Book - Physiology of the Fetus 4
| Embryology - 7 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Windle WF. Physiology of the Fetus. (1940) Saunders, Philadelphia.
1940 Physiology of the Fetus: 1 Introduction | 2 Heart | 3 Circulation | 4 Blood | 5 Respiration | 6 Respiratory Movements | 7 Digestive | 8 Renal - Skin | 9 Muscles | 10 Neural Genesis | 11 Neural Activity | 12 Motor Reactions and Reflexes | 13 Senses | 14 Endocrine | 15 Nutrition and Metabolism | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter IV The Blood of the Fetus
General opinions regarding development of the blood are reviewed in textbooks of embryology and histology 1, 2 and a more extensive consideration of this subject will be found in Downey’s Handbook of Hematology. 3 The genesis of blood cells begins in all mammals shortly after the formation of germ layers. It starts in the wall of the yollc sac where mesenchymal cells retract their processes to assume more compact rounded forms freed from attachment to other cells. All types of blood cells arise from these hemoblasts. A similar transformation is encountered later in the body mesenchyme, liver, spleen and bone marrow gis these Structures develop. The first red blood corpuscles, derived from the primitive stem-cells of the yollc sac, play a transient functional role and are ultimately replaced by blood elements derived from other regions. Not until late embryonic stages do we normally find a division of blood forming tissues into myeloid for elaboration of red corpuscles and granulocytes, and lymphatic tissues for production of lymphocytes. Abnormally even after the adult stage has been reached lymphatic tissues and the loose connective tissue may lose their speciftcity and give rise to types of blood cells other than usually formed by them.
The Red Blood Corpuscles
During the early part of prenatal life oxygen transport is effected by nucleated erythrocytes. As time goes on these are re— placed by red blood corpuscles which have lost. their nuclei, but some immature elements, the reticulocytes, are still observed in blood smears at birth. The total number of hemoglobin containing cells and corpuscles is small at first, for the blood is highly fluid, but increases as the end of gestation approaches. The diameter of the fetal red corpuscles is greater than in the adult, diminishing as development proceeds. 4-6
These few facts have been known for many years and it is surprising that no one accepted their challenge to investigate this interesting subject systematically until recently. All but the most recent contributions have been reviewed by Wintrobe and shumackerlss 20 who described their own careful comparative study in the pig, rat, rabbit, cat, dog and man.
At a period as early as it was possible to obtain adequate blood samples from fetuses they found that the number of red blood corpuscles, the amount of hemoglobin and the volume of paclced red corpuscles (hematocrit) are low in comparison with adult blood of the same species. The corpuscles are large, chiefly nucleated, and contain a correspondingly large amount of hemoglobin.
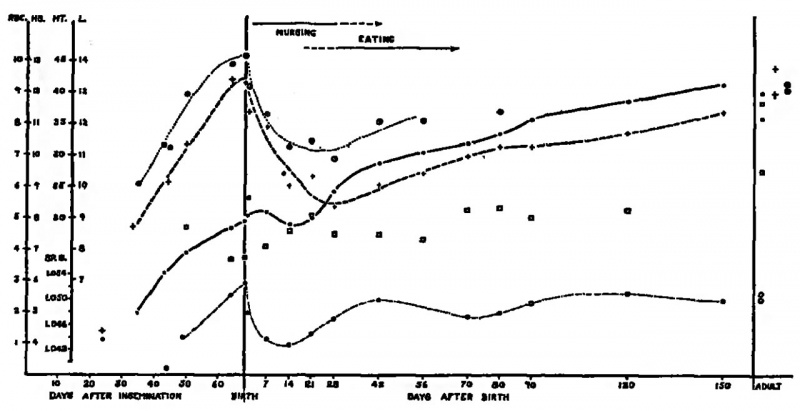
Fig. 19. Number of red blood corpuscles (●) in millions, leukocytes (□) in thousands, volume (hematocrit) of packed red corpuscles (⚉) in per cent, amount of hemoglobin (+) in grams per 100 cc., and specific gravity (0) of the blood of cats during prenatal and postnatal life until maturity Adult values for males are higher than for females in all except specific gravity. Each point represents an average.
