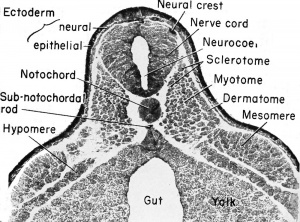
As the neural folds close they leave a column of cells between the
fold and the dorsal ectoderm. These are known as the cranial or
neural crests, depending upon the level under consideration. The cranial crests of the brain level are different from the neural crests of the
spinal cord level in that they become divided into four large pairs
known as the cranial placodes. These consist of thickened patches of
neural ectoderm of the head, one of which, the auditory placode, has
been described already. The original cellular connection between each
cranial crest segment and the brain remains as the sheath of the nerve
fibers to be developed in that region. At the spinal cord level the segments are metameric and will be as abundant as the spinal nerves developed therefrom.
Four ganglionic masses are found in the development of the frog
cranial nerves as early as the 6 mm. stage. These include the first
or semilunar ganglion of the fifth nerve. The second is the acusticofacialis (VII-VIII) ganglion which becomes associated with the
auditory placode. The third is the glossopharyngeal (IX) and the
fourth is the vagus (X). The third and fourth arise together as the postotic ganglionic masses and each becomes associated with an epibranchial placode.
Embryologically the rudiments of all of the cranial nerves are more
complex than are the rudiments of the spinal nerves. They are made
up of cell masses from the neural crests, from ectodermal patches on
the surface of the head, and from cell processes extending out from
the ventro-lateral brain neuroblasts. The spinal nerves do not have
the surface ectodermal constituent. Following is a summary of the
cranial nerves of the frog embryo:
I. Olfactory: This is sensory, arising after hatching (11 mm.) from neuroblasts of the olfactory lobe (telencephalon) and growing to the olfactory placode.
II. Optic: This is sensory, arising at an early stage (6 mm.) from
the neuroblasts of the retinal layer of the optic cup to grow
along the ventral wall of the optic stalk to join the lateral
wall of the diencephalon.
III. Oculomotor: This is motor, arising Just before hatching from
the ventro-lateral wall of the mesencephalon (crura cerebri)
to innervate four muscles of the eye. These are the rectus
superior and the ciliary ganglion, the rectus inferior, the
rectus medialis, and the obliquus inferior.
IV. Trochlear: This is motor and remains very small. It arises late
in development from near the roof of the mesencephalon between the optic lobes and the cerebellum just posterior to the
valvula cerebelli and the posterior commissure. It innervates
the superior oblique muscles of the eye.
V. Trigeminal: This is a mixed nerve, also known as the trifacial
ganglion, which appears as early as the 5 mm. stage and has
a complicated origin from the dorsal (superficial) ganglionic
elements of the first cranial crest segment and the ganglionic
Segments of neural crest
Nerves derived from them
I
V
ramus ophthalmicus
II
V
main
branch
III
VII and VIII
IV
IX
V
X
VI
dorsal root of
1st spinal
Mesodermal
Somites
Ventral roots
Nerves
derived
from them
Head Somites
Trunk Somites
Premandibular
I
III
Mandibular
II
IV
Hyoid
III VI
1st
IV
disappears
2nd
V
disappears
3rd
VI
ventral
root of
1st spinal
4th
(Courtesy, Jenkinson: "Vertebrate Embryology," Oxford, The Clarendon Press.)
portion of the first cranial placode. It is well developed by the 9 mm. stage. The level is at the anterior end of the medulla. This is a very large and tri-radiate ganglion, including an Ophthalmic branch from the placode whose fibers pass to the skin and snout, and a forked Gasserian ganglion which arises from both the crest and placode elements and gives rise to the maxillary (upper) and mandibular (lower) branches of the fifth nerve. The entire mixed trigeminal nerve ganglion (V) joins the dorso-lateral wall of the medulla oblongata. The outer or non-nervous portion of the placode disappears and it is believed that the non-nervous portion of the crest segment may contribute some of the mesenchyme to the mandibular arch. It is certainly difficult to distinguish between them.
VI. Abducens: This is a motor nerve arising late (11 mm. stage)
from the neuroblasts of the ventral side of the medulla and
innervating the lateral or external rectus and retractor bulbi
muscles of the eye.
VII. Facial: This is a mixed nerve, arising by the 5 mm. stage from
the anterior ganglionic portion of the second or acusticofacialis (second cranial) crest segment and placode and is associated with neuroblasts coming from the medulla just posterior to cranial nerve V. The motor (efferent) fibers are divided
between the hyoid and the palatine (mouth) branches, but a
good portion of the crest segment, from which these fibers
arise, is non-nervous and is presumed to give rise to mesenchyme of the hyoid arch.
VIII. Auditory: This is a sensory nerve arising by the 5 mm. stage
from the posterior ganglionic portion of the second (acusticofacialis) crest ganglion, in close association with the auditory
placode, and innervates only the inner ear.
IX. Glossopharyngeal: This is a mixed ganglion, arising by the 9 mm. stage from the ganglionic portion of the third cranial crest segment and placode. Fibers of the ninth and tenth cranial ganglia enter the sides of the medulla together, but peripherally the ninth or glossopharyngeal nerve supplies the first branchial arch, the mouth, the tongue, and the pharynx. This ganglion is incompletely separated from the vagus by the anterior cardinal vein.
X. Vagus or pneumogastric gangHon: This is mixed and arises by the 9 mm. stage from the fourth cranial crest segment and
placode and becomes associated with the neuroblasts from
the medulla. Peripherally the branches feed the second
and third branchial arches, the tympanum, the muscles of
the shoulders, the viscera (including larynx, oesophagus,
stomach, lungs, and heart), and the lateral line system. The
non-nervous portion of the crest segment forms mesenchyme
and the superficial non-nervous elements of the placode
then disappear.
Summary of Derivatives of the Cranial Nerves in Tabular Form
Cranial Crest Segment Cranial Nerves Derived Therefrom
( V ophthalmic branch V mandibular branch II VII and VIII
III IX
IV X
The Spinal Nerves
The spinal nerves, aside from their component parts, differ from the cranial nerves in that they are related to mesodermal somites rather than visceral clefts.
The spinal nerves arise from the pair of continuous neural crests.
These are elongated strands of neural ectoderm left outside of the
dorsal neural tube, between it and the dorsal ectoderm, at the time of
closure of the neural tube. These crests become metamerically segmented and are paired as a result of the development of the somites. In
frontal sections of the 7 mm. stage these crests will be seen as dorsal
root ganglia of the spinal nerves. No placodes are developed in association with any part of the neural crests, as were described in association with some of the homologous cranial crests.
Each crest segment is made up of many neuroblasts which send fibers to the dorso-lateral wall of the spinal cord to form the dorsal root or ramus of the spinal nerve. This connects the dorsal root ganglion (original neural crest) with the spinal cord. Also, other fibers grow ventro-laterally from this ganglionic crest to become the afferent tracts from the skin and other sensory organs.
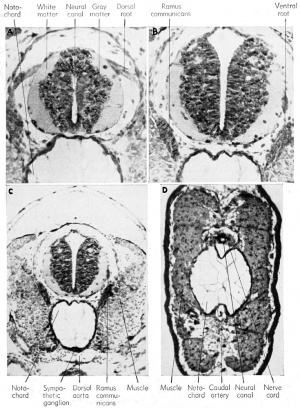
Within the spinal cord, as early as the 4 mm. stage, neuroblasts develop and send out bundles of efferent axons (fibers) which emerge
from the ventro-lateral wall of the spinal cord and almost immediately co-mingle with the afferent fibers, already described. The ventral
nerve roots or rami are entirely efferent, the dorsal nerve roots are entirely afferent, but the nerve trunks contain, within a common
sheath, both the afferent and efferent fiber trunks. Many of these spinal
nerve trunks fork to send both types of fibers dorsally to the sense
organs and ventro-laterally to the muscles and to the limbs.