As development proceeds, the number of corpuscles, amount of hemoglobin and volume of paclced corpuscles increase, not in a linear order, but more rapidly in early stages than later on near term. The mean corpuscular volume, mean corpuscular hemoglobin and proportion ok immature forms of red blood corpuscles decrease similarly. But the meanrorpuscular hemoglobin concentration remains about the same throughout the period studied. The amount of hemoglobin available for transporting oxygen is a function ok the size and number of red blood corpuscles and its concentration does not vary within individual corpuscles. It was observed that the period during which the greatest changes took place seemed to correspond to the time during which hemopoiesis is most active in the fetal liver. The development of blood corpuscles and of hemoglobin in the cat, not only during prenatal life but onward to maturity, is illustrated in Fig. 19.21
In the species whose gestation periods are short, such as the rat,18 the blood at birth is less lilce that of the adult than in species with long gestation periods, such as man. Even in the human fetus at birth, the number of red blood corpuscles in umbilical cord blood does not exceed that of the adult.19- 20 This view may seem to bö opposed to that which is generally taught, that the newborn has a higher red corpuscle count than the adult.22 Actually it is not, for the infant has more red corpuscles per cubic millimeter of blood in circulation twenty minutes to half an hour after birth than it had in utero.23 Some investigators have reported more than seven million per cmm.24- 25 and six million is not an excessive average iigure for the first day of life. If there is any delay between birth and talcing blood samples, a true picture of the condition in utero at the end of fetal life will not be obtained.
A sharp increase in red blood corpuscles is encountered in the cat on the day of birth.2I This persists for a weelc, but the number declines to nearly the birth level by two weelcs, rising again only after the lcittens begin to feed themselves at three weelcs (Fig. 19) . Wintrobe and shumaclcer suggested that high human red corpuscle counts after birth may be explained on the basis of dehydration, but ·we believe that the role played by dehydration on the first day of life of the cat is negligible. The specific gravity of the kitten’s blood does not increase at birth, but on the contrary, de— creases.21 The transient rise in red corpuscle counts may be partly explained on the basis of splenic contractions, which have been demonstrated to start at birth in the sheep.26
The human fetus has about as many red blood corpuscles at the end of gestation as the adult· but the corpuscles are of greater sizes consequently the mean hemoglobin content and the total amount of hemoglobin in the blood are higher than in the adult.. The amount of hemoglobin in the blood of the newborn has been reported by most investigators to be high; 20 gm. per 100 cc. of blood or even more is not uncommon.27 The most recent estimations made in blood drawn from the umbilical cord gave an average of 15.36 gm. per 1ooJcc.28 This value correlates well with the oxygen capacity of human cord blood (see Chapter V).
- Statement based partly on a study now in progress.
Hemoglobin decreases rapidly after birth. The high values for hemoglobin and the great number of corpuscles in the early postnatal period are undoubtedly related but there are other, poorly understood factors involved in this difference In the lcitten the curves for hemoglobin and corpuscles are not parallel until the fourth week of life. Perhaps this may be related to a substitution of new adult hemoglobin for the older fetal type.
An important conception of the development of blood has been proposed by Wintrobe and shumaclcen They observed that the developing fetal blood resembles that of patients with per— nicious anemia who are.being subjected to an eikective, continuous and extremely potent stimulus to blood formation. They suggest that the anti-pernicious anemia factor of Castle may be the same or very similar to the substance which causes the blood of the fetus to develop. It is possible that the fetus obtains this from the mother. If too little is available to supply the needs of both mother and fetus a deiiciency should manifest itself. Apparently it does so in "pernicious anemia of pregnancy" which can be controlled by administering liver extract and which even when untreated is relieved spontaneously by birth.29- Z? If an inadequate amount of the anti-pernicious anemia factor is available to the fetus one might expect to observe efkects upon the infant at birth. It has been shown that the incidence of primary anemia of the newborn is more common in multiple births, especially in prema— ture multiple births, than in single births at full term.31
Wintrobe and his colleagues were unable to bring about changes in the blood of rabbit fetuses by feeding liver extract to the m0thers or by injecting it into the placentas.32 They concluded that the fetuses were talcing up all the anti-pernicious anemia principle they could from normal maternal sources. 0ther investigators have created pregnant rats with human and swine gastric juices, which contain the antdpernicious anemia factor, and found that the diameter and mean volume of the fetal red blood corpuscles were reduced signilicantly at birthFHZ When the gastric juice had been inactivated by heating it produced no effect. 0n the other hand Wigodslcy and Ivy, using large doses of potent anti-pernicious anemia factor, have been unable to confirm these results. 36
Wintrobe and his colleagues made assays of fetal hog liver and placenta and found these Organs laclcing in physiologically demonstrable amounts of anti-pernicious anemia factor.32 This suggested that a true physiological deliciency exists in the fetus and must be made up by drawing upon maternal sources. More recently however the livers of fetal calves have been demonstrated to contain large amounts of the active principle.37
Oxygen Carrying Power of Fetal Blood
Although oxygen transport will be considered in greater detail in the next chapter it should be pointed out now that a true difference between fetal and adult hemoglobin has been found in several species of animals. This was suspected for some time38-4I and McCarthy has proved that fetal hemoglobin of the goat takes up oxygen more readily at low gas tensions than does maternal hemoc globin. This is illustrated in the oxygen dissociation curves of fetal and maternal hemoglobin reproduced in Fig. 20. The difference becomes less noticeable progressively until about five weelcs after birth when the kid’s hemoglobin attains adult characteristics. 42 Ha11 obtained similar results in fetal goats, rabbits and even chicks. 48 A very pronounced avidity for oxygen at low gas tensions has been demonstrated szin whole blood of calves near term. It was shown that differences in shape and Position of fetal oxygen dissociation curves of whole blood in relation to those of the mother are inexplainable solely on the basis of greater alkalinity of the mother’s blood; they are governecl by true chemical differences between the hemoglobins.
Fig. 20. Oxygen dissociation curves for hernoglobin (c) of sheep fetuses, 44 to 137 days gestation and (b) of their mothers. Limits fall within the shaded are-as. Approximate temperature 190 c; pH 9.3. (I-Iill; from Barcrofn Proc. Roy. Soc Vol. us, 1935 London.)
The human fetus at full term difkers from all others which have been studied, for it has been shown that its hemoglobin when studied in clilute solution takes up oxygen less readily than does that of the motherksss 40 This will be seen in Fig. 2 1 which should be contrasted with Fig. 20. However, buffered (PH 7.4) suspensions of the fetal corpuscles, like whole fetal blood, become more highly saturated with oxygen at low partial pressure than similar suspensions of maternal corpusclesÆ The oxygen dissociation curve of human fetal whole blood (see Fig. 27, F. 66) lies to the left of that of the mother which in turn lies to the right of that for normal human subjects. 41
Fig. 21. Oxygen dissociation curves for hemoglobin of the human fetus (tight) and mother (left) . stiaurowitzx Ztschn Physiol. check, Vol. EIN, 1g35.)
Species differences in avidity of the hemoglobin for oxygen may be related to the degree of maturity reached at birth. It would be interesting to construct dissociation curves for human fetal hemoglobin at the fifth or sixth month of gestation One can not doubt that the fetus of every species possesses a mechanism for assuring adequate intrauterine respiratory function throughout prenatal life. Even though the number of red blood corpuscles may be less than that of the adult they are larger and contain more hemoglobin. 1n those species with great deftciency in number of corpuscles at birth there is a compensatory increase in the avidity with which their hemoglobins combine with oxygen.
The Leukocytes And Platelets
At the time in early embryonic life when secondary erythrocytes are being formed by the yolk sac mesenchyme a few atypical megakaryocytes and leukocytes are si1nilarly produced. Later, with intensive production of erythroblasts in the embryonic liver, granular leukocytes proliferate between the endothelium and the liver cells and a few megakaryocytes are formed there. The pseudopodia of the megakaryocytes produce the blood platelets. Neither granular leukocytes nor platelets are formed in great numbers until fetal hemopoietic activity shifts to the bone marrow about the third month. The lymphocytes of the body develop relatively 1ater. They are formed by transformation of mesenchymal cells in the neighborhood of 1ymphatic vesse1s or in the walls of primitive lymph sacs.