The tadpole may have a total of 40 or more pairs of spinal nerves, but only the anterior 10 pairs remain after the degeneration of the tail at metamorphosis. The first pair (hypoglossal) emerges from between the first and the second vertebrae, to innervate the tongue and some of the muscles of the hyoid arch. The second pair (brachial) emerges from between the second and the third vertebrae and are very large. They have connections with the first and third spinal nerves and thus form the large brachial plexus, innervating the forelimbs and the muscles of the back. The third pair has the aforementioned connection with the brachial but it then supplies the external oblique and transverse muscles and the skin. The fourth, fifth, and sixth spinal nerves supply the skin and muscles of the abdominal wall. The seventh, eighth, and ninth spinal nerves together form the lumbosacral or sciatic plexus which innervates the posterior abdominal region and the hind legs. The tenth, with a branch from the ninth, forms the ischio-coccygeal plexus which sends branches to the urinary bladder, cloaca, genital ducts, and posterior lymph hearts.
The Neural Crest Derivatives
The Sympathetic Nervous System
The development of the sympathetic nervous system is not understood clearly but it is believed also to arise from the neural crests, by a downgrowth along the afferent trunk to the spinal nerve and thence to the dorsal aorta and the viscera beginning at about the 6 mm. stage. The sheath cells probably come from the neural tube itself. Just before hatching there may be seen clusters of cells around the junction of the dorsal and ventral nerve roots, and these clusters will give rise to the sympathetic ganglia which become associated with the dorsal aorta and the viscera. These ganglia are also interconnected by paired longitudinal strands on either side of the spinal cord. The fibers which join the sympathetic ganglia with the spinal nerve are known collectively as the ramus commuiiicans.
The sympathetic system arises with the vagus (cranial nerve X), and its connecting fibers extend through the jugular foramen of the skull and along each side of the spinal cord to receive these connecting rami from the segmental spinal nerves. Both independent sympathetic nerves and plexi are developed and are found often quite removed from their source of origin.
The Adrenal Medulla
The large brown cells of the inner portion of this endocrine organ arise some time before metamorphosis by migration from the sympathetic ganglia and hence are of ectodermal origin. They have lighter staining nuclei and are scattered among the more abundant cortical cells. These latter cells arise from the mesoderm and will be discussed subsequently.
The Stomodeum and Proctodeum
The stomodeum and proctodeum are also of ectodermal origin, each being represented by an invagination of ectoderm at either end of the digestive tract. The stomodeal ectoderm meets the oral endodermal evagination to form the oral plate which at the 6 mm stage ruptures through to form the mouth. Therefore, a portion of the mouth (to the tongue) is lined with ectoderm. The proctodeum is a similar caudal invagination of ectoderm which meets the evagination of the anal endoderm resulting in the formation of a temporary anal plate. This shortly ruptures. The anus is thus partially lined with ectoderm.
Summary of Embryonic Development of the 11 mm Frog Tadpole
Ectodermal Derivatives
Epidermis — thickened, ciliated on tail only.
Central Nervous System — differentiated into four brain vesicles and spinal cord.
Prosencephalon (Forebrain)
Telencephalon — anterior to optic recess; thin-roofed.
Cerebral hemispheres develop around each of lateral ventricles.
Anterior choroid plexus as median dorsal invagination into telocoel which becomes vascularized.
Olfactory (I) nerve from floor of cerebral hemisphere to mesial side of nasal tube.
Diencephalon — from epiphysis to dorsal thickening to tuberculum posterius.
Epiphysis as mid-dorsal evagination just posterior to choroid plexus.
Optic recess — just posterior to torus transversus or region of lamina terminalis (anterior fusion of neural folds) as median ventral evagination.
Optic chiasma — thickened floor where optic nerve enters (posterior to recess).
Optic (II) nerve — from chiasma to eyes, may be seen entering retina.
Infundibulum — bulbous evagination in floor of diencephalon between optic chiasma and tuberculum posterius, ventral to notochord.