Little is known about the number of leulcocytes and platelets or of the numerica1 relations between granular and nongranular leukocytes. Kindred and Coreyls attempted to determine the number of leukocytes in rat fetuses of different ages but found difficulty in differentiating them from immature forms of the red blood cells. It was apparent however that they increase about seven fold between the 16 and 35 mm. stages. It has been reported that both lymphocytes and polymorphonuclear cells are present in dog embryos at the time the limbs begin to grow out. But in fetuses 26 to 41 mm. long, at which time true neutrophilic leukocytes made their Hrst appearancq the total white cell count was only 1,500 to 2,500 per cmm. Jof blood, with lymphocytes forming the greatest proportions Total nucleated cell counts at the middle of gestation in the cat seem to be about 16,000 per cmmEI The number decreases toward full term reaching 7,500 to 9,000 at birth. Thereafter it remains fairly constant at 8,000 to 9,000 until 120 days, as compared with 10,500 to 12,700 in the adult (Fig. 19). Bruner and his colleagues found less than one third as many leulcocytes in the rat at birth as in the adult rat.18
At the time ok birth ok the human inkant it has been reported that the blood contains gsreat numbers ok leukocytesz in kact one often hears the term, leukocytosis ok birth. Counts greater than 19,000 on the first day after birth, decreasing to about 9,000 by the tenth day, have been reported-Z Japha found 36,000 leulcocytes in the human newborn.24 What factors, other than infections, may be responsible kor the elaboration ok such great numbers ok leukocytes are unknown. Bayer has reported that inhalation ok carbon dioxide led to increasing the number ok granulocytes and breathing oxygen resulted in the formation of more lymphocytes.
The number ok blood platelets at birth has been reported to Huctuate widely. Results ok a number ok investigators have been summarized by Merritt and Davidson.22 Nothing is known about them in prenatal like. The clotting time ok the blood in early prenatal like appears to be long.
The Plasma
The blood has a high water content which declines as the time ok birth approaches. It has been reported that water korms about 88 per cent ok the human ketal blood at the third month ok gestation and only 67 per cent at the ninth lunar month.49 The umbilical vein blood has a water content of 79.5 per cent, as compared with 8 1 .5 per cent in the mothers.ko A sharp postnatal increase in water was observed, reaching a maximum ok about 83 per cent 60 days after birth.
A kew observations in the cat indicate that the speciiic gravity ok the ketal blood is lower (about 1.04) at about the middle of fetal like than it is at kull term (1.052). It is as high or slightly higher near the end oksgestation than in the adult, diminishing gradually thereakter to reach its lowest level (1.043) about two weelcs postnatally.21
The osmotic pressure ok ketal blood colloids appears to be low in some species of animals, but data are incomplete. The subject will be discussed in Chapter VIII in relation to urine formation in the fetal kidneys.
The Cerebrospinal Fluid
A recent study of the cerebrospinal fluid in dog fetuses has shed light upon the nature ok its formation in the adult blood plasma and· the cerebrospinal fluid are alike in respect to their contents of chlorine, sodium and urea during the early part of fetal like. Between 40 and 43 days (fetuses 50 to 60 mm. long) the two fluids cease to be in balance. It is at this time that the cerebrospinal fluid is thought to change from an ultrafiltrate of the blood to a true secretion.
References Cited
1. Arey, L. B. 1940. Developmental Anatomy. saunders, Philadelphia.
2. Bloom, W.
3. Downey. H.
e9.
so. . Parsons, L. G. 1931. J.A.M.A» 97 (e): 973.
. Wintrobe, M. M. se I-I. B. shumaclcen . sweet, M. se W. F. Windle. 194o. Unpub1ished. . Merritt, K. K. se L. T. Davidson. I933. Am. Dis. Child» 46: 99o.
. Japha, A.
. Waugh, T. R» F. T. Merchant se G. B. Maughan.
1938. Maximows Textbook ok Histology, saunders, Philas delphia
1938. Handbook of Hematology, Hoeberz N. Y.
Malassez L. i875. Arch. Physiol. norm. path» set. e, e: est.
cohnsteim J. se N. Zuntz 1884. Pklügeks Arch» 34: 173.
Malassez L. 1889. Comph Rend. soc. Biol., 4I: e.
Jo1ly, J. 19o6. Ibid» 6o: 564.