Mesencephalon — bounded by dorsal thickening and tuberculum posterius.
Optic lobes from dorsal thickening, bi-lobed, become corpora bigemina.
Cavity — mesocoel, iter, or aqueduct of Sylvius.
Oculomotor (III) nerve — mesio-lateral, from floor.
Trochlear (IV) nerve — dorso-lateral, just posterior to optic lobes and difficult to find, being small.
Rhombencephalon — from dorsal thickening to spinal cord, metencephalon and myelencephalon not distinguishable in the frog.
Cerebellum — from thickened roof, anterior.
Posterior choroid plexus — from thin, vascular posterior roof.
Otic vesicles — found at level of cerebellum.
Cranial nerves V to X are associated with this portion of the brain.
Trigeminal (V) nerve — anterior to otocyst, large, sends branches to mandibular and maxillary processes of first visceral arch.
Abducens (VI) nerve — from floor of rhombencephalon, anterior and ventral to origin of trigeminal. Supplies eye muscles as do oculomotor and trochlear.
Facial (VII) and auditory (VIII) nerve — arises as a single ganglion, mesioposterior to otocyst, supplying facial muscles and saccule and utricle of ear.
Glossopharyngeal (IX) nerve — arises posterior to otocyst, sending branches to first branchial arch. Ganglion.
Vagus (X) nerve — arises with IX, sending branches to second, third, and fourth branchial arches; to lateral line organs and to viscera.
Cranial Crest Segments — visceral cartilages, parts of the cranial cartilages from head mesenchyme originally derived from ectoderm.
Spinal Cord — from rhombencephalon into tail, with continuous cavity.
Neural (central) canal — original neurocoel, constricted centrally.
Ependymal layer — elongate, ciliated cells lining the canal.
Mantle layer — gray matter consisting of compact cell bodies, lateral.
Marginal layer — white matter, consisting of outermost axons.
Dorsal root — afferent fibers passing dorso-mesially to join dorsal root ganglion.
Dorsal root ganglion — paired thick bundle of neurons dorso-lateral to spinal cord, derived from neural crests.
Ventral root — efferent fibers passing ventro-laterally from spinal cord to spinal nerve trunk.
Spinal nerve — fusion of afferent and efferent fibers associated with distal organs (muscles, etc.).
Dorsal ramus — branch of spinal nerve to dorsal muscles.
Ventral ramus — branch of spinal nerve to ventral muscles.
Communicating ramus — fibers from spinal nerve to sympathetic ganglion.
Sympathetic ganglion — nerve cells lateral to dorsal aorta, derived from neural crest.
Special Sense Organs — found in the head region.
Eye — well developed in 11 mm. stage, some parts mesodermal.
Retinal layer — sensory portion consisting of rods and cones (inner and
outer granular layers). Pigmented layer — thinner layer outside of retinal layer. Lens — arising as vesicle, now a solid ball lying in opening of optic cup.
Sagittal section through the anterior end of the 8 mm. frog larva.
| Part or all of these structures are derived from mesoderm. |
|---|
|
Ear — arising from auditory placode, then otocyst.
Utricle — mesial and dorsal, giving rise to the three semi-circular canals, two of which have developed, i.e., anterior dorso-ventral and outer horizontal canals.
Saccule — outer and ventral portion of original otocyst.
Endolymphatic duct — between utricle and brain, joining saccule.
Cochlea — mesial portion of saccule, pigmented and ciliated.
Nose — arising from olfactory placodes.
External nares — glandular organ of Jacobson, opened to exterior.
Internal nares — extension of tubular opening from external nares into pharynx, called choana.
Stomodeum and Proctodeum — ectodermally lined openings into mouth and cloaca.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Reference
Rugh R. Book - The Frog Its Reproduction and Development. (1951) The Blakiston Company.
Cite this page: Hill, M.A. (2026, Mayıs 22) Embryology Book - The Frog Its Reproduction and Development 11. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_The_Frog_Its_Reproduction_and_Development_11
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G