. Jolly, J. 19o9· Ibid» 66: I36. . Nicho1as, J. s. se E. B. Boswortlx 19e8. Am. J. Physiol» 83: 499.
KnolL W. 19e9. Ztschn milc.—anat. Forsch» is: IV.
. Zeidberg, L. D. 19e9. Am. J. Physiol» 9o: —i7e. . Deseö, D. 19e9. Pllügeks Arch» ee1: ges. . lcindred, J. E. se E. L. Corey. 193o. Anat. Rec» 47: e13.
Kindred, J. E. se E. L. Corey. I931. Physiol. Zool» 4: e94.
. srnith, c. 193e. J. Path. Bact» 35: 717.
Jones, J. M» M. E. shipp se T. A. sonder. se Med., 34: 873.
Baker-oft, J. se J. A. Kennedzk 1939 J. Physiol» 95: i73.
Brunerx H. D» J. van de Erve se A. J. Carlson. I938. Am. J. Physiol» 1e4: 6eo.
Wintrobe, M. M. se I-I. B. shumaclcen 1935. clin. Inv» 14: 837.
1936. Am. J. Anat» 58: gis.
1936. Proc. soc. Exp. Bio1.
Schiff, E. 189e. Jahrlx Icinderh1lc» 34: 159. 191e. In Pkaundler se schlossmanns Diseases ok chi1dren, e: Ist, Lippincoth Philadelphia. » Feldmam W. M. 19eo. Ante-Nara! and Post-Nara! child Physiologyz Longmans Circen, London.
Taylor, D. B. se T. Gotsew 1938. Cited hy Barcrokh in The Brain and . Its Environmenh Yale Univ. Press, New I-Iaven.
Uppmam H. s. 19e4. Am. J. Dis. child» e7: 473. · I939. Am. Med. sei» 198: 646. -
Bland, P. B» L. Goldstein se A. First. I93o. Ibid» 179: 48. Beard, I-I. I-I. se V. C. Me«yers. 1933. Arn. Physiol» Io6: 449.
Wintrobe, M. M» R. E. Kinsey, R. c. B1ount se W. Trager. 1937. Am. J. Med. sei» IV: 449. . stasney, J. sc G. M. Eis-Zins. . stasney, J. sc F. C. Mann« 1937. Ibicl., is: 699.
. Schliche, C. P. 1939. Ibid., 14: 145.
. Wigodslcx I-I. s. sc A. C. 1vy. 1938. Proc. soc. Exp. Biol. sc Med., 38: 787. . Wigodslcy, H. s.,· 0. Richter sc A. C. Ivy. 19Z8. Am. Physiol» us: 21 z. . Kriigetz F. sc W. Gerlach. 1927. Ztschtx f. ges. expetz Med., 54: 653.
. Njcoletti, F. 193o. Axch. AntropoL Crim. Psictiiac Mal. Legale, so: 386. . Perrier, C. sc P. Janelli. 1931. Arch. Fisiol., 29: 289.
. Hentscheh I-I. 1928. Münclx mal. Wchnschty 75: 1237.
. McCarthy, E. F. 19Z3. J. Physiol» 8o: Los.
. Ball, F. G. 1934. . Roos, J. sc C. Romijtx . Haurowitz F. 1935. Hoppesey Ztschtz Physiol. Chem., Erz-s: us.
. Eastmarh N. J., E. M. K. Geiling sc A. M. DeLawder.
1937. stakk Proc., Mayo Clin., is: 49o.
1bid., Sz- ges. 1938. 1bid., ge: 249.
I-Iill, R. 1935. Cited by J. Baker-oft. Proc. Roy. soc. Loncl., B, us: 242.
1933. Johns Hoplcins Hosp. Ball» 53: 246.
. Bayen W. 1932 Jahrb. Kinclerhlk., 134: 3o4. . Talcakusm s., K. Kuroda sc K. Li. . Kurocla, K. sc K. Li.
. Flexnety L. B. 1938. Am. J. Physiol» 124»: ist.
1937. Keijo J. Med., 8: 58. 1937. Ibid., 8: 40.
Cite this page: Hill, M.A. (2024, May 7) Embryology Book - Physiology of the Fetus 4. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Physiology_of_the_Fetus_4
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G